Lifitegrast Ophthalmic Solution 5% Is a Safe and Efficient Eyedrop for Dry Eye Disease: A Systematic Review and Meta-Analysis
Abstract
1. Introduction
2. Methods
2.1. Literature Search
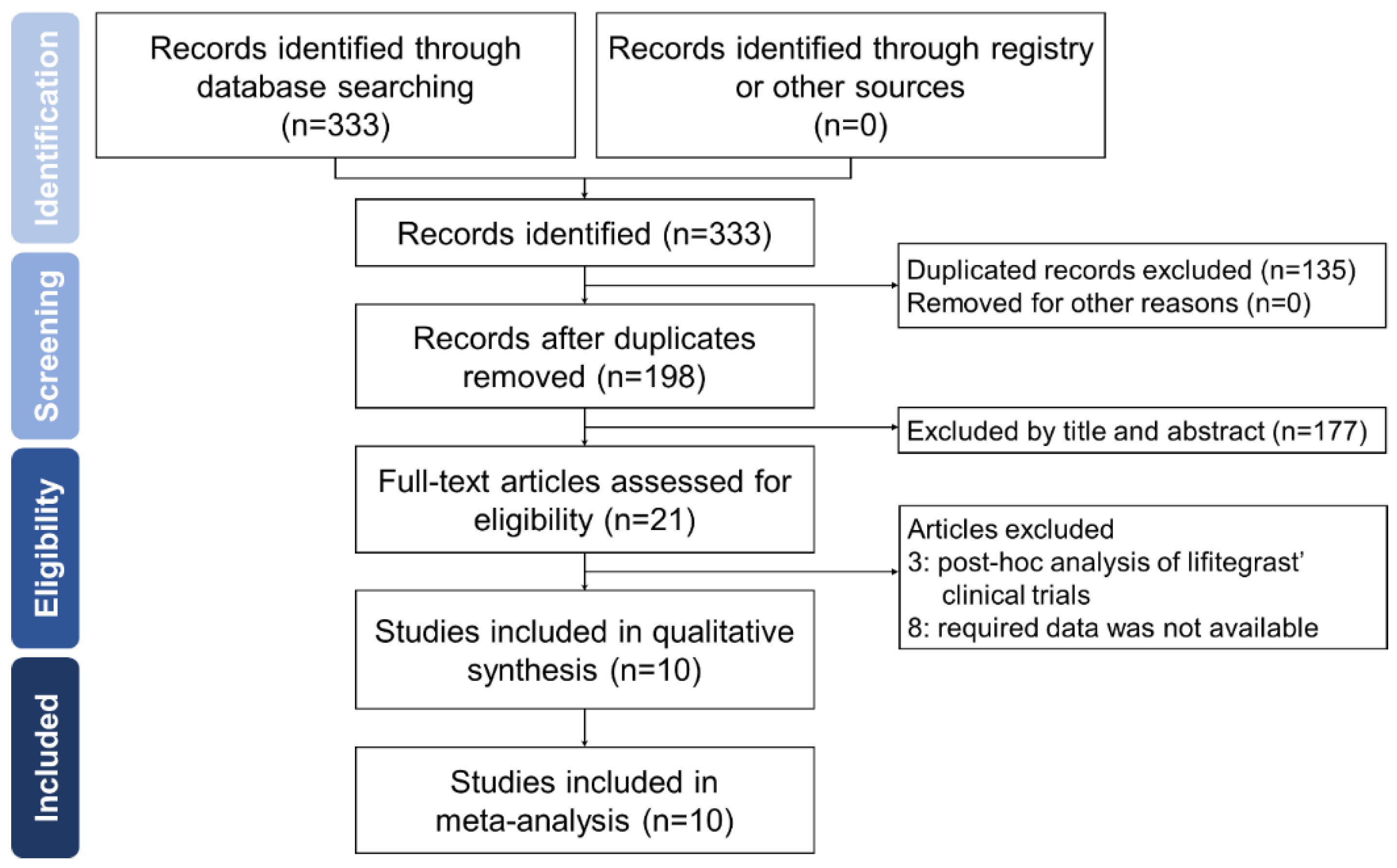
2.2. Study Selection
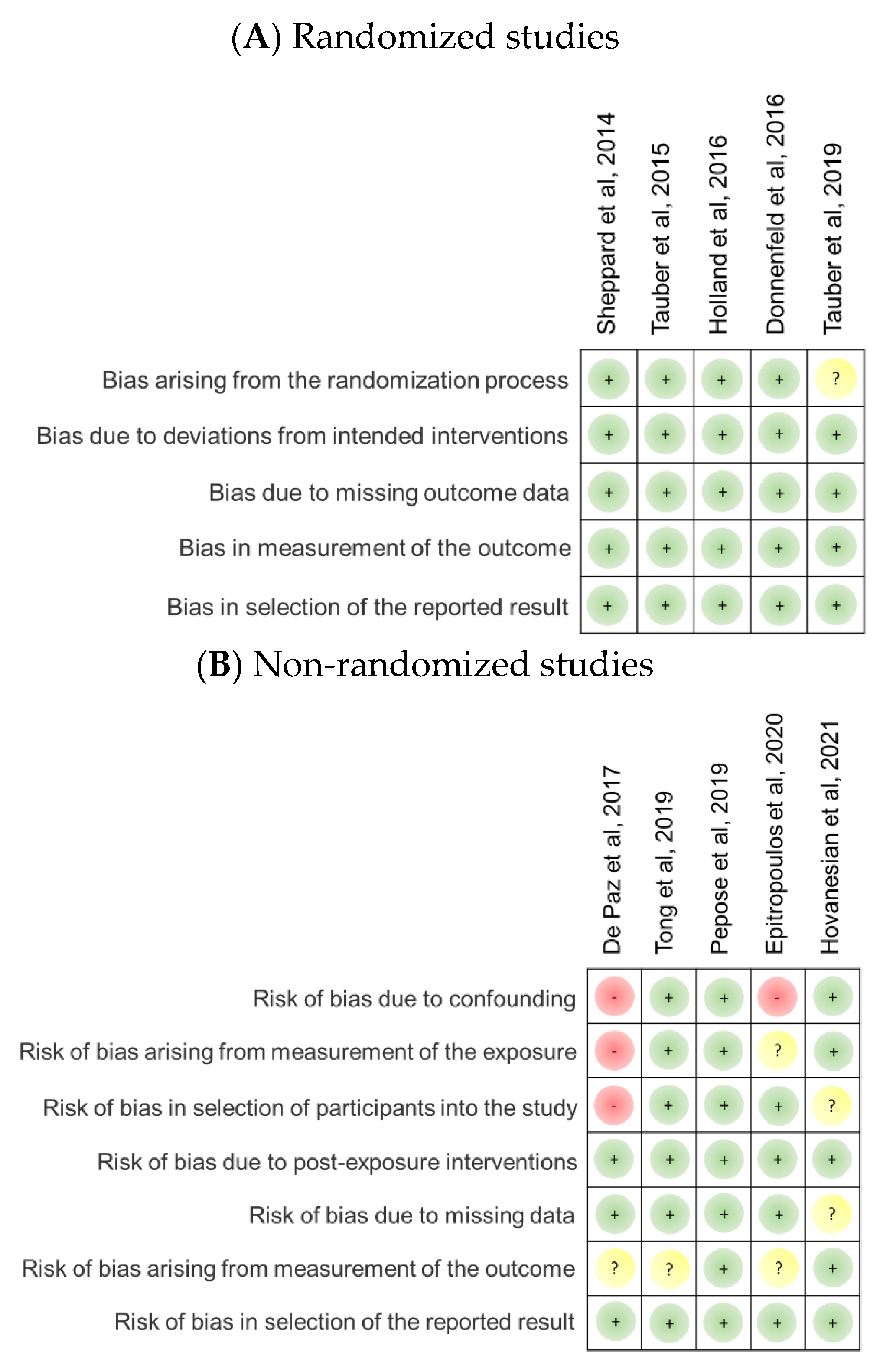
2.3. Data Extraction and Risk-of-Bias Assessment
2.4. Statistical Analysis
3. Results
3.1. Characteristic of Included Studies
Risk of Bias of Included Studies
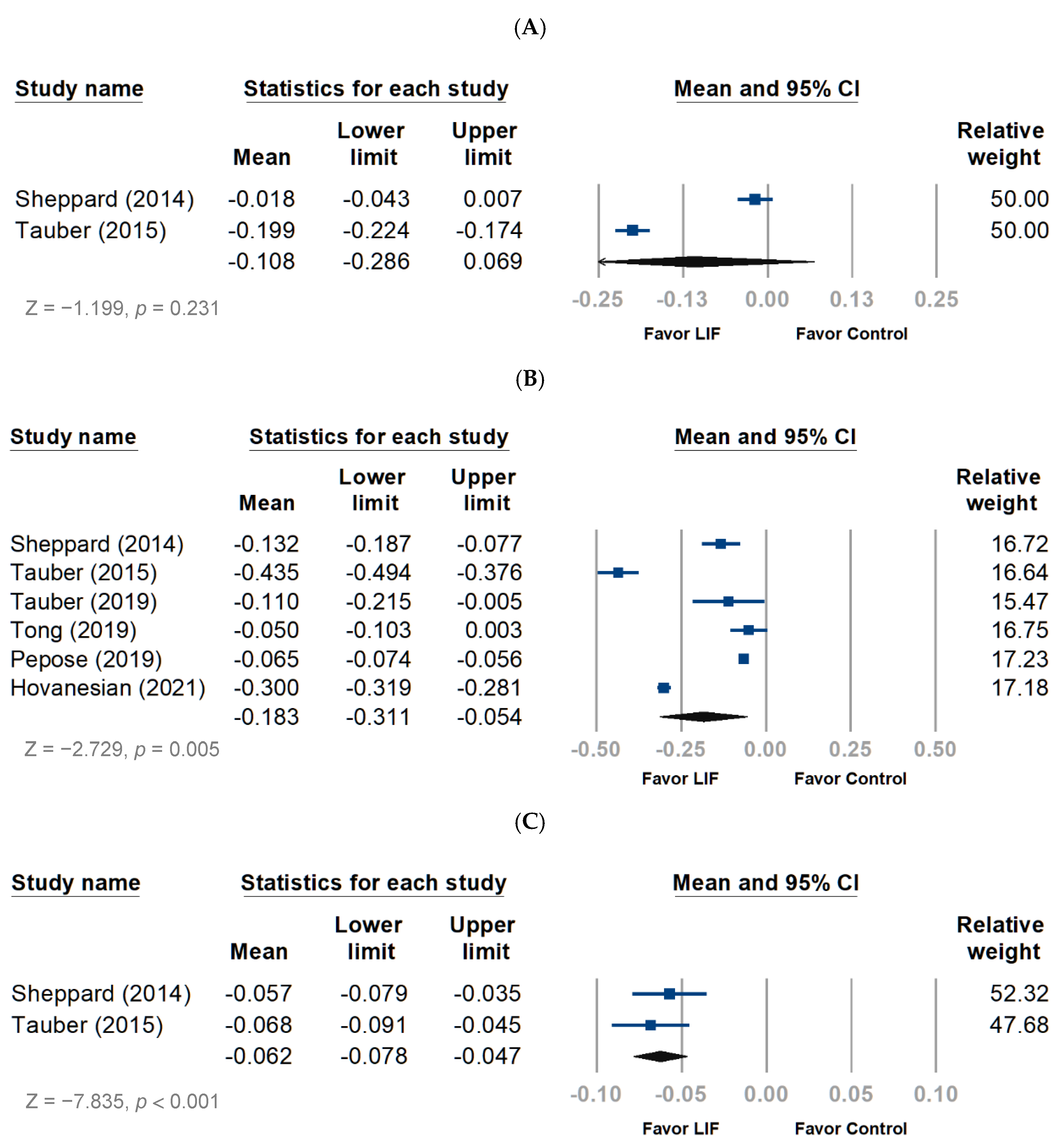
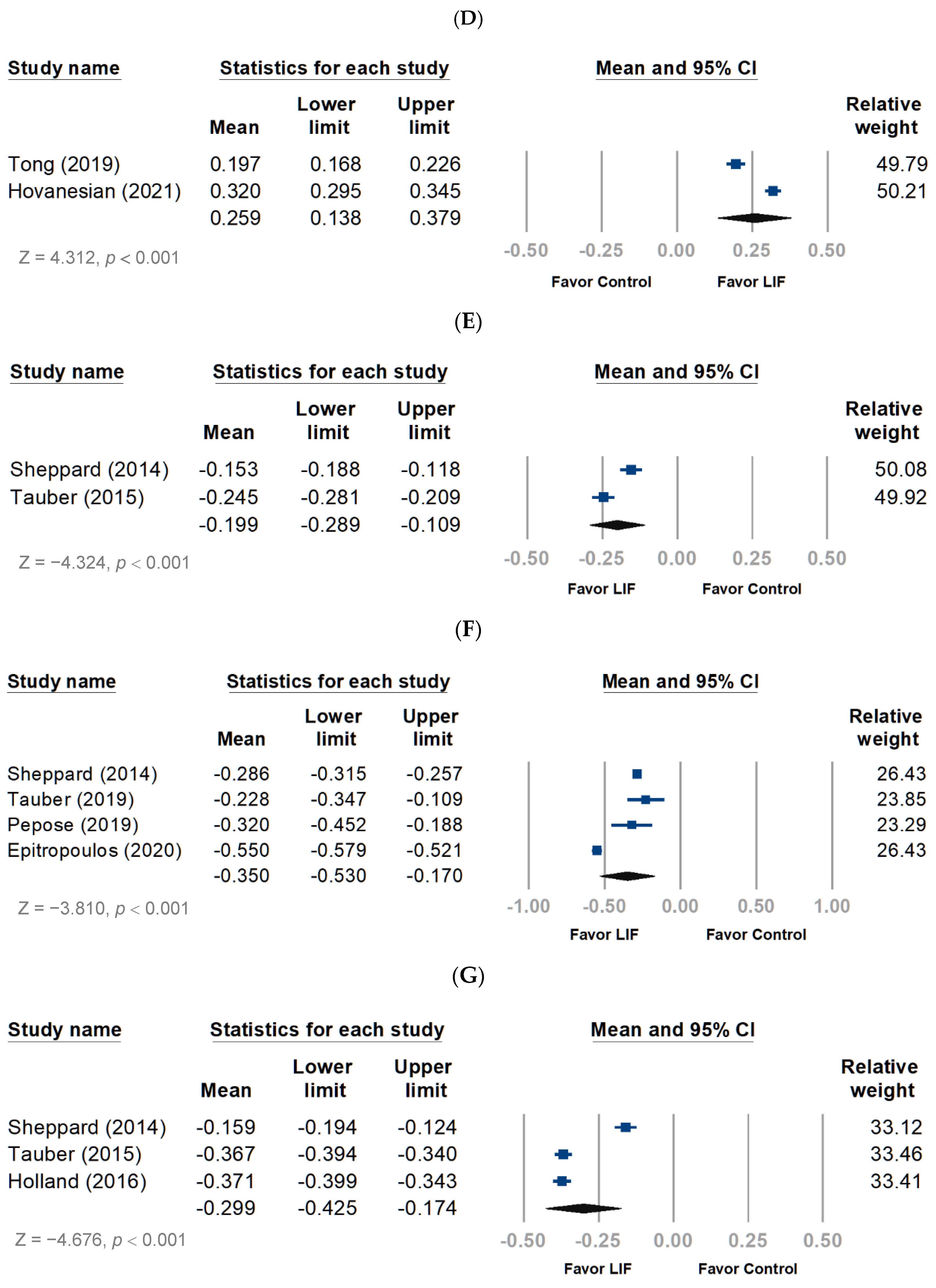
3.2. Efficacy Outcomes
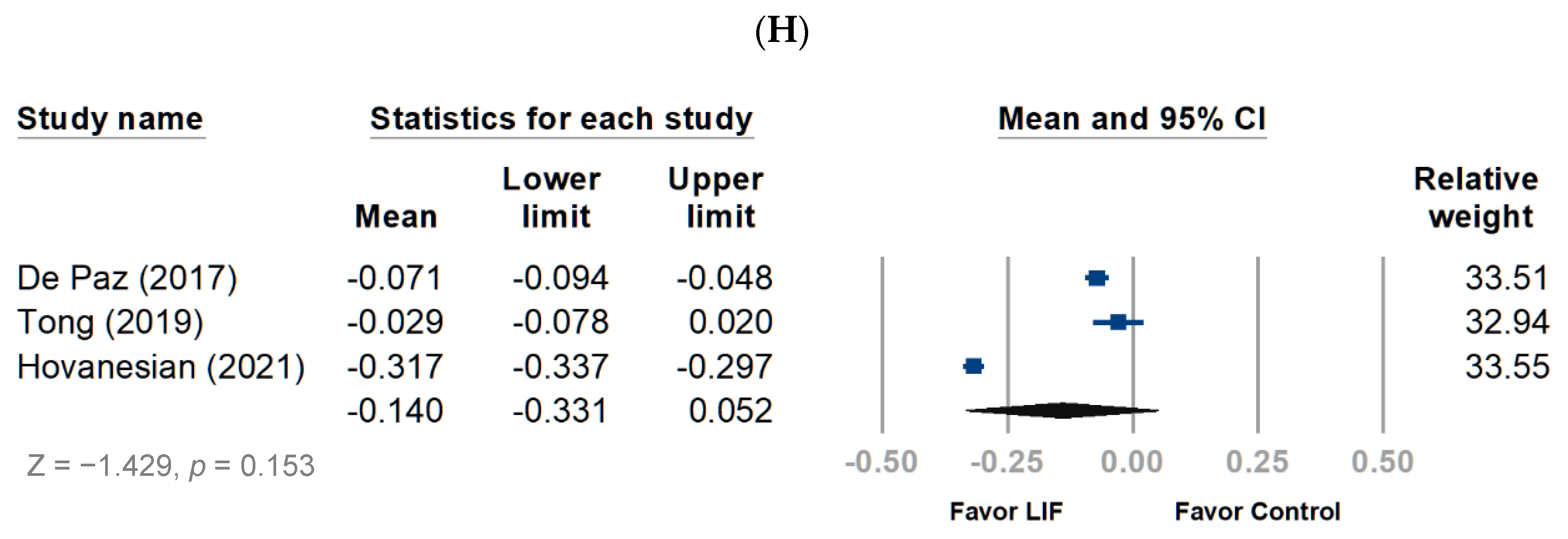
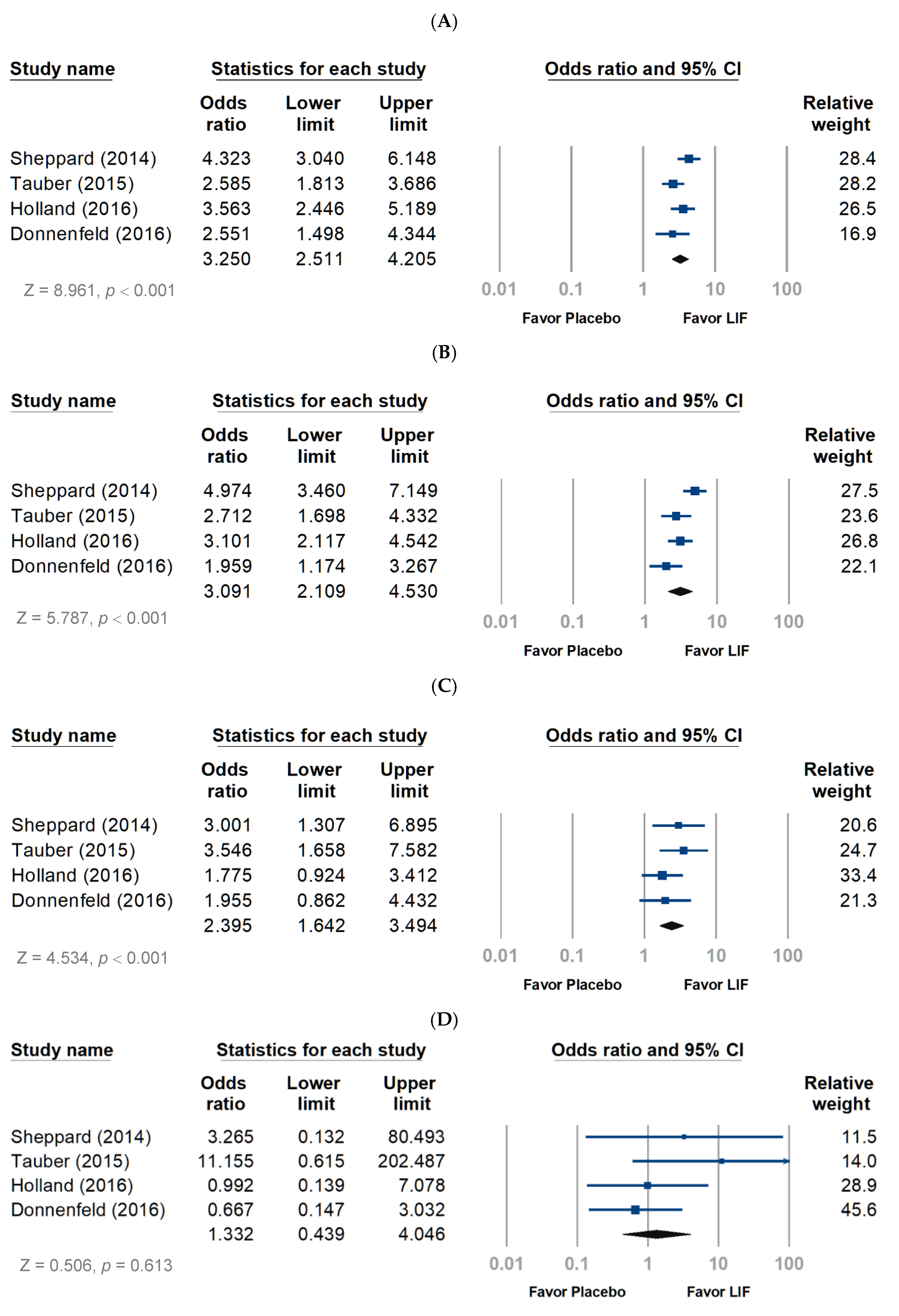
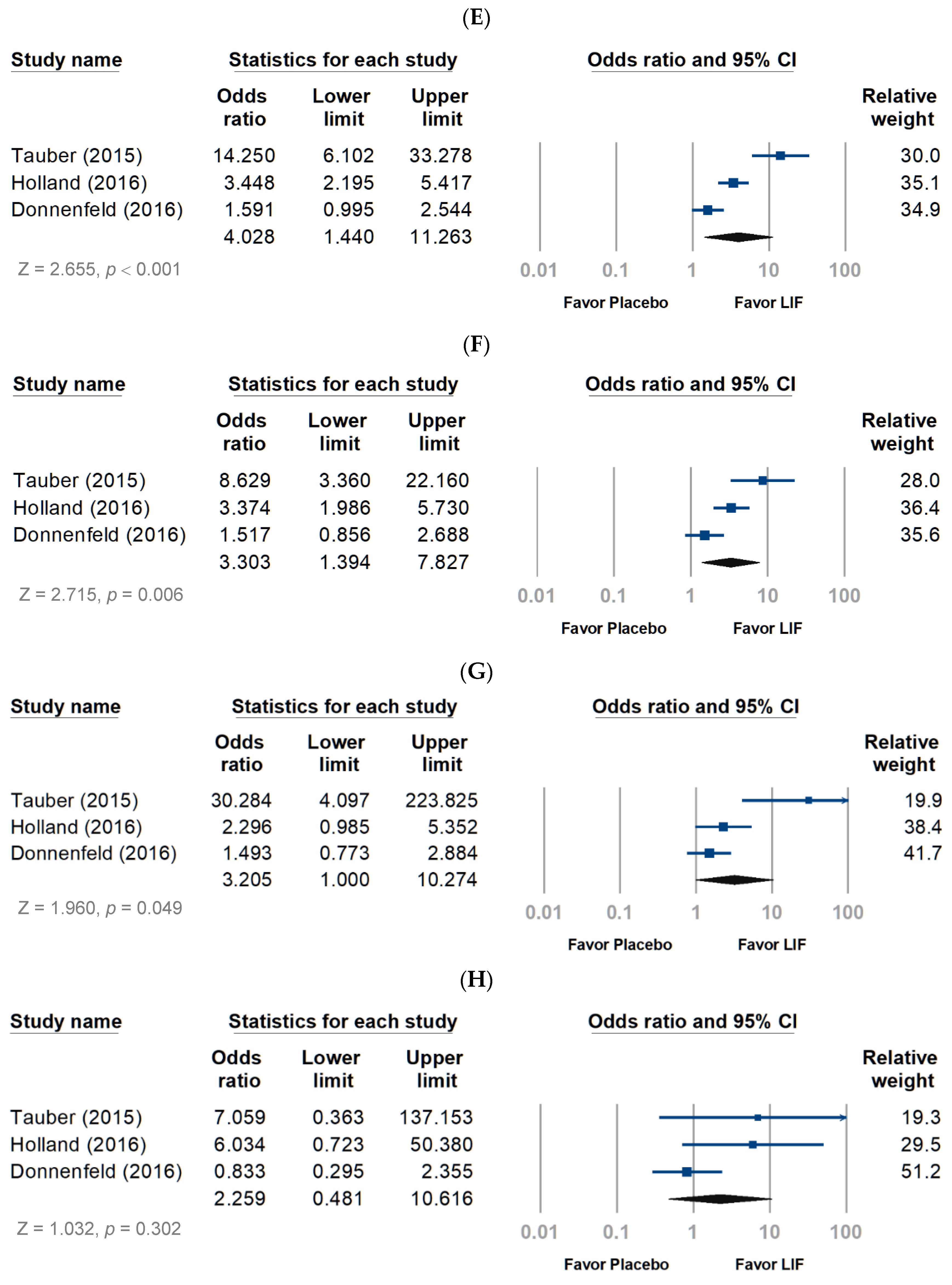
3.2.1. Objective Evaluation
3.2.2. Subjective Evaluation
3.3. Safety Outcomes
4. Discussion
Strengths and Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
References
- Perez, V.L.; Pflugfelder, S.C.; Zhang, S.; Shojaei, A.; Haque, R. Lifitegrast, a Novel Integrin Antagonist for Treatment of Dry Eye Disease. Ocul. Surf. 2016, 14, 207–215. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Volpe, E.A.; Gandhi, N.B.; Schaumburg, C.S.; Siemasko, K.F.; Pangelinan, S.B.; Kelly, S.D.; Hayday, A.C.; Li, D.-Q.; Stern, M.E.; et al. NK Cells Promote Th-17 Mediated Corneal Barrier Disruption in Dry Eye. PLoS ONE 2012, 7, e36822. [Google Scholar] [CrossRef]
- De Paiva, C.S.; Chotikavanich, S.; Pangelinan, S.B.; Pitcher, J.D.; Fang, B.; Zheng, X.; Ma, P.; Farley, W.J.; Siemasko, K.F.; Niederkorn, J.Y.; et al. IL-17 disrupts corneal barrier following desiccating stress. Mucosal Immunol. 2009, 2, 243–253. [Google Scholar] [CrossRef] [PubMed]
