Locally Recurrent Rectal Cancer According to a Standardized MRI Classification System: A Systematic Review of the Literature
Abstract
:1. Introduction
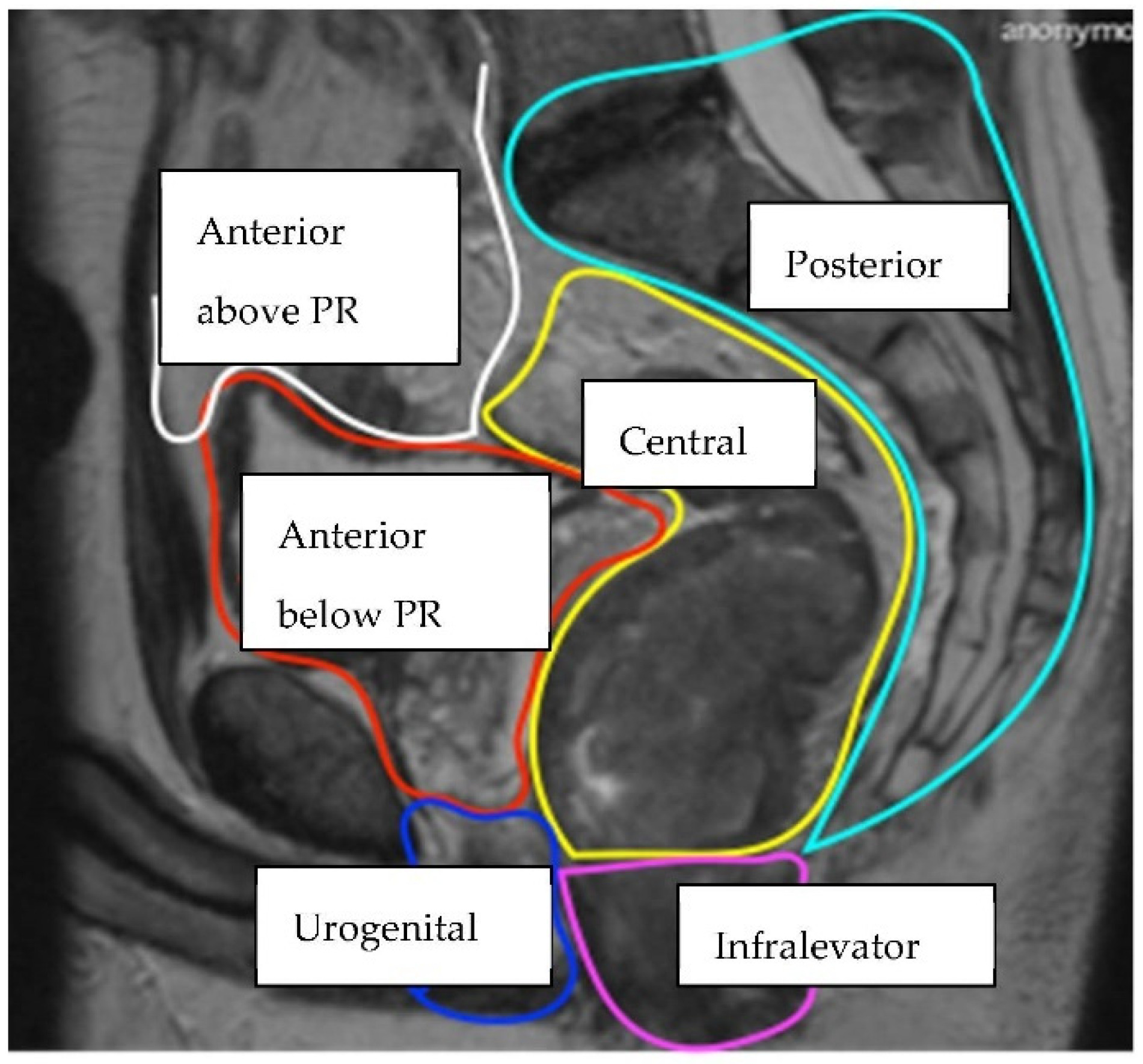
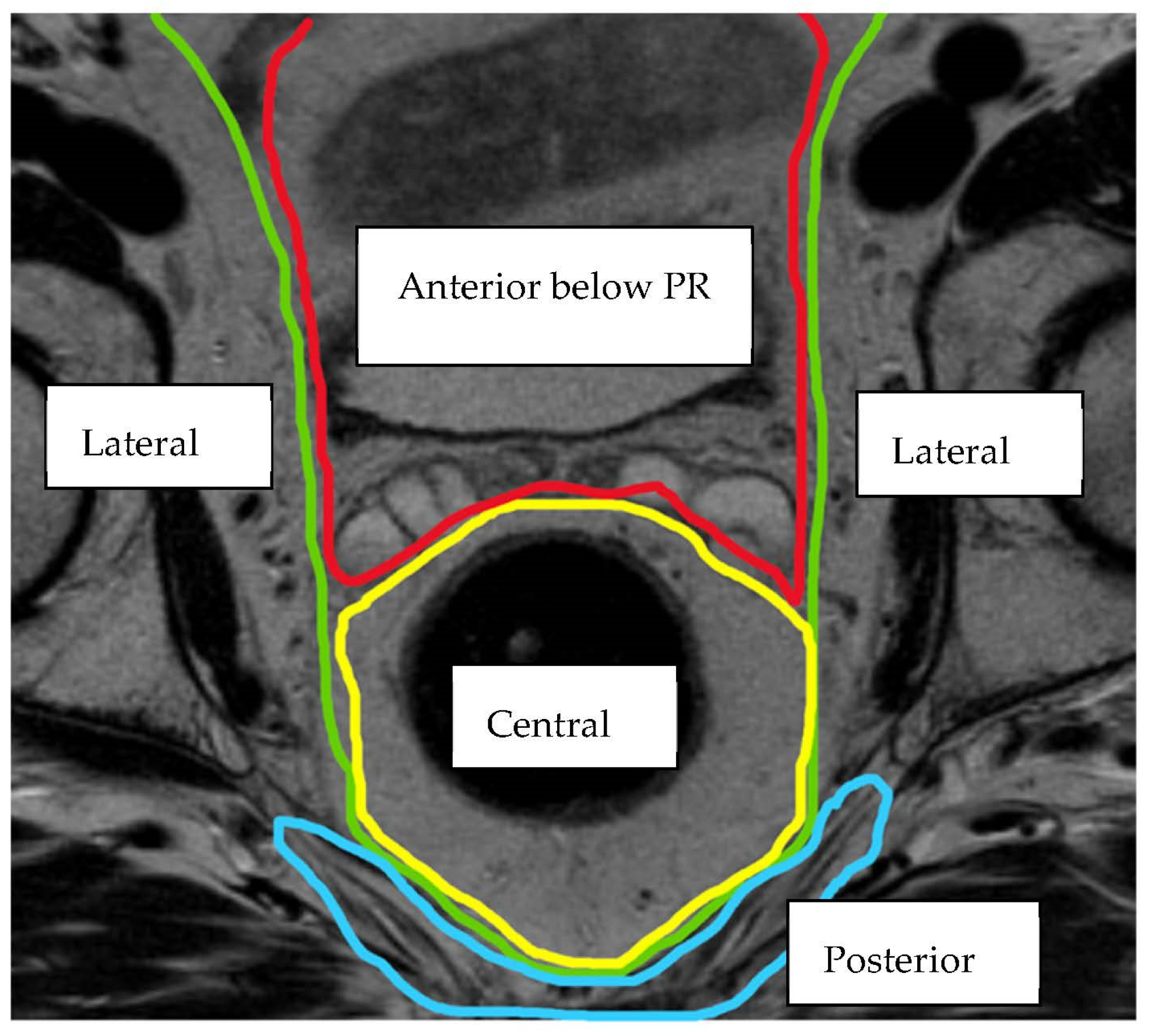
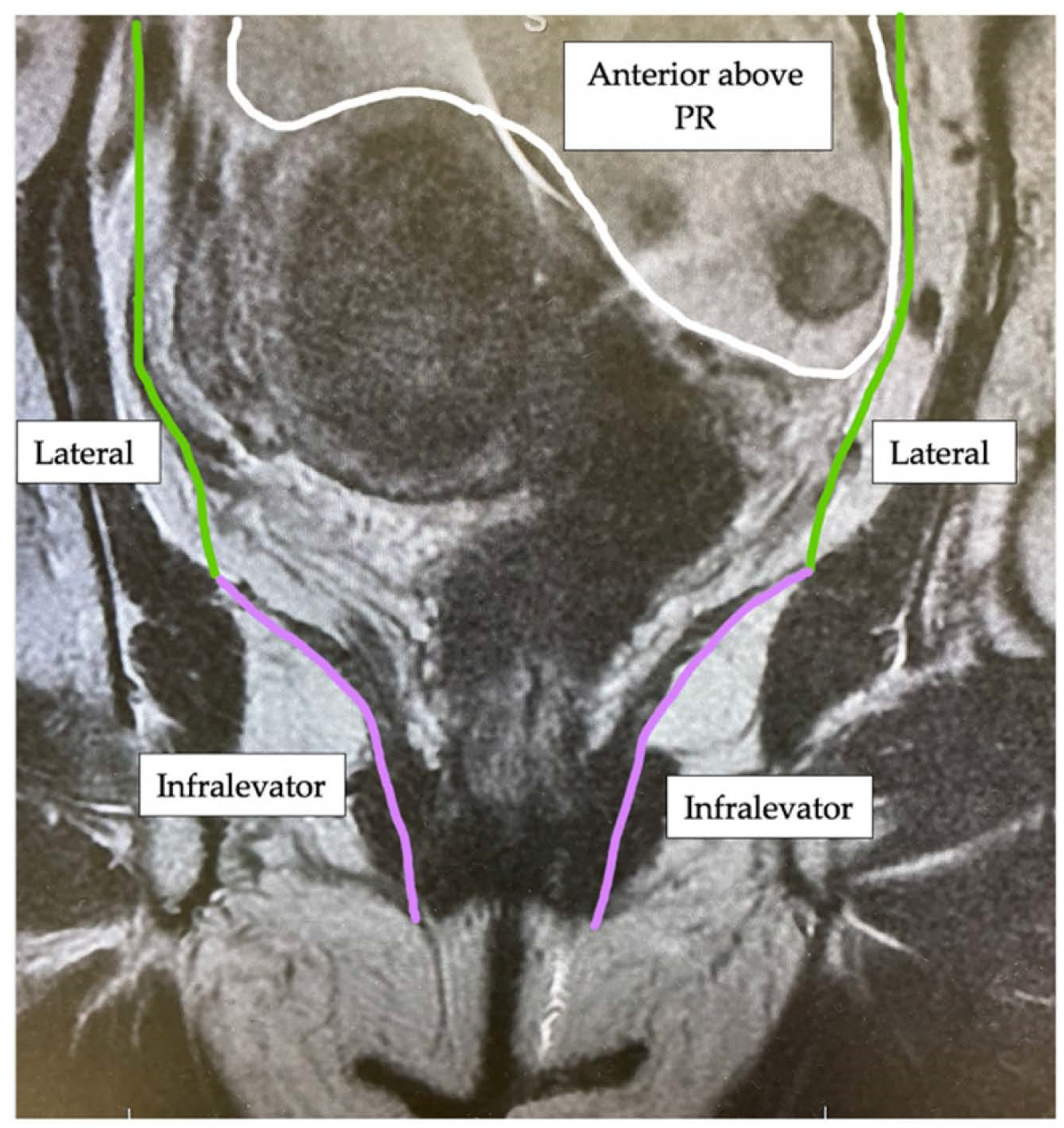
2. Materials and Methods
2.1. Search Strategy
2.2. Selection Criteria
- Reported on patients with LRRC or rectosigmoid cancer who underwent previous ‘curative’ surgery (R0 resection).
- Reported on patients where the anatomical location of LR or an established classification system for describing LRRC was documented.
2.3. Outcomes of Interest
2.3.1. Primary Outcome
2.3.2. Secondary Outcomes
- Height of the primary tumour;
- The primary surgical procedure performed;
- Tumour node metastasis (TNM) staging, extramural vascular invasion (EMVI), and nodal status of the primary tumour;
- Perioperative treatment received for the primary tumour.
3. Results
3.1. Study Stratification
3.1.1. Eligible Studies
3.1.2. Included Studies
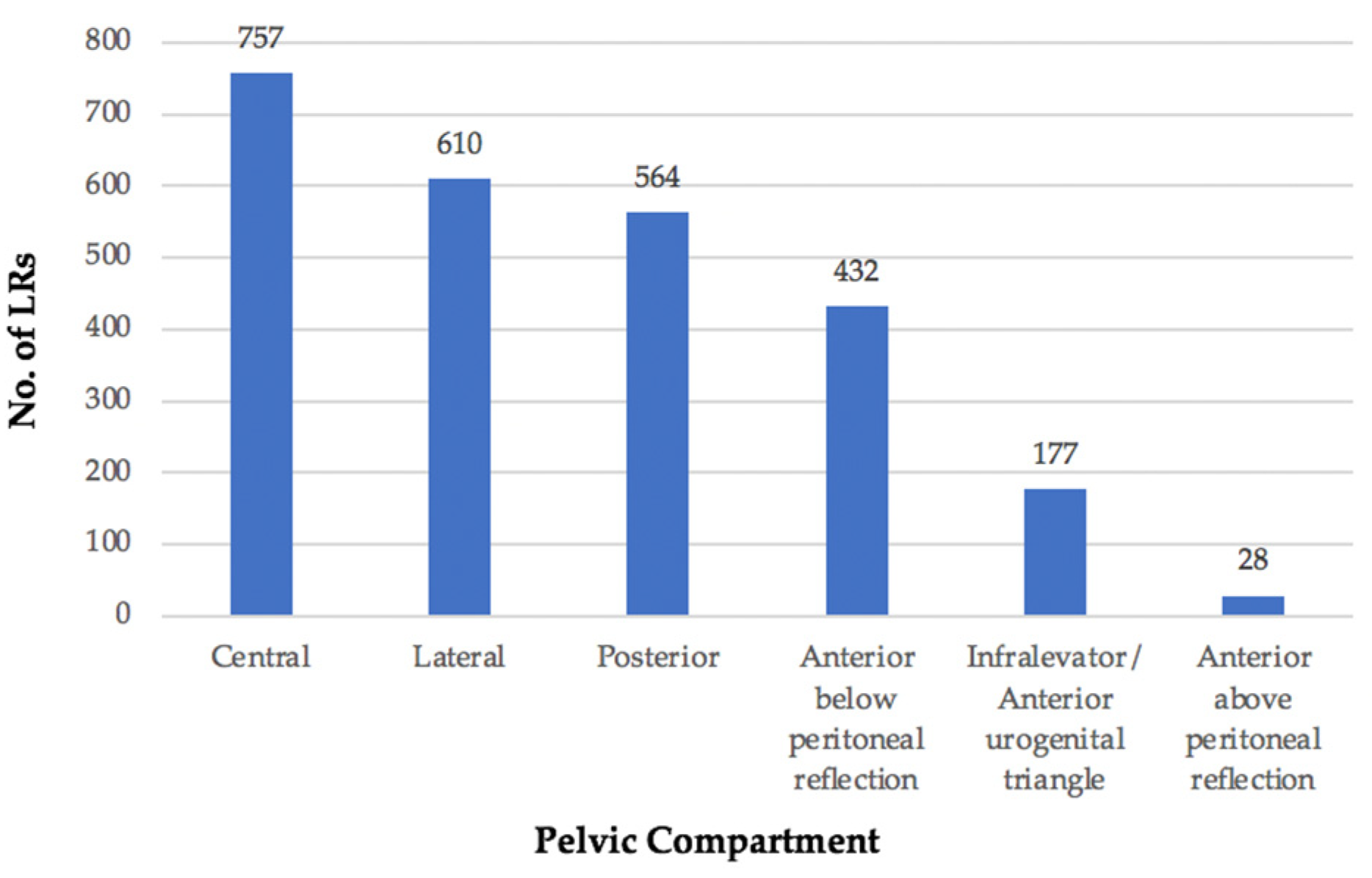
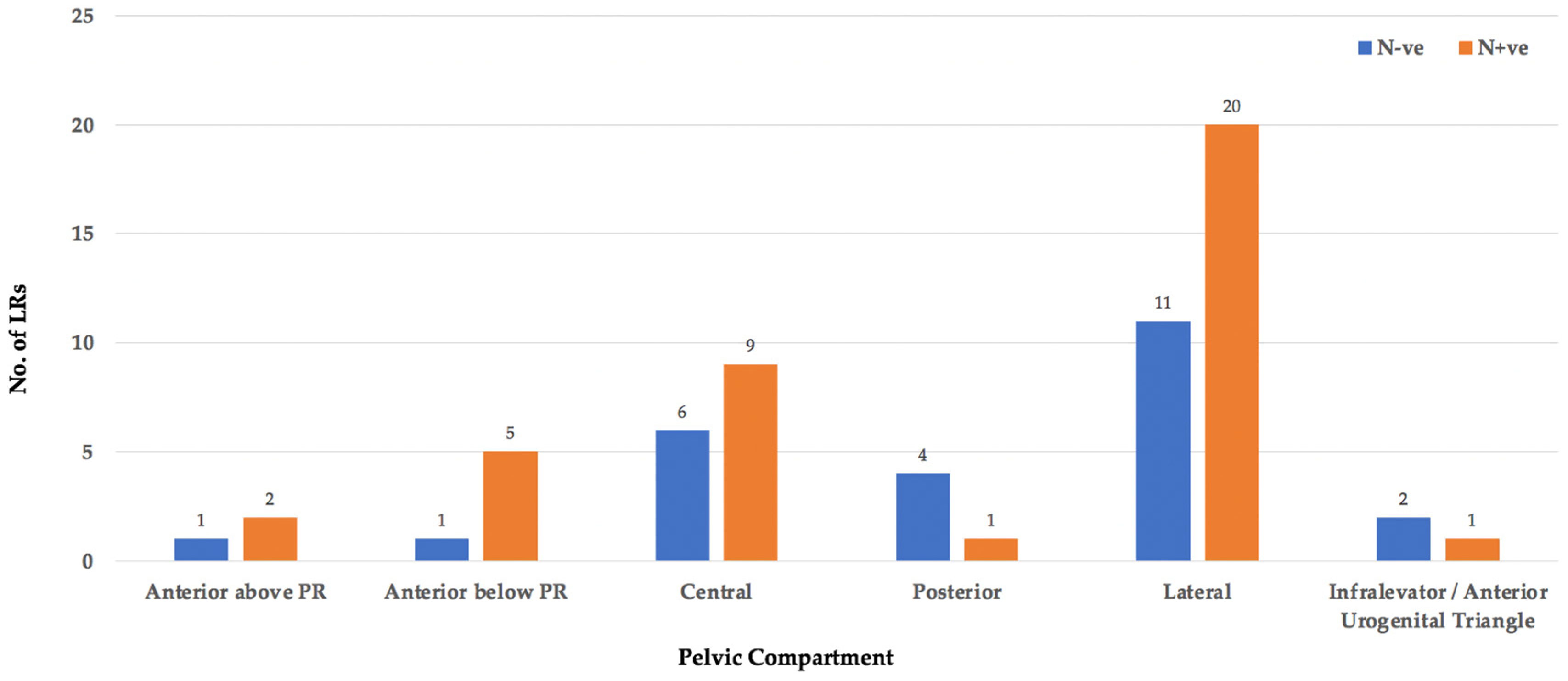
3.2. Location
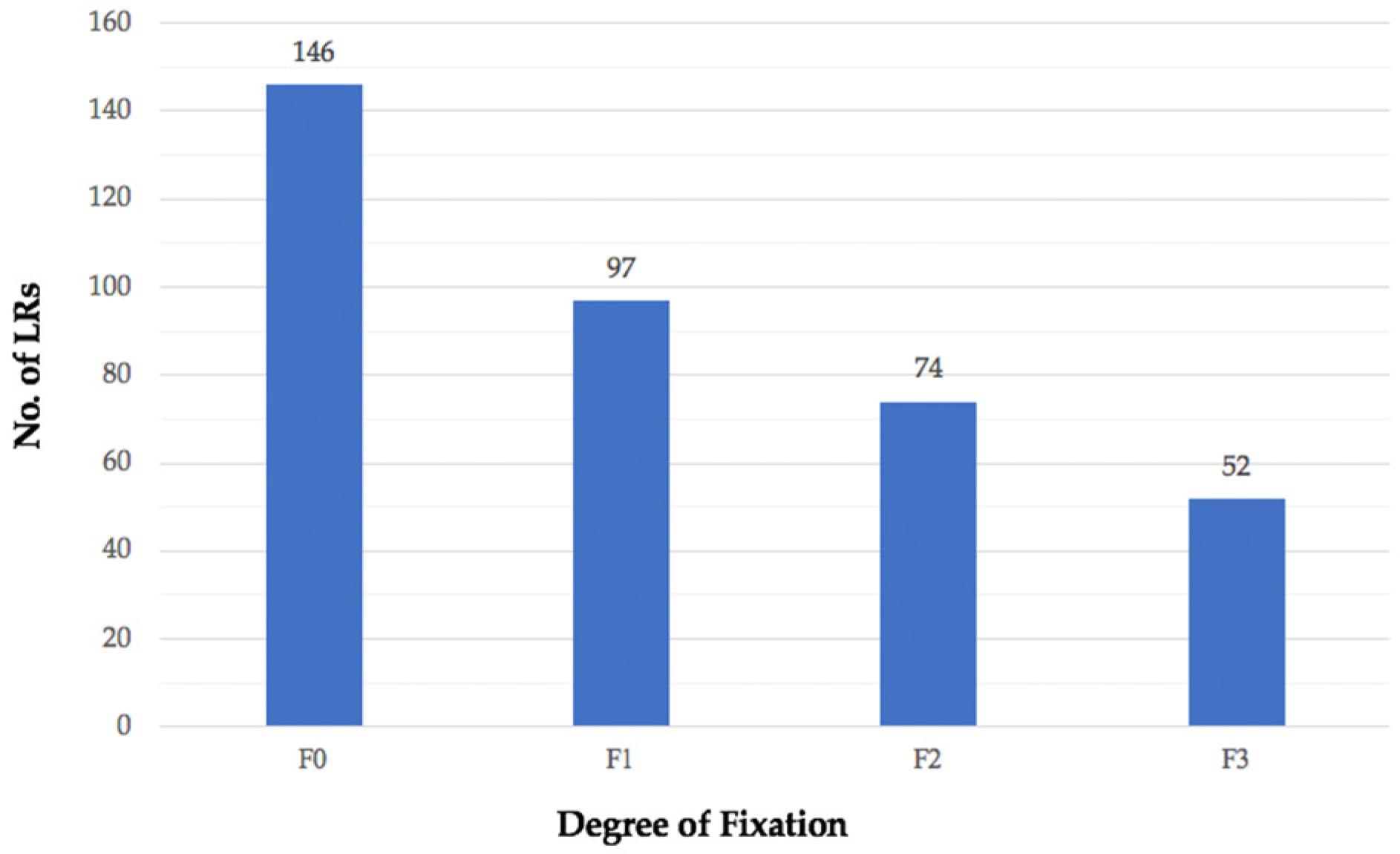
3.3. Height of Primary Tumour
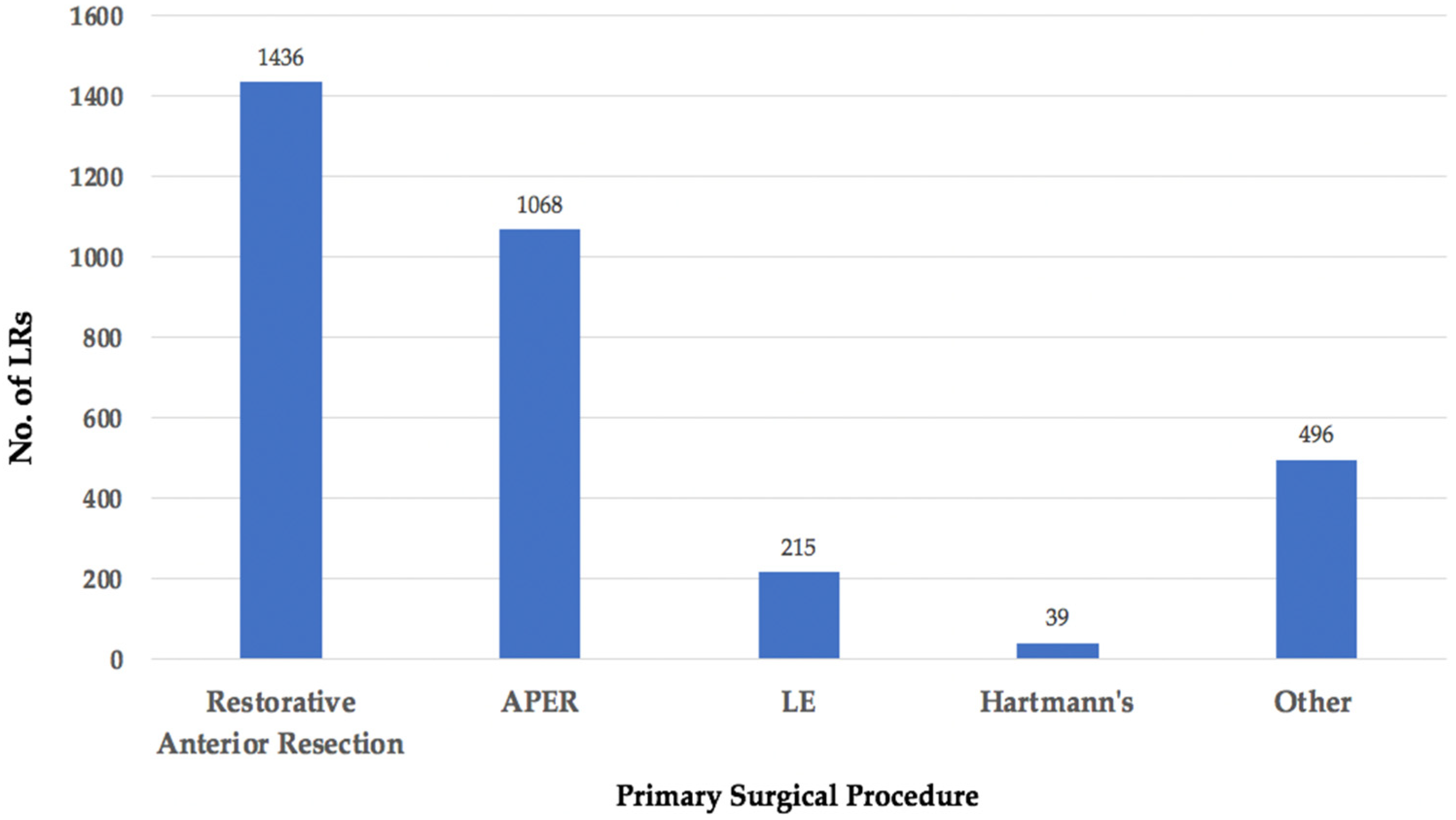
3.4. Primary Surgery
3.5. TNM Stage
3.6. EMVI Status
3.7. Nodal Status
3.8. Perioperative Treatment
4. Discussion
4.1. Location
4.2. Height of Primary Tumour, Primary Surgery and T-Stage
4.3. Perioperative Treatment, EMVI and Nodal Status
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- PelvEx Collaborative. Factors affecting outcomes following pelvic exenteration for locally recurrent rectal cancer. J. Br. Surg. 2018, 105, 650–657. [Google Scholar] [CrossRef] [PubMed]
- Jayne, D.G.; Thorpe, H.C.; Copeland, J.; Quirke, P.; Brown, J.M.; Guillou, P.J. Five-year follow-up of the Medical Research Council CLASICC trial of laparoscopically assisted versus open surgery for colorectal cancer. J. Br. Surg. 2010, 97, 1638–1645. [Google Scholar] [CrossRef] [PubMed]
- Bhangu, A.; Ali, S.M.; Brown, G.; Nicholls, R.J.; Tekkis, P. Indications and outcome of pelvic exenteration for locally advanced primary and recurrent rectal cancer. Ann. Surg. 2014, 259, 315–322. [Google Scholar] [CrossRef] [PubMed]
- Beyond TME Collaborative. Consensus statement on the multidisciplinary management of patients with recurrent and primary rectal cancer beyond total mesorectal excision planes. J. Br. Surg. 2013, 100, 1009–1014. [Google Scholar] [CrossRef]
- Georgiou, P.A.; Tekkis, P.P.; Constantinides, V.A.; Patel, U.; Goldin, R.D.; Darzi, A.W.; Nicholls, R.J.; Brown, G. Diagnostic accuracy and value of magnetic resonance imaging (MRI) in planning exenterative pelvic surgery for advanced colorectal cancer. Eur. J. Cancer 2013, 49, 72–81. [Google Scholar] [CrossRef]
- Suzuki, K.; Dozois, R.R.; Devine, R.M.; Nelson, H.; Weaver, A.L.; Gunderson, L.L.; Ilstrup, D.M. Curative reoperations for locally recurrent rectal cancer. Dis. Colon Rectum 1996, 39, 730–736. [Google Scholar] [CrossRef]
- Yamada, K.; Ishizawa, T.; Niwa, K.; Chuman, Y.; Akiba, S.; Aikou, T. Patterns of pelvic invasion are prognostic in the treatment of locally recurrent rectal cancer. Br. J. Surg. 2001, 88, 988–993. [Google Scholar] [CrossRef]
