Adolescent, Pregnant, and HIV-Infected: Risk of Adverse Pregnancy and Perinatal Outcomes in Young Women from Southern Mozambique
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design and Data Sources
2.2. Study Population
2.3. Study Area
2.4. Study Variables
2.5. Data Cleaning and Analysis
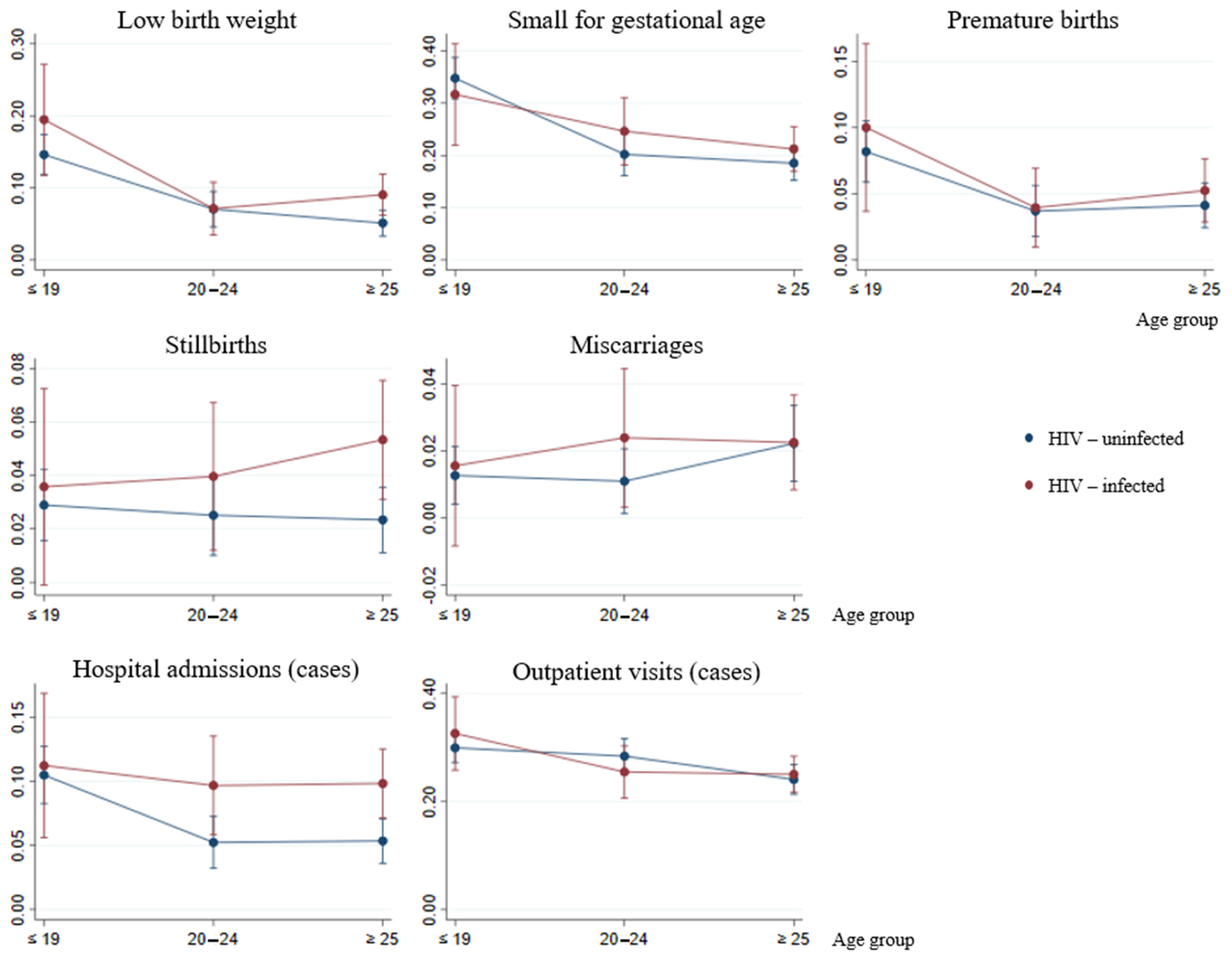
3. Results
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Darroch, J.E.; Woog, V.; Bankoleand, A.; Ashford, L.S. Adding It Up: Costs and Benefits of Meeting the Contraceptive Needs of Adolescents. 2016. Available online: https://www.guttmacher.org/report/adding-it-meeting-contraceptive-needs-of-adolescents (accessed on 24 December 2020).
