Blood Transfusion Reactions—A Comprehensive Review of the Literature including a Swiss Perspective
Abstract
1. Introduction
2. Review Design and Methods
3. Transfusion Reactions
3.1. Epidemiology
3.2. Definition of Blood Transfusion Reactions and Initial Management
| Region | Transfused Blood Products in 2020 | Reported TR | Imputability | Severity Grade 2–3 | Deaths | Mortality (Death/Transfused Blood Product) | Reporting Rate | Death/Reported TR |
|---|---|---|---|---|---|---|---|---|
| CH | 275,343 | 2032 | 1910 1 | 1486 | 3 | 0.001% | 0.74% | 0.14% |
| F | 2,806,774 | 9060 | 7062 2 | 610 | 5 | 0.0002% | 0.32% | 0.06% |
| D | 4,400,164 | 921 | 621 3 | n/a | 7 | 0.0002% | 0.02% | 0.76% |
| UK | 2,074,517 | 4063 | 2881 4 | n/a | 39 | 0.002% | 0.2% | 0.95% |
3.3. Acute Hemolytic Transfusion Reaction
3.4. Febrile Non-Hemolytic Transfusion Reaction (FNHTR)
3.5. Anaphylactic Transfusion Reaction (ATR) and Minor Allergic Transfusion Reaction
3.6. Lung Transfusion Complications
3.7. Transfusion-Related Acute Lung Injury (TRALI)
3.8. Transfusion-Associated Circulatory Overload (TACO)
3.9. Massive Transfusion-Associated Complications
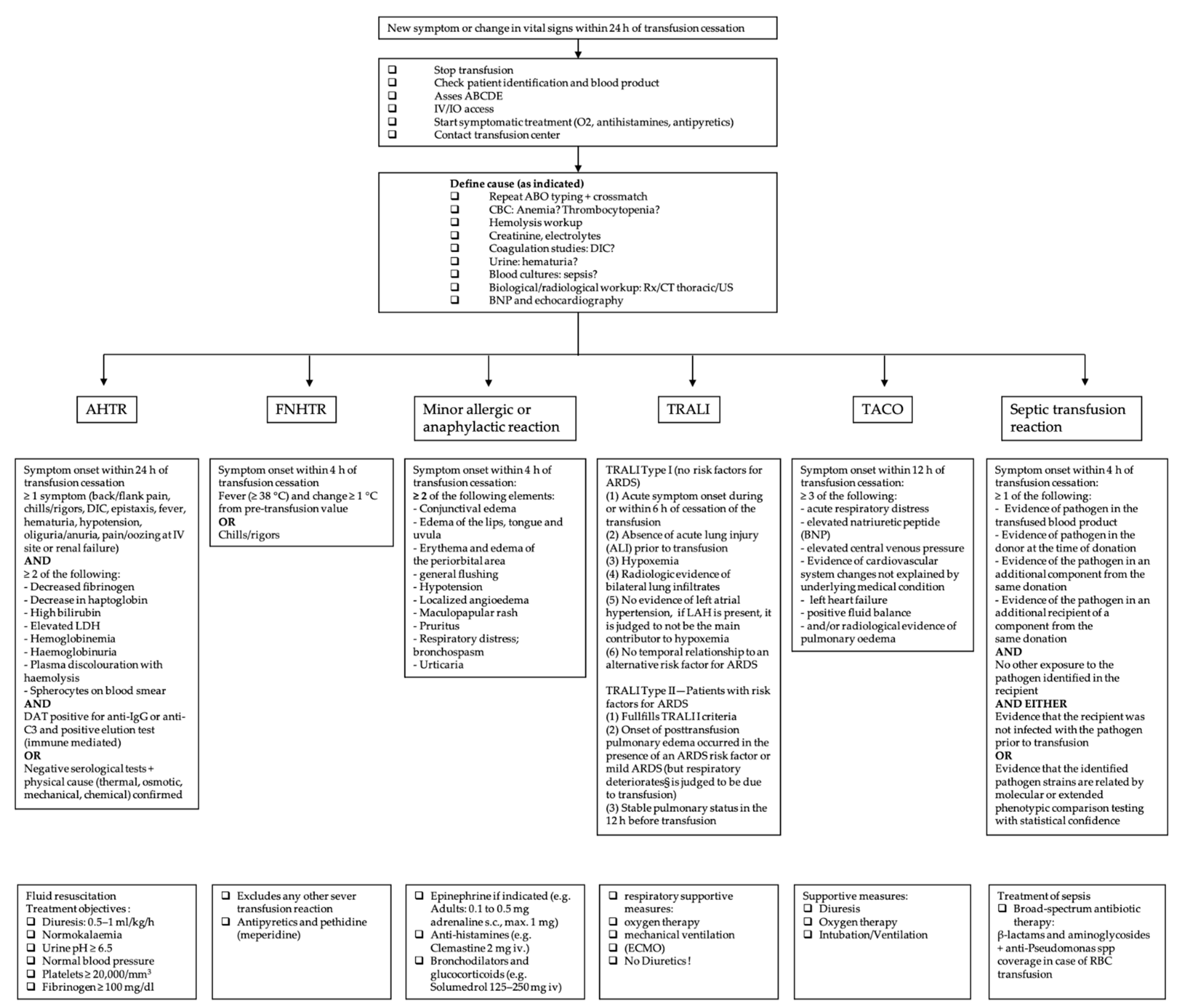

3.10. Septic Transfusion Reaction
3.11. Adverse Transfusion Reactions and COVID-19
3.12. Rare Transfusion Reactions
Acute Pain Transfusion Reaction
3.13. Prevention of Transfusion Reactions
3.14. Hemovigilance Reporting
3.15. Blood Transfusion Quality
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Swissmedic. Analyse des Annonces D’hémovigilance. 2020. Available online: https://www.swissmedic.ch/swissmedic/fr/home/humanarzneimittel/marktueberwachung/haemovigilance/haemovigilance-publications-events/haemovigilance-report-2020.html (accessed on 31 January 2022).
