Regression-Based Normative Data for the Montreal Cognitive Assessment (MoCA) and Its Memory Index Score (MoCA-MIS) for Individuals Aged 18–91
Abstract
:1. Introduction
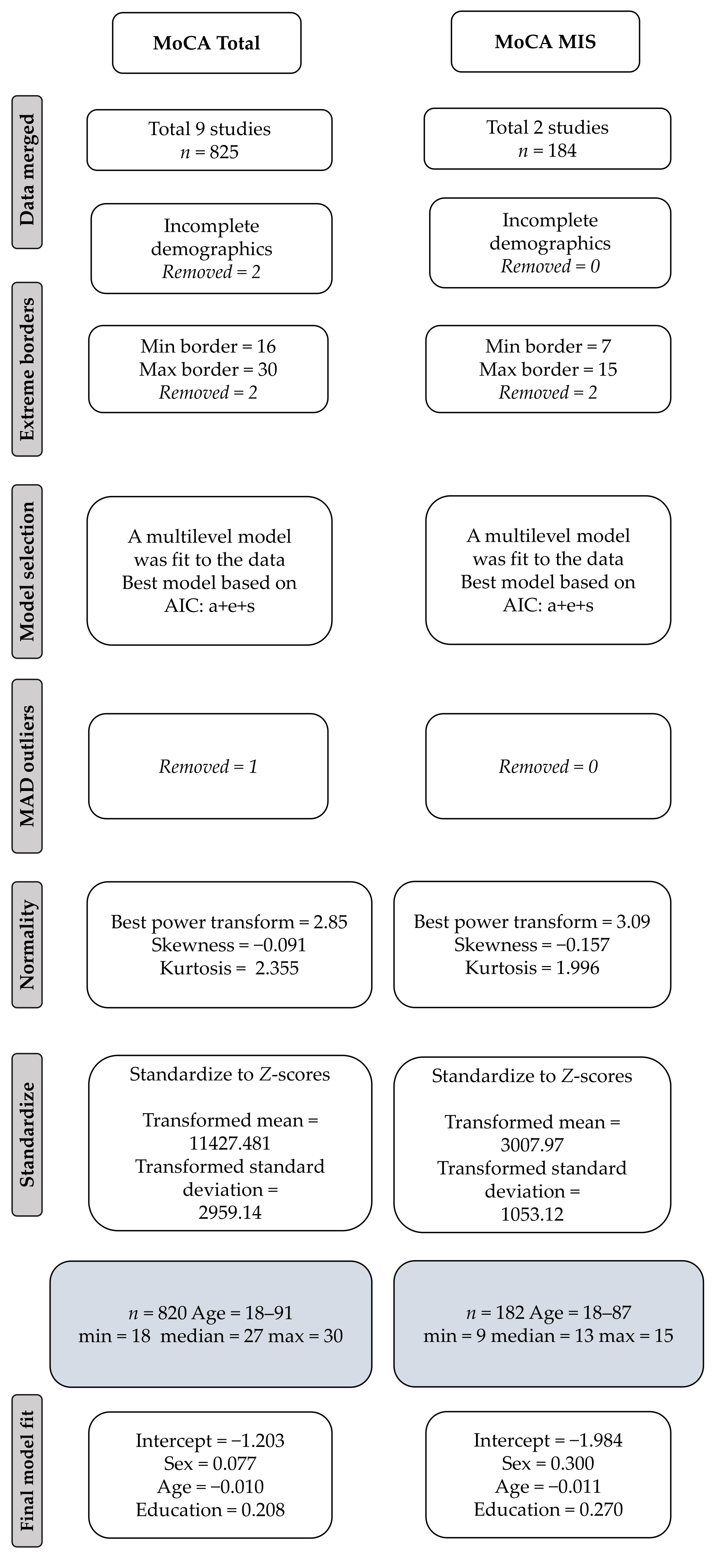
2. Materials and Methods
2.1. Participants
2.2. Procedure
2.3. Data Processing and Analyses
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
| Age Groups 18–59 | ||||||||||||||||||||||||
| 18–29 | 30–39 | 40–49 | 50–59 | |||||||||||||||||||||
| Education | Low | Av | High | Low | Av | High | Low | Av | High | Low | Av | High | ||||||||||||
| Sex | M | W | M | W | M | W | M | W | M | W | M | W | M | W | M | W | M | W | M | W | M | W | M | W |
| MoCA Score | ||||||||||||||||||||||||
| 30 | 98 | 98 | 91 | 91 | 86 | 86 | 99 | 99 | 93 | 93 | 89 | 89 | 99 | 99 | 94 | 94 | 91 | 91 | 99 | 99 | 96 | 95 | 93 | 93 |
| 29 | 94 | 94 | 80 | 80 | 72 | 72 | 96 | 96 | 83 | 83 | 77 | 76 | 97 | 97 | 86 | 86 | 80 | 80 | 97 | 97 | 89 | 89 | 83 | 83 |
| 28 | 86 | 86 | 64 | 64 | 54 | 54 | 89 | 89 | 69 | 69 | 60 | 60 | 91 | 91 | 73 | 73 | 65 | 64 | 93 | 93 | 77 | 77 | 69 | 69 |
| 27 | 75 | 75 | 47 | 47 | 37 | 37 | 79 | 79 | 52 | 52 | 43 | 42 | 82 | 82 | 57 | 57 | 47 | 47 | 85 | 85 | 62 | 62 | 52 | 52 |
| 26 | 59 | 59 | 31 | 31 | 23 | 23 | 65 | 65 | 36 | 36 | 27 | 27 | 69 | 69 | 41 | 40 | 31 | 31 | 74 | 73 | 45 | 45 | 36 | 35 |
| 25 | 44 | 44 | 18 | 18 | 13 | 13 | 50 | 49 | 23 | 22 | 16 | 16 | 54 | 54 | 26 | 26 | 19 | 19 | 59 | 59 | 30 | 30 | 22 | 22 |
| 24 | 30 | 30 | 10 | 10 | 6 | 6 | 35 | 35 | 13 | 13 | 8 | 8 | 40 | 39 | 16 | 15 | 11 | 10 | 44 | 44 | 19 | 19 | 13 | 13 |
| 23 | 19 | 19 | 5 | 5 | 3 | 3 | 23 | 23 | 7 | 7 | 4 | 4 | 27 | 27 | 9 | 9 | 5 | 5 | 31 | 31 | 11 | 11 | 7 | 7 |
| 22 | 11 | 11 | 3 | 3 | 1 | 1 | 14 | 14 | 4 | 3 | 2 | 2 | 17 | 17 | 5 | 5 | 3 | 3 | 21 | 20 | 6 | 6 | 4 | 3 |
| 21 | 7 | 7 | 1 | 1 | 1 | 1 | 9 | 8 | 2 | 2 | 1 | 1 | 11 | 10 | 2 | 2 | 1 | 1 | 13 | 13 | 3 | 3 | 2 | 2 |
| 20 | 4 | 4 | 1 | 1 | 0 | 0 | 5 | 5 | 1 | 1 | 0 | 0 | 6 | 6 | 1 | 1 | 1 | 1 | 8 | 8 | 2 | 2 | 1 | 1 |
| 19 | 2 | 2 | 0 | 0 | 0 | 0 | 3 | 3 | 0 | 0 | 0 | 0 | 4 | 4 | 1 | 1 | 0 | 0 | 5 | 5 | 1 | 1 | 0 | 0 |
| 18 | 1 | 1 | 0 | 0 | 0 | 0 | 2 | 1 | 0 | 0 | 0 | 0 | 2 | 2 | 0 | 0 | 0 | 0 | 3 | 3 | 0 | 0 | 0 | 0 |
| 17 | 1 | 1 | 0 | 0 | 0 | 0 | 1 | 1 | 0 | 0 | 0 | 0 | 1 | 1 | 0 | 0 | 0 | 0 | 2 | 2 | 0 | 0 | 0 | 0 |
| ≤16 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 1 | 1 | 0 | 0 | 0 | 0 | 1 | 1 | 0 | 0 | 0 | 0 |
| Age Groups 60–91 | ||||||||||||||||||||||||
| 60–69 | 70–79 | ≥80 | ||||||||||||||||||||||
| Education | Low | Av | High | Low | Av | High | Low | Av | High | |||||||||||||||
| Sex | M | W | M | W | M | W | M | W | M | W | M | W | M | W | M | W | M | W | ||||||
| MoCA Score | ||||||||||||||||||||||||
| 30 | 99 | 99 | 97 | 97 | 94 | 94 | 100 | 100 | 97 | 97 | 95 | 95 | 100 | 100 | 98 | 98 | 96 | 96 | ||||||
| 29 | 98 | 98 | 91 | 91 | 86 | 86 | 98 | 99 | 93 | 93 | 88 | 88 | 99 | 99 | 94 | 94 | 91 | 91 | ||||||
| 28 | 95 | 95 | 80 | 80 | 73 | 73 | 95 | 96 | 84 | 84 | 77 | 77 | 96 | 97 | 86 | 86 | 80 | 80 | ||||||
| 27 | 88 | 88 | 66 | 66 | 57 | 57 | 87 | 90 | 70 | 70 | 61 | 61 | 89 | 92 | 72 | 74 | 66 | 66 | ||||||
| 26 | 77 | 77 | 50 | 50 | 40 | 40 | 76 | 80 | 55 | 55 | 45 | 45 | 79 | 84 | 56 | 59 | 50 | 50 | ||||||
| 25 | 63 | 63 | 34 | 34 | 26 | 26 | 62 | 68 | 39 | 39 | 30 | 30 | 66 | 72 | 40 | 44 | 34 | 34 | ||||||
| 24 | 49 | 49 | 22 | 22 | 15 | 15 | 47 | 54 | 26 | 26 | 18 | 18 | 51 | 58 | 27 | 30 | 22 | 22 | ||||||
| 23 | 35 | 35 | 13 | 13 | 9 | 9 | 33 | 40 | 16 | 16 | 11 | 11 | 38 | 44 | 17 | 19 | 13 | 13 | ||||||
| 22 | 24 | 24 | 7 | 7 | 4 | 4 | 23 | 28 | 9 | 9 | 6 | 6 | 26 | 32 | 10 | 11 | 7 | 7 | ||||||
| 21 | 15 | 15 | 4 | 4 | 2 | 2 | 15 | 19 | 5 | 5 | 3 | 3 | 18 | 22 | 6 | 6 | 4 | 4 | ||||||
| 20 | 10 | 10 | 2 | 2 | 1 | 1 | 9 | 12 | 3 | 3 | 2 | 2 | 11 | 14 | 3 | 4 | 2 | 2 | ||||||
| 19 | 6 | 6 | 1 | 1 | 1 | 1 | 6 | 7 | 1 | 1 | 1 | 1 | 7 | 9 | 2 | 2 | 1 | 1 | ||||||
| 18 | 4 | 4 | 1 | 1 | 0 | 0 | 4 | 5 | 1 | 1 | 0 | 0 | 5 | 6 | 1 | 1 | 1 | 1 | ||||||
| 17 | 2 | 2 | 0 | 0 | 0 | 0 | 2 | 3 | 0 | 0 | 0 | 0 | 3 | 4 | 1 | 1 | 0 | 0 | ||||||
| ≤16 | 1 | 1 | 0 | 0 | 0 | 0 | 2 | 2 | 0 | 0 | 0 | 0 | 2 | 2 | 0 | 0 | 0 | 0 | ||||||
| Age Groups 18–59 | ||||||||||||||||||||||||
| 18–29 | 30–39 | 40–49 | 50–59 | |||||||||||||||||||||
| Education | Low | Av | High | Low | Av | High | Low | Av | High | Low | Av | High | ||||||||||||
| Sex | M | W | M | W | M | W | M | W | M | W | M | W | M | W | M | W | M | W | M | W | M | W | M | W |
| MIS | ||||||||||||||||||||||||
| 15 | 99 | 99 | 94 | 89 | 88 | 81 | 100 | 99 | 95 | 92 | 91 | 85 | 100 | 99 | 96 | 93 | 93 | 88 | 100 | 100 | 97 | 95 | 94 | 90 |
