A Different rTMS Protocol for a Different Type of Depression: 20.000 rTMS Pulses for the Treatment of Bipolar Depression Type II
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Sample
2.2. Study Sample Subgroups
2.3. Procedure of rTMS
2.4. BDI and SCL-90-R Questionnaires
2.5. Statistical Procedure
3. Results
3.1. Tolerability
3.2. Clinical Efficacy
3.3. Beck Depression Inventory
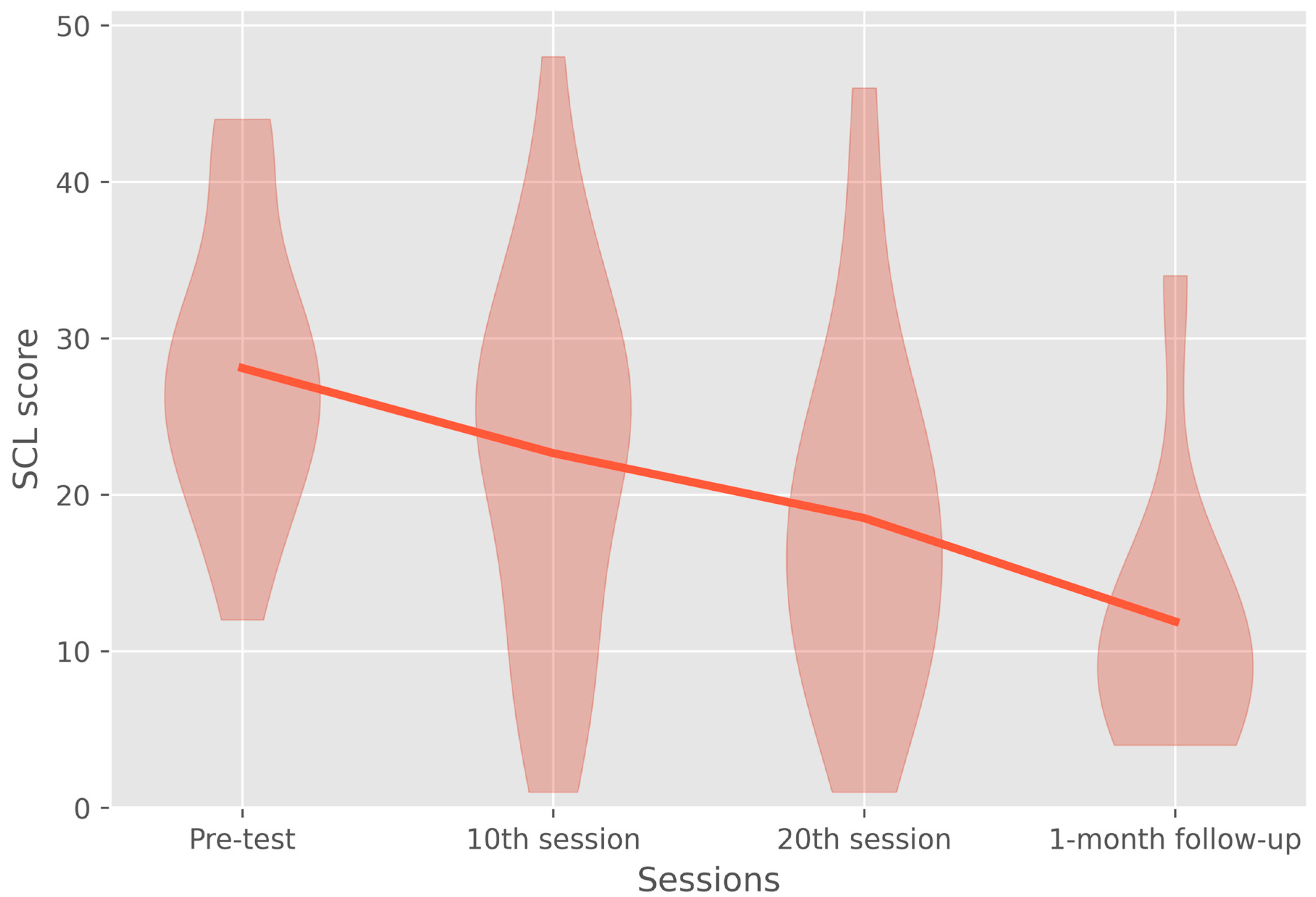
3.4. Symptom Checklist-90-Revised Depression Subscale
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Fagiolini, A.; Forgione, R.; Maccari, M.; Cuomo, A.; Morana, B.; Dell’Osso, M.C.; Pellegrini, F.; Rossi, A. Prevalence, chronicity, burden and borders of bipolar disorder. J. Affect. Disord. 2013, 148, 161–169. [Google Scholar] [CrossRef] [PubMed]
- Pompili, M.; Harnic, D.; Gonda, X.; Forte, A.; Dominici, G.; Innamorati, M.; Fountoulakis, K.N.; Serafini, G.; Sher, L.; Janiri, L.; et al. Impact of living with bipolar patients: Making sense of caregivers’ burden. World J. Psychiatry 2014, 4, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Greenberg, S.; Rosenblum, K.L.; McInnis, M.G.; Muzik, M. The role of social relationships in bipolar disorder: A review. Psychiatry Res. 2014, 219, 248–254. [Google Scholar] [CrossRef] [PubMed]
