Trend and Geographic Disparities in the Mortality Rates of Primary Systemic Vasculitis in the United States from 1999 to 2019: A Population-Based Study
Abstract
1. Introduction
2. Materials and Methods
3. Results
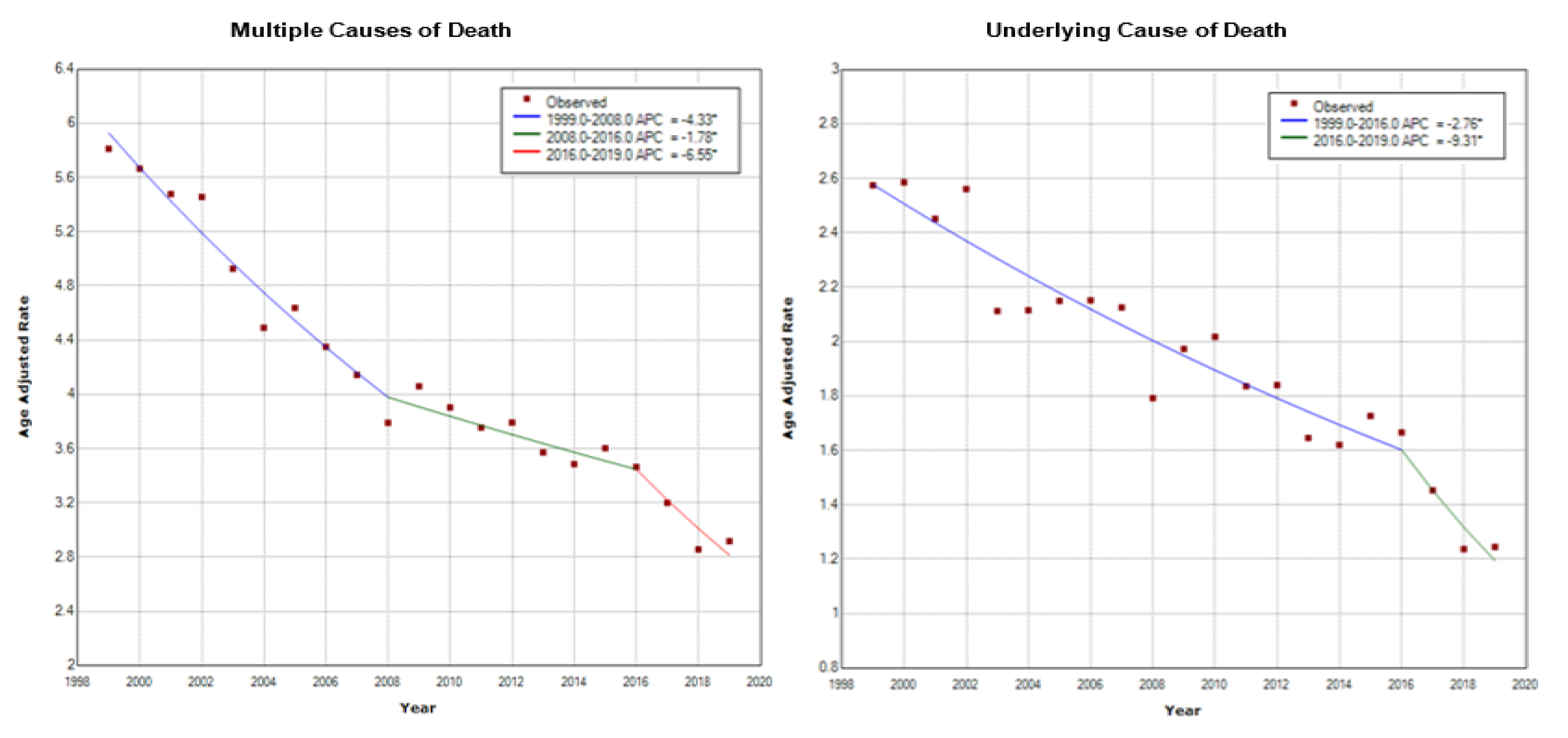
3.1. Overall Age-Adjusted Mortality Rates
3.2. Mortality Rates by Gender
3.3. Mortality Rates by Race
3.4. Mortality Rates by the Hispanic or Non-Hispanic or Latino Groups
3.5. Mortality Rates by Race and Gender
3.6. Mortality Rates by Vasculitis Type
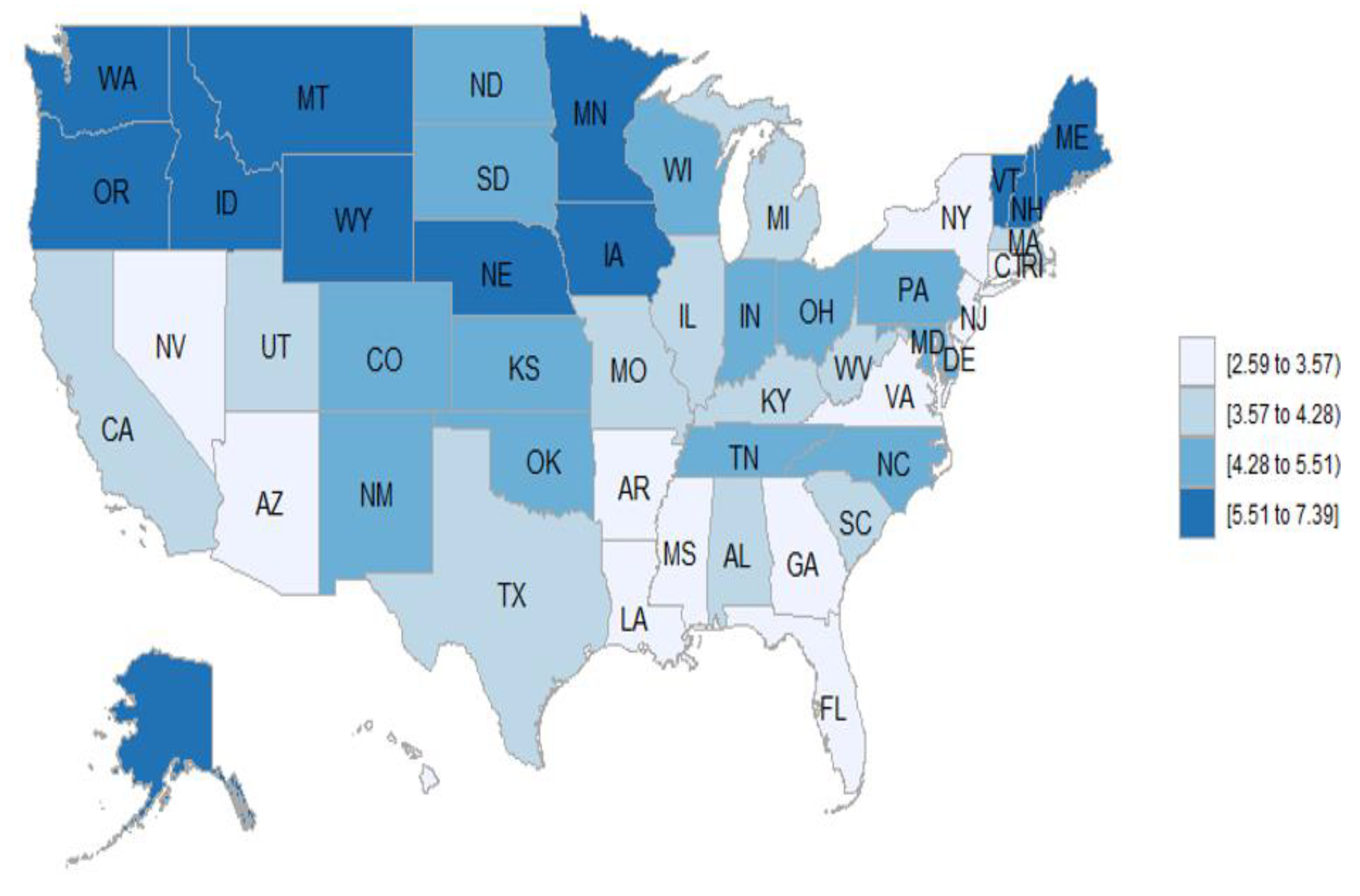
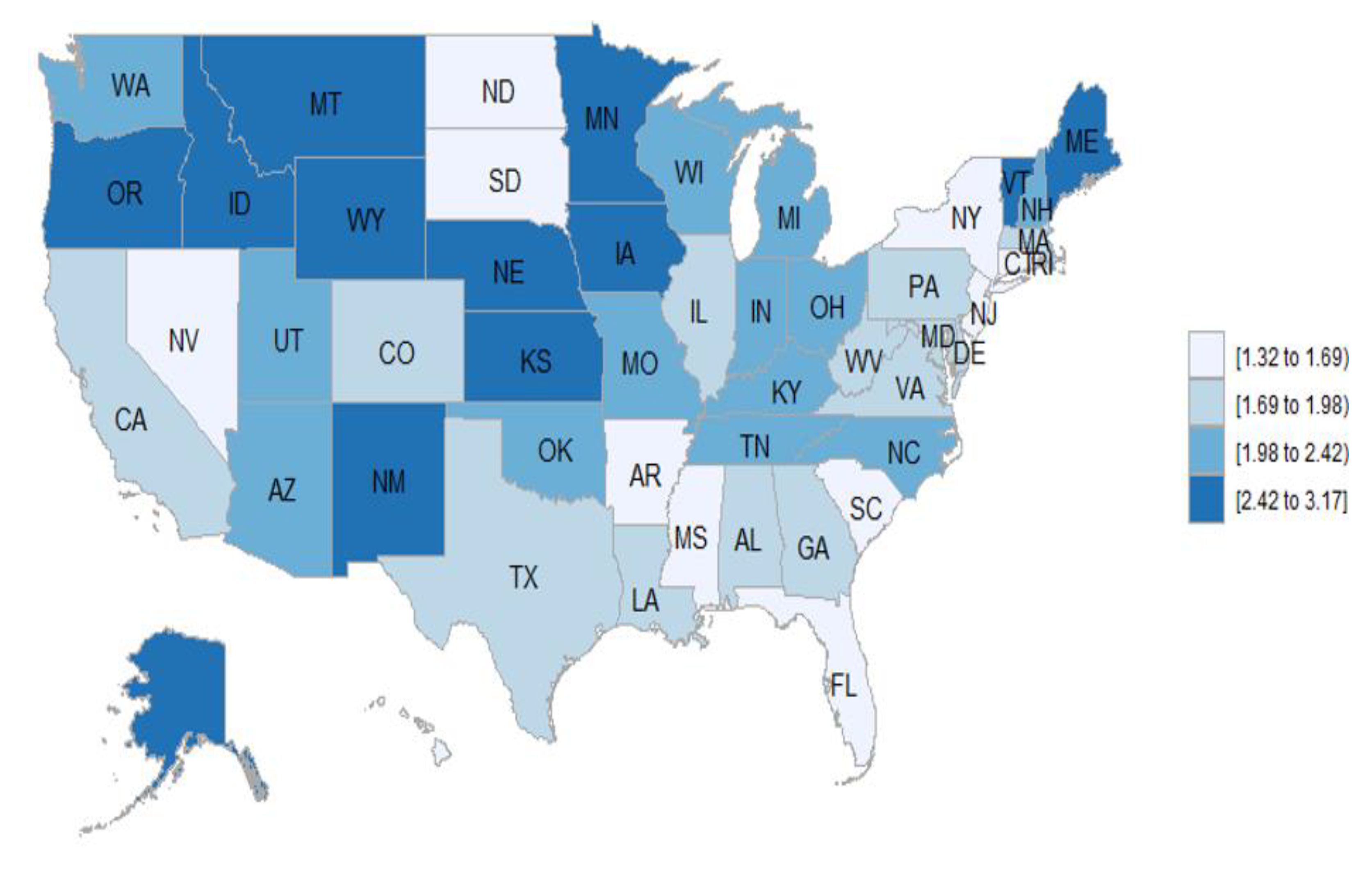
3.7. Geographic Distribution of the Vasculitis Mortality Rates
3.8. Distribution of Vasculitis Mortality Rates According to the Urban–Rural Continuum
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Jennette, J.C.; Falk, R.J.; Bacon, P.A.; Basu, N.; Cid, M.C.; Ferrario, F.; Flores-Suarez, L.F.; Gross, W.L.; Guillevin, L.; Hagen, E.C.; et al. 2012 revised International Chapel Hill Consensus Conference Nomenclature of Vasculitides. Arthritis Rheum. 2013, 65, 1–11. [Google Scholar] [CrossRef]
- Phillip, R.; Luqmani, R. Mortality in systemic vasculitis: A systematic review. Clin. Exp. Rheumatol. 2008, 26, S94–S104. [Google Scholar]