- World Health Organization. Adolescents: Health Risks and Solutions. 2018. Available online: https://www.who.int/news-room/fact-sheets/detail/adolescents-health-risks-and-solutions (accessed on 4 February 2021).
- United Nations. The Global Strategy for Women’s, Children’s and Adolescents’ Health (2016–2030). 2015. Available online: https://www.who.int/life-course/partners/global-strategy/globalstrategyreport2016-2030-lowres.pdf (accessed on 24 December 2020).
- WHO. Core Competencies in Adolescent Health and Development for Primary Care Providers: Including a Tool to Assess the Adolescent Health And Development Component in Pre-Service Education of Health-Care Providers. 2015. Available online: https://www.who.int/maternal_child_adolescent/documents/core_competencies/en/ (accessed on 24 December 2020).
- Ganchimeg, T.; Ota, E.; Morisaki, N.; Laopaiboon, M.; Lumbiganon, P.; Zhang, J.; Yamdamsuren, B.; Temmerman, M.; Say, L.; Tunçalp, Ö.; et al. Pregnancy and childbirth outcomes among adolescent mothers: A World Health Organization multicountry study. BJOG Int. J. Obstet. Gynaecol. 2014, 121, 40–48. [Google Scholar] [CrossRef] [PubMed]
- Ganchimeg, T.; Mori, R.; Ota, E.; Koyanagi, A.; Gilmour, S.; Shibuya, K.; Torloni, M.R.; Betran, A.P.; Seuc, A.; Vogel, J.; et al. Maternal and perinatal outcomes among nulliparous adolescents in low- and mid-dle-income countries: A multi-country study. BJOG 2013, 120, 1622–1630. [Google Scholar] [CrossRef] [PubMed]
- Kozuki, N.; Lee, A.C.C.; Silveira, M.F.; Sania, A.; Vogel, J.P.; Adair, L.; Barros, F.; Caulfield, L.E.; Christian, P.; Fawzi, W.; et al. The associations of parity and maternal age with small-for-gestational-age, preterm, and neonatal and infant mortality: A meta-analysis. BMC Public Health 2013, 13, S2. [Google Scholar] [CrossRef] [PubMed]
- Mombo-Ngoma, G.; Mackanga, J.R.; González, R.; Ouedraogo, S.; Kakolwa, M.A.; Manego, R.Z.; Basra, A.; Rupérez, M.; Cot, M.; Kabanywany, A.M.; et al. Young adolescent girls are at high risk for adverse pregnancy outcomes in sub-Saharan Africa: An observational multicountry study. BMJ Open 2016, 6, e011783. [Google Scholar] [CrossRef] [PubMed]
- Grønvik, T.; Sandøy, I.F. Complications associated with adolescent childbearing in Sub-Saharan Africa: A systematic literature review and meta-analysis. PLoS ONE 2018, 13, e0204327. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.K.; Wen, S.W.; Fleming, N.; Demissie, K.; Rhoads, G.G.; Walker, M. Teenage pregnancy and adverse birth outcomes: A large population based retro-spective cohort study. Int. J. Epidemiol. 2007, 36, 368–373. [Google Scholar] [CrossRef]
- Fraser, A.M.; Brockert, J.E.; Ward, R. Association of Young Maternal Age with Adverse Reproductive Outcomes. N. Engl. J. Med. 1995, 332, 1113–1118. [Google Scholar] [CrossRef]
- UNAIDS. AIDSinfo. 2020. Available online: https://aidsinfo.unaids.org/ (accessed on 24 December 2020).