- Lee, R.S.; Hermens, D.F.; Scott, J.; Redoblado-Hodge, M.A.; Naismith, S.L.; Lagopoulos, J.; Griffiths, K.R.; Porter, M.A.; Hickie, I.B. A meta-analysis of neuropsychological functioning in first-episode bipolar disorders. J. Psychiatr. Res. 2014, 57, 1–11. [Google Scholar] [CrossRef]
- Merikangas, K.R.; Jin, R.; He, J.P.; Kessler, R.C.; Lee, S.; Sampson, N.A.; Viana, M.C.; Andrade, L.H.; Hu, C.; Karam, E.G.; et al. Prevalence and Correlates of Bipolar Spectrum Disorder in the World Mental Health Survey Initiative. Arch. Gen. Psychiatry 2011, 68, 241. [Google Scholar] [CrossRef]
- Weinstock, L.M.; Gaudiano, B.A.; Epstein-Lubow, G.; Tezanos, K.; Celis-deHoyos, C.E.; Miller, I.W. Medication burden in bipolar disorder: A chart review of patients at psychiatric hospital admission. Psychiatry Res. 2014, 216, 24–30. [Google Scholar] [CrossRef]
- Butler, M.; Urosevic, S.; Desai, P.; Sponheim, S.R.; Popp, J.; Nelson, V.A.; Thao, V.; Sunderlin, B. Chapter 1, Introduction. In Treatment for Bipolar Disorder in Adults: A Systematic Review [Internet]; Comparative Effectiveness Review, No. 208; Agency for Healthcare Research and Quality (US): Rockville, MD, USA, 2018; pp. 1–9. Available online: https://www.ncbi.nlm.nih.gov/books/NBK532193/ (accessed on 15 July 2022).
