Trends of Stem Cell Therapies in Age-Related Macular Degeneration
Abstract
1. Introduction
2. History of RPE Cell Therapy for Age-Related Macular Degeneration
3. Cell Therapy for Age-Related Macular Degeneration Using Pluripotent Stem Cell-Derived RPE Cells
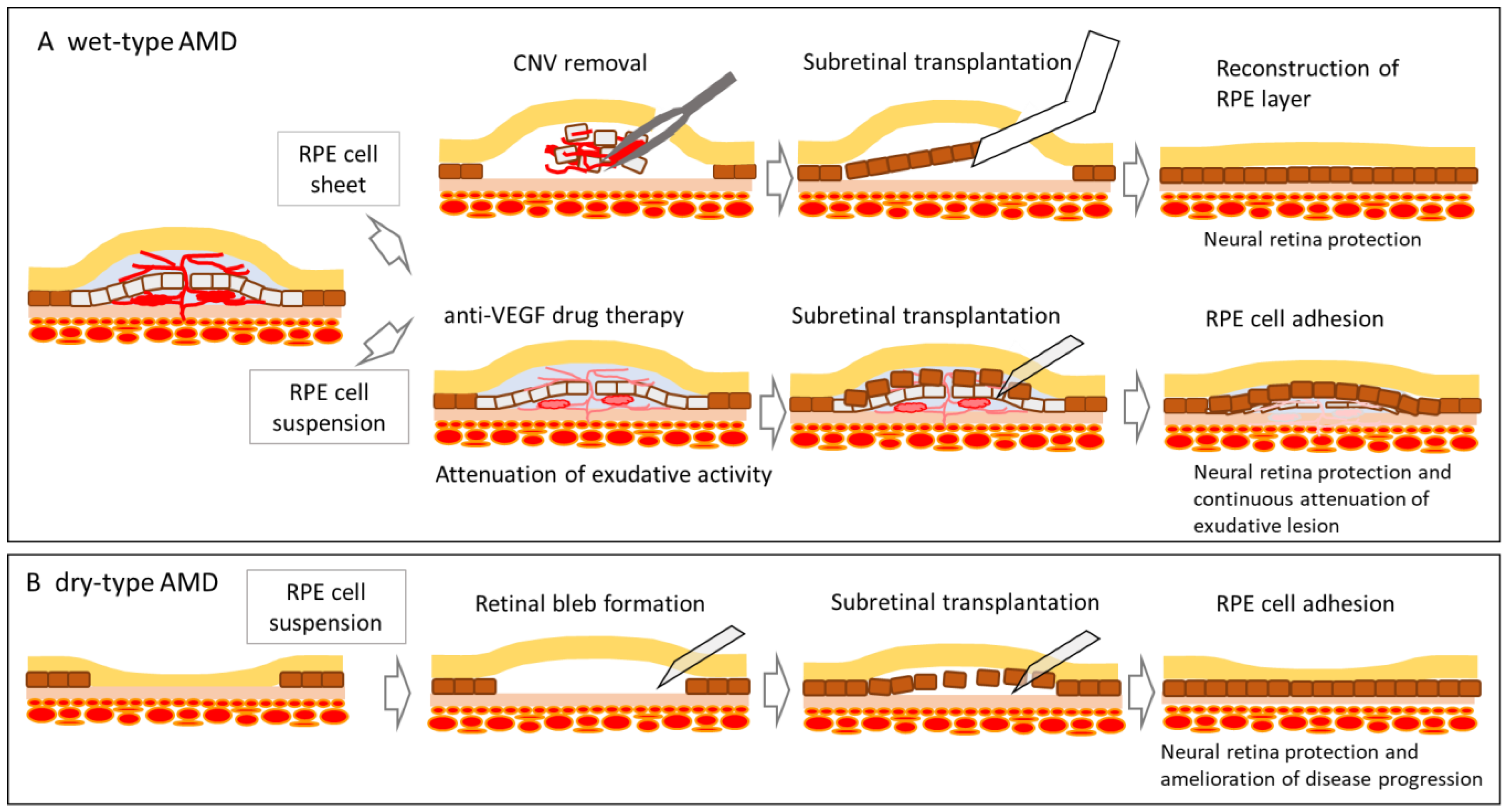
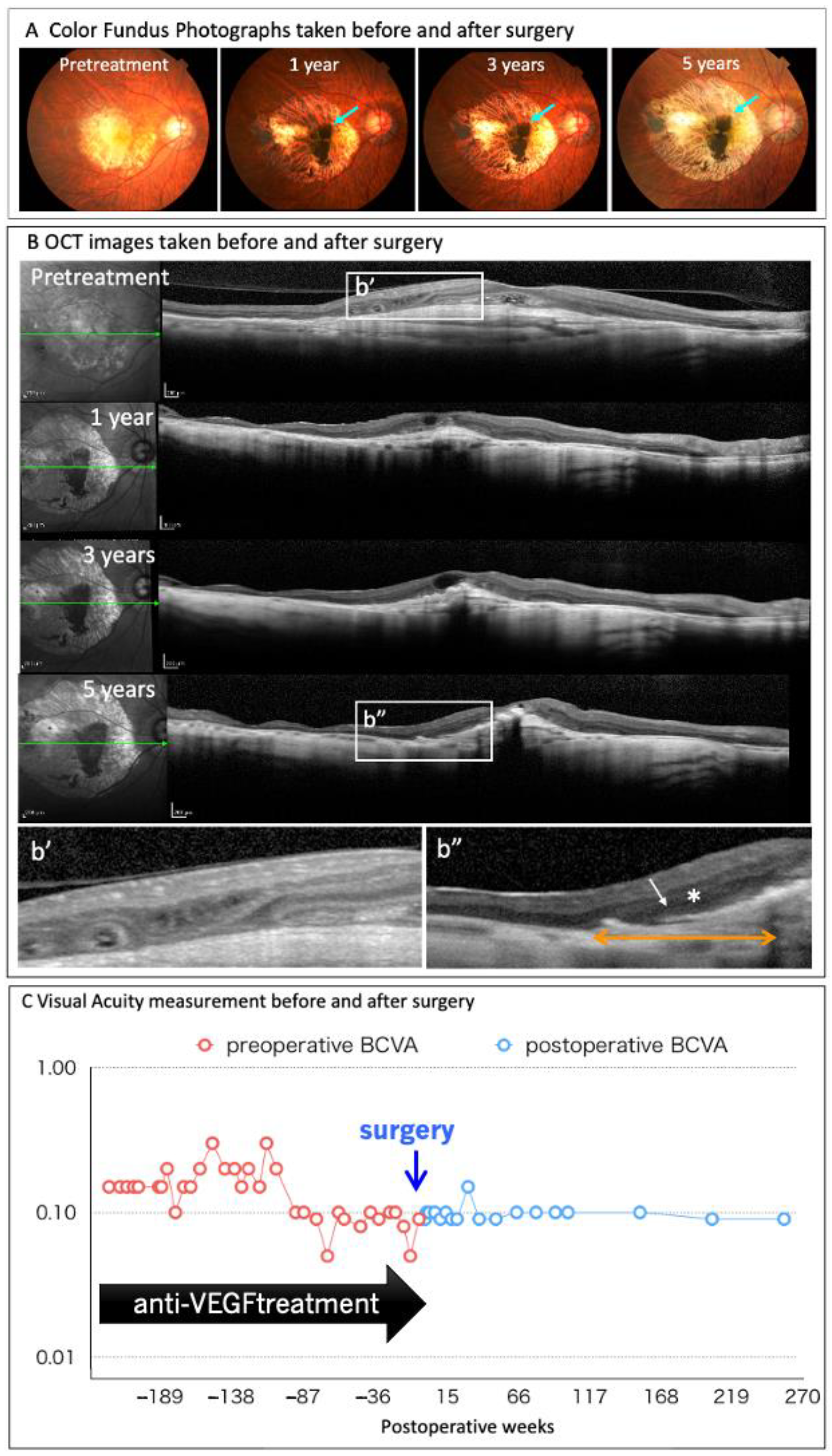
3.1. Autologous iPS Cell-Derived RPE Cell Sheet Transplantation
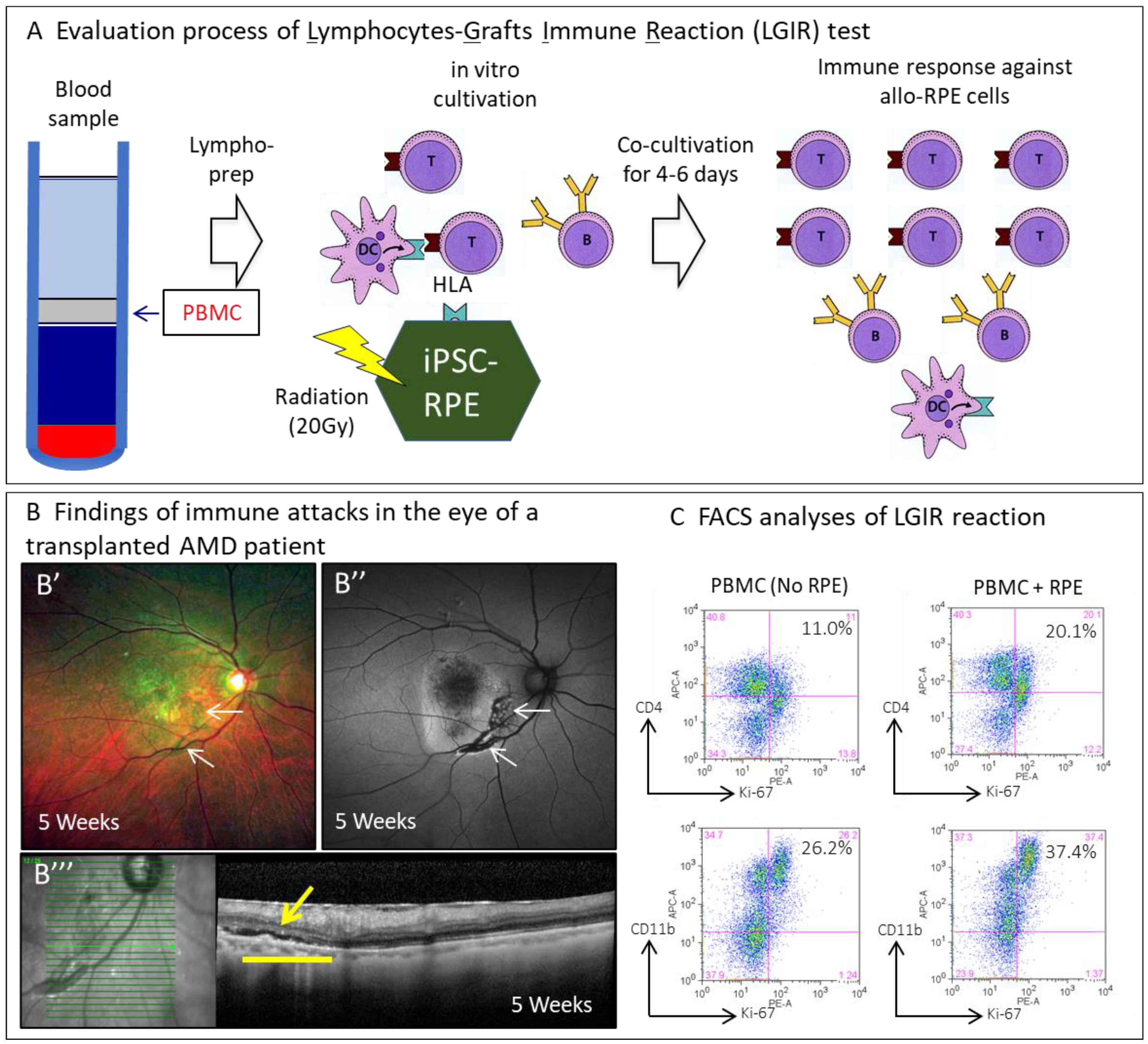
3.2. Allogeneic iPS Cell-Derived RPE Cell Suspension Transplantation
- The raw material was established from an HLA 6-locus homozygote donor manufactured by the Center for iPS Cell Research and Application, Kyoto University.
- A cell suspension that is easy to store and transport and is considered less invasive by transplantation was selected as the dosage form.
- A frozen stock of RPE cells was created as an intermediate. This was then thawed k based on the patient’s date of transplantation. A recovery culture was performed for two weeks, after which the cell suspension was prepared using a dedicated transplant medium.
- At the time of transplantation, removal of the CNV, performed in autologous RPE transplantation was not conducted, and a commercially available ophthalmic cannula (PolyTip® cannula 25 g/38 g, MedOne, FL, USA) was used as the dedicated transplantation device for the cell suspension.
- Immune rejection after transplantation was evaluated using the LGIR and donor-specific antibodies tests to detect the response of the transplanted patients’ peripheral lymphocytes to the transplanted cells in vitro and the presence of RPE-specific antibodies. Optical coherence tomography (OCT) imaging was utilized to detect exudative findings in RPE-transplanted lesions.