- Swissmedic 2019 © Copyright. The Swiss Haemovigilance Reporting System—Fundamentals. Swissmedic-Analyse des Annonces D’hémovigilance. 2018. Available online: https://www.swissmedic.ch/swissmedic/de/home/humanarzneimittel/marktueberwachung/haemovigilance/haemovigilance-publications-events.html (accessed on 2 May 2022).
- Swissmedic 2019 © Copyright. The Swiss Haemovigilance Reporting System—Fundamentals. Swissmedic-Analyse des Annonces D’hémovigilance. 2017. Available online: https://www.swissmedic.ch/swissmedic/de/home/humanarzneimittel/marktueberwachung/haemovigilance/haemovigilance-publications-events.html (accessed on 2 May 2022).
- L’Agence Nationale de Sécurité du Médicament et des Produits de Santé-18eme Rapport National D’hémovigilance. December 2021. Available online: https://ansm.sante.fr/uploads/2021/12/08/20211208-rapport-hemovigilance-2020-vf.pdf (accessed on 23 March 2022).
- Funk, M.B.; Heiden, M.; Muller, S. Hämovigilanz-Bericht des Paul-Ehrlich-Instituts 2020: Auswertung der Meldungen von Reaktionen und Zwischenfällen nach § 63i AMG. 2021. Available online: www.pei.de/haemovigilanzbericht (accessed on 26 March 2022).
- Annual SHOT Report 2020. Available online: https://www.shotuk.org/wp-content/uploads/myimages/Interactive_SHOT-REPORT-2020_V2.1.pdf (accessed on 26 March 2022).
- Annual SHOT Report 2020—Supplementary information Chapter 2: Participation in UK Haemovigilance. Available online: https://www.shotuk.org/wp-content/uploads/myimages/Participation-Data-Supplementary-material-2020.pdf (accessed on 27 March 2022).
- National Healthcare Safety Network Biovigilance Component Hemovigilance Module Surveillance Protocol. Available online: https://www.cdc.gov/nhsn/pdfs/biovigilance/bv-hv-protocol-current.pdf (accessed on 11 February 2022).
- Rogers, M.A.M.; Rohde, J.M.; Blumberg, N. Haemovigilance of reactions associated with red blood cell transfusion: Comparison across 17 Countries. Vox Sang. 2016, 110, 266–277. [Google Scholar] [CrossRef] [PubMed]
- Elliott, M.; Coventry, A. Critical care: The eight vital signs of patient monitoring. Br. J. Nurs. 2012, 21, 621–625. [Google Scholar] [CrossRef] [PubMed]
- Hoffbrand, A.V.; Higgs, D.R.; Keeling, D.M.; Mehta, A.B. Postgraduate Haematology, 7th ed.; John Wiley & Sons: Hoboken, NJ, USA, 2011; pp. 214–246. [Google Scholar]
- Delaney, M.; Wendel, S.; Bercovitz, R.S.; Cid, J.; Cohn, C.; Dunbar, N.M.; Apelseth, T.O.; Popovsky, M.; Stanworth, S.J.; Tinmouth, A.; et al. Transfusion reactions: Prevention, diagnosis, and treatment. Lancet 2016, 388, 2825–2836. [Google Scholar] [CrossRef]
- Strobel, E. Hemolytic Transfusion Reactions. Transfus. Med. Hemother. 2008, 35, 346–353. [Google Scholar] [CrossRef] [PubMed]
- Panch, S.R.; Montemayor-Garcia, C.; Klein, H.G. Hemolytic Transfusion Reactions. N. Engl. J. Med. 2019, 381, 150–162. [Google Scholar] [CrossRef] [PubMed]
- Arthur, C.M.; Chonat, S.; Fasano, R.; Yee, M.; Josephson, C.D.; Roback, J.D.; Stowell, S.R. Examining the Role of Complement in Predicting, Preventing, and Treating Hemolytic Transfusion Reactions. Transfus. Med. Rev. 2019, 33, 217–224. [Google Scholar] [CrossRef] [PubMed]
- Fiorellino, J.; Elahie, A.L.; Warkentin, T.E. Acute haemolysis, DIC and renal failure after transfusion of uncross-matched blood during trauma resuscitation: Illustrative case and literature review. Transfus. Med. 2018, 28, 319–325. [Google Scholar] [CrossRef]
- Namikawa, A.; Shibuya, Y.; Ouchi, H.; Takahashi, H.; Furuto, Y. A case of ABO-incompatible blood transfusion treated by plasma exchange therapy and continuous hemodiafiltration. CEN Case Rep. 2018, 7, 114–120. [Google Scholar] [CrossRef]
- Deveci, B.; Saba, R.; Altunay, H.; Toptas, T.; Kublashvilli, G.; Karadogan, I. Severe Acute Hemolytic Transfusion Reaction Treated with Ruxolitinib and Plasma Exchange. Transfus. Med. Hemother. 2021, 48, 250–253. [Google Scholar] [CrossRef]