| 14 | 95 | 92 | 74 | 64 | 62 | 51 | 97 | 94 | 79 | 70 | 68 | 57 | 97 | 95 | 82 | 74 | 72 | 62 | 98 | 96 | 86 | 78 | 77 | 67 |
| 13 | 83 | 75 | 47 | 36 | 34 | 24 | 86 | 79 | 53 | 41 | 39 | 29 | 89 | 83 | 58 | 47 | 44 | 33 | 91 | 86 | 63 | 52 | 50 | 38 |
| 12 | 63 | 52 | 24 | 16 | 15 | 9 | 69 | 58 | 29 | 20 | 19 | 12 | 73 | 63 | 34 | 24 | 23 | 15 | 77 | 68 | 39 | 29 | 27 | 18 |
| 11 | 43 | 33 | 12 | 7 | 6 | 3 | 49 | 38 | 15 | 9 | 8 | 5 | 55 | 43 | 18 | 12 | 11 | 6 | 60 | 49 | 22 | 14 | 13 | 8 |
| 10 | 28 | 20 | 6 | 3 | 3 | 1 | 34 | 24 | 7 | 4 | 4 | 2 | 39 | 28 | 9 | 6 | 5 | 3 | 44 | 33 | 12 | 7 | 6 | 4 |
| 9 | 19 | 12 | 3 | 1 | 1 | 1 | 23 | 15 | 4 | 2 | 2 | 1 | 27 | 19 | 5 | 3 | 2 | 1 | 32 | 22 | 7 | 4 | 3 | 2 |
| 8 | 13 | 8 | 2 | 1 | 1 | 0 | 16 | 10 | 2 | 1 | 1 | 0 | 20 | 13 | 3 | 2 | 1 | 1 | 24 | 16 | 4 | 2 | 2 | 1 |
| 7 | 10 | 6 | 1 | 0 | 0 | 0 | 12 | 7 | 1 | 1 | 1 | 0 | 15 | 9 | 2 | 1 | 1 | 0 | 19 | 12 | 3 | 1 | 1 | 1 |
| 6 | 8 | 4 | 1 | 0 | 0 | 0 | 10 | 6 | 1 | 0 | 0 | 0 | 12 | 7 | 1 | 1 | 1 | 0 | 15 | 10 | 2 | 1 | 1 | 0 |
| 5 | 6 | 4 | 1 | 0 | 0 | 0 | 8 | 5 | 1 | 0 | 0 | 0 | 11 | 6 | 1 | 1 | 0 | 0 | 13 | 8 | 2 | 1 | 1 | 0 |
| 4 | 6 | 3 | 0 | 0 | 0 | 0 | 8 | 4 | 1 | 0 | 0 | 0 | 10 | 6 | 1 | 0 | 0 | 0 | 12 | 7 | 1 | 1 | 1 | 0 |
| 3 | 5 | 3 | 0 | 0 | 0 | 0 | 7 | 4 | 1 | 0 | 0 | 0 | 9 | 5 | 1 | 0 | 0 | 0 | 12 | 7 | 1 | 1 | 1 | 0 |
| 2 | 5 | 3 | 0 | 0 | 0 | 0 | 7 | 4 | 1 | 0 | 0 | 0 | 9 | 5 | 1 | 0 | 0 | 0 | 11 | 7 | 1 | 1 | 1 | 0 |
| 1 | 5 | 3 | 0 | 0 | 0 | 0 | 7 | 4 | 1 | 0 | 0 | 0 | 9 | 5 | 1 | 0 | 0 | 0 | 11 | 7 | 1 | 1 | 1 | 0 |
| Age Groups 60–91 | ||||||||||||||||||||||||
| 60–69 | 70–79 | ≥80 | ||||||||||||||||||||||
| Education | Low | Av | High | Low | Av | High | Low | Av | High | |||||||||||||||
| Sex | M | W | M | W | M | W | M | W | M | W | M | W | M | W | M | W | M | W | ||||||
| MIS | ||||||||||||||||||||||||
| 15 | 100 | 100 | 98 | 96 | 96 | 92 | 100 | 100 | 99 | 97 | 97 | 94 | 100 | 100 | 99 | 98 | 98 | 96 | ||||||
| 14 | 99 | 97 | 88 | 82 | 80 | 72 | 99 | 98 | 91 | 85 | 84 | 76 | 99 | 99 | 93 | 88 | 87 | 80 | ||||||
| 13 | 93 | 89 | 68 | 57 | 55 | 44 | 95 | 91 | 73 | 62 | 50 | 49 | 96 | 93 | 77 | 67 | 65 | 54 | ||||||
| 12 | 81 | 73 | 44 | 33 | 31 | 22 | 85 | 77 | 50 | 38 | 36 | 26 | 88 | 81 | 55 | 44 | 41 | 31 | ||||||
| 11 | 65 | 54 | 26 | 18 | 16 | 10 | 70 | 59 | 30 | 21 | 20 | 13 | 74 | 64 | 35 | 25 | 24 | 16 | ||||||
| 10 | 49 | 38 | 15 | 9 | 8 | 5 | 55 | 43 | 18 | 12 | 10 | 6 | 60 | 49 | 22 | 14 | 13 | 8 | ||||||
| 9 | 37 | 27 | 9 | 5 | 4 | 2 | 42 | 31 | 11 | 7 | 6 | 3 | 47 | 36 | 14 | 8 | 8 | 4 | ||||||
| 8 | 28 | 19 | 5 | 3 | 3 | 1 | 33 | 23 | 7 | 4 | 4 | 2 | 38 | 27 | 9 | 5 | 5 | 2 | ||||||
| 7 | 22 | 15 | 4 | 2 | 2 | 1 | 26 | 18 | 5 | 3 | 2 | 1 | 31 | 22 | 6 | 4 | 3 | 2 | ||||||
| 6 | 19 | 12 | 3 | 1 | 1 | 1 | 22 | 15 | 4 | 2 | 2 | 1 | 27 | 18 | 5 | 3 | 2 | 1 | ||||||
| 5 | 16 | 10 | 3 | 1 | 1 | 0 | 20 | 13 | 3 | 2 | 1 | 1 | 24 | 16 | 4 | 2 | 2 | 1 | ||||||
| 4 | 15 | 9 | 2 | 1 | 1 | 0 | 18 | 12 | 3 | 1 | 1 | 1 | 22 | 15 | 4 | 2 | 2 | 1 | ||||||
| 3 | 14 | 9 | 2 | 1 | 1 | 0 | 18 | 11 | 3 | 1 | 1 | 1 | 21 | 14 | 3 | 2 | 2 | 1 | ||||||
| 2 | 14 | 9 | 2 | 1 | 1 | 0 | 17 | 11 | 2 | 1 | 1 | 1 | 21 | 14 | 3 | 2 | 2 | 1 | ||||||
| 1 | 14 | 9 | 2 | 1 | 1 | 0 | 17 | 11 | 2 | 1 | 1 | 0 | 21 | 14 | 3 | 2 | 2 | 1 | ||||||
References
- Kessels, R.P.C.; Hendriks, M.P.H. Neuropsychological assessment. In Encyclopedia of Mental Health, 3rd ed.; Elsevier: Oxford, UK, 2022. [Google Scholar] [CrossRef]
- Nieuwenhuis-Mark, R.E. The death knoll for the MMSE: Has it outlived its purpose? J. Geriatr. Psychiatry Neurol. 2010, 23, 151–157. [Google Scholar] [CrossRef] [PubMed]
- Appels, B.A.; Scherder, E. The diagnostic accuracy of dementia-screening instruments with an administration time of 10 to 45 minutes for use in secondary care: A systematic review. Am. J. Alzheimers Dis. Other Demen. 2010, 25, 301–316. [Google Scholar] [CrossRef] [PubMed]
- Nasreddine, Z.S.; Phillips, N.A.; Bédirian, V.; Charbonneau, S.; Whitehead, V.; Collin, I.; Cummings, J.L.; Chertkow, H. The Montreal Cognitive Assessment, MoCA: A brief screening tool for mild cognitive impairment. J. Am. Geriatr. Soc. 2005, 53, 695–699. [Google Scholar] [CrossRef]
- Blanco-Campal, A.; Diaz-Orueta, U.; Navarro-Prados, A.B.; Burke, T.; Libon, D.J.; Lamar, M. Features and psychometric properties of the Montreal Cognitive Assessment: Review and proposal of a process-based approach version (MoCA-PA). Appl. Neuropsychol. Adult 2021, 28, 658–672. [Google Scholar] [CrossRef] [PubMed]