3.3. Allogeneic ES Cell-Derived RPE Cell Suspension Transplantation
3.4. Allogeneic ES Cell-Derived RPE Cell Sheet
3.5. Possibility of Pluripotent Stem Cell-Derived RPE Cell/Photoreceptor Complex Product
3.6. Treatment with Somatic Stem Cells
4. Future Prospective in Retinal Regenerative Medicine
4.1. Expanding Indications for RPE Cell Transplantation
4.2. Examination of Appropriate Efficacy Evaluation Method
4.3. Optimization of Cell Resources
4.4. Rise of New Technology for Manufacturing
4.5. Regarding Regulatory Issues Related to Regenerative Medicine
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Wong, W.L.; Su, X.; Li, B.X.; Cheung, C.M.G.; Klein, B.E.; Cheng, C.-Y.; Wong, T.Y. Global prevalence of age-related macular degeneration and disease burden projection for 2020 and 2040: A systematic review and meta-analysis. Lancet Glob. Health 2014, 2, e106–e116. [Google Scholar] [CrossRef]
- Singh, M.S.; Park, S.S.; Albini, T.A.; Canto-Soler, M.V.; Klassen, H.; MacLaren, R.E.; Takahashi, M.; Nagiel, A.; Schwartz, S.D.; Bharti, K. Retinal stem cell transplantation: Balancing safety and potential. Prog. Retin. Eye Res. 2020, 75, 100779. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Tang, Z.; Gu, P. Stem/progenitor cell-based transplantation for retinal degeneration: A review of clinical trials. Cell Death Dis. 2020, 11, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Maeda, A.; Mandai, M.; Takahashi, M. Gene and Induced Pluripotent Stem Cell Therapy for Retinal Diseases. Annu. Rev. Genom. Hum. Genet. 2019, 20, 201–216. [Google Scholar] [CrossRef] [PubMed]
- Scholl, H.P.N.; Strauss, R.W.; Singh, M.S.; Dalkara, D.; Roska, B.; Picaud, S.; Sahel, J.-A. Emerging therapies for inherited retinal degeneration. Sci. Transl. Med. 2016, 8, 368rv6. [Google Scholar] [CrossRef]
- Mandai, M.; Watanabe, A.; Kurimoto, Y.; Hirami, Y.; Morinaga, C.; Daimon, T.; Fujihara, M.; Akimaru, H.; Sakai, N.; Shibata, Y.; et al. Autologous Induced Stem-Cell–Derived Retinal Cells for Macular Degeneration. N. Engl. J. Med. 2017, 376, 1038–1046. [Google Scholar] [CrossRef] [PubMed]
- Da Cruz, L.; Fynes, K.; Georgiadis, O.; Kerby, J.; Luo, Y.H.; Ahmado, A.; Vernon, A.; Daniels, J.T.; Nommiste, B.; Hasan, S.M.; et al. Phase 1 clinical study of an embryonic stem cell–derived retinal pigment epithelium patch in age-related macular degeneration. Nat. Biotechnol. 2018, 36, 328–337. [Google Scholar] [CrossRef]
- Kashani, A.H.; Lebkowski, J.S.; Rahhal, F.M.; Avery, R.L.; Salehi-Had, H.; Dang, W.; Lin, C.-M.; Mitra, D.; Zhu, D.; Thomas, B.B.; et al. A bioengineered retinal pigment epithelial monolayer for advanced, dry age-related macular degeneration. Sci. Transl. Med. 2018, 10, eaao4097. [Google Scholar] [CrossRef] [PubMed]
- Sugita, S.; Mandai, M.; Hirami, Y.; Takagi, S.; Maeda, T.; Fujihara, M.; Matsuzaki, M.; Yamamoto, M.; Iseki, K.; Hayashi, N.; et al. HLA-Matched Allogeneic iPS Cells-Derived RPE Transplantation for Macular Degeneration. J. Clin. Med. 2020, 9, 2217. [Google Scholar] [CrossRef]
- Schwartz, S.D.; Regillo, C.D.; Lam, B.L.; Eliott, D.; Rosenfeld, P.J.; Gregori, N.Z.; Hubschman, J.-P.; Davis, J.L.; Heilwell, G.; Spirn, M.; et al. Human embryonic stem cell-derived retinal pigment epithelium in patients with age-related macular degeneration and Stargardt’s macular dystrophy: Follow-up of two open-label phase 1/2 studies. Lancet 2015, 385, 509–516. [Google Scholar] [CrossRef]
- Schwartz, S.D.; Hubschman, J.-P.; Heilwell, G.; Franco-Cardenas, V.; Pan, C.K.; Ostrick, R.M.; Mickunas, E.; Gay, R.; Klimanskaya, I.; Lanza, R. Embryonic stem cell trials for macular degeneration: A preliminary report. Lancet 2012, 379, 713–720. [Google Scholar] [CrossRef]
- Takagi, S.; Mandai, M.; Gocho, K.; Hirami, Y.; Yamamoto, M.; Fujihara, M.; Sugita, S.; Kurimoto, Y.; Takahashi, M. Evaluation of Transplanted Autologous Induced Pluripotent Stem Cell-Derived Retinal Pigment Epithelium in Exudative Age-Related Macular Degeneration. Ophthalmol. Retin. 2019, 3, 850–859. [Google Scholar] [CrossRef] [PubMed]
- Mehat, M.S.; Sundaram, V.; Ripamonti, C.; Robson, A.G.; Smith, A.J.; Borooah, S.; Robinson, M.; Rosenthal, A.N.; Innes, W.; Weleber, R.G.; et al. Transplantation of Human Embryonic Stem Cell-Derived Retinal Pigment Epithelial Cells in Macular Degeneration. Ophthalmology 2018, 125, 1765–1775. [Google Scholar] [CrossRef]
- Sung, Y.; Lee, M.J.; Choi, J.; Jung, S.Y.; Chong, S.Y.; Sung, J.H.; Shim, S.H.; Song, W.K. Long-term safety and tolerability of subretinal transplantation of embryonic stem cell-derived retinal pigment epithelium in Asian Stargardt disease patients. Br. J. Ophthalmol. 2020, 10, 1136. [Google Scholar] [CrossRef]