- Weinstock, C.; Möhle, R.; Dorn, C.; Weisel, K.; Höchsmann, B.; Schrezenmeier, H.; Kanz, L. Successful use of eculizumab for treatment of an acute hemolytic reaction after ABO-incompatible red blood cell transfusion. Transfusion 2015, 55, 605–610. [Google Scholar] [CrossRef]
- Goel, R.; Tobian, A.A.R.; Shaz, B.H. Noninfectious transfusion-associated adverse events and their mitigation strategies. Blood 2019, 133, 1831–1839. [Google Scholar] [CrossRef] [PubMed]
- Addas-Carvalho, M.; Salles, T.S.I.; Saad, S.T.O. The association of cytokine gene polymorphisms with febrile non-hemolytic transfusion reaction in multitransfused patients. Transfus. Med. 2006, 16, 184–191. [Google Scholar] [CrossRef] [PubMed]
- Friedlander, M.; Noble, W.H. Meperidine to control shivering associated with platelet transfusion reaction. Can. J. Anaesth. 1989, 36, 460–462. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Kellerman, R.; Rakel, D. Blood Component Therapy and Transfusion Reactions. In Conn’s Current Therapy, 1st ed.; Kellerman, R., Ed.; Elsevier Health Sciences: Amsterdam, The Netherlands, 2021; pp. 402–409. [Google Scholar]
- Winqvist, I. Meperidine (pethidine) to control shaking chills and fever associated with non-hemolytic transfusion reactions. Eur. J. Haematol. 1991, 47, 154–155. [Google Scholar] [CrossRef]
- Ning, S.; Solh, Z.; Arnold, D.M.; Morin, P.A. Premedication for the prevention of nonhemolytic transfusion reactions: A systematic review and meta-analysis. Transfusion 2019, 59, 3609–3616. [Google Scholar] [CrossRef]
- Ibojie, J.; Greiss, M.A.; Urbaniak, S.J. Limited efficacy of universal leucodepletion in reducing the incidence of febrile nonhaemolytic reactions in red cell transfusions. Transfus. Med. 2002, 12, 181–185. [Google Scholar] [CrossRef]
- Patterson, B.J.; Freedman, J.; Blanchette, V.; Sher, G.; Pinkerton, P.; Hannach, B.; Meharchand, J.; Lau, W.; Boyce, N.; Pinchefsky, E.; et al. Effect of premedication guidelines and leukoreduction on the rate of febrile nonhaemolytic platelet transfusion reactions. Transfus. Med. 2000, 10, 199–206. [Google Scholar] [CrossRef]
- Hirayama, F. Current understanding of allergic transfusion reactions: Incidence, pathogenesis, laboratory tests, prevention and treatment. Br. J. Haematol. 2013, 160, 434–444. [Google Scholar] [CrossRef]
- Food and Drug Administration. Fatalities Reported to FDA Following Blood Collection and Transfusion Annual Summary for FY2019. Available online: https://www.fda.gov/media/147628/download (accessed on 28 March 2022).
- Kleinman, S.; Caulfield, T.; Chan, P.; Davenport, R.; McFarland, J.; McPhedran, S.; Meade, M.; Morrison, D.; Pinsent, T.; Robillard, P.; et al. Toward an understanding of transfusion-related acute lung injury: Statement of a consensus panel. Transfusion 2004, 44, 1774–1789. [Google Scholar] [CrossRef]
- Vlaar, A.P.J.; Toy, P.; Fung, M.; Looney, M.R.; Juffermans, N.P.; Bux, J.; Bolton-Maggs, P.; Peters, A.L.; Silliman, C.C.; Kor, D.J.; et al. A consensus redefinition of transfusion-related acute lung injury. Transfusion 2019, 59, 2465–2476. [Google Scholar] [CrossRef]
- Van den Akker, T.A.; Grimes, Z.M.; Friedman, M.T. Transfusion-Associated Circulatory Overload and Transfusion-Related Acute Lung Injury. Am. J. Clin. Pathol. 2021, 156, 529–539. [Google Scholar] [CrossRef] [PubMed]
- Looney, M.R.; Matthay, M.A. Animal models of transfusion-related acute lung injury. Crit. Care Med. 2006, 34 (Suppl. S5), S132–S136. [Google Scholar] [CrossRef] [PubMed]
- Kuldanek, S.A.; Kelher, M.; Silliman, C.C. Risk factors, management and prevention of transfusion-related acute lung injury: A comprehensive update. Expert Rev. Hematol. 2019, 12, 773–785. [Google Scholar] [CrossRef] [PubMed]