- Yatham, L.N.; Kennedy, S.H.; Parikh, S.V.; Schaffer, A.; Bond, D.J.; Frey, B.N.; Sharma, V.; Goldstein, B.I.; Rej, S.; Beaulieu, S.; et al. Canadian Network for Mood and Anxiety Treatments (CANMAT) and International Society for Bipolar Disorders (ISBD) 2018 guidelines for the management of patients with bipolar disorder. Bipolar Disord. 2018, 20, 97–170. [Google Scholar] [CrossRef]
- Goldberg, J.F.; Freeman, M.P.; Balon, R.; Citrome, L.; Thase, M.E.; Kane, J.M.; Fava, M. The american society of clinical psychopharmacology survey of psychopharmacologists’ practice patterns for the treatment of mood disorders: Research Article: Mood Disorders Prescribing Survey. Depress. Anxiety 2015, 32, 605–613. [Google Scholar] [CrossRef]
- Ghaemi, S.N. Treatment of Rapid-Cycling Bipolar Disorder: Are Antidepressants Mood Destabilizers? Am. J. Psychiatry 2008, 165, 300–302. [Google Scholar] [CrossRef]
- Pilhatsch, M.; Stamm, T.J.; Stahl, P.; Lewitzka, U.; Berghöfer, A.; Sauer, C.; Gitlin, M.; Frye, M.A.; Whybrow, P.C.; Bauer, M. Treatment of bipolar depression with supraphysiologic doses of levothyroxine: A randomized, placebo-controlled study of comorbid anxiety symptoms. Int. J. Bipolar Disord. 2019, 7, 21. [Google Scholar] [CrossRef]
- Andy, Z.; Robin, R.; Alexander, B.-A.; Abbi, L.; Christos, K. High-Dose Levothyroxine for Bipolar Disorder; the Potential Role of Thyroid Function and Genetic Tests. Report from Twenty Cases. Int. J. Psychiatry Res. 2021, 4, 1–7. [Google Scholar] [CrossRef]
- Suttajit, S.; Srisurapanont, M.; Maneeton, N.; Maneeton, B. Quetiapine for acute bipolar depression: A systematic review and meta-analysis. Drug Des Devel. Ther. 2014, 8, 827. [Google Scholar] [CrossRef]
- Koutsomitros, T.; Evagorou, O.; Schuhmann, T.; Zamar, A.; Sack, A.T. Advances in transcranial magnetic stimulation (TMS) and its applications in resistant depression. Psychiatriki 2021, 32 (Suppl. S1), 90–98. [Google Scholar] [CrossRef]
- Cohen, S.L.; Bikson, M.; Badran, B.W.; George, M.S. A visual and narrative timeline of US FDA milestones for Transcranial Magnetic Stimulation (TMS) devices. Brain Stimul. 2022, 15, 73–75. [Google Scholar] [CrossRef]
- Bermudes, R.A.; Lanocha, K.I.; Janicak, P.G. (Eds.) Transcranial Magnetic Stimulation: Clinical Applications for Psychiatric Practice, 1st ed.; American Psychiatric Association Publishing: Arlington, VA, USA, 2018; 216p. [Google Scholar]
- Milev, R.V.; Giacobbe, P.; Kennedy, S.H.; Blumberger, D.M.; Daskalakis, Z.J.; Downar, J.; Modirrousta, M.; Patry, S.; Vila-Rodriguez, F.; Lam, R.W.; et al. Canadian Network for Mood and Anxiety Treatments (CANMAT) 2016 Clinical Guidelines for the Management of Adults with Major Depressive Disorder: Section 4. Neurostimulation Treatments. Can. J. Psychiatry 2016, 61, 561–575. [Google Scholar] [CrossRef]
- Taylor, D.M.; Barnes, T.R.E.; Young, A.H. The Maudsley Prescribing Guidelines in Psychiatry, 14th ed.; The maudsley prescribing guidelines series; John Wiley & Sons: Hoboken, NJ, USA, 2021. [Google Scholar]