- Iacovino, J.R. Long-term survival of patients with Wegener’s granulomatosis. J. Insur. Med. 2000, 32, 249–253. [Google Scholar]
- Norton, W.L.; Suki, W.; Strunk, S. Combined corticosteroid and azathioprine therapy in 2 patients with Wegener’s granulomatosis. Arch. Intern Med. 1968, 121, 554–560. [Google Scholar] [CrossRef]
- Novack, S.N.; Pearson, C.M. Cyclophosphamide therapy in Wegener’s granulomatosis. N. Engl. J. Med. 1971, 284, 938–942. [Google Scholar] [CrossRef]
- Fauci, A.S.; Wolff, S.M.; Johnson, J.S. Effect of cyclophosphamide upon the immune response in Wegener’s granulomatosis. N. Engl. J. Med. 1971, 285, 1493–1496. [Google Scholar] [CrossRef]
- Stone, J.H.; Merkel, P.A.; Spiera, R.; Seo, P.; Langford, C.A.; Hoffman, G.S.; Kallenberg, C.G.M.; Clair, E.W.S.; Turkiewicz, A.; Tchao, N.K.; et al. Rituximab versus Cyclophosphamide for ANCA-Associated Vasculitis. N. Engl. J. Med. 2010, 363, 221–232. [Google Scholar] [CrossRef] [PubMed]
- Auguet, T.; Barragan, P.; Ramírez, R.; Quer, J.C.; Sirvent, J.J.; Richart, C. Lamivudine in the Treatment of Hepatitis B Virus-Related Polyarteritis Nodosa. JCR J. Clin. Rheumatol. 2007, 13, 298–299. [Google Scholar] [CrossRef] [PubMed]