- Bruijnen, C.J.W.H.; Dijkstra, B.A.G.; Walvoort, S.J.W.; Budy, M.J.J.; Beurmanjer, H.; De Jong, C.A.J.; Kessels, R.P.C. Psychometric properties of the Montreal Cognitive Assessment (MoCA) in healthy participants aged 18–70. Int. J. Psychiatry Clin. Pract. 2020, 24, 293–300. [Google Scholar] [CrossRef]
- Thissen, A.J.A.M.; van Bergen, F.; de Jonghe, J.F.M.; Kessels, R.P.C.; Dautzenberg, P.L.J. Bruikbaarheid en validiteit van de Nederlandse versie van de Montreal Cognitive Assessment (MoCA-D) bij het diagnosticeren van Mild Cognitive Impairment. Tijdschr. Gerontol. Geriatr. 2010, 41, 231–240. [Google Scholar] [CrossRef]
- Serrano, C.M.; Sorbara, M.; Minond, A.; Finlay, J.B.; Arizaga, R.L.; Iturry, M.; Martinez, P.; Heinemann, G.; Gagliardi, C.; Serra, A.; et al. Validation of the Argentine version of the Montreal Cognitive Assessment Test (MOCA): A screening tool for mild cognitive impairment and mild dementia in elderly. Dement. Neuropsychol. 2020, 14, 145–152. [Google Scholar] [CrossRef]
- O’Caoimh, R.; Timmons, S.; Molloy, D.W. Screening for mild cognitive impairment: Comparison of “MCI specific” screening instruments. J. Alzheimers Dis. 2016, 51, 619–629. [Google Scholar] [CrossRef] [Green Version]
- Davis, D.H.; Creavin, S.T.; Yip, J.L.; Noel-Storr, A.H.; Brayne, C.; Cullum, S. Montreal Cognitive Assessment for the diagnosis of Alzheimer’s disease and other dementias. Cochrane Database Syst. Rev. 2015, 2015, CD010775. [Google Scholar] [CrossRef] [Green Version]
- Konstantopoulos, K.; Vogazianos, P.; Doskas, T. Normative data of the Montreal Cognitive Assessment in the Greek population and Parkinsonian dementia. Arch. Clin. Neuropsychol. 2016, 31, 246–253. [Google Scholar] [CrossRef] [Green Version]
- Carlew, A.R.; Smith, E.E.; Goette, W.; Lippe, B.; Lacritz, L.; Rossetti, H. Montreal Cognitive Assessment (MoCA) scores in medically compromised patients: A scoping review. Health Psychol. 2021, 40, 717–726. [Google Scholar] [CrossRef]
- Janssen, M.A.M.; Meulenbroek, O.; Steens, S.C.; Góraj, B.; Bosch, M.; Koopmans, P.P.; Kessels, R.P.C. Cognitive functioning, wellbeing and brain correlates in HIV-1 infected patients on long-term combination antiretroviral therapy. AIDS 2015, 29, 2139–2148. [Google Scholar] [CrossRef]
- Rosenblum, S.; Meyer, S.; Gemerman, N.; Mentzer, L.; Richardson, A.; Israeli-Korn, S.; Livneh, V.; Karmon, T.F.; Nevo, T.; Yahalom, G.; et al. The Montreal Cognitive Assessment: Is it suitable for identifying mild cognitive impairment in Parkinson’s Disease? Mov. Disord. Clin. Pract. 2020, 7, 648–655. [Google Scholar] [CrossRef] [PubMed]
- Rosca, E.C.; Simu, M. Montreal Cognitive Assessment for evaluating cognitive impairment in multiple sclerosis: A systematic review. Acta Neurol. Belg. 2020, 120, 1307–1321. [Google Scholar] [CrossRef] [PubMed]
- Potocnik, J.; Ovcar Stante, K.; Rakusa, M. The validity of the Montreal Cognitive Assessment (MoCA) for the screening of vascular cognitive impairment after ischemic stroke. Acta Neurol. Belg. 2020, 120, 681–685. [Google Scholar] [CrossRef]
- Coleman, K.K.; Coleman, B.L.; MacKinley, J.D.; Pasternak, S.H.; Finger, E.C. Detection and differentiation of frontotemporal dementia and related disorders from Alzheimer disease using the Montreal Cognitive Assessment. Alzheimer Dis. Assoc. Disord. 2016, 30, 258–263. [Google Scholar] [CrossRef] [PubMed]
- Bruijnen, C.J.W.H.; Jansen, M.; Dijkstra, B.A.G.; Walvoort, S.J.W.; Lugtmeijer, S.; Markus, W.; De Jong, C.A.J.; Kessels, R.P.C. The Montreal Cognitive Assessment as a cognitive screen in addiction health care: A validation study for clinical practice. J. Subst. Use 2018, 24, 47–54. [Google Scholar] [CrossRef] [Green Version]
- Luque, B.; García, V.; Tabernero, C. Depression and cognitive impairment in a spanish sample of psychoactive substance users receiving mental health care. Healthcare 2022, 10, 887. [Google Scholar] [CrossRef]
- van Gils, P.; van Heugten, C.; Hofmeijer, J.; Keijzer, H.; Nutma, S.; Duits, A. The Montreal Cognitive Assessment is a valid cognitive screening tool for cardiac arrest survivors. Resuscitation 2022, 172, 130–136. [Google Scholar] [CrossRef]
- Murillo-Garcia, A.; Leon-Llamas, J.L.; Villafaina, S.; Rohlfs-Dominguez, P.; Gusi, N. MoCA vs. MMSE of fibromyalgia patients: The possible role of dual-task tests in detecting cognitive impairment. J. Clin. Med. 2021, 10, 125. [Google Scholar] [CrossRef]
- Rosca, E.C.; Simu, M. Montreal cognitive assessment for evaluating cognitive impairment in Huntington’s disease: A systematic review. CNS Spectr. 2022, 27, 27–45. [Google Scholar] [CrossRef] [PubMed]
- de Ruiter, S.C.; de Jonghe, J.; Germans, T.; Ruiter, J.H.; Jansen, R. Cognitive impairment is very common in elderly patients with syncope and unexplained falls. J Am. Med. Dir. Assoc. 2017, 18, 409–413. [Google Scholar] [CrossRef] [PubMed]