- Aggarwal, S.; Peck, T.; Olsakovsky, L. Dry eye syndrome in menopause and perimenopausal age group. J. Mid-life Health 2017, 8, 51–54. [Google Scholar] [CrossRef]
- Stern, M.E.; Schaumburg, C.S.; Pflugfelder, S.C. Dry Eye as a Mucosal Autoimmune Disease. Int. Rev. Immunol. 2013, 32, 19–41. [Google Scholar] [CrossRef]
- Mandell, J.T.; Idarraga, M.; Kumar, N.; Galor, A. Impact of Air Pollution and Weather on Dry Eye. J. Clin. Med. 2020, 9, 3740. [Google Scholar] [CrossRef]
- Gayton, J.L. Etiology, prevalence, and treatment of dry eye disease. Clin. Ophthalmol. 2009, 3, 405–412. [Google Scholar] [CrossRef]
- Semba, C.P.; Torkildsen, G.L.; Lonsdale, J.D.; McLaurin, E.B.; Geffin, J.A.; Mundorf, T.K.; Kennedy, K.S.; Ousler, G.W. A Phase 2 Randomized, Double-Masked, Placebo-Controlled Study of a Novel Integrin Antagonist (SAR 1118) for the Treatment of Dry Eye. Am. J. Ophthalmol. 2012, 153, 1050–1060. [Google Scholar] [CrossRef]
- Sheppard, J.D.; Torkildsen, G.L.; Lonsdale, J.D.; D’Ambrosio, F.A.; McLaurin, E.B.; Eiferman, R.A.; Kennedy, K.S.; Semba, C.P. Lifitegrast Ophthalmic Solution 5.0% for Treatment of Dry Eye Disease: Results of the OPUS-1 phase 3 study. Ophthalmology 2014, 121, 475–483. [Google Scholar] [CrossRef]
- Tauber, J.; Karpecki, P.; Latkany, R.; Luchs, J.; Martel, J.; Sall, K.; Raychaudhuri, A.; Smith, V.; Semba, C.P. Lifitegrast Ophthalmic Solution 5.0% versus Placebo for Treatment of Dry Eye Disease: Results of the Randomized Phase III OPUS-2 Study. Ophthalmology 2015, 122, 2423–2431. [Google Scholar] [CrossRef]