- Yang, Z.G.; Lei, X.L.; Li, X.L. Early application of low-dose glucocorticoid improves acute respiratory distress syndrome: A meta-analysis of randomized controlled trials. Exp. Ther. Med. 2017, 13, 1215–1224. [Google Scholar] [CrossRef]
- Goldberg, A.D.; Kor, D.J. State of the art management of transfusion-related acute lung injury (TRALI). Curr. Pharm. Des. 2012, 18, 3273–3284. [Google Scholar] [CrossRef]
- Wallis, J.P.; Lubenko, A. Wells AW, Chapman CE. Single hospital experience of TRALI. Transfusion 2003, 43, 1053–1059. [Google Scholar] [CrossRef]
- Djalali, A.G.; Moore, K.A.; Kelly, E. Report of a patient with severe transfusion-related acute lung injury after multiple transfusions, resuscitated with albumin. Resuscitation 2005, 66, 225–230. [Google Scholar] [CrossRef]
- Caudrillier, A.; Looney, M.R. Platelet-neutrophil interactions as a target for prevention and treatment of transfusion-related acute lung injury. Curr. Pharm. Des. 2012, 18, 3260–3266. [Google Scholar] [CrossRef]
- Müller, M.C.A.; Stroo, I.; Wouters, D.; Zeerleder, S.S.; Roelofs, J.J.T.H.; Boon, L.; Vroom, M.B.; Juffermans, N.P. The effect of C1-inhibitor in a murine model of transfusion-related acute lung injury. Vox Sang. 2014, 107, 71–75. [Google Scholar] [CrossRef]
- Semple, J.W.; Rebetz, J.; Kapur, R. Transfusion-associated circulatory overload and transfusion-related acute lung injury. Blood 2019, 133, 1840–1853. [Google Scholar] [CrossRef]
- Semple, J.W.; McVey, M.J.; Kim, M.; Rebetz, J.; Kuebler, W.M.; Kapur, R. Targeting Transfusion-Related Acute Lung Injury: The Journey From Basic Science to Novel Therapies. Crit. Care Med. 2018, 46, e452–e458. [Google Scholar] [CrossRef] [PubMed]
- Transfusion-Associated Circulatory Overload (TACO) Definition. 2018. Available online: https://www.aabb.org/docs/default-source/default-document-library/resources/taco-2018-definition.pdf?sfvrsn=e1bcfce4_0 (accessed on 28 March 2022).
- Parmar, N.; Pendergrast, J.; Lieberman, L.; Lin, Y.; Callum, J.; Cserti-Gazdewich, C. The association of fever with transfusion-associated circulatory overload. Vox Sang. 2017, 112, 70–78. [Google Scholar] [CrossRef] [PubMed]
- Blumberg, N.; Heal, J.M.; Gettings, K.F.; Phipps, R.P.; Masel, D.; Refaai, M.A.; Kirkley, S.A.; Fialkow, L.B. An association between decreased cardiopulmonary complications (transfusion-related acute lung injury and transfusion-associated circulatory overload) and implementation of universal leukoreduction of blood transfusions. Transfusion 2010, 50, 2738–2744. [Google Scholar] [CrossRef] [PubMed]
- Roubinian, N.; Murphy, E.L. Adjusting the Focus on Transfusion-associated Circulatory Overload. Anesthesiology 2017, 126, 363–365. [Google Scholar] [CrossRef]
- Clifford, L.; Jia, Q.; Subramanian, A.; Yadav, H.; Schroeder, D.R.; Kor, D.J. Risk Factors and Clinical Outcomes Associated with Perioperative Transfusion-associated Circulatory Overload. Anesthesiology 2017, 126, 409–418. [Google Scholar] [CrossRef]
- Patil, V.; Shetmahajan, M. Massive transfusion and massive transfusion protocol. Indian J. Anaesth. 2014, 58, 590–595. [Google Scholar] [CrossRef]
- Guerado, E.; Medina, A.; Mata, M.I.; Galvan, J.M.; Bertrand, M.L. Protocols for massive blood transfusion: When and why, and potential complications. Eur. J. Trauma Emerg. Surg. 2016, 42, 283–295. [Google Scholar] [CrossRef]
- Yücel, N.; Lefering, R.; Maegele, M.; Vorweg, M.; Tjardes, T.; Ruchholtz, S.; Neugebauer, E.A.; Wappler, F.; Bouillon, B.; Rixen, D.; et al. Trauma Associated Severe Hemorrhage (TASH)-Score: Probability of mass transfusion as surrogate for life threatening hemorrhage after multiple trauma. J. Trauma 2006, 60, 1228–1237. [Google Scholar] [CrossRef]
- Rainer, T.H.; Ho, A.M.-H.; Yeung, J.H.H.; Cheung, N.K.; Wong, R.S.M.; Tang, N.; Ng, S.K.; Wong, G.K.; Lai, P.B.; Graham, C.A. Early risk stratification of patients with major trauma requiring massive blood transfusion. Resuscitation 2011, 82, 724–729. [Google Scholar] [CrossRef]