- Hoche, F.; Guell, X.; Vangel, M.G.; Sherman, J.C.; Schmahmann, J.D. The cerebellar cognitive affective/Schmahmann syndrome scale. Brain 2018, 141, 248–270. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rosca, E.C.; Cornea, A.; Simu, M. Montreal Cognitive Assessment for evaluating the cognitive impairment in patients with schizophrenia: A systematic review. Gen. Hosp. Psychiatry 2020, 65, 64–73. [Google Scholar] [CrossRef]
- Early, M.L.; Linton, E.; Bosch, A.; Campbell, T.; Hill-Briggs, F.; Pecker, L.H.; Lance, E.I.; Lanzkron, S. The Montreal cognitive assessment as a cognitive screening tool in sickle cell disease: Associations with clinically significant cognitive domains. Br. J. Haematol. 2022, in press. [CrossRef]
- Machii, N.; Kudo, A.; Saito, H.; Tanabe, H.; Iwasaki, M.; Hirai, H.; Masuzaki, H.; Shimabukuro, M. Walking speed is the sole determinant criterion of sarcopenia of mild cognitive impairment in Japanese elderly patients with type 2 diabetes mellitus. J. Clin. Med. 2020, 9, 2133. [Google Scholar] [CrossRef]
- Aiello, E.N.; Gramegna, C.; Esposito, A.; Gazzaniga, V.; Zago, S.; Difonzo, T.; Maddaluno, O.; Appollonio, I.; Bolognini, N. The Montreal Cognitive Assessment (MoCA): Updated norms and psychometric insights into adaptive testing from healthy individuals in Northern Italy. Aging Clin. Exp. Res. 2022, 34, 375–382. [Google Scholar] [CrossRef]
- Aiello, E.N.; Fiabane, E.; Manera, M.R.; Radici, A.; Grossi, F.; Ottonello, M.; Pain, D.; Pistarini, C. Screening for cognitive sequelae of SARS-CoV-2 infection: A comparison between the Mini-Mental State Examination (MMSE) and the Montreal Cognitive Assessment (MoCA). Neurol. Sci. 2022, 43, 81–84. [Google Scholar] [CrossRef]
- Apolinario, D.; Dos Santos, M.F.; Sassaki, E.; Pegoraro, F.; Pedrini, A.; Cestari, B.; Amaral, A.H.; Mitt, M.; Müller, M.B.; Suemoto, C.K.; et al. Normative data for the Montreal Cognitive Assessment (MoCA) and the Memory Index Score (MoCA-MIS) in Brazil: Adjusting the nonlinear effects of education with fractional polynomials. Int. J. Geriatr. Psychiatry 2018, 33, 893–899. [Google Scholar] [CrossRef]
- Bartos, A.; Fayette, D. Validation of the Czech Montreal Cognitive Assessment for Mild Cognitive Impairment due to Alzheimer disease and Czech norms in 1,552 Elderly persons. Dement. Geriatr. Cogn. Disord. 2018, 46, 335–345. [Google Scholar] [CrossRef]
- Borland, E.; Nagga, K.; Nilsson, P.M.; Minthon, L.; Nilsson, E.D.; Palmqvist, S. The Montreal Cognitive Assessment: Normative data from a large Swedish population-based cohort. J. Alzheimers Dis. 2017, 59, 893–901. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cesar, K.G.; Yassuda, M.S.; Porto, F.H.G.; Brucki, S.M.D.; Nitrini, R. MoCA Test: Normative and diagnostic accuracy data for seniors with heterogeneous educational levels in Brazil. Arq. Neuropsiquiatr. 2019, 77, 775–781. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Classon, E.; van den Hurk, W.; Lyth, J.; Johansson, M.M. Montreal Cognitive Assessment: Normative data for cognitively healthy Swedish 80- to 94-year-olds. J. Alzheimers Dis. 2022, 87, 1335–1344. [Google Scholar] [CrossRef] [PubMed]
- Conti, S.; Bonazzi, S.; Laiacona, M.; Masina, M.; Coralli, M.V. Montreal Cognitive Assessment (MoCA)-Italian version: Regression based norms and equivalent scores. Neurol. Sci. 2015, 36, 209–214. [Google Scholar] [CrossRef]
- Engedal, K.; Gjøra, L.; Benth, J.Š.; Wagle, J.; Rønqvist, T.K.; Selbæk, G. The Montreal Cognitive Assessment: Normative data from a large, population-based sample of cognitive healthy older adults in Norway: The HUNT study. J. Alzheimers Dis. 2022, 86, 589–599. [Google Scholar] [CrossRef]
- Freitas, S.; Simões, M.R.; Alves, L.; Santana, I. Montreal Cognitive Assessment (MoCA): Normative study for the Portuguese population. J. Clin. Exp. Neuropsychol. 2011, 33, 989–996. [Google Scholar] [CrossRef]
- Gaete, M.; Jorquera, S.; Bello-Lepe, S.; Mendoza, Y.M.; Véliz, M.; Alonso-Sanchez, M.F.; Lira, J. Standardized results of the Montreal Cognitive Assessment (MoCA) for neurocognitive screening in a Chilean population. Neurologia, 2020, in press. [CrossRef]
- Gluhm, S.; Goldstein, J.; Loc, K.; Colt, A.; Liew, C.V.; Corey-Bloom, J. Cognitive performance on the Mini-Mental State Examination and the Montreal Cognitive Assessment across the healthy adult lifespan. Cogn. Behav. Neurol. 2013, 26, 1–5. [Google Scholar] [CrossRef] [Green Version]
- Hayek, M.; Tarabey, L.; Abou-Mrad, T.; Fadel, P.; Abou-Mrad, F. Normative data for the Montreal Cognitive Assessment in a Lebanese older adult population. J. Clin. Neurosci. 2020, 74, 81–86. [Google Scholar] [CrossRef]