- Holland, E.J.; Luchs, J.; Karpecki, P.M.; Nichols, K.K.; Jackson, M.A.; Sall, K.; Tauber, J.; Roy, M.; Raychaudhuri, A.; Shojaei, A. Lifitegrast for the Treatment of Dry Eye Disease: Results of a Phase III, Randomized, Double-Masked, Placebo-Controlled Trial (OPUS-3). Ophthalmology 2017, 124, 53–60. [Google Scholar] [CrossRef] [PubMed]
- Donnenfeld, E.D.; Karpecki, P.M.; Majmudar, P.A.; Nichols, K.K.; Raychaudhuri, A.; Roy, M.; Semba, C.P. Safety of Lifitegrast Ophthalmic Solution 5.0% in Patients with Dry Eye Disease: A 1-Year, Multicenter, Randomized, Placebo-Controlled Study. Cornea 2016, 35, 741–748. [Google Scholar] [CrossRef] [PubMed]
- De Paz, C.J.; Gonzalez, A.L.; Ngo, C. SIDRE: Symptomatic Improvement of Dry Eye Study. Clin. Optom. 2017, 9, 49–54. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Tauber, J. A 6-Week, Prospective, Randomized, Single-Masked Study of Lifitegrast Ophthalmic Solution 5% Versus Thermal Pulsation Procedure for Treatment of Inflammatory Meibomian Gland Dysfunction. Cornea 2020, 39, 403–407. [Google Scholar] [CrossRef]
- Tong, A.Y.; Passi, S.F.; Gupta, P.K. Clinical Outcomes of Lifitegrast 5% Ophthalmic Solution in the Treatment of Dry Eye Disease. Eye Contact Lens 2020, 46 (Suppl. S1), S20–S24. [Google Scholar] [CrossRef]
- Pepose, J.S.; Qazi, M.A.; Devries, D.K. Longitudinal changes in dry eye symptoms and signs following lifitegrast therapy and relationship to tear osmolarity. Clin. Ophthalmol. 2019, 13, 571–579. [Google Scholar] [CrossRef]
- Epitropoulos, A.T.; Therattil, A.; Periman, L.M.; Rosenberg, E.D. Improving Tolerance and Compliance with Topical Immunomodulators Using Micro-Emulsion Lipid Layer Artificial Tears. Clin. Ophthalmol. 2020, 14, 1921–1929. [Google Scholar] [CrossRef]