- Wanebo, H.J.; Antoniuk, P.; Koness, R.J.; Levy, A.; Vezeridis, M.; Cohen, S.I.; Wrobleski, D.E. Pelvic resection of recurrent rectal cancer—Technical considerations and outcomes. Dis. Colon Rectum 1999, 42, 1438–1448. [Google Scholar] [CrossRef]
- Moore, H.G.; Shoup, M.; Riedel, E.; Minsky, B.D.; Alektiar, K.M.; Ercolani, M.; Paty, P.B.; Wong, D.W.; Guillem, J.G. Colorectal cancer pelvic recurrences: Determinants of resectability. Dis. Colon Rectum 2004, 47, 1599–1606. [Google Scholar] [CrossRef]
- Roels, S.; Duthoy, W.; Haustermans, K.; Penninckx, F.; Vandecaveye, V.; Boterberg, T.; De Neve, W. Definition and delineation of the clinical target volume for rectal cancer. Int. J. Radiat. Oncol. Biol. Phys. 2006, 65, 1129–1142. [Google Scholar] [CrossRef]
- Pilipshen, S.J.; Heilweil, M.; Quan, S.H.; Sternberg, S.S.; Enker, W.E. Patterns of pelvic recurrence following definitive resections of rectal cancer. Cancer 1984, 53, 1354–1362. [Google Scholar] [CrossRef]
- Boyle, K.M.; Sagar, P.M.; Chalmers, A.G.; Sebag-Montefiore, D.; Cairns, A.; Eardley, I. Surgery for locally recurrent rectal cancer. Dis. Colon Rectum 2005, 48, 929–937. [Google Scholar] [CrossRef] [PubMed]
- Hruby, G.; Barton, M.; Miles, S.; Carroll, S.; Nasser, E.; Stevens, G. Sites of local recurrence after surgery, with or without chemotherapy, for rectal cancer: Implications for radiotherapy field design. Int. J. Radiat. Oncol. Biol. Phys. 2003, 55, 138–143. [Google Scholar] [CrossRef]
- Georgiou, P.A.; Tekkis, P.P.; Brown, G. Pelvic colorectal recurrence: Crucial role of radiologists in oncologic and surgical treatment options. Cancer Imaging Off. Publ. Int. Cancer Imaging Soc. 2011, 11, S103–S111. [Google Scholar] [CrossRef] [Green Version]
- Rokan, Z.; Simillis, C.; Kontovounisios, C.; Moran, B.J.; Tekkis, P.; Brown, G. Systematic review of classification systems for locally recurrent rectal cancer. BJS Open 2021, 5, zrab024. [Google Scholar] [CrossRef]
- Moher, D.; Shamseer, L.; Clarke, M.; Ghersi, D.; Liberati, A.; Petticrew, M.; Shekelle, P.; Stewart, L.A. Preferred reporting items for systematic review and meta-analysis protocols (PRISMA-P) 2015 statement. Syst. Rev. 2015, 4, 1. [Google Scholar] [CrossRef] [Green Version]
- Shea, B.J.; Reeves, B.C.; Wells, G.; Thuku, M.; Hamel, C.; Moran, J.; Moher, D.; Tugwell, P.; Welch, V.; Kristjansson, E.; et al. AMSTAR 2: A critical appraisal tool for systematic reviews that include randomised or non-randomised studies of healthcare interventions, or both. BMJ 2017, 358, j4008. [Google Scholar] [CrossRef] [Green Version]
- Adachi, W.; Nishio, A.; Watanabe, H.; Igarashi, J.; Yazawa, K.; Nimura, Y.; Koide, N.; Matsushita, A.; Monma, T.; Hanazaki, K.; et al. Reresection for local recurrence of rectal cancer. Surg. Today 1999, 29, 999–1003. [Google Scholar] [CrossRef]
- Adloff, M.; Arnaud, J.P.; Schloegel, M.; Thibaud, D. Factors influencing local recurrence after abdominoperineal resection for cancer of the rectum. Dis. Colon Rectum 1985, 28, 413–415. [Google Scholar] [CrossRef]