- Ihle-Hansen, H.; Vigen, T.; Berge, T.; Einvik, G.; Aarsland, D.; Rønning, O.M.; Thommessen, B.; Røsjø, H.; Tveit, A.; Ihle-Hansen, H. Montreal Cognitive Assessment in a 63- to 65-year-old Norwegian cohort from the general population: Data from the Akershus Cardiac Examination 1950 Study. Dement. Geriatr. Cogn. Disord. Extra 2017, 7, 318–327. [Google Scholar] [CrossRef] [Green Version]
- Kang, J.M.; Cho, Y.S.; Park, S.; Lee, B.H.; Sohn, B.K.; Choi, C.H.; Choi, J.S.; Jeong, H.Y.; Cho, S.J.; Lee, J.H.; et al. Montreal cognitive assessment reflects cognitive reserve. BMC Geriatr. 2018, 18, 261. [Google Scholar] [CrossRef] [PubMed]
- Kenny, R.A.; Coen, R.F.; Frewen, J.; Donoghue, O.A.; Cronin, H.; Savva, G.M. Normative values of cognitive and physical function in older adults: Findings from the Irish Longitudinal Study on Ageing. J. Am. Geriatrr. Soc. 2013, 61 (Suppl. 2), S279–S290. [Google Scholar] [CrossRef]
- Kopecek, M.; Stepankova, H.; Lukavsky, J.; Ripova, D.; Nikolai, T.; Bezdicek, O. Montreal cognitive assessment (MoCA): Normative data for old and very old Czech adults. Appl. Neuropsychol. Adult 2017, 24, 23–29. [Google Scholar] [CrossRef] [PubMed]
- Larouche, E.; Tremblay, M.P.; Potvin, O.; Laforest, S.; Bergeron, D.; Laforce, R.; Monetta, L.; Boucher, L.; Tremblay, P.; Belleville, S.; et al. Normative data for the Montreal Cognitive Assessment in middle-aged and elderly Quebec-French people. Arch. Clin. Neuropsychol. 2016, 31, 819–826. [Google Scholar] [CrossRef] [Green Version]
- Lee, J.Y.; Lee, D.W.; Cho, S.J.; Na, D.L.; Jeon, H.J.; Kim, S.K.; Lee, Y.R.; Youn, J.H.; Kwon, M.; Lee, J.H.; et al. Brief screening for mild cognitive impairment in elderly outpatient clinic: Validation of the Korean version of the Montreal Cognitive Assessment. J. Geriatr. Psychiatry Neurol. 2008, 21, 104–110. [Google Scholar] [CrossRef]
- Lu, J.; Li, D.; Li, F.; Zhou, A.; Wang, F.; Zuo, X.; Jia, X.F.; Song, H.; Jia, J. Montreal cognitive assessment in detecting cognitive impairment in Chinese elderly individuals: A population-based study. J. Geriatr. Psychiatry Neurol. 2011, 24, 184–190. [Google Scholar] [CrossRef] [PubMed]
- Malek-Ahmadi, M.; Powell, J.J.; Belden, C.M.; O’Connor, K.; Evans, L.; Coon, D.W.; Nieri, W. Age- and education-adjusted normative data for the Montreal Cognitive Assessment (MoCA) in older adults age 70–99. Aging Neuropsychol. Cogn. 2015, 22, 755–761. [Google Scholar] [CrossRef]
- Muayqil, T.A.; Alamri, N.K.; Alqahtani, A.M.; Julaidan, S.S.; Alsuhaibani, R.; Nafisah, I.; Alkeridy, W.A.; Aljafen, B.N.; Alanazy, M.H. Normative and equated data of the original and basic versions of the Montreal Cognitive Assessment among community dwelling Saudi Arabians. Behav. Neurol. 2021, 2021, 5395627. [Google Scholar] [CrossRef]
- Narazaki, K.; Nofuji, Y.; Honda, T.; Matsuo, E.; Yonemoto, K.; Kumagai, S. Normative data for the Montreal Cognitive Assessment in a Japanese community-dwelling older population. Neuroepidemiology 2013, 40, 23–29. [Google Scholar] [CrossRef]
- Ojeda, N.; Del Pino, R.; Ibarretxe-Bilbao, N.; Schretlen, D.J.; Pena, J. Test de evaluacion cognitiva de Montreal: Normalizacion y estandarizacion de la prueba en poblacion española [Montreal Cognitive Assessment Test: Normalization and standardization for Spanish population]. Rev. Neurol. 2016, 63, 488–496. [Google Scholar] [CrossRef]
- Pereiro, A.X.; Ramos-Lema, S.; Lojo-Seoane, C.; Guàrdia-Olmos, J.; Facal-Mayo, D.; Juncos-Rabadán, O. Normative data for the Montreal Cognitive Assessment (MOCA) in a Spanish sample of community-dweller adults. Eur. Geriatr. Med. 2017, 8, 240–244. [Google Scholar] [CrossRef]
- Pinto, T.; Machado, L.; Bulgacov, T.M.; Rodrigues-Júnior, A.L.; Costa, M.; Ximenes, R.; Sougey, E.B. Influence of age and education on the performance of elderly in the Brazilian version of the Montreal Cognitive Assessment battery. Dement. Geriatr. Cogn. Disord. 2018, 45, 290–299. [Google Scholar] [CrossRef]
- Rossetti, H.C.; Lacritz, L.H.; Cullum, C.M.; Weiner, M.F. Normative data for the Montreal Cognitive Assessment (MoCA) in a population-based sample. Neurology 2011, 77, 1272–1275. [Google Scholar] [CrossRef]
- Rossetti, H.C.; Lacritz, L.H.; Hynan, L.S.; Cullum, C.M.; Van Wright, A.; Weiner, M.F. Montreal Cognitive Assessment performance among community-dwelling African Americans. Arch. Clin. Neuropsychol. 2017, 32, 238–244. [Google Scholar] [CrossRef] [Green Version]
- Sachs, B.C.; Chelune, G.J.; Rapp, S.R.; Couto, A.M.; Willard, J.J.; Williamson, J.D.; Sink, K.M.; Coker, L.H.; Gaussoin, S.A.; Gure, T.R.; et al. Robust demographically-adjusted normative data for the Montreal Cognitive Assessment (MoCA): Results from the systolic blood pressure intervention trial. Clin. Neuropsychol. 2022, in press. [CrossRef] [PubMed]