- Hovanesian, J.A.; Nichols, K.K.; Jackson, M.; Katz, J.; Chan, A.; Glassberg, M.B.; Sloesen, B.; Korves, C.; Nguyen, C.; Syntosi, A. Real-World Experience with Lifitegrast Ophthalmic Solution (Xiidra®) in the US and Canada: Retrospective Study of Patient Characteristics, Treatment Patterns, and Clinical Effectiveness in 600 Patients with Dry Eye Disease. Clin. Ophthalmol. 2021, 15, 1041–1054. [Google Scholar] [CrossRef]
- Bowden, A.; Foxrushby, J.; Nyandieka, L.; Kangeri, J. This journal issue contains abstracts that have been accepted for presentation as the 8th Annual Conference of the International Society for Quality of Life Research (ISOQOL). Qual. Life Res. 2001, 10, 193–306. [Google Scholar] [CrossRef][Green Version]
- Holland, E.J.; Jackson, M.A.; Donnenfeld, E.; Piccolo, R.; Cohen, A.; Barabino, S.; Rolando, M.; Figueiredo, F.C. Efficacy of Lifitegrast Ophthalmic Solution, 5.0%, in Patients with Moderate to Severe Dry Eye Disease: A Post Hoc Analysis of 2 Randomized Clinical Trials. JAMA Ophthalmol. 2021, 139, 1200. [Google Scholar] [CrossRef]
- Atallah, R.T.; Castanos, M.V.; Najac, R.; Donnenfeld, E. Six months’ treatment with lifitegrast in patients with moderate-to-severe symptomatic dry eye: A retrospective chart review. Clin. Ophthalmol. 2019, 13, 1033–1037. [Google Scholar] [CrossRef] [PubMed]
- Semba, C.P.; Swearingen, D.; Smith, V.L.; Newman, M.S.; O’Neill, C.A.; Burnier, J.P.; Haughey, D.B.; Gadek, T.R. Safety and Pharmacokinetics of a Novel Lymphocyte Function-associated Antigen-1 Antagonist Ophthalmic Solution (SAR 1118) in Healthy Adults. J. Ocul. Pharmacol. Ther. 2011, 27, 99–104. [Google Scholar] [CrossRef] [PubMed]