- Friedman, K.G.; Gauvreau, K.; Baker, A.; Son, M.B.; Sundel, R.; Dionne, A.; Giorgio, T.; De Ferranti, S.; Newburger, J.W. Primary adjunctive corticosteroid therapy is associated with improved outcomes for patients with Kawasaki disease with coronary artery aneurysms at diagnosis. Arch. Dis. Child. 2021, 106, 247–252. [Google Scholar] [CrossRef] [PubMed]
- Kermani, T.A.; Warrington, K.J.; Crowson, C.S.; Hunder, G.G.; Ytterberg, S.R.; Gabriel, S.E.; Matteson, E.L. Predictors of Dissection in Aortic Aneurysms from Giant Cell Arteritis. JCR J. Clin. Rheumatol. 2016, 22, 184–187. [Google Scholar] [CrossRef]
- Lanska, D.J. Geographic distribution of stroke mortality in the United States: 1939–1941 to 1979–1981. Neurology 1993, 43, 1839. [Google Scholar] [CrossRef]
- Akiyama, M. Trial of Tocilizumab in Giant-Cell Arteritis. N. Engl. J. Med. 2017, 377, 1493–1495. [Google Scholar] [CrossRef]
- Hechter, H.H.; Borhani, N.O. Mortality and Geographic Distribution of Arteriosclerotic Heart Disease: Recent Changes 1950–1960. Public Health Rep. (1896–1970) 1965, 80, 11. [Google Scholar] [CrossRef]