- Baek, J.Y.; Yu, J.I.; Park, H.C.; Choi, O.H.; Yoo, G.S.; Cho, W.K.; Lee, W.-Y.; Yun, S.H.; Cho, Y.B.; Park, Y.A.; et al. Risk factors for locoregional recurrence in patients with pathologic T3N0 rectal cancer with negative resection margin treated by surgery alone. Radiat. Oncol. J. 2019, 37, 110–116. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Beppu, N.; Kimura, F.; Aihara, T.; Doi, H.; Tomita, N.; Yanagi, H.; Yamanaka, N. Patterns of Local Recurrence and Oncologic Outcomes in T3 Low Rectal Cancer (≤5 cm from the Anal Verge) Treated With Short-Course Radiotherapy With Delayed Surgery: Outcomes in T3 Low Rectal Cancer Treated With Short-Course Radiotherapy With Delayed Surgery. Ann. Surg. Oncol. 2017, 24, 219–226. [Google Scholar] [PubMed]
- Beppu, N.; Okamoto, R.; Nakamoto, Y.; Kimura, F.; Ikeda, M.; Tomita, N.; Yanagi, H.; Yamanaka, N. Risk factors and patterns of local recurrence in T3 rectal cancer treated with short-course hyperfractionated accelerated chemoradiotherapy with delayed surgery. Acta Oncol. 2018, 57, 1723–1727. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bikhchandani, J.; Ong, G.K.; Dozois, E.J.; Mathis, K.L. Outcomes of salvage surgery for cure in patients with locally recurrent disease after local excision of rectal cancer. Dis. Colon Rectum 2015, 58, 283–287. [Google Scholar] [CrossRef] [PubMed]
- Bird, T.G.; Ngan, S.Y.; Chu, J.; Kroon, R.; Lynch, A.C.; Heriot, A.G. Outcomes and prognostic factors of multimodality treatment for locally recurrent rectal cancer with curative intent. Int. J. Colorectal Dis. 2018, 33, 393–401. [Google Scholar] [CrossRef] [PubMed]
- Brown, W.E.; Koh, C.E.; Badgery-Parker, T.; Solomon, M.J. Validation of MRI and Surgical Decision Making to Predict a Complete Resection in Pelvic Exenteration for Recurrent Rectal Cancer. Dis. Colon Rectum 2017, 60, 144–151. [Google Scholar] [CrossRef]
- Choi, S.H.; Chang, J.S.; Yoon, H.I.; Jang, D.-S.; Kim, N.K.; Lim, J.S.; Min, B.S.; Huh, H.; Shin, S.J.; Ahn, J.B.; et al. Mapping of lateral pelvic lymph node recurrences in rectal cancer: A radiation oncologist’s perspective. J. Cancer Res. Clin. Oncol. 2018, 144, 1119–1128. [Google Scholar] [CrossRef]
- Dresen, R.C.; Kusters, M.; Daniels-Gooszen, A.W.; Cappendijk, V.C.; Nieuwenhuijzen, G.A.P.; Kessels, A.G.H.; de Bruïne, A.P.; Beets, G.L.; Rutten, H.J.T.; Beets-Tan, R.G.H. Absence of tumor invasion into pelvic structures in locally recurrent rectal cancer: Prediction with preoperative MR imaging. Radiology 2010, 256, 143–150. [Google Scholar] [CrossRef] [Green Version]
- Even-Sapir, E.; Parag, Y.; Lerman, H.; Gutman, M.; Levine, C.; Rabau, M.; Figer, A.; Metser, U. Detection of recurrence in patients with rectal cancer: PET/CT after abdominoperineal or anterior resection. Radiology 2004, 232, 815–822. [Google Scholar] [CrossRef] [PubMed]
- Fujii, S.; Shimada, H.; Yamagishi, S.; Ota, M.; Ichikawa, Y.; Kunisaki, C.; Ike, H.; Ohki, S. Surgical Strategy for Local Recurrence after Resection of Rectal Cancer. Hepato-Gastroenterology 2009, 56, 667–671. [Google Scholar]
- Gleeson, F.C.; Larson, D.W.; Dozois, E.J.; A Boardman, L.; E Clain, J.; Rajan, E.; Topazian, M.D.; Wang, K.K.; Levy, M.J. Local Recurrence Detection Following Transanal Excision Facilitated by EUS-FNA. Hepato-Gastroenterology 2012, 59, 1102–1107. [Google Scholar] [CrossRef]