- Santangelo, G.; Siciliano, M.; Pedone, R.; Vitale, C.; Falco, F.; Bisogno, R.; Siano, P.; Barone, P.; Grossi, D.; Santangelo, F.; et al. Normative data for the Montreal Cognitive Assessment in an Italian population sample. Neurol. Sci. 2015, 36, 585–591. [Google Scholar] [CrossRef] [Green Version]
- Siciliano, M.; Chiorri, C.; Passaniti, C.; Sant’Elia, V.; Trojano, L.; Santangelo, G. Comparison of alternate and original forms of the Montreal Cognitive Assessment (MoCA): An Italian normative study. Neurol. Sci. 2019, 40, 691–702. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sink, K.M.; Craft, S.; Smith, S.C.; Maldjian, J.A.; Bowden, D.W.; Xu, J.; Freedman, B.I.; Divers, J. Montreal Cognitive Assessment and Modified Mini Mental State Examination in African Americans. J. Aging Res. 2015, 2015, 872018. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Thomann, A.E.; Goettel, N.; Monsch, R.J.; Berres, M.; Jahn, T.; Steiner, L.A.; Monsch, A.U. The Montreal Cognitive Assessment: Normative data from a German-speaking cohort and comparison with international normative samples. J. Alzheimers Dis. 2018, 64, 643–655. [Google Scholar] [CrossRef] [Green Version]
- Julayanont, P.; Brousseau, M.; Chertkow, H.; Phillips, N.; Nasreddine, Z.S. Montreal Cognitive Assessment Memory Index Score (MoCA-MIS) as a predictor of conversion from mild cognitive impairment to Alzheimer’s disease. J. Am. Geriatr. Soc. 2014, 62, 679–684. [Google Scholar] [CrossRef]
- Kaur, A.; Edland, S.D.; Peavy, G.M. The MoCA-Memory Index Score: An efficient alternative to paragraph recall for the detection of amnestic mild cognitive impairment. Alzheimer Dis. Assoc. Disord. 2018, 32, 120–124. [Google Scholar] [CrossRef] [PubMed]
- Bruijnen, C.J.W.H.; Walvoort, S.J.W.; Dijkstra, B.A.G.; de Jong, C.A.J.; Kessels, R.P.C. The course of cognitive performance during inpatient treatment in patients with alcohol use disorder with no, mild or major neurocognitive disorders. Alcohol Alcohol. 2021, 56, 89–100. [Google Scholar] [CrossRef]
- Oosterhuis, H.E.; van der Ark, L.A.; Sijtsma, K. Sample size requirements for traditional and regression-based norms. Assessment 2016, 23, 191–202. [Google Scholar] [CrossRef] [PubMed]
- Heaton, R.K.; Matthews, C.G.; Grant, I.; Avitable, N. Demographic Corrections with Comprehensive Norms: An Overzealous Attempt or a Good Start? J. Clin. Exp. Neuropsychol. 1996, 18, 449–458. [Google Scholar] [CrossRef]
- Duits, A.; Kessels, R. Schatten van het premorbide functioneren. In Neuropsychologische Diagnostiek: De Klinische Praktijk; Hendriks, M., Kessels, R., Gorissen, M., Schmand, B., Duits, A., Eds.; Boom: Amsterdam, The Netherlands, 2014; pp. 173–186. [Google Scholar]
- Hochstenbach, J.; Mulder, T.; van Limbeek, J.; Donders, R.; Schoonderwaldt, H. Cognitive decline following stroke: A comprehensive study of cognitive decline following stroke. J. Clin. Exp. Neuropsychol. 1998, 20, 503–517. [Google Scholar] [CrossRef] [PubMed]
- Kasse, E.; Oudman, E.; Olivier, M.; Wijnia, J.W.; Postma, A. Subtle object location perception deficits in Korsakoff’s syndrome. J. Exp. Clin. Neuropsychol. 2019, 41, 881–887. [Google Scholar] [CrossRef] [Green Version]
- Oudman, E.; Postma, A.; Van der Stigchel, S.; Appelhof, B.; Wijnia, J.W.; Nijboer, T.C. The Montreal Cognitive Assessment (MoCA) is superior to the Mini Mental State Examination (MMSE) in detection of Korsakoff’s syndrome. Clin. Neuropsychol. 2014, 28, 1123–1132. [Google Scholar] [CrossRef]
- Derks-Dijkman, M.W.; Schaefer, R.S.; Stegeman, M.; Van Tilborg, I.D.A.; Kessels, R.P.C. Effects of musical mnemonics on working memory performance in cognitively unimpaired young and older adults. Exp. Aging Res. 2022; revision submitted. [Google Scholar]
- Janssen, M.A.M.; Bosch, M.; Koopmans, P.P.; Kessels, R.P.C. Validity of the Montreal Cognitive Assessment and the HIV Dementia Scale in the assessment of cognitive impairment in HIV-1 infected patients. J. Neurovirol. 2015, 21, 383–390. [Google Scholar] [CrossRef] [Green Version]
- Sutter, K.; Oostwoud Wijdenes, L.; Van Beers, R.J.; Claassen, J.A.H.R.; Kessels, R.P.C.; Medendorp, W.P. Does early-stage Alzheimer’s disease affect the dynamics of motor adaptation? In 2022 Neuroscience Meeting Planner (Online Abstracts); Society for Neuroscience: Chicago, IL, USA, 2022. [Google Scholar]
- UNESCO. International Standard Classification of Education (ISCED 2011); UNESCO Institute for Statistics: Montreal, QC, Canada, 2011. [Google Scholar]