- Rodriguez-Pla, A.; Simms, R.W. Geographic disparity in systemic sclerosis mortality in the United States: 1999–2017. J. Scleroderma Relat. Disord. 2019. [Google Scholar] [CrossRef]
- Centers for Disease Control and Prevention; National Center for Health Statistics. Multiple Cause of Death 1999–2019. On CDC WONDER Online Database, Released in 2020. Data are from the Multiple Cause of Death Files, 1999–2019, as Compiled from Data Provided by the 57 Vital Statistics Jurisdictions through the Vital Statistics Cooperative Program. Available online: http://wonder.cdc.gov/mcd-icd10.html (accessed on 25 December 2020).
- Centers for Disease Control and Prevention; National Center for Health Statistics. Underlying Cause of Death 1999–2019. On CDC WONDER Online Database, Released in 2020. Data are from the Multiple Cause of Death Files, 1999–2019, as Compiled from Data Provided by the 57 Vital Statistics Jurisdictions through the Vital Statistics Cooperative Program. Available online: http://wonder.cdc.gov/ucd-icd10.html (accessed on 25 December 2020).
- Joinpoint Regression Program, VA, 2020. Statistical Research and Applications Branch. National Cancer Institute. Available online: https://surveillance.cancer.gov/joinpoint/ (accessed on 25 December 2020).