- Paskowitz, D.M.; Nguyen, Q.D.; Gehlbach, P.; Handa, J.T.; Solomon, S.; Stark, W.; Shaikh, O.; Semba, C.; Gadek, T.R.; Do, D.V. Safety, tolerability, and bioavailability of topical SAR 1118, a novel antagonist of lymphocyte function-associated antigen-1: A phase 1b study. Eye 2012, 26, 944–949. [Google Scholar] [CrossRef][Green Version]
- Pflugfelder, S.C.; de Paiva, C.S. The Pathophysiology of Dry Eye Disease: What We Know and Future Directions for Research. Ophthalmology 2017, 124, S4–S13. [Google Scholar] [CrossRef]
- Brewitt, H.; Sistani, F. Dry Eye Disease: The Scale of the Problem. Surv. Ophthalmol. 2001, 45 (Suppl. S2), S199–S202. [Google Scholar] [CrossRef]
- Giannaccare, G.; Taroni, L.; Senni, C.; Scorcia, V. Intense Pulsed Light Therapy in the Treatment of Meibomian Gland Dysfunction: Current Perspectives. Clin. Optom. 2019, 11, 113–126. [Google Scholar] [CrossRef] [PubMed]
- Dell, S.J.; Desai, N.R. IPL + Thermal Pulsation: A Thorough Approach to Dry Eye. 2018. Available online: https://www.ophthalmologytimes.com/view/ipl-thermal-pulsation-thorough-approach-dry-eye (accessed on 22 July 2022).
- Knop, E.; Knop, N.; Millar, T.; Obata, H.; Sullivan, D.A. The International Workshop on Meibomian Gland Dysfunction: Report of the Subcommittee on Anatomy, Physiology, and Pathophysiology of the Meibomian Gland. Investig. Opthalmol. Vis. Sci. 2011, 52, 1938–1978. [Google Scholar] [CrossRef] [PubMed]
- Yavuz, B.; Pehlivan, S.B.; Ünlü, N. An Overview on Dry Eye Treatment: Approaches for Cyclosporin a Delivery. Sci. World J. 2012, 2012, 194848. [Google Scholar] [CrossRef]