- Shih, A.W.; Al Khan, S.; Wang, A.Y.-H.; Dawe, P.; Young, P.Y.; Greene, A.; Hudoba, M.; Vu, E. Systematic reviews of scores and predictors to trigger activation of massive transfusion protocols. J. Trauma Acute Care Surg. 2019, 87, 717–729. [Google Scholar] [CrossRef]
- Camazine, M.N.; Hemmila, M.R.; Leonard, J.C.; Jacobs, R.A.; Horst, J.A.; Kozar, R.A.; Bochicchio, G.V.; Nathens, A.B.; Cryer, H.M.; Spinella, P.C. Massive transfusion policies at trauma centers participating in the American College of Surgeons Trauma Quality Improvement Program. J. Trauma Acute Care Surg. 2015, 78 (Suppl. S1), S48–S53. [Google Scholar] [CrossRef] [PubMed]
- Bawazeer, M.; Ahmed, N.; Izadi, H.; McFarlan, A.; Nathens, A.; Pavenski, K. Compliance with a massive transfusion protocol (MTP) impacts patient outcome. Injury 2015, 46, 21–28. [Google Scholar] [CrossRef] [PubMed]
- Cotton, B.A.; Au, B.K.; Nunez, T.C.; Gunter, O.L.; Robertson, A.M.; Young, P.P. Predefined massive transfusion protocols are associated with a reduction in organ failure and postinjury complications. J. Trauma 2009, 66, 41–49. [Google Scholar] [CrossRef] [PubMed]
- Johnson, J.W.; Gracias, V.H.; Schwab, C.W.; Reilly, P.M.; Kauder, D.R.; Shapiro, M.B.; Dabrowski, G.P.; Rotondo, M.F. Evolution in damage control for exsanguinating penetrating abdominal injury. J. Trauma 2001, 51, 261–271. [Google Scholar] [CrossRef]
- Smith, H.M.; Farrow, S.J.; Ackerman, J.D.; Stubbs, J.R.; Sprung, J. Cardiac arrests associated with hyperkalemia during red blood cell transfusion: A case series. Anesth. Analg. 2008, 106, 1062–1069. [Google Scholar] [CrossRef]
- Dunne, J.R.; Malone, D.; Tracy, J.K.; Gannon, C.; Napolitano, L.M. Perioperative anemia: An independent risk factor for infection, mortality, and resource utilization in surgery. J. Surg. Res. 2002, 102, 237–244. [Google Scholar] [CrossRef]
- Savioli, G.; Ceresa, I.F.; Caneva, L.; Gerosa, S.; Ricevuti, G. Trauma-Induced Coagulopathy: Overview of an Emerging Medical Problem from Pathophysiology to Outcomes. Medicines 2021, 8, 16. [Google Scholar] [CrossRef]
- Simmons, J.W.; Powell, M.F. Acute traumatic coagulopathy: Pathophysiology and resuscitation. Br. J. Anaesth. 2016, 117 (Suppl. S3), iii31–iii43. [Google Scholar] [CrossRef]
- Brecher, M.E.; Hay, S.N. Bacterial contamination of blood components. Clin. Microbiol. Rev. 2005, 18, 195–204. [Google Scholar] [CrossRef]
- Perez, P.; Salmi, L.R.; Folléa, G.; Schmit, J.L.; de Barbeyrac, B.; Sudre, P.; Salamon, R.; BACTHEM Group, French Haemovigilance Network. Determinants of transfusion-associated bacterial contamination: Results of the French BACTHEM Case-Control Study. Transfusion 2001, 41, 862–872. [Google Scholar] [CrossRef]
- Williamson, L.M.; Lowe, S.; Love, E.M.; Cohen, H.; Soldan, K.; McClelland, D.B.; Skacel, P.; Barbara, J.A. Serious hazards of transfusion (SHOT) initiative: Analysis of the first two annual reports. BMJ 1999, 319, 16–19. [Google Scholar] [CrossRef] [PubMed]
- Levy, J.H.; Neal, M.D.; Herman, J.H. Bacterial contamination of platelets for transfusion: Strategies for prevention. Crit. Care 2018, 22, 271. [Google Scholar] [CrossRef] [PubMed]
- Martin, I.W.; Cohn, C.S.; Delaney, M.; Fontaine, M.J.; Shih, A.W.; Dunbar, N.M.; SCARED Study Investigators on behalf of the Biomedical Excellence for Safer Transfusion (BEST) Collaborative. Limitations of current practices in detection of bacterially contaminated blood products associated with suspected septic transfusion reactions. Transfusion 2021, 61, 2414–2420. [Google Scholar] [CrossRef] [PubMed]
- L’Agence Nationale de Sécurité du Médicament et des Produits de Santé-17eme Rapport National D’hémovigilance. July 2020. Available online: https://ansm.sante.fr/actualites/lansm-publie-le-rapport-dactivite-hemovigilance-2019 (accessed on 11 February 2022).