- Hahnloser, D.; Nelson, H.; Gunderson, L.L.; Hassan, I.; Haddock, M.G.; O’Connell, M.J.; Cha, S.; Sargent, D.J.; Horgan, A. Curative potential of multimodality therapy for locally recurrent rectal cancer. Ann. Surg. 2003, 237, 502–508. [Google Scholar] [CrossRef] [PubMed]
- Hasegawa, S.; Takahashi, R.; Hida, K.; Kawada, K.; Sakai, Y. Revisiting the treatment strategy for rectal cancer through the pattern of local recurrence. Eur. J. Surg. Oncol. 2016, 42, 1674–1679. [Google Scholar] [CrossRef] [PubMed]
- Ingle, P.; Bal, M.; Engineer, R.; Ostwal, V.; Desouza, A.; Saklani, A. Do Acellular Mucin Pools in Resection Margins for Rectal Cancer Influence Outcomes? Indian J. Surg. Oncol. 2019, 10, 515–519. [Google Scholar] [CrossRef] [PubMed]
- Kanemitsu, Y.; Hirai, T.; Komori, K.; Kato, T. Prediction of residual disease or distant metastasis after resection of locally recurrent rectal cancer. Dis. Colon Rectum 2010, 53, 779–789. [Google Scholar] [CrossRef]
- Killingback, M.; Barron, P.; Dent, O.F. Local recurrence after curative resection of cancer of the rectum without total mesorectal excision. Dis. Colon Rectum 2001, 44, 473–483. [Google Scholar] [CrossRef]
- Kim, T.H.; Jeong, S.-Y.; Choi, D.H.; Kim, D.Y.; Jung, K.H.; Moon, S.H.; Chang, H.J.; Lim, S.-B.; Choi, H.-S.; Park, J.-G. Lateral lymph node metastasis is a major cause of locoregional recurrence in rectal cancer treated with preoperative chemoradiotherapy and curative resection. Ann. Surg. Oncol. 2008, 15, 729–737. [Google Scholar] [CrossRef]
- Kim, T.G.; Park, W.; Choi, D.H.; Park, H.C.; Kim, S.-H.; Cho, Y.B.; Yun, S.H.; Kim, H.C.; Lee, W.Y.; Lee, J.; et al. Factors associated with lateral pelvic recurrence after curative resection following neoadjuvant chemoradiotherapy in rectal cancer patients. Int. J. Colorectal Dis. 2014, 29, 193–200. [Google Scholar] [CrossRef]
- Kusters, M.; Beets, G.; van de Velde, C.J.H.; Beets-Tan, R.G.H.; Marijnen, C.; Rutten, H.J.T.; Putter, H.; Moriya, Y. A comparison between the treatment of low rectal cancer in Japan and the Netherlands, focusing on the patterns of local recurrence. Ann. Surg. 2009, 249, 229–235. [Google Scholar] [CrossRef]
- Kusters, M.; Holman, F.A.; Martijn, H.; Nieuwenhuijzen, G.A.; Creemers, G.J.; Daniels-Gooszen, A.W.; van den Berg, H.A.; van den Brule, A.J.; van de Velde, C.J.H.; Rutten, H.J.T. Patterns of local recurrence in locally advanced rectal cancer after intra-operative radiotherapy containing multimodality treatment. Radiother. Oncol. 2009, 92, 221–225. [Google Scholar] [CrossRef]
- Kusters, M.; Marijnen, C.; van de Velde, C.; Rutten, H.; Lahaye, M.; Kim, J.; Beets-Tan, R.; Beets, G. Patterns of local recurrence in rectal cancer; a study of the Dutch TME trial. Eur. J. Surg. Oncol. 2010, 36, 470–476. [Google Scholar] [CrossRef] [Green Version]
- Lambregts, D.M.; Cappendijk, V.C.; Maas, M.; Beets, G.L.; Beets-Tan, R.G. Value of MRI and diffusion-weighted MRI for the diagnosis of locally recurrent rectal cancer. Eur. Radiol. 2011, 21, 1250–1258. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Luna-Perez, P.; Rodriguez-Ramirez, S.; Gonzalez-Macouzet, J.; Rodriguez-Coria, D.F.; Delgado, S.; Lopez, M.J. The influence of pre-operative radiation therapy on the patterns of recurrence in rectal adenocarcinoma. Semin. Surg. Oncol. 1999, 17, 199–205. [Google Scholar] [CrossRef]