- Strauss, G.; Osen, W.; Debatin, K.M. Induction of apoptosis and modulation of activation and effector function in T cells by immunosuppressive drugs. Clin. Exp. Immunol. 2002, 128, 255–266. [Google Scholar] [CrossRef]
- Westera, L.; Drylewicz, J.; Braber, I.D.; Mugwagwa, T.; Van Der Maas, I.; Kwast, L.; Volman, T.; Van De Weg-Schrijver, E.H.R.; Bartha, I.; Spierenburg, G.; et al. Closing the gap between T-cell life span estimates from stable isotope-labeling studies in mice and humans. Blood 2013, 122, 2205–2212. [Google Scholar] [CrossRef]
| Author, Year (Study Name) | N | Comparison | Age Mean | Age SD | Female (%) | Country | Duration of Study | Study Design | Blindness | ITT | Outcome Measurement |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Sheppard et al., 2014 [9] (OPUS-1) | 588 | vehicle | 60.7 | 12.0 | 69.2 | US | 12 weeks | RCT | double | N/M | CFS, EDS, LGS, ODS, OSDI, TEAE, VAS |
| Tauber et al., 2015 [10] (OPUS-2) | 718 | vehicle | 58.8 | 14.1 | 76.6 | US | 12 weeks | RCT | double | Yes | CFS, EDS, LGS, ODS, TEAE, VAS |
| Holland et al., 2016 [11] (OPUS-3) | 711 | vehicle | 58.7 | 14.5 | 75.5 | US | 12 weeks | RCT | double | Yes | EDS, ODS, TEAE, VAS |
| Donnenfeld et al., 2016 [12] (SONATA) | 332 | vehicle | 59.5 | 12.7 | 75.3 | US | 360 days | RCT | double | N/M | post-instillation comfort, TEAE |
| De Paz et al., 2017 [13] | 14 | - | 44.9 | 3.1 | 85.8 | US | 4 weeks | case-control | none | N/M | ODSI |
| Tauber et al., 2019 [14] | 50 | TPP | 65.8 | 8.9 | 80.0 | US | 6 weeks | RCT | single | N/M | BCVA, bulbar conjunctival injection, CFS, lipid layer thickness, meibomian gland patency, MGD score, MMP-9, VAS |