- Eder, A.F.; Goldman, M. How do I investigate septic transfusion reactions and blood donors with culture-positive platelet donations? Transfusion 2011, 51, 1662–1668. [Google Scholar] [CrossRef] [PubMed]
- Ramirez-Arcos, S. Bacterial contamination. In Tranfusion Reactions, 4th ed.; Popovsky, M.D., Mark, A., Eds.; AABB (Association for the Advancement of Blood & Biotherapies): Bethesda, MD, USA, 2012. [Google Scholar]
- Haass, K.A.; Sapiano, M.R.P.; Savinkina, A.; Kuehnert, M.J.; Basavaraju, S.V. Transfusion-transmitted Infections reported to the National Healthcare Safety Network Hemovigilance Module. Transfus. Med. Rev. 2019, 33, 84–91. [Google Scholar] [CrossRef]
- Heroes, A.-S.; Ndalingosu, N.; Kalema, J.; Luyindula, A.; Kashitu, D.; Akele, C.; Kabinda, J.; Lagrou, K.; Vandekerckhove, P.; Jacobs, J.; et al. Bacterial contamination of blood products for transfusion in the Democratic Republic of the Congo: Temperature monitoring, qualitative and semi-quantitative culture. Blood Transfus. 2020, 18, 348–358. [Google Scholar] [CrossRef]
- Kuehnert, M.J.; Roth, V.R.; Haley, N.R.; Gregory, K.R.; Elder, K.V.; Schreiber, G.B.; Arduino, M.J.; Holt, S.C.; Carson, L.A.; Banerjee, S.N.; et al. Transfusion-transmitted bacterial infection in the United States, 1998 through 2000. Transfusion 2001, 41, 1493–1499. [Google Scholar] [CrossRef]
- DeSimone, R.A.; Costa, V.A.; Kane, K.; Sepulveda, J.L.; Ellsworth, G.B.; Gulick, R.M.; Zucker, J.; Sobieszcyk, M.E.; Schwartz, J.; Cushing, M.M. Blood component utilization in COVID-19 patients in New York City: Transfusions do not follow the curve. Transfusion 2021, 61, 692–698. [Google Scholar] [CrossRef]
- Grandone, E.; Mastroianno, M.; di Mauro, L.; Caroli, A.; Tiscia, G.; Ostuni, A. Blood supply, transfusion demand and mortality in Italian patients hospitalised during nine months of COVID-19 pandemic. Blood Transfus. 2021. Online ahead of print. [Google Scholar] [CrossRef]
- Perelman, S.I.; Shander, A.; Mabry, C.; Ferraris, V.A. Preoperative anemia management in the coronavirus disease (COVID-19) era. Jtcvs Open 2021, 5, 85–94. [Google Scholar] [CrossRef]
- Nguyen, F.T.; van den Akker, T.; Lally, K.; Lam, H.; Lenskaya, V.; Liu, S.T.H.; Bouvier, N.M.; Aberg, J.A.; Rodriguez, D.; Krammer, F.; et al. Transfusion reactions associated with COVID-19 convalescent plasma therapy for SARS-CoV-2. Transfusion 2021, 61, 78–93. [Google Scholar] [CrossRef] [PubMed]
- Amrutiya, V.; Patel, R.; Baghal, M.; Patel, B.; Waykole, T.; Patel, H.; Govil, S.; Lo, A. Transfusion-related acute lung injury in a COVID-19-positive convalescent plasma recipient: A case report. J. Int. Med. Res. 2021, 49, 3000605211032814. [Google Scholar] [CrossRef] [PubMed]
- Coronavirus Disease 2019 (COVID-19) and Supply of Substances of Human Origin in the EU/EEA—Second Update. Available online: https://www.ecdc.europa.eu/sites/default/files/documents/Supply-SoHO-COVID-19--second-update-erratum-Feb-2021.pdf (accessed on 29 March 2022).
- Corman, V.M.; Rabenau, H.F.; Adams, O.; Oberle, D.; Funk, M.B.; Keller-Stanislawski, B.; Timm, J.; Drosten, C.; Ciesek, S. SARS-CoV-2 asymptomatic and symptomatic patients and risk for transfusion transmission. Transfusion 2020, 60, 1119–1122. [Google Scholar] [CrossRef] [PubMed]
- Zaid, Y.; Puhm, F.; Allaeys, I.; Naya, A.; Oudghiri, M.; Khalki, L.; Limami, Y.; Zaid, N.; Sadki, K.; Ben El Haj, R.; et al. Platelets Can Associate with SARS-Cov-2 RNA and Are Hyperactivated in COVID-19. Circ. Res. 2020, 127, 1404–1418. [Google Scholar] [CrossRef]
- Zhang, S.; Liu, Y.; Wang, X.; Yang, L.; Li, H.; Wang, Y.; Liu, M.; Zhao, X.; Xie, Y.; Yang, Y.; et al. SARS-CoV-2 binds platelet ACE2 to enhance thrombosis in COVID-19. J. Hematol. Oncol. 2020, 13, 120. [Google Scholar] [CrossRef]
- Manne, B.K.; Denorme, F.; Middleton, E.A.; Portier, I.; Rowley, J.W.; Stubben, C.; Petrey, A.C.; Tolley, N.D.; Guo, L.; Cody, M.; et al. Platelet gene expression and function in patients with COVID-19. Blood 2020, 136, 1317–1329. [Google Scholar] [CrossRef]
- Koupenova, M.; Corkrey, H.A.; Vitseva, O.; Tanriverdi, K.; Somasundaran, M.; Liu, P.; Soofi, S.; Bhandari, R.; Godwin, M.; Parsi, K.M.; et al. SARS-CoV-2 Initiates Programmed Cell Death in Platelets. Circ. Res. 2021, 129, 631–646. [Google Scholar] [CrossRef]