- McDermott, F.T.; Hughes, E.S.; Pihl, E.; Johnson, W.R.; Price, A.B. Local recurrence after potentially curative resection for rectal cancer in a series of 1008 patients. Br. J. Surg. 1985, 72, 34–37. [Google Scholar] [CrossRef]
- Mendenhall, W.M.; Million, R.R.; Pfaff, W.W. Patterns of recurrence in adenocarcinoma of the rectum and rectosigmoid treated with surgery alone: Implications in treatment planning with adjuvant radiation therapy. Int. J. Radiat. Oncol. Biol. Phys. 1983, 9, 977–985. [Google Scholar] [CrossRef]
- Messiou, C.; Chalmers, A.G.; Boyle, K.; Wilson, D.; Sagar, P. Pre-operative MR assessment of recurrent rectal cancer. Br. J. Radiol. 2008, 81, 468–473. [Google Scholar] [CrossRef] [PubMed]
- Nagasaki, T.; Akiyoshi, T.; Ueno, M.; Fukunaga, Y.; Nagayama, S.; Fujimoto, Y.; Konishi, T.; Yamaguchi, T. Laparoscopic salvage surgery for locally recurrent rectal cancer. J. Gastrointest. Surg. 2014, 18, 1319–1326. [Google Scholar] [CrossRef] [PubMed]
- Novell, F.; Pascual, S.; Viella, P.; Trias, M. Endorectal ultrasonography in the follow-up of rectal cancer. Is it a better way to detect early local recurrence? Int. J. Colorectal Dis. 1997, 12, 78–81. [Google Scholar] [CrossRef]
- Pergolizzi, S.; Settineri, N.; Santacaterina, A.; Maisano, R.; Frosina, P.; Loria, F.; Nardella, G.; Garufi, G.; Sansotta, G.; De Renzis, C. Prognostic factors in ambulatory patients with inoperable locoregionally recurrent rectal cancer following curative surgery. Anticancer Res. 1999, 19, 1383–1390. [Google Scholar]
- Rahbari, N.N.; Ulrich, A.B.; Bruckner, T.; Munter, M.; Nickles, A.; Contin, P.; Löffler, T.; Reissfelder, C.; Koch, M.; Büchler, M.; et al. Surgery for locally recurrent rectal cancer in the era of total mesorectal excision: Is there still a chance for cure? Ann. Surg. 2011, 253, 522–533. [Google Scholar] [CrossRef]
- Rich, T.; Gunderson, L.L.; Lew, R.; Galdibini, J.J.; Cohen, A.M.; Donaldson, G. Patterns of recurrence of rectal cancer after potentially curative surgery. Cancer 1983, 52, 1317–1329. [Google Scholar] [CrossRef]
- Roodbeen, S.X.; Spinelli, A.; Bemelman, W.A.; Di Candido, F.; Cardepont, M.; Denost, Q.; D’Hoore, A.; Houben, B.; Knol, J.J.; Martín-Pérez, B.; et al. Local Recurrence After Transanal Total Mesorectal Excision for Rectal Cancer: A Multicenter Cohort Study. Ann Surg. 2021, 274, 359–366. [Google Scholar] [CrossRef] [PubMed]
- Safioleas, M.C.; Moulakakis, K.G.; Stamatakos, M.; Kountouras, J.; Lygidakis, N.J. Local recurrence following curative low anterior resection for rectal carcinoma. Hepato-Gastroenterology 2005, 52, 94–96. [Google Scholar] [PubMed]
- Scialpi, M.; Andreatta, R.; Agugiaro, S.; Zottele, F.; Niccolini, M.; Dalla Palma, F. Rectal carcinoma: Preoperative staging and detection of postoperative local recurrence with transrectal and transvaginal ultrasound. Abdom. Imaging 1993, 18, 381–389. [Google Scholar] [CrossRef] [PubMed]