- Rodriguez-Pla, A.; Monach, P.A.; Rossello-Urgell, J. Vasculitis as Underlying Cause of Death in the United States: 1999–2010. Arthritis Rheumatol. 2014, 66, 1786. [Google Scholar]
- Rodriguez-Pla, A. Geographic Disparities in Mortality Rates of Vasculitis in the United States: 1999 to 2017 (abstract). Arthritis Rheumatol. 2019, 71, 1718. [Google Scholar]
- Wallace, Z.S.; Lu, N.; Unizony, S.; Stone, J.H.; Choi, H.K. Improved survival in granulomatosis with polyangiitis: A general population-based study. Semin. Arthritis Rheum. 2016, 45, 483–489. [Google Scholar] [CrossRef]
- Steinberg, A.W.; Wechsler, M.E.; Pérez, E.R.F. Trends in Antineutrophil Cytoplasmic Autoantibody–Associated Vasculitis-Related Mortality in the United States, 1999 to 2017. Ann. Intern. Med. 2019, 172, 160. [Google Scholar] [CrossRef]
- Tan, J.A.; Dehghan, N.; Chen, W.; Xie, H.; Esdaile, J.M.; Avina-Zubieta, J.A. Mortality in ANCA-associated vasculitis: Ameta-analysis of observational studies. Ann. Rheum. Dis. 2017, 76, 1566–1574. [Google Scholar] [CrossRef]
- Holle, J.U.; Gross, W.L.; Latza, U.; Nölle, B.; Ambrosch, P.; Heller, M.; Fertmann, R.; Reinhold-Keller, E. Improved outcome in 445 patients with Wegener’s granulomatosis in a German vasculitis center over four decades. Arthritis Rheum. 2011, 63, 257–266. [Google Scholar] [CrossRef]
- Bierman, A.S. Sex matters: Gender disparities in quality and outcomes of care. Can. Med. Assoc. J. 2007, 177, 1520–1521. [Google Scholar] [CrossRef]
- Birnie, D.H.; Sambell, C.; Johansen, H.; Williams, K.; Lemery, R.; Green, M.S.; Gollob, M.H.; Lee, U.S.; Tang, A.S. Use of implantable cardioverter defibrillators in Canadian and US survivors of out-of-hospital cardiac arrest. Can. Med. Assoc. J. 2007, 177, 41–46. [Google Scholar] [CrossRef]
- Scott, D.G.I.; Watts, R.A. Epidemiology and clinical features of systemic vasculitis. Clin. Exp. Nephrol. 2013, 17, 607–610. [Google Scholar] [CrossRef]
- Cao, Y.; Schmitz, J.L.; Yang, J.; Hogan, S.L.; Bunch, D.; Hu, Y.; Jennette, C.E.; Berg, E.A.; Arnett, F.C.; Jennette, J.C.; et al. DRB1*15 Allele is a Risk Factor for PR3-ANCA disease in African Americans. J. Am. Soc. Nephrol. 2011, 22, 1161–1167. [Google Scholar] [CrossRef] [PubMed]
- O’Shaughnessy, M.M.; Hogan, S.L.; Poulton, C.J.; Falk, R.J.; Singh, H.K.; Nickeleit, V.; Jennette, J.C. Temporal and Demographic Trends in Glomerular Disease Epidemiology in the Southeastern United States, 1986–2015. Clin. J. Am. Soc. Nephrol. 2017, 12, 614–623. [Google Scholar] [CrossRef] [PubMed]
- Chandran, A.K.; Udayakumar, P.D.; Crowson, C.S.; Warrington, K.J.; Matteson, E.L. The incidence of giant cell arteritis in Olmsted County, Minnesota, over a 60-year period 1950–2009. Scand. J. Rheumatol. 2014, 44, 215–218. [Google Scholar] [CrossRef] [PubMed]
- Smith, C.A.; Fidler, W.J.; Pinals, R.S. The epidemiology of giant cell arteritis. Report of a Ten-Year Study in Shelby County, Tennessee. Arthritis Rheum. 1983, 26, 1214–1219. [Google Scholar] [CrossRef] [PubMed]
- Mader, T.H.; Werner, R.P.; Chamberlain, D.G.; Doornbos, D. Giant cell arteritis in Alaska Natives. Can. J. Ophthalmol. 2009, 44, 53–56. [Google Scholar] [CrossRef]