- Song, W.K.; Park, K.-M.; Kim, H.-J.; Lee, J.H.; Choi, J.; Chong, S.Y.; Shim, S.H.; Del Priore, L.V.; Lanza, R. Treatment of Macular Degeneration Using Embryonic Stem Cell-Derived Retinal Pigment Epithelium: Preliminary Results in Asian Patients. Stem Cell Rep. 2015, 4, 860–872. [Google Scholar] [CrossRef] [PubMed]
- Gouras, P.; Flood, M.T.; Kjeldbye, H.; Bilek, M.K.; Eggers, H. Transplantation of cultured human retinal epithelium to Bruch’s membrane of the owl monkey’s eye. Curr. Eye Res. 1985, 4, 253–265. [Google Scholar] [CrossRef] [PubMed]
- Peyman, A.G.; Blinder, K.J.; Paris, C.L.; Alturki, W.; Nelson, J.N.C.; Desai, U. A Technique for Retinal Pigment Epithelium Transplantation for Age-Related Macular Degeneration Secondary to Extensive Subfoveal Scarring. Ophthalmic Surg. Lasers Imaging Retin. 1991, 22, 102–108. [Google Scholar] [CrossRef]
- Algvere, P.V.; Berglin, L.; Gouras, P.; Sheng, Y. Transplantation of fetal retinal pigment epithelium in age-related macular degeneration with subfoveal neovascularization. Graefe’s Arch. Clin. Exp. Ophthalmol. 1994, 232, 707–716. [Google Scholar] [CrossRef] [PubMed]
- Algvere, P.V.; Berglin, L.; Gouras, P.; Sheng, Y. Transplantation of RPE in age-related macular degeneration: Observations in disciform lesions and dry RPE atrophy. Graefe’s Arch. Clin. Exp. Ophthalmol. 1997, 235, 149–158. [Google Scholar] [CrossRef]
- Weisz, J.M.; Humayun, M.S.; De Juan, E.; Del Cerro, M.; Sunness, J.S.; Dagnelie, G.; Soylu, M.; Rizzo, L.; Nussenblatt, R.B. Allogenic fetal retinal pigment epithelial cell transplant in a patient with geographic atrophy. Retina 1999, 19, 540–545. [Google Scholar] [CrossRef]
- Del Priore, L.V.; Kaplan, H.J.; Tezel, T.H.; Hayashi, N.; Berger, A.S.; Green, W. Retinal pigment epithelial cell transplantation after subfoveal membranectomy in age-related macular degeneration. Am. J. Ophthalmol. 2001, 131, 472–480. [Google Scholar] [CrossRef]
- Joussen, A.M.; Heussen, F.M.; Joeres, S.; Llacer, H.; Prinz, B.; Rohrschneider, K.; Maaijwee, K.J.; Van Meurs, J.; Kirchhof, B. Autologous Translocation of the Choroid and Retinal Pigment Epithelium in Age-related Macular Degeneration. Am. J. Ophthalmol. 2006, 142, 17–30. [Google Scholar] [CrossRef]
- MacLaren, R.E.; Uppal, G.S.; Balaggan, K.S.; Tufail, A.; Munro, P.M.; Milliken, A.B.; Ali, R.R.; Rubin, G.S.; Aylward, G.W.; Da Cruz, L. Autologous Transplantation of the Retinal Pigment Epithelium and Choroid in the Treatment of Neovascular Age-Related Macular Degeneration. Ophthalmology 2007, 114, 561–570. [Google Scholar] [CrossRef] [PubMed]
- Maaijwee, K.; Heimann, H.; Missotten, T.; Mulder, P.; Joussen, A.; Van Meurs, J. Retinal pigment epithelium and choroid translocation in patients with exudative age-related macular degeneration: Long-term results. Graefe’s Arch. Clin. Exp. Ophthalmol. 2007, 245, 1681–1689. [Google Scholar] [CrossRef] [PubMed]
- Haruta, M.; Sasai, Y.; Kawasaki, H.; Amemiya, K.; Ooto, S.; Kitada, M.; Suemori, H.; Nakatsuji, N.; Ide, C.; Honda, Y.; et al. In vitro and in vivo characterization of pigment epithelial cells differentiated from primate embryonic stem cells. Investig. Opthalmol. Vis. Sci. 2004, 45, 1020–1025. [Google Scholar] [CrossRef] [PubMed]
- Osakada, F.; Ikeda, H.; Sasai, Y.; Takahashi, M. Stepwise differentiation of pluripotent stem cells into retinal cells. Nat. Protoc. 2009, 4, 811–824. [Google Scholar] [CrossRef]
- Hirami, Y.; Osakada, F.; Takahashi, K.; Okita, K.; Yamanaka, S.; Ikeda, H.; Yoshimura, N.; Takahashi, M. Generation of retinal cells from mouse and human induced pluripotent stem cells. Neurosci. Lett. 2009, 458, 126–131. [Google Scholar] [CrossRef] [PubMed]
- Butler, D.; Callaway, E.; Check Hayden, E.; Cyranoski, D.; Hand, E.; Nosengo, N.; Samuel Reich, E.; Tollefson, J.; Yahia, M. 365 days: Nature’s 10. Nat. Cell Biol. 2014, 516, 311–319. [Google Scholar] [CrossRef]
- Sugita, S.; Iwasaki, Y.; Makabe, K.; Kamao, H.; Mandai, M.; Shiina, T.; Ogasawara, K.; Hirami, Y.; Kurimoto, Y.; Takahashi, M. Successful Transplantation of Retinal Pigment Epithelial Cells from MHC Homozygote iPSCs in MHC-Matched Models. Stem Cell Rep. 2016, 7, 635–648. [Google Scholar] [CrossRef]
- Sugita, S.; Iwasaki, Y.; Makabe, K.; Kimura, T.; Futagami, T.; Suegami, S.; Takahashi, M. Lack of T Cell Response to iPSC-Derived Retinal Pigment Epithelial Cells from HLA Homozygous Donors. Stem Cell Rep. 2016, 7, 619–634. [Google Scholar] [CrossRef]