- UNAIDS. Start Free Stay Free AIDS Free. 2019. Available online: https://www.unaids.org/sites/default/files/media_asset/20190722_UNAIDS_SFSFAF_2019_en.pdf (accessed on 24 December 2020).
- Callahan, T.; Modi, S.; Swanson, J.; Ng’Eno, B.; Broyles, L.N. Pregnant adolescents living with HIV: What we know, what we need to know, where we need to go. J. Int. AIDS Soc. 2017, 20, 21858. [Google Scholar] [CrossRef]
- WHO. Consolidated Guidelines on the Use of Antiretroviral Drugs for Treating and Preventing HIV Infection: Rec-Ommendations for a Public Health Approach; WHO: Geneva, Switzerland, 2016. [Google Scholar]
- Psaros, C.; Smit, J.A.; Mosery, N.; Bennett, K.; Coleman, J.N.; Bangsberg, D.R.; Safren, A.S. PMTCT Adherence in Pregnant South African Women: The Role of Depression, Social Support, Stigma, and Structural Barriers to Care. Ann. Behav. Med. 2020, 54, 626–636. [Google Scholar] [CrossRef]
- Mukosha, M.; Chiyesu, G.; Vwalika, B. Adherence to antiretroviral therapy among HIV infected pregnant women in public health sectors: A pilot of Chilenje level one Hospital Lusaka, Zambia. Pan Afr. Med. J. 2020, 35, 49. [Google Scholar] [CrossRef]
- Fatti, G.; Shaikh, N.; Eley, B.; Jackson, D.; Grimwood, A. Adolescent and young pregnant women at increased risk of mother-to-child transmission of HIV and poorer maternal and infant health outcomes: A cohort study at public facilities in the Nelson Mandela Bay Metropolitan district, Eastern Cape, South Africa. S. Afr. Med. J. 2014, 104, 874–880. [Google Scholar] [CrossRef]
- Ronen, K.; McGrath, C.J.; Langat, A.C.; Kinuthia, J.; Omolo, D.; Singa, B.; Katana, A.K.; Ng’Ang’, A.L.W.; John-Stewart, G. Gaps in Adolescent Engagement in Antenatal Care and Prevention of Moth-er-to-Child HIV Transmission Services in Kenya. J. Acquir. Immune Defic. Synd. 2017, 74, 30–37. [Google Scholar] [CrossRef] [PubMed]
- Ramraj, T.; Jackson, D.; Dinh, T.-H.; Olorunju, S.; Lombard, C.; Sherman, G.; Puren, A.; Ramokolo, V.; Noveve, N.; Singh, Y.; et al. Adolescent Access to Care and Risk of Early Mother-to-Child HIV Transmission. J. Adolesc. Health 2018, 62, 434–443. [Google Scholar] [CrossRef] [PubMed]
- Groves, A.K.; Maman, S.; Stankard, P.H.; Gebrekristos, L.T.; Amon, J.J.; Moodley, D. Addressing the unique needs of adolescent mothers in the fight against HIV. J. Int. AIDS Soc. 2018, 21, e25155. [Google Scholar] [CrossRef] [PubMed]
- Toska, E.; Laurenzi, C.A.; Roberts, K.J.; Cluver, L.; Sherr, L. Adolescent mothers affected by HIV and their children: A scoping review of evidence and experiences from sub-Saharan Africa. Glob. Public Health 2020, 15, 1655–1673. [Google Scholar] [CrossRef]
- Menéndez, C.; Bardají, A.; Sigauque, B.; Romagosa, C.; Sanz, S.; Serra-Casas, E.; Macete, E.; Berenguera, A.; David, C.; Dobaño, C.; et al. A Randomized Placebo-Controlled Trial of Intermittent Preventive Treatment in Pregnant Women in the Context of Insecticide Treated Nets Delivered through the Antenatal Clinic. PLoS ONE 2008, 3, e1934. [Google Scholar] [CrossRef] [PubMed]
- González, R.; Mombo-Ngoma, G.; Ouedraogo, S.; Kakolwa, M.A.; Abdulla, S.; Accrombessi, M.; Aponte, J.J.; Akerey-Diop, D.; Basra, A.; Briand, V.; et al. Intermittent Preventive Treatment of Malaria in Pregnancy with Mefloquine in HIV-Negative Women: A Multicentre Randomized Controlled Trial. PLoS Med. 2014, 11, e1001733. [Google Scholar] [CrossRef]
- González, R.; Desai, M.; Macete, E.; Ouma, P.; Kakolwa, M.A.; Abdulla, S.; Aponte, J.J.; Bulo, H.; Kabanywanyi, A.M.; Katana, A.; et al. Intermittent Preventive Treatment of Malaria in Pregnancy with Mefloquine in HIV-Infected Women Receiving Cotrimoxazole Prophylaxis: A Multicenter Randomized Placebo-Controlled Trial. PLoS Med. 2014, 11, e1001735. [Google Scholar] [CrossRef]
- WHO. Adolescent Pregnancy. 2020. Available online: https://www.who.int/news-room/fact-sheets/detail/adolescent-pregnancy (accessed on 4 February 2021).