- de Vent, N.R.; Agelink van Rentergem, J.A.; Schmand, B.A.; Murre, J.M.; ANDI Consortium; Huizenga, H.M. Advanced Neuropsychological Diagnostics Infrastructure (ANDI): A Normative database created from control datasets. Front. Psychol. 2016, 7, 1601. [Google Scholar] [CrossRef] [Green Version]
- Curran, P.J.; Hussong, A.M. Integrative data analysis: The simultaneous analysis of multiple data sets. Psychol. Methods 2009, 14, 81–100. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cohen, J.; Cohen, P.; West, S.G.; Aiken, L.S. Applied Multiple Regression/Correlation Analysis for the Behavioral Sciences; Lawrence Erlbaum Associates: Mahwah, NJ, USA, 2003. [Google Scholar]
- Leys, C.; Ley, C.; Klein, O.; Bernard, P.; Licata, L. Detecting outliers: Do not use standard deviation around the mean, use absolute deviation around the median. J. Exp. Soc. Psychol. 2013, 49, 764–766. [Google Scholar] [CrossRef] [Green Version]
- Crawford, J.R.; Howell, D.C. Comparing an individual’s test score against norms derived from small samples. Clin. Neuropsychol. 1998, 12, 482–486. [Google Scholar] [CrossRef]
- Lezak, M.D.; Howieson, D.B.; Bigler, E.D.; Tranel, D. Neuropsychological Assessment, 5th ed.; Oxford University Press: New York, NY, USA, 2012. [Google Scholar]
- Crawford, J.R.; Garthwaite, P.H.; Azzalini, A.; Howell, D.C.; Laws, K.R. Testing for a deficit in single-case studies: Effects of departures from normality. Neuropsychologia 2006, 44, 666–677. [Google Scholar] [CrossRef] [PubMed]
- Box, G.E.P.; Cox, D.R. An analysis of transformations. J. R. Stat. Soc. Ser. B 1964, 26, 211–252. [Google Scholar] [CrossRef]
- Sakia, R.M. The Box-Cox transformation technique: A review. Statistician 1992, 41, 169–178. [Google Scholar] [CrossRef]
- Curran, P.J.; West, S.G.; Finch, J.F. The robustness of test statistics to nonnormality and specification error in confirmatory factor analysis. Psychol. Methods 1996, 1, 16–29. [Google Scholar] [CrossRef]
- Van den Berg, E.; Nys, G.M.S.; Brands, A.M.A.; Ruis, C.; Van Zandvoort, M.J.E.; Kessels, R.P.C. The Brixton Spatial Anticipation Test as a test for executive function: Validity in patient groups and norms for older adults. J. Int. Neuropsychol. Soc. 2009, 15, 695–703. [Google Scholar] [CrossRef]
- RStudio Team. RStudio: Integrated Development for R; RStudio, PBC: Boston, MA, USA, 2020; Available online: http://www.rstudio.com/ (accessed on 20 June 2022).
- Guilmette, T.J.; Sweet, J.J.; Hebben, N.; Koltai, D.; Mahone, E.M.; Spiegler, B.J.; Stucky, K.; Westerveld, M.; Conference Participants. American Academy of Clinical Neuropsychology consensus conference statement on uniform labeling of performance test scores. Clin. Neuropsychol. 2020, 34, 437–453. [Google Scholar] [CrossRef] [Green Version]
- Strauss, E.; Sherman, E.M.S.; Spreen, O. A Compendium of Neuropsychological Tests: Administration, Norms, and Commentary, 3rd ed.; Oxford University Press: New York, NY, USA, 2006. [Google Scholar]
- Eikelboom, W.S.; Bertens, D.; Kessels, R.P.C. Cognitive rehabilitation in normal aging and individuals with subjective cognitive decline. In Cognitive Rehabilitation and Neuroimaging; Chiaravalloti, N., Weber, E., DeLuca, J., Eds.; Springer: Cham, Switzerland, 2020; pp. 37–67. [Google Scholar] [CrossRef]
- Asperholm, M.; Högman, N.; Rafi, J.; Herlitz, A. What did you do yesterday? a meta-analysis of sex differences in episodic memory. Psychol. Bull. 2019, 145, 785–821. [Google Scholar] [CrossRef]
- Asperholm, M.; van Leuven, L.; Herlitz, A. Sex differences in episodic memory variance. Front. Psychol. 2020, 11, 613. [Google Scholar] [CrossRef] [PubMed]
- Ratcliffe, L.N.; McDonald, T.; Robinson, B.; Sass, J.R.; Loring, D.W.; Hewitt, K.C. Classification statistics of the Montreal Cognitive Assessment (MoCA): Are we interpreting the MoCA correctly? Clin. Neuropsychol. 2022; ahead-of-print. [Google Scholar] [CrossRef] [PubMed]
- Thomann, A.E.; Berres, M.; Goettel, N.; Steiner, L.A.; Monsch, A.U. Enhanced diagnostic accuracy for neurocognitive disorders: A revised cut-off approach for the Montreal Cognitive Assessment. Alzheimers Res. Ther. 2020, 12, 39. [Google Scholar] [CrossRef] [PubMed]
- Khan, G.; Mirza, N.; Waheed, W. Developing guidelines for the translation and cultural adaptation of the Montreal Cognitive Assessment: Scoping review and qualitative synthesis. Br. J. Psychiatry Open 2022, 8, e21. [Google Scholar] [CrossRef]
| Study | Country | n | Age Range | Adjustment | Method | MIS |
|---|---|---|---|---|---|---|
| Aiello et al. [29] | Italy | 579 | 21–96 | Age, Edu | Regression | No |
| Apolinario et al. [30] | Brazil | 597 | 50–90 | Age, Edu | Regression | Yes |
| Bartos and Fayette [31] | Czech Republic | 1552 | 65–76 | Age, Edu | Regression | No |