- Essa, M.F.; Elbashir, E.; Batarfi, K.; Alharbi, M. Lack of transmission of SARS-CoV-2 by platelet transfusion from a COVID-19-positive donor in a hematopoietic stem cell transplantation patient. Pediatr. Blood Cancer 2021, 68, e28658. [Google Scholar] [CrossRef]
- Young, B.E.; Ong, S.W.X.; Kalimuddin, S.; Low, J.G.; Tan, S.Y.; Loh, J.; Ng, O.-T.; Marimuthu, K.; Ang, L.W.; Mak, T.M.; et al. Epidemiologic Features and Clinical Course of Patients Infected With SARS-CoV-2 in Singapore. JAMA 2020, 323, 1488–1494. [Google Scholar] [CrossRef]
- Al Mahmasani, L.; Hodroj, M.H.; Finianos, A.; Taher, A. COVID-19 pandemic and transfusion medicine: The worldwide challenge and its implications. Ann. Hematol. 2021, 100, 1115–1122. [Google Scholar] [CrossRef]
- Joyner, M.J.; Bruno, K.A.; Klassen, S.A.; Kunze, K.L.; Johnson, P.W.; Lesser, E.R.; Wiggins, C.C.; Senefeld, J.W.; Klompas, A.M.; Hodge, D.O.; et al. Safety Update: COVID-19 Convalescent Plasma in 20,000 Hospitalized Patients. Mayo Clin. Proc. 2020, 95, 1888–1897. [Google Scholar] [CrossRef] [PubMed]
- Duan, K.; Liu, B.; Li, C.; Zhang, H.; Yu, T.; Qu, J.; Zhou, M.; Chen, L.; Meng, S.; Hu, Y.; et al. Effectiveness of convalescent plasma therapy in severe COVID-19 patients. Proc. Natl. Acad. Sci. USA 2020, 117, 9490–9496. [Google Scholar] [CrossRef] [PubMed]
- Quaglietta, A.; Nicolucci, A.; Posata, R.; Frattari, A.; Parruti, G.; Accorsi, P. Impact of Covid-19 epidemic on the activities of a blood centre, transfusion support for infected patients and clinical outcomes. Transfus. Med. 2021, 31, 160–166. [Google Scholar] [CrossRef] [PubMed]
- Barriteau, C.M.; Bochey, P.; Lindholm, P.F.; Hartman, K.; Sumugod, R.; Ramsey, G. Blood transfusion utilization in hospitalized COVID-19 patients. Transfusion 2020, 60, 1919–1923. [Google Scholar] [CrossRef] [PubMed]
- Remakanth, R.; Abhishekh, B. Is it an acute pain transfusion reaction? Asian J. Transfus. Sci. 2021, 15, 97–99. [Google Scholar] [CrossRef]
- Hardwick, J.; Osswald, M.; Walker, D. Acute pain transfusion reaction. Oncol. Nurs. Forum. 2013, 40, 543–545. [Google Scholar] [CrossRef]
- Klein, H.G.; Spahn, D.R.; Carson, J.L. Red blood cell transfusion in clinical practice. Lancet 2007, 370, 415–426. [Google Scholar] [CrossRef]
- Carson, J.L.; Guyatt, G.; Heddle, N.M.; Grossman, B.J.; Cohn, C.S.; Fung, M.K.; Gernsheimer, T.; Holcomb, J.B.; Kaplan, L.J.; Katz, L.M.; et al. Clinical Practice Guidelines From the AABB: Red Blood Cell Transfusion Thresholds and Storage. JAMA 2016, 316, 2025–2035. [Google Scholar] [CrossRef]
- Carson, J.L.; Stanworth, S.J.; Dennis, J.A.; Trivella, M.; Roubinian, N.; Fergusson, D.A.; Triulzi, D.; Dorée, C.; Hébert, P.C. Transfusion thresholds for guiding red blood cell transfusion. Cochrane Database Syst. Rev. 2021, 12, CD002042. [Google Scholar] [CrossRef]
- Carson, J.L.; Terrin, M.L.; Noveck, H.; Sanders, D.W.; Chaitman, B.R.; Rhoads, G.G.; Nemo, G.; Dragert, K.; Beaupre, L.; Hildebrand, K.; et al. Liberal or restrictive transfusion in high-risk patients after hip surgery. N. Engl. J. Med. 2011, 365, 2453–2462. [Google Scholar] [CrossRef]
- Ducrocq, G.; Gonzalez-Juanatey, J.R.; Puymirat, E.; Lemesle, G.; Cachanado, M.; Durand-Zaleski, I.; Arnaiz, J.A.; Martínez-Sellés, M.; Silvain, J.; Ariza-Solé, A.; et al. Effect of a restrictive vs liberal blood transfusion strategy on major cardiovascular events among patients with acute myocardial infarction and anemia: The REALITY Randomized Clinical Trial. JAMA 2021, 325, 552–560. [Google Scholar] [CrossRef] [PubMed]
- Hébert, P.C.; Wells, G.; Blajchman, M.A.; Marshall, J.; Martin, C.; Pagliarello, G.; Tweeddale, M.; Schweitzer, I.; Yetisir, E. A Multicenter, Randomized, Controlled Clinical Trial of Transfusion Requirements in Critical Care. N. Engl. J. Med. 1999, 340, 409–417. [Google Scholar] [CrossRef] [PubMed]
- Lacroix, J.; Hébert, P.C.; Hutchison, J.S.; Hume, H.A.; Tucci, M.; Ducruet, T.; Gauvin, F.; Collet, J.P.; Toledano, B.J.; Robillard, P.; et al. Transfusion strategies for patients in pediatric intensive care units. N. Engl. J. Med. 2007, 356, 1609–1619. [Google Scholar] [CrossRef] [PubMed]
- Villanueva, C.; Colomo, A.; Bosch, A. Transfusion for acute upper gastrointestinal bleeding. N. Engl. J. Med. 2013, 368, 1362–1363. [Google Scholar] [CrossRef]