- Fujii, S.; Sugita, S.; Futatsugi, Y.; Ishida, M.; Edo, A.; Makabe, K.; Kamao, H.; Iwasaki, Y.; Sakaguchi, H.; Hirami, Y.; et al. A Strategy for Personalized Treatment of iPS-Retinal Immune Rejections Assessed in Cynomolgus Monkey Models. Int. J. Mol. Sci. 2020, 21, 3077. [Google Scholar] [CrossRef] [PubMed]
- Gandhi, J.K.; Mano, F.; Iezzi, R.; LoBue, S.A.; Holman, B.H.; Fautsch, M.P.; Olsen, T.W.; Pulido, J.S.; Marmorstein, A.D. Fibrin hydrogels are safe, degradable scaffolds for sub-retinal implantation. PLoS ONE 2020, 15, e0227641. [Google Scholar] [CrossRef] [PubMed]
- Gandhi, J.K.; Manzar, Z.; Bachman, L.A.; Andrews-Pfannkoch, C.; Knudsen, T.; Hill, M.; Schmidt, H.; Iezzi, R.; Pulido, J.S.; Marmorstein, A.D. Fibrin hydrogels as a xenofree and rapidly degradable support for transplantation of retinal pigment epithelium monolayers. Acta Biomater. 2018, 67, 134–146. [Google Scholar] [CrossRef] [PubMed]
- Eiraku, M.; Takata, N.; Ishibashi, H.; Kawada, M.; Sakakura, E.; Okuda, S.; Sekiguchi, K.; Adachi, T.; Sasai, Y. Self-organizing optic-cup morphogenesis in three-dimensional culture. Nature 2011, 472, 51–56. [Google Scholar] [CrossRef] [PubMed]
- Shirai, H.; Mandai, M.; Matsushita, K.; Kuwahara, A.; Yonemura, S.; Nakano, T.; Assawachananont, J.; Kimura, T.; Saito, K.; Terasaki, H.; et al. Transplantation of human embryonic stem cell-derived retinal tissue in two primate models of retinal degeneration. Proc. Natl. Acad. Sci. USA 2016, 113, E81–E90. [Google Scholar] [CrossRef] [PubMed]
- Mandai, M.; Fujii, M.; Hashiguchi, T.; Sunagawa, G.A.; Ito, S.-I.; Sun, J.; Kaneko, J.; Sho, J.; Yamada, C.; Takahashi, M. iPSC-Derived Retina Transplants Improve Vision in rd1 End-Stage Retinal-Degeneration Mice. Stem Cell Rep. 2017, 8, 69–83. [Google Scholar] [CrossRef]
- Iraha, S.; Tu, H.-Y.; Yamasaki, S.; Kagawa, T.; Goto, M.; Takahashi, R.; Watanabe, T.; Sugita, S.; Yonemura, S.; Sunagawa, G.A.; et al. Establishment of Immunodeficient Retinal Degeneration Model Mice and Functional Maturation of Human ESC-Derived Retinal Sheets after Transplantation. Stem Cell Rep. 2018, 10, 1059–1074. [Google Scholar] [CrossRef]
- Kuriyan, A.E.; Albini, T.A.; Townsend, J.H.; Rodriguez, M.; Pandya, H.K.; Leonard, R.E.; Parrott, M.B.; Rosenfeld, P.J.; Flynn, H.W.; Goldberg, J.L. Vision Loss after Intravitreal Injection of Autologous “Stem Cells” for AMD. N. Engl. J. Med. 2017, 376, 1047–1053. [Google Scholar] [CrossRef]
- Boulton, M.; Dayhaw-Barker, P. The role of the retinal pigment epithelium: Topographical variation and ageing changes. Eye 2001, 15, 384–389. [Google Scholar] [CrossRef]
- Hicks, D.; Hamel, C.P. The Retinal Pigment Epithelium in Health and Disease. Curr. Mol. Med. 2010, 10, 802–823. [Google Scholar] [CrossRef]
- Palczewski, K. Chemistry and Biology of the Initial Steps in Vision: The Friedenwald Lecture. Investig. Opthalmol. Vis. Sci. 2014, 55, 6651–6672. [Google Scholar] [CrossRef]
- Hageman, G.S. An Integrated Hypothesis That Considers Drusen as Biomarkers of Immune-Mediated Processes at the RPE-Bruch’s Membrane Interface in Aging and Age-Related Macular Degeneration. Prog. Retin. Eye Res. 2001, 20, 705–732. [Google Scholar] [CrossRef]
- Ach, T.; Huisingh, C.; McGwin, G.; Messinger, J.D.; Zhang, T.; Bentley, M.J.; Gutierrez, D.B.; Ablonczy, Z.; Smith, R.T.; Sloan, K.R.; et al. Quantitative Autofluorescence and Cell Density Maps of the Human Retinal Pigment Epithelium. Investig. Opthalmol. Vis. Sci. 2014, 55, 4832–4841. [Google Scholar] [CrossRef]
- Hollander, A.I.D.; Roepman, R.; Koenekoop, R.K.; Cremers, F.P. Leber congenital amaurosis: Genes, proteins and disease mechanisms. Prog. Retin. Eye Res. 2008, 27, 391–419. [Google Scholar] [CrossRef]
- Tanna, P.; Strauss, R.W.; Fujinami, K.; Michaelides, M. Stargardt disease: Clinical features, molecular genetics, animal models and therapeutic options. Br. J. Ophthalmol. 2016, 101, 25–30. [Google Scholar] [CrossRef]
- Nakamura, M.; Skalet, J.; Miyake, Y. RDH5 gene mutations and electroretinogram in fundus albipunctatus with or without macular dystrophy. Doc. Ophthalmol. 2003, 107, 3–11. [Google Scholar] [CrossRef]
- Maguire, A.M.; Russell, S.; Wellman, J.A.; Chung, D.C.; Yu, Z.-F.; Tillman, A.; Wittes, J.; Pappas, J.; Elci, O.; Marshall, K.A.; et al. Efficacy, Safety, and Durability of Voretigene Neparvovec-rzyl in RPE65 Mutation–Associated Inherited Retinal Dystrophy. Ophthalmolpgy 2019, 126, 1273–1285. [Google Scholar] [CrossRef] [PubMed]
- Lukovic, D.; Castro, A.A.; Delgado, A.B.G.; Bernal, M.D.L.A.M.; Pelaez, N.L.; Lloret, A.D.; Espejo, R.P.; Kamenarova, K.; Sánchez, L.F.; Cuenca, N.; et al. Human iPSC derived disease model of MERTK-associated retinitis pigmentosa. Sci. Rep. 2015, 5, 12910. [Google Scholar] [CrossRef]