| Borland et al. [32] | Sweden | 758 | 65–85 | Age, Edu, Sex | Regression | No |
| Cesar et al. [33] | Brazil | 385 | 60–80+ | Age, Edu | Stratified | No |
| Classon et al. [34] | Sweden | 181 | 80–94 | Age, Edu | Regression | No |
| Conti et al. [35] | Italy | 225 | 60–80 | Age, Edu | Regression | No |
| Engedal et al. [36] | Norway | 4780 | 70–90 | Age, Edu, Sex | Regression | No |
| Freitas et al. [37] | Portugal | 650 | 25–91 | Age, Edu | Regression | No |
| Gaete et al. [38] | Chile | 526 | 18–90 | Age, Edu | Regression | No |
| Gluhm et al. [39] | USA | 254 | 20–89 | Age | Stratified | No |
| Hayek et al. [40] | Lebanon | 164 | 60–87 | Age, Edu | Regression | No |
| Ihle-Hansen et al. [41] | Norway | 3413 | 63–65 | Edu | Mean + SD | No |
| Kang et al. [42] | Korea | 211 | 60–90 | Age, Edu, Sex | Stratified | No |
| Kenny et al. [43] | Ireland | 5802 | 50–85 | Age, Edu | GAMLSS | No |
| Konstantopoulos et al. [11] | Greece | 710 | 20–85 | Age, Edu, Sex | Regression | No |
| Kopecek et al. [44] | Czech Republic | 540 | 60–98 | Age, Edu | Regression | No |
| Larouche et al. [45] | Canada | 1019 | 41–98 | Age, Edu, Sex | Regression | No |
| Lee et al. [46] | Korea | 115 | 69.1 (±6.1) † | Edu ‡ | Mean + SD | No |
| Lu et al. [47] | China | 6283 | 65–100 | Age, Edu | Stratified | No |
| Malek-Ahmadi et al. [48] | USA | 205 | 70–99 | Age, Edu | Stratified | No |
| Muayqil et al. [49] | Saudi Arabia | 311 | 18–80 | Age, Edu | Stratified | No |
| Narazaki et al. [50] | Japan | 1977 | 65–96 | Age, Edu | Regression | No |
| Nasreddine et al. [4] | Canada | 90 | 72.8 (±7.0) † | Edu ‡ | Mean + SD | No |
| Ojeda et al. [51] | Spain | 700 | 18–86 | Age, Edu | Regression | No |
| Pereiro et al. [52] | Spain | 563 | 50–97 | Age, Edu | Regression | No |
| Pinto et al. [53] | Brazil | 110 | 65–88 | Age, Edu | Regression | No |
| Rossetti et al. [54] | USA | 2653 | 18–85 | Age, Edu | Stratified | No |
| Rossetti et al. [55] | USA | 1118 | 18–75 | Age, Edu | Stratified | No |
| Sachs et al. [56] | USA | 5338 | 55–85 | Age, Edu, Sex, Race | Regression | No |
| Santangelo et al. [57] | Italy | 415 | 21–95 | Age, Edu | Regression | No |
| Serrano et al. [8] | Argentina | 155 | 60–91 | Age, Edu | Stratified | No |
| Siciliano et al. [58] | Italy | 302/413 # | 20–89 | Age, Edu | Regression | No |
| Sink et al. [59] | USA | 414 | 35–83 | Age, Edu | Stratified | No |
| Thomann et al. [60] | Switzerland | 283 | 65–91 | Age, Edu, Sex | Regression | No |
| Current Study | Netherlands | 820 | 18–91 | Age, Edu, Sex | Regression | Yes |
| Education | Low | Average | High | |||
|---|---|---|---|---|---|---|
| Age Group | M | W | M | W | M | W |
| 18–29 | 0 | 0 | 33 | 54 | 30 | 65 |
| 30–39 | 4 | 0 | 11 | 22 | 22 | 20 |
| 40–49 | 7 | 2 | 19 | 29 | 15 | 27 |
| 50–59 | 21 | 9 | 22 | 27 | 39 | 25 |
| 60–69 | 16 | 28 | 25 | 29 | 43 | 31 |
| 70–79 | 9 | 20 | 13 | 23 | 25 | 27 |
| ≥80 | 4 | 7 | 2 | 8 | 5 | 6 |
| Percentile | Diagnostic Label |
|---|---|
| ≥98 | Exceptionally high |
| 91–97 | Above average |
| 75–90 | High average |
| 25–74 | Average |
| 9–24 | Low average |
| 2–8 | Below average |
| <2 | Exceptionally low |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kessels, R.P.C.; de Vent, N.R.; Bruijnen, C.J.W.H.; Jansen, M.G.; de Jonghe, J.F.M.; Dijkstra, B.A.G.; Oosterman, J.M. Regression-Based Normative Data for the Montreal Cognitive Assessment (MoCA) and Its Memory Index Score (MoCA-MIS) for Individuals Aged 18–91. J. Clin. Med. 2022, 11, 4059. https://doi.org/10.3390/jcm11144059
Kessels RPC, de Vent NR, Bruijnen CJWH, Jansen MG, de Jonghe JFM, Dijkstra BAG, Oosterman JM. Regression-Based Normative Data for the Montreal Cognitive Assessment (MoCA) and Its Memory Index Score (MoCA-MIS) for Individuals Aged 18–91. Journal of Clinical Medicine. 2022; 11(14):4059. https://doi.org/10.3390/jcm11144059
Chicago/Turabian StyleKessels, Roy P. C., Nathalie R. de Vent, Carolien J. W. H. Bruijnen, Michelle G. Jansen, Jos F. M. de Jonghe, Boukje A. G. Dijkstra, and Joukje M. Oosterman. 2022. "Regression-Based Normative Data for the Montreal Cognitive Assessment (MoCA) and Its Memory Index Score (MoCA-MIS) for Individuals Aged 18–91" Journal of Clinical Medicine 11, no. 14: 4059. https://doi.org/10.3390/jcm11144059
APA StyleKessels, R. P. C., de Vent, N. R., Bruijnen, C. J. W. H., Jansen, M. G., de Jonghe, J. F. M., Dijkstra, B. A. G., & Oosterman, J. M. (2022). Regression-Based Normative Data for the Montreal Cognitive Assessment (MoCA) and Its Memory Index Score (MoCA-MIS) for Individuals Aged 18–91. Journal of Clinical Medicine, 11(14), 4059. https://doi.org/10.3390/jcm11144059