- Schlaepfer, T.E.; George, M.S.; Helen Mayberg on behalf of the WFSBP Task Force on Brain Stimulation. WFSBP Guidelines on Brain Stimulation Treatments in Psychiatry. World J. Biol. Psychiatry 2010, 11, 2–18. [Google Scholar] [CrossRef]
- Hallett, M. Transcranial Magnetic Stimulation: A Primer. Neuron 2007, 55, 187–199. [Google Scholar] [CrossRef]
- Pascual-Leone, A.; Valls-Solé, J.; Wassermann, E.M.; Hallett, M. Responses to rapid-rate transcranial magnetic stimulation of the human motor cortex. Brain 1994, 117, 847–858. [Google Scholar] [CrossRef]
- Baeken, C.; Brem, A.K.; Arns, M.; Brunoni, A.R.; Filipčić, I.; Ganho-Ávila, A.; Langguth, B.; Padberg, F.; Poulet, E.; Rachid, F.; et al. Repetitive transcranial magnetic stimulation treatment for depressive disorders: Current knowledge and future directions. Curr. Opin. Psychiatry 2019, 32, 409–415. [Google Scholar] [CrossRef]
- Cohen, D.A.; Freitas, C.; Tormos, J.M.; Oberman, L.; Eldaief, M.; Pascual-Leone, A. Enhancing plasticity through repeated rTMS sessions: The benefits of a night of sleep. Clin. Neurophysiol. 2010, 121, 2159–2164. [Google Scholar] [CrossRef]
- Lefaucheur, J.P.; Aleman, A.; Baeken, C.; Benninger, D.H.; Brunelin, J.; Di Lazzaro, V.; Filipović, S.R.; Grefkes, C.; Hasan, A.; Hummel, F.C.; et al. Evidence-based guidelines on the therapeutic use of repetitive transcranial magnetic stimulation (rTMS): An update (2014–2018). Clin. Neurophysiol. 2020, 131, 474–528. [Google Scholar] [CrossRef] [PubMed]
- Brunoni, A.R.; Chaimani, A.; Moffa, A.H.; Razza, L.B.; Gattaz, W.F.; Daskalakis, Z.J.; Carvalho, A.F. Repetitive Transcranial Magnetic Stimulation for the Acute Treatment of Major Depressive Episodes: A Systematic Review with Network Meta-analysis. JAMA Psychiatry 2017, 74, 143. [Google Scholar] [CrossRef] [PubMed]
- Gold, A.K.; Ornelas, A.C.; Cirillo, P.; Caldieraro, M.A.; Nardi, A.E.; Nierenberg, A.A.; Kinrys, G. Clinical applications of transcranial magnetic stimulation in bipolar disorder. Brain Behav. 2019, 9, e01419. Available online: https://onlinelibrary.wiley.com/doi/10.1002/brb3.1419 (accessed on 13 July 2022). [CrossRef] [PubMed]
- McGirr, A.; Karmani, S.; Arsappa, R.; Berlim, M.T.; Thirthalli, J.; Muralidharan, K.; Yatham, L.N. Clinical efficacy and safety of repetitive transcranial magnetic stimulation in acute bipolar depression. World Psychiatry 2016, 15, 85–86. [Google Scholar] [CrossRef]
- Nguyen, T.D.; Hieronymus, F.; Lorentzen, R.; McGirr, A.; Østergaard, S.D. The efficacy of repetitive transcranial magnetic stimulation (rTMS) for bipolar depression: A systematic review and meta-analysis. J. Affect. Disord. 2020, 279, 250–255. [Google Scholar] [CrossRef]
- Judd, L.L.; Akiskal, H.S.; Schettler, P.J.; Endicott, J.; Maser, J.; Solomon, D.A.; Leon, A.C.; Rice, J.A.; Keller, M.B. The Long-term Natural History of the Weekly Symptomatic Status of Bipolar I Disorder. Arch. Gen. Psychiatry 2002, 59, 530–537. [Google Scholar] [CrossRef]
- Forte, A.; Baldessarini, R.J.; Tondo, L.; Vázquez, G.H.; Pompili, M.; Girardi, P. Long-term morbidity in bipolar-I, bipolar-II, and unipolar major depressive disorders. J. Affect. Disord. 2015, 178, 71–78. [Google Scholar] [CrossRef]