- Sacoor, C.; Nhacolo, A.; Nhalungo, D.; Aponte, J.J.; Bassat, Q.; Augusto, O.; Mandomando, I.; Sacarlal, J.; Lauchande, N.; Sigaúque, B.; et al. Profile: Manhica Health Research Centre (Manhica HDSS). Int. J. Epidemiol. 2012, 42, 1309–1318. [Google Scholar] [CrossRef]
- González, R.; Munguambe, K.; Aponte, J.; Bavo, C.; Nhalungo, D.; Macete, E.; Alonso, P.; Menéndez, C.; Naniche, D. High HIV prevalence in a southern semi-rural area of Mozambique: A community-based survey. HIV Med. 2012, 13, 581–588. [Google Scholar] [CrossRef]
- González, R.; Augusto, O.J.; Munguambe, K.; Pierrat, C.; Pedro, E.N.; Sacoor, C.; De Lazzari, E.; Aponte, J.J.; Macete, E.; Alonso, P.L.; et al. HIV Incidence and Spatial Clustering in a Rural Area of Southern Mozambique. PLoS ONE 2015, 10, e0132053. [Google Scholar] [CrossRef] [PubMed]
- Firth, D. Bias reduction of maximum likelihood estimates. Biometrika 1993, 80, 27–38. [Google Scholar] [CrossRef]
- StataCorp. Stata Statistical Software: Release 16; StataCorp LLC: College Station, TX, USA, 2019. [Google Scholar]
- Naniche, D.; Lahuerta, M.; Bardaji, A.; Sigauque, B.; Romagosa, C.; Berenguera, A.; Mandomando, I.; David, C.; Sanz, S.; Aponte, J.; et al. Mother-to-child transmission of HIV-1: Association with malaria prevention, anaemia and placental malaria. HIV Med. 2008, 9, 757–764. [Google Scholar] [CrossRef]
- Rollins, N.C.; Coovadia, H.M.; Bland, R.M.; Coutsoudis, A.; Bennish, M.L.; Patel, D.; Newell, M.-L. Pregnancy Outcomes in HIV-Infected and Uninfected Women in Rural and Urban South Africa. JAIDS J. Acquir. Immune Defic. Syndr. 2007, 44, 321–328. [Google Scholar] [CrossRef]
- Santosa, W.B.; Staines-Urias, E.; Tshivuila-Matala, C.O.; Norris, S.A.; Hemelaar, J. Perinatal outcomes associated with maternal HIV and an-tiretroviral therapy in pregnancies with accurate gestational age in South Africa. AIDS 2019, 33, 1623–1633. [Google Scholar] [CrossRef]
- Xiao, P.-L.; Zhou, Y.-B.; Chen, Y.; Yang, M.-X.; Song, X.-X.; Shi, Y.; Jiang, Q.-W. Association between maternal HIV infection and low birth weight and prematurity: A meta-analysis of cohort studies. BMC Pregnancy Childbirth 2015, 15, 246. [Google Scholar] [CrossRef]
- González, R.; Rupérez, M.; Sevene, E.; Vala, A.; Maculuve, S.; Bulo, H.; Nhacolo, A.; Mayor, A.; Aponte, J.J.; Macete, E.; et al. Effects of HIV infection on maternal and neonatal health in southern Mozambique: A prospective cohort study after a decade of antiretroviral drugs roll out. PLoS ONE 2017, 12, e0178134. [Google Scholar] [CrossRef]