- Li, M.; Dolz-Marco, R.; Huisingh, C.; Messinger, J.D.; Feist, R.M.; Ferrara, D.; Freund, K.B.; Curcio, C.A. Clinicopathologic correlation of geographic atrophy secondary to age-related macular degeneration. Retina 2019, 39, 802–816. [Google Scholar] [CrossRef] [PubMed]
- Koenekoop, R.K. SuccessfulRPE65Gene Replacement and Improved Visual Function in Humans. Ophthalmic Genet. 2008, 29, 89–91. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Hayashi, T.; Takeuchi, T.; Gekka, T.; Ueoka, Y.; Kitahara, K.; Goto-Omoto, S. Compound heterozygous RDH5 mutations in familial fleck retina with night blindness. Acta Ophthalmol. Scand. 2005, 84, 254–258. [Google Scholar] [CrossRef] [PubMed]
- Young, M.; Chui, L.; Fallah, N.; Or, C.; Merkur, A.B.; Kirker, A.W.; Albiani, D.A.; Forooghian, F. Exacerbation of choroidal and retinal pigment epithelial atrophy after anti–vascular endothelial growth factor treatment in neovascular age-related macular degeneration. Retina 2014, 34, 1308–1315. [Google Scholar] [CrossRef] [PubMed]
- Kuroda, Y.; Yamashiro, K.; Tsujikawa, A.; Ooto, S.; Tamura, H.; Oishi, A.; Nakanishi, H.; Miyake, M.; Yoshikawa, M.; Yoshimura, N. Retinal Pigment Epithelial Atrophy in Neovascular Age-Related Macular Degeneration After Ranibizumab Treatment. Am. J. Ophthalmol. 2016, 161, 94–103.e1. [Google Scholar] [CrossRef] [PubMed]
- Kuroda, Y.; Yamashiro, K.; Miyake, M.; Yoshikawa, M.; Nakanishi, H.; Oishi, A.; Tamura, H.; Ooto, S.; Tsujikawa, A.; Yoshimura, N. Factors Associated with Recurrence of Age-Related Macular Degeneration after Anti-Vascular Endothelial Growth Factor Treatment. Ophthalmology 2015, 122, 2303–2310. [Google Scholar] [CrossRef] [PubMed]
- Daniel, E.; Maguire, M.G.; Grunwald, J.E.; Toth, C.A.; Jaffe, G.J.; Martin, D.F.; Ying, G.-S. For the Comparison of Age-Related Macular Degeneration Treatments Trials Research Group Incidence and Progression of Nongeographic Atrophy in the Comparison of Age-Related Macular Degeneration Treatments Trials (CATT) Clinical Trial. JAMA Ophthalmol. 2020, 138, 510. [Google Scholar] [CrossRef]
- Nagiel, A.; Freund, K.B.; Spaide, R.F.; Munch, I.C.; Larsen, M.; Sarraf, D. Mechanism of Retinal Pigment Epithelium Tear Formation Following Intravitreal Anti–Vascular Endothelial Growth Factor Therapy Revealed by Spectral-Domain Optical Coherence Tomography. Am. J. Ophthalmol. 2013, 156, 981–988. [Google Scholar] [CrossRef] [PubMed]
- Gardiner, K.L.; Cideciyan, A.V.; Swider, M.; Dufour, V.L.; Sumaroka, A.; Komáromy, A.M.; Hauswirth, W.W.; Iwabe, S.; Jacobson, S.G.; Beltran, W.A.; et al. Long-Term Structural Outcomes of Late-Stage RPE65 Gene Therapy. Mol. Ther. 2020, 28, 266–278. [Google Scholar] [CrossRef] [PubMed]
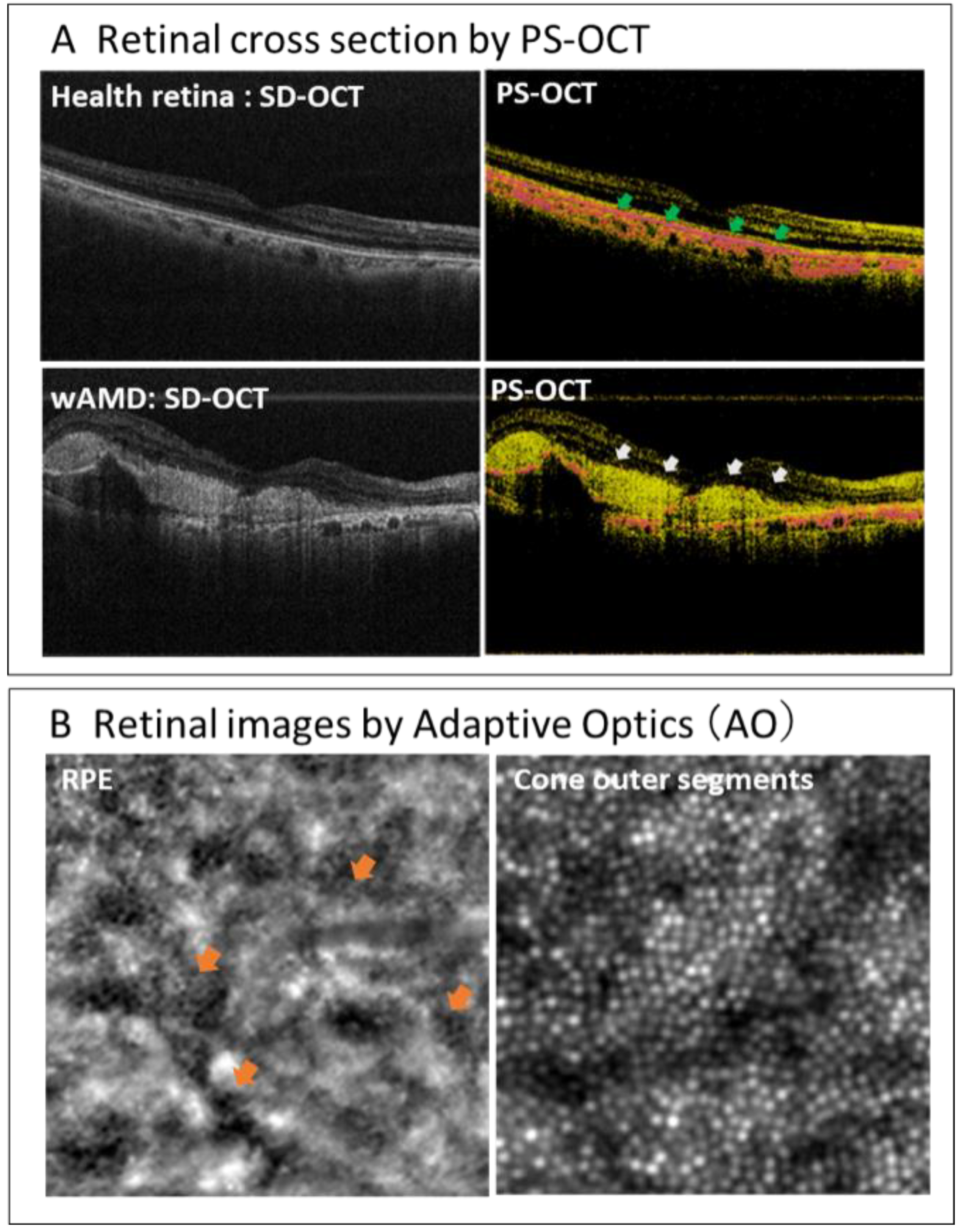
- Matsuzaki, M.; Mandai, M.; Yamanari, M.; Totani, K.; Nishida, M.; Sugita, S.; Maeda, T.; Koide, N.; Takagi, S.; Hirami, Y.; et al. Polarization-sensitive optical coherence tomography for estimating relative melanin content of autologous induced stem-cell derived retinal pigment epithelium. Sci. Rep. 2020, 10, 7656. [Google Scholar] [CrossRef]
| No. | Study Title | Sponsor/Collaborators | Intervention | Age | Phases | No. of Subjects | Start/Completion Date | Status | Study ID |
|---|---|---|---|---|---|---|---|---|---|
| 1 | A Study of transplantation of autologous induced pluripotent stem cell (iPSC) derived retinal pigment epithelium (RPE) cell sheet in subjects with exudative age-related macular degeneration | the Laboratory for Retinal Regeneration, RIKEN Center for Developmental Biology | autologous hiPSC derived RPE cell sheet | 50 years and older | P1 | 1 | October 2013 /September 2018 | completed | UMIN000011929 |
| 2 | Autologous Transplantation of Induced Pluripotent Stem Cell-Derived Retinal Pigment Epithelium for Geographic Atrophy Associated With Age-Related Macular Degeneration | National Institutes of Health Clinical Center, Bethesda, Maryland, U.S. | Combination Product: hiPSC-derived RPE/PLGA scaffold | 55 years and older | P1 | 20 | July 2020 /March 2029 | Recruiting | NCT04339764 |
| 3 | A Study Of Implantation Of Retinal Pigment Epithelium In Subjects With Acute Wet Age Related Macular Degeneration | Moorfields Eye Hospital NHS Foundation Trust, London, U.K. | PF-05206388: RPE living tissue equivalent for intraocular use in the form of a monolayer of RPE cells immobilized on a polyester membrane. | 60 years and older | P1 | 2 | July 2020 /March 2029 | Recruiting | NCT04339764 |