- Barkun, A.N.; Almadi, M.; Kuipers, E.J.; Laine, L.; Sung, J.; Tse, F.; Leontiadis, G.I.; Abraham, N.S.; Calvet, X.; Chan, F.; et al. Management of nonvariceal upper gastrointestinal bleeding: Guideline recommendations from the international consensus group. Ann. Intern. Med. 2019, 171, 805–822. [Google Scholar] [CrossRef]
- Hajjar, L.A.; Vincent, J.-L.; Galas, F.R.B.G.; Nakamura, R.E.; Silva, C.M.P.; Santos, M.H.; Fukushima, J.; Kalil Filho, R.; Sierra, D.B.; Lopes, N.H.; et al. Transfusion requirements after cardiac surgery: The TRACS randomized controlled trial. JAMA 2010, 304, 1559–1567. [Google Scholar] [CrossRef]
- Mazer, C.D.; Whitlock, R.P.; Fergusson, D.A.; Hall, J.; Belley-Cote, E.; Connolly, K.; Khanykin, B.; Gregory, A.J.; de Médicis, É.; McGuinness, S.; et al. Restrictive or Liberal Red-Cell Transfusion for Cardiac Surgery. N. Engl. J. Med. 2017, 377, 2133–2144. [Google Scholar] [CrossRef]
- Norfolk, D. Handbook of Transfusion Medicine, 5th ed.; Norfolk, D., Ed.; The Stationery Office: London, UK, 2013. [Google Scholar]
- Amended by No I of the FA of 18 March 2016, in Force since 1 Jan. 2019 (AS 2017 2745, 2018 3575; BBl 2013 1), Art. 59 Mandatory Notification, Notification System and the Right to Notify. Available online: https://www.fedlex.admin.ch/eli/cc/2001/422/en#fn-d6e5532 (accessed on 20 March 2022).
- Swissmedic. The Swiss Haemovigilance Reporting System—Fundamentals. 2019. Available online: https://www.swissmedic.ch/swissmedic/en/home/humanarzneimittel/marktueberwachung/haemovigilance/haemovigilance-meldesystem.html (accessed on 25 March 2022).
- Réseau National d’Hémovigilance Déclaration et Gestion des Évènements Indésirables Transfusionnels RNHV—eFIT. Available online: https://e-fit.ansm.sante.fr/rnhv/rnhv/loginAccueil.html (accessed on 26 March 2022).
- Paul-Ehrlich-Institut. Meldeformulare. Available online: https://www.pei.de/DE/arzneimittelsicherheit/haemovigilanz/meldeformulare-online-meldung/meldeformulare-online-meldung-node.html;jsessionid=4355D9493116DCC74983F41E29DB3C58.intranet241 (accessed on 26 March 2022).
- Glynn, S.A. The red blood cell storage lesion: A method to the madness. Transfusion 2010, 50, 1164–1169. [Google Scholar] [CrossRef]
- Storch, E.K.; Custer, B.S.; Jacobs, M.R.; Menitove, J.E.; Mintz, P.D. Review of current transfusion therapy and blood banking practices. Blood Rev. 2019, 38, 100593. [Google Scholar] [CrossRef]
- Noordin, S.S.; Karim, F.A.; Mohammad, W.M.Z.; Bin, W.; Hussein, A.R. Coagulation Factor Activities Changes Over 5 Days in Thawed Fresh Frozen Plasma Stored at Different Initial Storage Temperatures. Indian J. Hematol. Blood Transfus. 2018, 34, 510–516. [Google Scholar] [CrossRef]
- Bassuni, W.Y.; Blajchman, M.A.; Al-Moshary, M.A. Why implement universal leukoreduction? Hematol. Oncol. Stem. Cell 2008, 1, 106–123. [Google Scholar] [CrossRef]
- European Directorate for the Quality of Medicines & HealthCare. Recommendation No. R (95) 15. In Guide to the Preparation, Use and Quality Assurance of Blood Components, 20th ed.; Keitel, S., Ed.; European Directorate for the Quality of Medicines & HealthCare: Strasbourg, France, 2020; pp. 13–453. [Google Scholar]
- Blood Guide. European Directorate for the Quality of Medicines & HealthCare. Available online: https://www.edqm.eu/en/ (accessed on 8 May 2022).
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ackfeld, T.; Schmutz, T.; Guechi, Y.; Le Terrier, C. Blood Transfusion Reactions—A Comprehensive Review of the Literature including a Swiss Perspective. J. Clin. Med. 2022, 11, 2859. https://doi.org/10.3390/jcm11102859
Ackfeld T, Schmutz T, Guechi Y, Le Terrier C. Blood Transfusion Reactions—A Comprehensive Review of the Literature including a Swiss Perspective. Journal of Clinical Medicine. 2022; 11(10):2859. https://doi.org/10.3390/jcm11102859
Chicago/Turabian StyleAckfeld, Theresa, Thomas Schmutz, Youcef Guechi, and Christophe Le Terrier. 2022. "Blood Transfusion Reactions—A Comprehensive Review of the Literature including a Swiss Perspective" Journal of Clinical Medicine 11, no. 10: 2859. https://doi.org/10.3390/jcm11102859
APA StyleAckfeld, T., Schmutz, T., Guechi, Y., & Le Terrier, C. (2022). Blood Transfusion Reactions—A Comprehensive Review of the Literature including a Swiss Perspective. Journal of Clinical Medicine, 11(10), 2859. https://doi.org/10.3390/jcm11102859