| Tong et al., 2019 [15] | 121 | - | 60.5 | 14.4 | 87.6 | US | 12 weeks | retrospective cohort | none | N/M | CFS, MGD score, OSDI, TBUT, TEAE |
| Pepose et al., 2019 [16] | 30 | - | 67.4 | - | 88.5 | US | 12 weeks | longitudinal | none | Yes | CFS, MGD score, TBUT, tear osmolality, VAS |
| Epitropoulos et al., 2020 [17] | 33 | CYC | 69.3 | 4.2 | 78.8 | US | 3 weeks | retrospective cohort | none | N/M | VAS |
| Hovanesian et al., 2021 [18] | 600 | - | 57.1 | - | 75.8 | US/Canada | 12 months | retrospective cohort | none | N/M | CFS, DEQ-5, ODSI, Schirmer score, SPEED, TBUT |
| Variables | Studies Included | Lifitegrast | Placebo | N | Risk Ratio | 95% Cl | p | |||
|---|---|---|---|---|---|---|---|---|---|---|
| Event | Total | Event | Total | Lower Limit | Upper Limit | |||||
| Subjects with ≥1 TEAE | 3 | 504 | 936 | 238 | 824 | 1760 | 2.87 | 2.36 | 3.50 | <0.001 |
| Subjects with ≥1 ocular TEAE | 4 | 552 | 1229 | 233 | 1119 | 2348 | 3.10 | 2.58 | 3.72 | <0.001 |
| Instillation site irritation | 4 | 195 | 1229 | 33 | 1119 | 2348 | 6.21 | 4.25 | 9.06 | <0.001 |
| Instillation site pain | 1 | 63 | 293 | 11 | 295 | 588 | 7.07 | 3.64 | 13.73 | <0.001 |
| Instillation site reaction | 4 | 149 | 1229 | 37 | 1119 | 2348 | 4.03 | 2.79 | 5.84 | <0.001 |
| Instillation site pruritus | 1 | 19 | 293 | 6 | 295 | 588 | 3.34 | 1.31 | 8.49 | 0.011 |
| Visual acuity reduced | 3 | 55 | 872 | 43 | 765 | 1637 | 1.13 | 0.75 | 1.71 | 0.559 |
| Eye pain | 1 | 6 | 293 | 5 | 295 | 588 | 1.21 | 0.37 | 4.02 | 0.753 |
| Lacrimation increased | 1 | 7 | 293 | 1 | 295 | 588 | 7.20 | 0.88 | 58.86 | 0.066 |
| Eye pruritus | 1 | 5 | 293 | 2 | 295 | 588 | 2.54 | 0.49 | 13.22 | 0.267 |