- Sinaei, M.; Swallow, C.; Milot, L.; Moghaddam, P.A.; Smith, A.; Atri, M. Patterns and signal intensity characteristics of pelvic recurrence of rectal cancer at MR imaging. Radiographics 2013, 33, E171–E187. [Google Scholar] [CrossRef]
- Syk, E.; Torkzad, M.R.; Blomqvist, L.; Nilsson, P.J.; Glimelius, B. Local recurrence in rectal cancer: Anatomic localization and effect on radiation target. Int. J. Radiat. Oncol. Biol. Phys. 2008, 72, 658–664. [Google Scholar] [CrossRef]
- Uehara, K.; Ito, Z.; Yoshino, Y.; Arimoto, A.; Kato, T.; Nakamura, H.; Imagama, S.; Nishida, Y.; Nagino, M. Aggressive surgical treatment with bony pelvic resection for locally recurrent rectal cancer. Eur. J. Surg. Oncol. 2015, 41, 413–420. [Google Scholar] [CrossRef]
- Valentini, V.; Morganti, A.G.; De Franco, A.; Coco, C.; Ratto, C.; Battista Doglietto, G.; Trodella, L.; Ziccarelli, L.; Picciocchi, A.; Cellini, N. Chemoradiation with or without intraoperative radiation therapy in patients with locally recurrent rectal carcinoma: Prognostic factors and long term outcome. Cancer 1999, 86, 2612–2624. [Google Scholar] [CrossRef]
- Vigliotti, A.; Rich, T.A.; Romsdahl, M.M.; Withers, H.R.; Oswald, M.J. Postoperative adjuvant radiotherapy for adenocarcinoma of the rectum and rectosigmoid. Int. J. Radiat. Oncol. Biol. Phys. 1987, 13, 999–1006. [Google Scholar] [CrossRef]
- Westberg, K.; Palmer, G.; Hjern, F.; Nordenvall, C.; Johansson, H.; Holm, T.; Martling, A. Population-based study of factors predicting treatment intention in patients with locally recurrent rectal cancer. Br. J. Surg. 2017, 104, 1866–1873. [Google Scholar] [CrossRef] [Green Version]
- Wieldraaijer, T.; Bruin, P.; Duineveld, L.A.; Tanis, P.; Smits, A.B.; Van Weert, H.C.; Wind, J. Clinical Pattern of Recurrent Disease during the Follow-Up of Rectal Carcinoma. Dig. Surg. 2018, 35, 35–41. [Google Scholar] [CrossRef] [Green Version]
- Wiig, J.N.; Wolff, P.A.; Tveit, K.M.; Giercksky, K.E. Location of pelvic recurrence after ‘curative’ low anterior resection for rectal cancer. Eur. J. Surg. Oncol. 1999, 25, 590–594. [Google Scholar] [CrossRef]
- Wong, C.S.; Cummings, B.J.; Brierley, J.D.; Catton, C.N.; McLean, M.; Catton, P.; Hao, Y. Treatment of locally recurrent rectal carcinoma–results and prognostic factors. Int. J. Radiat. Oncol. Biol. Phys. 1998, 40, 427–435. [Google Scholar] [CrossRef]
- You, Y.N.; Roses, R.E.; Chang, G.J.; Rodriguez-Bigas, M.A.; Feig, B.W.; Slack, R.; Nguyen, S.; Skibber, J.M. Multimodality Salvage of Recurrent Disease After Local Excision for Rectal Cancer. Dis. Colon Rectum 2012, 55, 1213–1219. [Google Scholar] [CrossRef] [PubMed]
- Yu, T.-K.; Bhosale, P.R.; Crane, C.H.; Iyer, R.B.; Skibber, J.M.; Rodriguez-Bigas, M.A.; Feig, B.W.; Chang, G.J.; Eng, C.; Wolff, R.A.; et al. Patterns of locoregional recurrence after surgery and radiotherapy or chemoradiation for rectal cancer. Int. J. Radiat. Oncol. 2008, 71, 1175–1180. [Google Scholar] [CrossRef]
- Yun, J.-A.; Huh, J.W.; Kim, H.C.; Park, Y.A.; Cho, Y.B.; Yun, S.H.; Lee, W.Y.; Chun, H.-K. Local recurrence after curative resection for rectal carcinoma: The role of surgical resection. Medicine 2016, 95, e3942. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Du, C.Z.; Sun, Y.S.; Gu, J. Patterns and prognosis of locally recurrent rectal cancer following multidisciplinary treatment. World J. Gastroenterol. 2012, 18, 7015–7020. [Google Scholar] [CrossRef] [PubMed]