- Fitzgerald, P.B.; Hoy, K.E.; Elliot, D.; McQueen, S.; Wambeek, L.E.; Daskalakis, Z.J. A negative double-blind controlled trial of sequential bilateral rTMS in the treatment of bipolar depression. J. Affect. Disord. 2016, 198, 158–162. [Google Scholar] [CrossRef]
- Rachid, F. Repetitive Transcranial Magnetic Stimulation and Treatment-emergent Mania and Hypomania: A Review of the Literature. J. Psychiatr. Pract. 2017, 23, 150–159. [Google Scholar] [CrossRef]
- Blumberger, D.M.; Maller, J.J.; Thomson, L.; Mulsant, B.H.; Rajji, T.K.; Maher, M.; Brown, P.E.; Downar, J.; Vila-Rodriguez, F.; Fitzgerald, P.B.; et al. Unilateral and bilateral MRI-targeted repetitive transcranial magnetic stimulation for treatment- resistant depression: A randomized controlled study. J. Psychiatry Neurosci. 2016, 41, E58–E66. [Google Scholar] [CrossRef]
- Herwig, U.; Cardenas-Morales, L.; Connemann, B.J.; Kammer, T.; Schönfeldt-Lecuona, C. Sham or real--post hoc estimation of stimulation condition in a randomized transcranial magnetic stimulation trial. Neurosci. Lett. 2010, 471, 30–33. [Google Scholar] [CrossRef]
- Herwig, U.; Fallgatter, A.J.; Höppner, J.; Eschweiler, G.W.; Kron, M.; Hajak, G.; Padberg, F.; Naderi-Heiden, A.; Abler, B.; Eichhammer, P.; et al. Antidepressant effects of augmentative transcranial magnetic stimulation: Randomised multicentre trial. Br. J. Psychiatry 2007, 191, 441–448. [Google Scholar] [CrossRef]
- Sayar, G.H.; Ozten, E.; Tan, O.; Tarhan, N. Transcranial magnetic stimulation for treating depression in elderly patients. Neuropsychiatr. Dis. Treat. 2013, 9, 501–504. [Google Scholar] [CrossRef]
- Sayar, G.H.; Ozten, E.; Tufan, E.; Cerit, C.; Kağan, G.; Dilbaz, N.; Tarhan, N. Transcranial magnetic stimulation during pregnancy. Arch. Women’s Ment. Health 2013, 17, 311–315. [Google Scholar] [CrossRef]
- Wideman, T.H.; Sullivan, M.J.L.; Inada, S.; McIntyre, D.; Kumagai, M.; Yahagi, N.; Turner, J.R.; Upton, J.; Burns, R.J.; Rothman, A.J.; et al. Beck Depression Inventory (BDI). In Encyclopedia of Behavioral Medicine; Gellman, M.D., Turner, J.R., Eds.; Springer: New York, NY, USA, 2013; pp. 178–179. Available online: http://link.springer.com/10.1007/978-1-4419-1005-9_441 (accessed on 10 July 2022).
- Richter, P.; Werner, J.; Heerlein, A.; Kraus, A.; Sauer, H. On the Validity of the Beck Depression Inventory. Psychopathology 1998, 31, 160–168. [Google Scholar] [CrossRef]
- Richter, P. Zur Konstruktvalidität des Beck-Depressionsinventars (BDI) bei der Erfassung Depressiver Verläufe: Ein Empirischer und Methodischer Beitrag (Psychologie); Theorie und Forschung; Roderer Verlag: Regensburg, Germany, 1991; 306p. [Google Scholar]
- Beck, A.T.; Steer, R.A.; Carbin, M.G. Psychometric properties of the Beck Depression Inventory: Twenty-five years of evaluation. Clin. Psychol. Rev. 1988, 8, 77–100. [Google Scholar] [CrossRef]
- Derogatis, L. SCL-90-R: SCL 90 R Administration, Scoring and Procedures Manual II for the Revised Version and Other Instruments of the Psychopathology Rating Scale Series; Clinical Psychometric Research: Towson, MD, USA, 1992. [Google Scholar]