- Ramstead, C.L.; Patel, A.D. Giant cell arteritis in a neuro-ophthalmology clinic in Saskatoon, 1998–2003. Can. J. Ophthalmol. 2007, 42, 295–298. [Google Scholar] [CrossRef]
- Pereira, L.S.; Yoon, M.K.; Hwang, T.N.; E Hong, J.; Ray, K.; Porco, T.; McCulley, T.J. Giant cell arteritis in Asians: A comparative study. Br. J. Ophthalmol. 2010, 95, 214–216. [Google Scholar] [CrossRef]
- Lam, B.L.; Wirthlin, R.S.; Gonzalez, A.; Dubovy, S.R.; Feuer, W.J. Giant Cell Arteritis Among Hispanic Americans. Am. J. Ophthalmol. 2007, 143, 161–163. [Google Scholar] [CrossRef]
- Sreih, A.G.; Mandhadi, R.; Aldaghlawi, F.; Khan, A.; Irshad, V.; Finn, K.; Block, J.A. ANCA-associated vasculitis in Hispanic Americans: An unrecognized severity. Clin. Rheumatol. 2014, 34, 943–948. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Injean, P.; Tran, P.; Panikkath, D.; Downey, C. ANCA-associated Vasculitis in Caucasian and Hispanics of the Inland Empire of Southern California. Arthritis Rheum. 2020, 72, 0060. [Google Scholar]
- Lyons, P.A.; Rayner, T.F.; Trivedi, S.; Holle, J.U.; Watts, R.A.; Jayne, D.R.; Baslund, B.; Brenchley, P.; Bruchfeld, A.; Chaudhry, A.N.; et al. Genetically distinct subsets within ANCA-associated vasculitis. N. Engl. J. Med. 2012, 367, 214–223. [Google Scholar] [CrossRef] [PubMed]
- Merkel, P.A.; Xie, G.; Monach, P.A.; Ji, X.; Ciavatta, D.J.; Byun, J.; Pinder, B.D.; Zhao, A.; Zhang, J.; Tadesse, Y.; et al. Identification of Functional and Expression Polymorphisms Associated with Risk for Antineutrophil Cytoplasmic Autoantibody–Associated Vasculitis. Arthritis Rheumatol. 2017, 69, 1054–1066. [Google Scholar] [CrossRef] [PubMed]
- Barra, L.; E Pope, J.; Pequeno, P.; E Saxena, F.; Bell, M.; Haaland, D.; Widdifield, J. Incidence and prevalence of giant cell arteritis in Ontario, Canada. Rheumatology 2020, 59, 3250–3258. [Google Scholar] [CrossRef]
- Mohammad, A.J.; Nilsson, J.-Å.; Jacobsson, L.T.H.; Merkel, P.A.; Turesson, C. Incidence and mortality rates of biopsy-proven giant cell arteritis in southern Sweden. Ann. Rheum. Dis. 2014, 74, 993–997. [Google Scholar] [CrossRef]
- Gonzalez-Gay, M.A.; Miranda-Filloy, J.A.; Lopez-Diaz, M.J.; Perez-Alvarez, R.; Gonzalez-Juanatey, C.; Sanchez-Andrade, A.; Martin, J.; Llorca, J. Giant Cell Arteritis in Northwestern Spain. Medicine 2007, 86, 61–68. [Google Scholar] [CrossRef]
- Bas-Lando, M.; Breuer, G.S.; Berkun, Y.; Mates, M.; Sonnenblick, M.; Nesher, G. The incidence of giant cell arteritis in Jerusalem over a 25-year period: Annual and seasonal fluctuations. Clin. Exp. Rheumatol. 2007, 25, 15–17. [Google Scholar]
- Matteson, E.L.; Gold, K.N.; Bloch, D.A.; Hunder, G.G. Long-term survival of patients with giant cell arteritis in the american college of rheumatology giant cell arteritis classification criteria cohort. Am. J. Med. 1996, 100, 193–196. [Google Scholar] [CrossRef]
- Andersen, J.B.; Myklebust, G.; Haugeberg, G.; Pripp, A.H.; Diamantopoulos, A.P. Incidence Trends and Mortality of Giant Cell Arteritis in Southern Norway. Arthritis Rheum. 2021, 73, 409–414. [Google Scholar] [CrossRef]
- Unizony, S.; Menendez, M.E.; Rastalsky, N.; Stone, J.H. Inpatient complications in patients with giant cell arteritis: Decreased mortality and increased risk of thromboembolism, delirium and adrenal insufficiency. Rheumatology 2015, 54, 1360–1368. [Google Scholar] [CrossRef] [PubMed]