- Zhu, X.G.; Li, Y.H.; Li, X.F.; Cai, Y. Intensity-modulated radiation therapy for pelvic oligo-recurrence from rectal cancer: Long-term results from a single institution. Am. J. Transl. Res. 2016, 8, 1265–1272. [Google Scholar] [PubMed]
- Iversen, H.; Martling, A.; Johansson, H.; Nilsson, P.J.; Holm, T. Pelvic local recurrence from colorectal cancer: Surgical challenge with changing preconditions. Colorectal Dis. 2018, 20, 399–406. [Google Scholar] [CrossRef]
- Gilbertsen, V.A. Adenocarcinoma of the Rectum: Incidence and Locations of Recurrent Tumor Following Present-day Operations Performed for Cure. Ann. Surg. 1960, 151, 340–348. [Google Scholar] [CrossRef]
- Shao, H.; Ma, X.; Gao, Y.; Wang, J.; Wu, J.; Wang, B.; Li, J.; Tian, J. Comparison of the diagnostic efficiency for local recurrence of rectal cancer using CT, MRI, PET and PET-CT: A systematic review protocol. Medicine 2018, 97, e12900. [Google Scholar] [CrossRef]
- Heald, R.J. The ‘Holy Plane’ of rectal surgery. J. R. Soc. Med. 1988, 81, 503–508. [Google Scholar] [CrossRef] [PubMed]
- Taylor, F.G.; Quirke, P.; Heald, R.J.; Moran, B.J.; Blomqvist, L.; Swift, I.R.; Sebag-Montefiore, D.; Tekkis, P.; Brown, G. Preoperative magnetic resonance imaging assessment of circumferential resection margin predicts disease-free survival and local recurrence: 5-year follow-up results of the MERCURY study. J. Clin. Oncol. 2014, 32, 34–43. [Google Scholar] [CrossRef] [PubMed]
- Heald, R.J.; Husband, E.M.; Ryall, R.D. The mesorectum in rectal cancer surgery–the clue to pelvic recurrence? Br. J. Surg. 1982, 69, 613–616. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Liu, J.; Wang, S.; Zhao, H.; Ge, S.; Wang, W. Adverse Effects of Anastomotic Leakage on Local Recurrence and Survival After Curative Anterior Resection for Rectal Cancer: A Systematic Review and Meta-analysis. World J. Surg. 2017, 41, 277–284. [Google Scholar] [CrossRef]
- Enriquez-Navascues, J.M.; Borda, N.; Lizerazu, A.; Placer, C.; Elosegui, J.L.; Ciria, J.P.; Lacasta, A.; Bujanda, L. Patterns of local recurrence in rectal cancer after a multidisciplinary approach. World J. Gastroenterol. 2011, 17, 1674–1684. [Google Scholar] [CrossRef]
- Fan, W.-H.; Xiao, J.; An, X.; Jiang, W.; Li, L.-R.; Gao, Y.-H.; Chen, G.; Kong, L.-H.; Lin, J.-Z.; Wang, J.-P.; et al. Patterns of recurrence in patients achieving pathologic complete response after neoadjuvant chemoradiotherapy for rectal cancer. J. Cancer Res. Clin. Oncol. 2017, 143, 1461–1467. [Google Scholar] [CrossRef] [Green Version]
- Kusters, M.; van de Velde, C.J.; Beets-Tan, R.G.; Akasu, T.; Fujita, S.; Yamamoto, S.; Moriya, Y. Patterns of local recurrence in rectal cancer: A single-center experience. Ann. Surg. Oncol. 2009, 16, 289–296. [Google Scholar] [CrossRef] [Green Version]
- Heald, R.J.; Moran, B.J.; Ryall, R.D.H.; Sexton, R.; MacFarlane, J.K. Rectal Cancer: The Basingstoke experience of total mesorectal excision, 1978–1997. Arch. Surg. 1998, 133, 894–899. [Google Scholar] [CrossRef] [Green Version]