| 4 | Study of Subretinal Implantation of Human Embryonic Stem Cell-Derived RPE Cells in Advanced Dry AMD | Retinal Arizona LTD, Phoenix, Arizona, U.S./Retina-Vitreous Associates Medical Group, Beverly Hills, California, U.S. and others | CPCB-RPE1 (Human Embryonic Stem Cell-Derived RPE Cells Seeded on a Polymeric Substrate) | 55 years to 85 years | P1/2a | 16 | July 2019 /June 2023 | Active, not recruiting | NCT02590692 |
| 5 | A Study of transplantation of allogenic induced pluripotent stem cell (iPSC) derived retinal pigment epithelium (RPE) cell suspension in subjects with neovascular age related macular degeneration | the Laboratory for Retinal Regeneration, RIKEN Center for Developmental Biology, Kobe, Japan/ Kobe City Medical Center General Hosital, Kobe, Japan | Subretinal transplantation of allogenic hiPSC derived RPE cells | 50 years to 85 years | P1 | 5 | February 2017 /October 2021 | Active, not recruiting | UMIN000026003 |
| 6 | Stem Cell Therapy for Outer Retinal Degenerations | Federal University of Sao Paulo, Sao Paulo, Brazil | injection of hESC derived RPE in suspension/Procedure: injection hESC derived RPE seeded in a substrate | 18 years to 90 years | P1/2 | 15 | September 2016 /July 2020 | Completed | NCT02903576 |
| 7 | Subretinal Transplantation of Retinal Pigment Epitheliums in Treatment of Age-related Macular Degeneration Diseases | Chinese Academy of Sciences/Beijing Tongren Hospital, China | hESC derived RPE | 55 years and older | P1/2 | 10 | January 2018 /December 2020 | Recruiting | NCT02755428 |
| 8 | Safety and Efficacy of Subretinal Transplantation of Clinical Human Embryonic Stem Cell Derived Retinal Pigment Epitheliums in Treatment of Retinitis Pigmentosa | Qi Zhou, Chinese Academy of Sciences | hESC derived RPE | 18 years and older | P1 | 10 | May 2020 /December 2021 | Recruiting | NCT03944239 |
| 9 | Treatment of Dry Age Related Macular Degeneration Disease With Retinal Pigment Epithelium Derived From Human Embryonic Stem Cells | Chinese Academy of Sciences/ The First Affiliated Hospital of Zhengzhou University, China | hESC derived RPE | 55 years and older | P1/2 | 15 | September 2017 /December 2020 | Recruiting | NCT03046407 |
| 10 | Safety and Tolerability of Sub-retinal Transplantation of Human Embryonic Stem Cell Derived Retinal Pigmented Epithelial (hESC-RPE) Cells in Patients With Stargardt’s Macular Dystrophy (SMD) | Astellas Institute for Regenerative Medicine/Astellas Pharma Inc., U.S. | hESC derived RPE (MA09-hRPE) | 18 years and older | P1/2 | 15 | November 2011 /September 2015 | completed | NCT01469832 |
| 11 | A Follow up Study to Determine the Safety and Tolerability of Sub-retinal Transplantation of Human Embryonic Stem Cell Derived Retinal Pigmented Epithelial (hESC-RPE) Cells in Patients With Stargardt’s Macular Dystrophy (SMD) | Astellas Institute for Regenerative Medicine/Astellas Pharma Inc., U.S. | hESC derived RPE (MA09-hRPE) | 18 years and older | 12 | January 2013 /October 2019 | completed | NCT02941991 | |
| 12 | Sub-retinal Transplantation of hESC Derived RPE(MA09-hRPE) Cells in Patients With Stargardt’s Macular Dystrophy | Astellas Institute for Regenerative Medicine/Astellas Pharma Inc., U.S. | hESC derived RPE (MA09-hRPE) | 18 years and older | P1/2 | 13 | April 2011 /August 2015 | completed | NCT01345006 |
| 13 | Safety and Tolerability of Sub-retinal Transplantation of hESC Derived RPE (MA09-hRPE) Cells in Patients With Advanced Dry Age Related Macular Degeneration | Astellas Institute for Regenerative Medicine/Astellas Pharma Inc., U.S. | hESC derived RPE (MA09-hRPE) | 55 years and older | P1/2 | 13 | April 2011 /August 2015 | completed | NCT01344993 |
| 14 | Long Term Follow Up of Sub-retinal Transplantation of hESC Derived RPE Cells in Stargardt Macular Dystrophy Patients | Astellas Institute for Regenerative Medicine/Astellas Pharma Inc., U.S.. | hESC derived RPE (MA09-hRPE) | 18 years and older | P1 | 13 | July 2012 /June 2019 | completed | NCT02445612 |
| 15 | Long Term Follow Up of Sub-retinal Transplantation of hESC Derived RPE Cells in Patients With AMD | Astellas Institute for Regenerative Medicine/Astellas Pharma Inc., U.S. | hESC derived RPE (MA09-hRPE) | 18 years and older | 11 | February 2013 /August 2019 | completed | NCT02463344 | |
| 16 | A Phase I/IIa, Open-Label, Single-Center, Prospective Study to Determine the Safety and Tolerability of Sub-retinal Transplantation of Human ES Cell Derived RPE (MA09-hRPE) Cells in Patients With Advanced Dry Age-related Macular Degeneration (AMD) | Astellas Institute for Regenerative Medicine/Astellas Pharma Inc., U.S. | hESC derived RPE (MA09-hRPE) | 55 years and older | P1/2a | 12 | September 2012 /June 2020 | Active, not recruiting | NCT01674829 |