- Ben-Shabat, N.; Tiosano, S.; Shovman, O.; Comaneshter, D.; Shoenfeld, Y.; Cohen, A.D.; Amital, H. Mortality among Patients with Giant Cell Arteritis: A Large-scale Population-based Cohort Study. J. Rheumatol. 2019, 47, 1385–1391. [Google Scholar] [CrossRef]
- Kaewput, W.; Thongprayoon, C.; Petnak, T.; Cato, L.D.; Chewcharat, A.; Boonpheng, B.; Bathini, T.; Vallabhajosyula, S.; Cheungpasitporn, W. Inpatient burden and mortality of heatstroke in the United States. Int. J. Clin. Pr. 2021, 75, e13837. [Google Scholar] [CrossRef]
- Gong, G.; Phillips, S.G.; Hudson, C.; Curti, D.; Philips, B.U. Higher US Rural Mortality Rates Linked to Socioeconomic Status, Physician Shortages, And Lack Of Health Insurance. Health Aff. 2019, 38, 2003–2010. [Google Scholar] [CrossRef]
- Foreman, K.J.; Naghavi, M.; Ezzati, M. Improving the usefulness of US mortality data: New methods for reclassification of underlying cause of death. Popul. Health Metr. 2016, 14, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Thomas, S.L.; Griffiths, C.; Smeeth, L.; Rooney, C.; Hall, A.J. Burden of Mortality Associated with Autoimmune Diseases Among Females in the United Kingdom. Am. J. Public Health 2010, 100, 2279–2287. [Google Scholar] [CrossRef] [PubMed]
| Multiple Causes of Death | Underlying Cause of Death | ||||||
|---|---|---|---|---|---|---|---|
| Race | Gender | Deaths | Age-Adjusted Rate (95% CI) | Percentage of Total Deaths | Deaths | Age-Adjusted Rate (95% CI) | Percentage of Total Deaths |
| White | Female | 15,256 |
4.469 (4.397–4.542) | 54.52% | 6622 |
2.006 (1.957–2.056) | 50.75% |
| Male | 10,583 |
4.176 (4.095–4.256) | 37.82% | 5298 |
2.052 (1.996–2.108) | 40.60% | |
| Total | 25,839 | 4.371 (4.317–4.424) | 92.34% | 11,920 | 2.032 (1.995–2.069) | 91.36% | |
| American Indian or Alaska Native | Female | 102 |
3.582 (2.851–4.313) | 0.37% | 70 |
2.429 (1.867–3.108) | 0.54% |
| Male | 80 |
3.275 (2.516–4.190) | 0.29% | 40 |
1.594 (1.090–2.250) | 0.31% | |
| Total | 182 | 3.451 (2.910–3.992) | 0.65% | 110 | 2.059 (1.643–2.475) | 0.84% | |
| Black or African American | Female | 843 |
2.088 (1.946–2.231) | 3.013% | 397 |
0.978 (0.881–1.076) | 3.04% |
| Male | 599 |
1.970 (1.800–2.140) | 2.14% | 348 |
1.099 (0.975–1.223) | 2.67% | |
| Total | 1442 | 2.037 (1.929–2.146) | 5.15% | 745 | 1.024 (0.948–1.099) | 5.71% | |
| Asian or Pacific Islander | Female | 295 |
1.892 (1.672–2.111) | 1.05% | 152 |
0.944 (0.791–1.097) | 1.17% |
| Male | 225 |
1.795 (1.548–2.041) | 0.80% | 121 |
0.944 (0.768–1.120) | 0.93% | |
| Total | 520 | 1.861 (1.696–2.026) | 1.86% | 273 | 0.941 (0.826–1.057) | 2.09% | |
| Total | 27,983 | 4.077 (4.029–4.125) | 100.00% | 13,048 | 1.888 (1.855–1.921) | 100.00% | |
| Multiple Causes of Death | Underlying Cause of Death | |||
|---|---|---|---|---|
| Type of Vasculitis | Deaths | Age-Adjusted Rate (95% CI) | Deaths | Age-Adjusted Rate (95% CI) |
| Granulomatosis with polyangiitis (GPA) | 10,388 |
1.514 (1.485–1.544) | 6547 |
0.962 (0.939–0.986) |
| Giant cell arteritis (GCA) | 9200 |
1.329 (1.302–1.356) | 1823 |
0.268 (0.256–0.281) |