- Nagtegaal, I.D.; van de Velde, C.J.; Marijnen, C.A.; van Krieken, J.H.; Quirke, P. Low rectal cancer: A call for a change of approach in abdominoperineal resection. J. Clin. Oncol. 2005, 23, 9257–9264. [Google Scholar] [CrossRef] [Green Version]
- Nagtegaal, I.D.; Quirke, P. What is the role for the circumferential margin in the modern treatment of rectal cancer? J. Clin. Oncol. 2008, 26, 303–312. [Google Scholar] [CrossRef]
- Adam, I.; Martin, I.; Finan, P.; Johnston, D.; Mohamdee, M.; Scott, N.; Dixon, M.; Quirke, P. Role of circumferential margin involvement in the local recurrence of rectal cancer. Lancet 1994, 344, 707–711. [Google Scholar] [CrossRef]
- Rasanen, M.; Carpelan-Holmstrom, M.; Mustonen, H.; Renkonen-Sinisalo, L.; Lepisto, A. Pattern of rectal cancer recurrence after curative surgery. Int. J. Colorectal Dis. 2015, 30, 775–785. [Google Scholar] [CrossRef] [PubMed]



| Compartment | Structures within Compartment |
|---|---|
| Anterior above Peritoneal Reflection | Ureters, iliac vessels above peritoneal reflection, sigmoid colon, small bowel, lateral pelvic sidewall fascia (peritoneal surface) |
| Anterior below Peritoneal Reflection | Genitourinary system (seminal vesicles, prostate, uterus, vagina, ovaries, bladder/vesico-ureteric junction, proximal urethra), pubic symphysis |
| Central | Rectum/neo-rectum (intra/extra-luminal), perirectal fat or mesorectal recurrence |
| Posterior | Coccyx, pre-sacral fascia, retro-sacral space, sacrum, sciatic nerve, sciatic notch, S1 and S2 nerve roots |
| Lateral | Internal and external iliac vessels, lateral pelvic lymph nodes, piriformis muscle, internal obturator muscle |
| Infralevator | Levator ani muscles, external sphincter complex, ischio-anal fossa |
| Anterior Urogenital triangle | Perineal body/perineal scar (if previous abdomino-perineal resection of rectum), vaginal introitus, distal urethra, crus penis |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rokan, Z.; Simillis, C.; Kontovounisios, C.; Moran, B.; Tekkis, P.; Brown, G. Locally Recurrent Rectal Cancer According to a Standardized MRI Classification System: A Systematic Review of the Literature. J. Clin. Med. 2022, 11, 3511. https://doi.org/10.3390/jcm11123511
Rokan Z, Simillis C, Kontovounisios C, Moran B, Tekkis P, Brown G. Locally Recurrent Rectal Cancer According to a Standardized MRI Classification System: A Systematic Review of the Literature. Journal of Clinical Medicine. 2022; 11(12):3511. https://doi.org/10.3390/jcm11123511
Chicago/Turabian StyleRokan, Zena, Constantinos Simillis, Christos Kontovounisios, Brendan Moran, Paris Tekkis, and Gina Brown. 2022. "Locally Recurrent Rectal Cancer According to a Standardized MRI Classification System: A Systematic Review of the Literature" Journal of Clinical Medicine 11, no. 12: 3511. https://doi.org/10.3390/jcm11123511
APA StyleRokan, Z., Simillis, C., Kontovounisios, C., Moran, B., Tekkis, P., & Brown, G. (2022). Locally Recurrent Rectal Cancer According to a Standardized MRI Classification System: A Systematic Review of the Literature. Journal of Clinical Medicine, 11(12), 3511. https://doi.org/10.3390/jcm11123511