| 17 | A Safety Surveillance Study in Subjects With Macular Degenerative Disease Treated With Human Embryonic Stem Cell-derived Retinal Pigment Epithelial Cell Therapy | Astellas Institute for Regenerative Medicine/Astellas Pharma Inc., U.S. | hESC derived RPE (MA09-hRPE) | 18 years and older | P1/2 | 36 | January 2018 /December 2029 | Enrolling by invitation | NCT03167203 |
| 18 | Retinal Pigment Epithelium Safety Study For Patients In B4711001 | Moorfields Eye Hospital NHS Foundation Trust, U.K. | hESC derived RPE | 60 years and older | 2 | September 2016 /October 2020 | Active, not recruiting | NCT03102138 | |
| 19 | Safety and Efficacy Study of OpRegen for Treatment of Advanced Dry-Form Age-Related Macular Degeneration | Lineage Cell Therapeutics, Inc./CellCure Neurosciences Ltd., Israel | OpRegen | 50 years and older | P1/2 | 24 | August 2015 /December 2024 | Recruiting | NCT02286089 |
| No. | Study Title | Sponsor/Collaborators | Intervention | Age | Phases | No. of Subjects | Start/Completion Date | Status | Study ID |
|---|---|---|---|---|---|---|---|---|---|
| 1 | Study of Human Central Nervous System Stem Cells (HuCNS-SC) in Age-Related Macular Degeneration (AMD) | Retina-Vitreaous Associates Medical Group, Los Angeles, California, United States/ Byers Eye Institute at Stanford, Stanford Hospital and Clinics, Palo Alto, California, United States/ New York Eye and Ear Infirmary, New York, United States and others | hCNS-SC cells | 50 years and older | P1/2 | 15 | June 2012 /June 2015 | completed | NCT01632527 |
| 2 | Long-Term Follow-up Safety Study of Human Central Nervous System Stem Cells in Subjects With Geographic Atrophy of Age-Related Macular Degeneration | Retina Foundation of the Southwest, Dallas, Texas, United States | hCNS-SC cells | 50 years and older | P1 | 8 | April 2014 /May 2016 | Terminated | NCT02137915 |
| 3 | Intravitreal Bone Marrow-Derived Stem Cells in Patients With Macular Degeneration | Rubens Siqueira Research Center, Sao Jose do Rio Preto, SP, Brazil | intravitreal injection of autologous bone marrow stem cell | 18 to 80 years old | P1 | 20 | August 2011 /September 2013 | Completed | NCT01518127 |
| 4 | Stem Cells Therapy in Degenerative Diseases of the Retina | Department of Ophthalmology, Szczecin, Poland | autologous bone marrow-isolated stem/ progenitor cells transplantation | 18 Years to 65 Years | P1 | 30 | December 2018 /February 2020 | Enrolling by invitation | NCT03772938 |
| 5 | Stem Cell Ophthalmology Treatment Study II | MD Stem Cells, Westport, Connecticut, United States/ MD Stem Cells, Coral Springs, Florida, United States/ Fakeeh University Hospital, Dubai, United Arab Emirates | autologous bone marrow derived stem cells | 18 Years and older | NA | 500 | January 2016 /January 2022 | Recruiting | NCT03011541 |
| 6 | Clinical Trial of Autologous Intravitreal Bone-marrow CD34+ Stem Cells for Retinopathy | University of California Davis, Sacramento, California, United States | CD34+ bone marrow stem cells intravitreal | 18 years and older | P1 | 15 | July 2012 /January 2022 | Enrolling by invitation | NCT01736059 |
| 7 | Stem Cell Ophthalmology Treatment Study | MD Stem Cells, Westport, Connecticut, United States | autologous bone marrow derived stem cells | 18 years and older | P1 | 300 | August 2012 /July 2020 | Enrolling by invitation | NCT01920867 |
| 8 | Study of Assess the safety and effects of cells injected in Dry Macular Degeneration | Bioheart Sunrise, Florida, United States | adipose derived stem dells | 50 years to 90 years | NA | 0 | December 2013 /December 2016 | Withdrawn | NCT02024269 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Maeda, T.; Sugita, S.; Kurimoto, Y.; Takahashi, M. Trends of Stem Cell Therapies in Age-Related Macular Degeneration. J. Clin. Med. 2021, 10, 1785. https://doi.org/10.3390/jcm10081785
Maeda T, Sugita S, Kurimoto Y, Takahashi M. Trends of Stem Cell Therapies in Age-Related Macular Degeneration. Journal of Clinical Medicine. 2021; 10(8):1785. https://doi.org/10.3390/jcm10081785
Chicago/Turabian StyleMaeda, Tadao, Sunao Sugita, Yasuo Kurimoto, and Masayo Takahashi. 2021. "Trends of Stem Cell Therapies in Age-Related Macular Degeneration" Journal of Clinical Medicine 10, no. 8: 1785. https://doi.org/10.3390/jcm10081785
APA StyleMaeda, T., Sugita, S., Kurimoto, Y., & Takahashi, M. (2021). Trends of Stem Cell Therapies in Age-Related Macular Degeneration. Journal of Clinical Medicine, 10(8), 1785. https://doi.org/10.3390/jcm10081785