- Holi, M. Assessment of Psychiatric Symptoms Using the SCL-90. Ph.D. Thesis, Department of Psychiatry, Helsinki University, Helsinki, Finland, 2003. [Google Scholar]
- Mann, J.J. The Medical Management of Depression. N. Engl. J. Med. 2005, 353, 1819–1834. [Google Scholar] [CrossRef]
- Gershon, A.A.; Dannon, P.N.; Grunhaus, L. Transcranial Magnetic Stimulation in the Treatment of Depression. Am. J. Psychiatry 2003, 160, 835–845. [Google Scholar] [CrossRef]
- Üçok, A.; Karaveli, D.; Kundakçi, T.; Yazici, O. Comorbidity of personality disorders with bipolar mood disorders. Compr. Psychiatry 1998, 39, 72–74. [Google Scholar] [CrossRef]
- Altshuler, L.; Suppes, T.; Black, D.; Nolen, W.A.; Keck, P.E.; Frye, M.A.; McElroy, S.; Kupka, R.; Grunze, H.; Walden, J.; et al. Impact of Antidepressant Discontinuation after Acute Bipolar Depression Remission on Rates of Depressive Relapse at 1-Year Follow-Up. Am. J. Psychiatry 2003, 160, 1252–1262. [Google Scholar] [CrossRef]
- Hunter, A.M.; Minzenberg, M.J.; Cook, I.A.; Krantz, D.E.; Levitt, J.G.; Rotstein, N.M.; Chawla, S.; Leuchter, A.F. Concomitant medication use and clinical outcome of repetitive Transcranial Magnetic Stimulation (rTMS) treatment of Major Depressive Disorder. Brain Behav. 2019, 9, e01275. [Google Scholar] [CrossRef] [PubMed]
- Ward, H.B.; Yip, A.; Siddiqui, R.; Morales, O.G.; Seiner, S.J.; Siddiqi, S.H. Borderline personality traits do not influence response to TMS. J. Affect. Disord. 2020, 281, 834–838. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.; Nassan, M.; Singh, B.; Croarkin, P.E.; Kung, S. Response rate underreports improvement in patients with major depressive disorder and comorbidities treated with repetitive transcranial magnetic stimulation (rTMS). Psychiatry Res. Commun. 2022, 2, 100033. [Google Scholar] [CrossRef]
- Gündoğmuş, İ.; Örnek, B.; Altınyay, N. The Impact of Repetetive Transcranial Magnetic Stimulation (rTMS) Treatment on Borderline Personality Disorder Symptoms: A Case Report. Psychiatry Behav. Sci. 2019, 9, 53–56. [Google Scholar] [CrossRef]

| Any Personality Disorder | Any Benzodiazepines | Total | ||
|---|---|---|---|---|
| No | Yes | |||
| No | 7 | 3 | 10 | |
| Females | 4 | 0 | 4 | |
| Males | 3 | 3 | 6 | |
| Yes | 5 | 8 | 13 | |
| Females | 1 | 7 | 8 | |
| Males | 4 | 1 | 5 | |
| Total | 12 | 11 | 23 | |
| Females | 5 | 7 | 12 | |
| Males | 7 | 4 | 11 | |
| Parameter | Numerator df | Denominator df | F-Statistic | p-Value |
|---|---|---|---|---|
| Session | 3 | 35.99 | 0.717 | 0.549 |
| Any personality disorder | 1 | 31.002 | 0.002 | 0.964 |
| Benzodiazepine use | 1 | 31.918 | 0.346 | 0.561 |
| Bidaily treatment | 1 | 19.059 | 0.153 | 0.7 |
| Age | 1 | 32.467 | 0.478 | 0.494 |
| Pre-test | 1 | 22.468 | 9.566 | 0.005 |
| Session * Any personality disorder | 3 | 35.564 | 0.08 | 0.97 |
| Session * Benzodiazepine use | 3 | 35.632 | 0.58 | 0.632 |
| Session * Bidaily treatment | 3 | 36 | 0.576 | 0.634 |
| Session * Age | 3 | 36.52 | 0.576 | 0.634 |
| Session * Pre-test | 3 | 35.938 | 6.187 | 0.002 |
| Session | Mean Difference | df | p-Value | |
|---|---|---|---|---|
| Pre-test | 10th session | 7.176 | 33.028 | 0.009 |