| Antiglomerular basement membrane (anti-GBM) disease | 1910 |
0.291 (0.277–0.304) | 1314 |
0.197 (0.186–0.209) |
| Polyarteritis nodosa (PAN) | 1694 |
0.239 (0.227–0.251) | 845 |
0.126 (0.117–0.135) |
| Eosinophilic granulomatosis with polyangiitis (EGPA) | 1261 |
0.178 (0.168–0.189) | 681 |
0.099 (0.091–0.107) |
| Cryoglobulinemia | 1353 |
0.195 (0.184–0.206) | 512 |
0.068 (0.062–0.074) |
| Microscopic polyangiitis (MPA) | 742 |
0.093 (0.086–0.100) | 508 |
0.063 (0.057–0.069) |
| Takayasu arteritis (TAK) | 533 |
0.079 (0.071–0.086) | 289 |
0.035 (0.030–0.040) |
| Behcet’s disease | 519 |
0.071 (0.064–0.078) | 252 |
0.033 (0.028–0.038) |
| Ig (immunoglobulin) A vasculitis (Henoch–Schönlein) (IgAV) | 384 |
0.042 (0.037–0.046) | 166 |
0.014 (0.011–0.017) |
| Kawasaki disease | 177 |
0.013 (0.010–0.016) | 102 |
0.008 (0.006–0.011) |
| Other conditions related to polyarteritis nodosa | 16 | Unreliable | 6 | Unreliable |
| Giant cell arteritis with polymyalgia Rheumatica | 12 | Unreliable | 2 | Unreliable |
| Juvenile polyarteritis | 1 | Unreliable | 1 | Unreliable |
| Total | 28,190 | 4.096 (4.048–4.144) | 13,048 | 1.888 (1.855–1.921) |
| Multiple Causes of Death | Underlying Cause of Death | |||||
|---|---|---|---|---|---|---|
| 2013 Urbanization * | Deaths | Age-Adjusted Rate (95% CI) | Percentage of Total Deaths | Deaths | Age-Adjusted Rate (95% CI) | Percentage of Total Deaths |
| Micropolitan | 3357 |
5.001 (4.831–5.172) | 12.00% | 1569 |
2.350 (2.232–2.468) | 12.03% |
| Small metro | 3321 |
4.930 (4.761–5.099) | 11.87% | 1525 |
2.265 (2.150–2.380) | 11.67% |
| Noncore | 2398 |
4.546 (4.362–4.731) | 8.57% | 1105 |
2.134 (2.006–2.262) | 8.47% |
| Medium metro | 6234 |
4.294 (4.186–4.401) | 22.28% | 2890 |
2.017 (1.943–2.092) | 22.15% |
| Large fringe metro | 6140 |
3.762 (3.667–3.857) | 21.94% | 2869 |
1.742 (1.677–1.806) | 21.99% |
| Large central metro | 6533 |
3.437 (3.353–3.521) | 23.35% | 3090 |
1.611 (1.554–1.668) | 23.68% |
| Total | 27,983 |
4.077 (4.029–4.125) | 100% | 13,048 |
1.888 (1.855–1.921) | 100% |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rodriguez-Pla, A.; Rossello-Urgell, J. Trend and Geographic Disparities in the Mortality Rates of Primary Systemic Vasculitis in the United States from 1999 to 2019: A Population-Based Study. J. Clin. Med. 2021, 10, 1759. https://doi.org/10.3390/jcm10081759
Rodriguez-Pla A, Rossello-Urgell J. Trend and Geographic Disparities in the Mortality Rates of Primary Systemic Vasculitis in the United States from 1999 to 2019: A Population-Based Study. Journal of Clinical Medicine. 2021; 10(8):1759. https://doi.org/10.3390/jcm10081759
Chicago/Turabian StyleRodriguez-Pla, Alicia, and Jose Rossello-Urgell. 2021. "Trend and Geographic Disparities in the Mortality Rates of Primary Systemic Vasculitis in the United States from 1999 to 2019: A Population-Based Study" Journal of Clinical Medicine 10, no. 8: 1759. https://doi.org/10.3390/jcm10081759
APA StyleRodriguez-Pla, A., & Rossello-Urgell, J. (2021). Trend and Geographic Disparities in the Mortality Rates of Primary Systemic Vasculitis in the United States from 1999 to 2019: A Population-Based Study. Journal of Clinical Medicine, 10(8), 1759. https://doi.org/10.3390/jcm10081759