| Ocular hyperemia | 1 | 7 | 293 | 4 | 295 | 588 | 1.78 | 0.52 | 6.15 | 0.362 |
| Subjects with ≥1 non-ocular TEAE | 3 | 284 | 936 | 114 | 824 | 1760 | 2.71 | 2.13 | 3.46 | <0.001 |
| Nervous system disorders | 1 | 63 | 359 | 11 | 359 | 588 | 6.73 | 3.48 | 13.01 | <0.001 |
| Dysgeusia | 3 | 140 | 936 | 4 | 824 | 1760 | 36.06 | 13.28 | 97.88 | <0.001 |
| Withdrawal due to ≥1 TEAE | 4 | 83 | 1229 | 25 | 1119 | 2348 | 3.17 | 2.01 | 4.99 | <0.001 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, J.-X.; Tsai, Y.-Y.; Lai, C.-T.; Li, Y.-L.; Wu, Y.-H.; Chiang, C.-C. Lifitegrast Ophthalmic Solution 5% Is a Safe and Efficient Eyedrop for Dry Eye Disease: A Systematic Review and Meta-Analysis. J. Clin. Med. 2022, 11, 5014. https://doi.org/10.3390/jcm11175014
Li J-X, Tsai Y-Y, Lai C-T, Li Y-L, Wu Y-H, Chiang C-C. Lifitegrast Ophthalmic Solution 5% Is a Safe and Efficient Eyedrop for Dry Eye Disease: A Systematic Review and Meta-Analysis. Journal of Clinical Medicine. 2022; 11(17):5014. https://doi.org/10.3390/jcm11175014
Chicago/Turabian StyleLi, Jing-Xing, Yi-Yu Tsai, Chun-Ting Lai, You-Ling Li, Ying-Hsuen Wu, and Chun-Chi Chiang. 2022. "Lifitegrast Ophthalmic Solution 5% Is a Safe and Efficient Eyedrop for Dry Eye Disease: A Systematic Review and Meta-Analysis" Journal of Clinical Medicine 11, no. 17: 5014. https://doi.org/10.3390/jcm11175014
APA StyleLi, J.-X., Tsai, Y.-Y., Lai, C.-T., Li, Y.-L., Wu, Y.-H., & Chiang, C.-C. (2022). Lifitegrast Ophthalmic Solution 5% Is a Safe and Efficient Eyedrop for Dry Eye Disease: A Systematic Review and Meta-Analysis. Journal of Clinical Medicine, 11(17), 5014. https://doi.org/10.3390/jcm11175014