| 20th session | 10.982 | 34.776 | <0.001 | |
| Follow-up | 18.187 | 38.873 | <0.001 | |
| 10th session | Pre-test | −7.176 | 33.028 | 0.009 |
| 20th session | 3.806 | 34.776 | 0.614 | |
| Follow-up | 11.011 | 38.873 | 0.050 | |
| 20th session | Pre-test | −10.982 | 34.776 | <0.001 |
| 10th session | −3.806 | 34.776 | 0.614 | |
| Follow-up | 7.205 | 38.789 | 0.497 | |
| Follow-up | Pre-test | −18.187 | 38.873 | <0.001 |
| 10th session | −11.011 | 38.873 | 0.050 | |
| 20th session | −7.205 | 38.789 | 0.497 | |
| Parameter | Numerator df | Denominator df | F-Statistic | p-Value |
|---|---|---|---|---|
| Session | 3 | 35.975 | 1.723 | 0.180 |
| Any personality disorder | 1 | 33.678 | 1.978 | 0.169 |
| Benzodiazepine use | 1 | 33.317 | 0.807 | 0.375 |
| Bidaily treatment | 1 | 18.855 | 0.000 | 0.995 |
| Age | 1 | 32.737 | 0.184 | 0.671 |
| Pre-test | 1 | 26.764 | 6.476 | 0.017 |
| Session * Any personality disorder | 3 | 36.199 | 0.963 | 0.420 |
| Session * Benzodiazepine use | 3 | 36.324 | 0.813 | 0.495 |
| Session * Bidaily treatment | 3 | 35.890 | 0.284 | 0.837 |
| Session * Age | 3 | 37.194 | 0.096 | 0.962 |
| Session * Pre-test | 3 | 36.695 | 5.309 | 0.004 |
| Session | Mean Difference | df | p-Value | |
|---|---|---|---|---|
| Pre-test | 10th session | 5.322 | 33.441 | 0.099 |
| 20th session | 9.814 | 35.353 | <0.001 | |
| Follow-up | 15.607 | 39.647 | 0.002 | |
| 10th session | Pre-test | −5.322 | 33.441 | 0.099 |
| 20th session | 4.492 | 35.353 | 0.355 | |
| Follow-up | 10.285 | 39.647 | 0.076 | |
| 20th session | Pre-test | −9.814 | 35.353 | <0.001 |
| 10th session | −4.492 | 35.353 | 0.355 | |
| Follow-up | 5.793 | 39.776 | 0.959 | |
| Follow-up | Pre-test | −15.607 | 39.647 | 0.002 |
| 10th session | −10.285 | 39.647 | 0.076 | |
| 20th session | −5.793 | 39.776 | 0.959 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Koutsomitros, T.; van der Zee, K.T.; Evagorou, O.; Schuhmann, T.; Zamar, A.C.; Sack, A.T. A Different rTMS Protocol for a Different Type of Depression: 20.000 rTMS Pulses for the Treatment of Bipolar Depression Type II. J. Clin. Med. 2022, 11, 5434. https://doi.org/10.3390/jcm11185434
Koutsomitros T, van der Zee KT, Evagorou O, Schuhmann T, Zamar AC, Sack AT. A Different rTMS Protocol for a Different Type of Depression: 20.000 rTMS Pulses for the Treatment of Bipolar Depression Type II. Journal of Clinical Medicine. 2022; 11(18):5434. https://doi.org/10.3390/jcm11185434
Chicago/Turabian StyleKoutsomitros, Theodoros, Kenneth T. van der Zee, Olympia Evagorou, Teresa Schuhmann, Antonis C. Zamar, and Alexander T. Sack. 2022. "A Different rTMS Protocol for a Different Type of Depression: 20.000 rTMS Pulses for the Treatment of Bipolar Depression Type II" Journal of Clinical Medicine 11, no. 18: 5434. https://doi.org/10.3390/jcm11185434
APA StyleKoutsomitros, T., van der Zee, K. T., Evagorou, O., Schuhmann, T., Zamar, A. C., & Sack, A. T. (2022). A Different rTMS Protocol for a Different Type of Depression: 20.000 rTMS Pulses for the Treatment of Bipolar Depression Type II. Journal of Clinical Medicine, 11(18), 5434. https://doi.org/10.3390/jcm11185434