- Moraleda, C.; de Deus, N.; Serna-Bolea, C.; Renom, M.; Quintó, L.; Macete, E.; Menéndez, C.; Naniche, D. Impact of HIV exposure on health outcomes in HIV-negative infants born to HIV-positive mothers in Sub-Saharan Africa. J. Acquir. Immune Defic. Syndr. 2014, 65, 182–189. [Google Scholar] [CrossRef] [PubMed]
- Goldman, G.; Budhram, S.; Budhram, S. A retrospective cohort study comparing pregnancy outcomes and neonatal char-acteristics between HIV-infected and HIV-non-infected mothers. S. Afr. Med. J. 2020, 110, 502–504. [Google Scholar]
- Twabi, H.S.; Manda, S.O.; Small, D.S. Assessing the effects of maternal HIV infection on pregnancy outcomes using cross-sectional data in Malawi. BMC Public Health 2020, 20, 974. [Google Scholar] [CrossRef]
- Theron, G.; Brummel, S.; Fairlie, L.; Pinilla, M.; McCarthy, K.; Owor, M.; Chinula, L.; Makanani, B.; Violari, A.; Moodley, D.; et al. Pregnancy Outcomes of Women Conceiving on Antiretroviral Therapy (ART) Compared to Those Commenced on ART During Pregnancy. Clin. Infect. Dis. 2020. [Google Scholar] [CrossRef] [PubMed]
- Tshivuila-Matala, C.O.; Honeyman, S.; Nesbitt, C.; Kirtley, S.; Kennedy, S.H.; Hemelaar, J. Adverse perinatal outcomes associated with antiretroviral therapy regimens: Systematic review and network meta-analysis. AIDS 2020, 34, 1643–1656. [Google Scholar] [CrossRef] [PubMed]
| Variable | Age in Years (Categories) N = 2352 | p-Value | |||
|---|---|---|---|---|---|
| ≤19 n = 729 | 20–24 n = 628 | ≥25 n = 995 | |||
| Demographic and clinical data | |||||
| HIV status 1 | Infected | 101 (14) | 195 (31) | 397 (40) | <0.001 |
| Uninfected | 628 (86) | 433 (69) | 598 (60) | ||
| Gravidity 1 | Primigravidae | 550 (75) | 89 (14) | 16 (2) | <0.001 |
| Multigravidas | 179 (25) | 539 (86) | 979 (98) | ||
| Weight at baseline (in kg) 2 | 58.1 (7.0) | 59.4 (7.8) | 62.3 (10.2) | <0.001 | |
| Gestational age at recruitment 2 | 21.1 (4.6) | 21.3 (5.0) | 21.7 (4.9) | 0.09 | |
| Anaemia (<11 g/dl Hb) 1 | Yes | 401 (55) | 331 (53) | 544 (55) | 0.66 |
| No | 328 (45) | 297 (48) | 451 (45) | ||
| MUAC at baseline 1 | Normal | 698 (96) | 616 (98) | 981 (99) | <0.001 |
| Lower or equal to 22 | 31 (4) | 12 (2) | 14 (1) | ||
| Literacy 1 | Literate | 592 (81) | 425 (68) | 548 (55) | <0.001 |
| Illiterate | 137 (19) | 203 (32) | 447 (45) | ||
| Data origin | |||||
| Trial 1 | Menéndez et al. 2008 | 216 (30) | 209 (33) | 256 (26) | <0.001 |
| González et al. 2014 (I) | 451 (62) | 276 (44) | 415 (42) | ||
| González et al. 2014 (II) | 62 (9) | 143 (23) | 324 (33) | ||
| Study arm 1 | Placebo | 132 (18) | 171 (27) | 295 (30) | <0.001 |
| MQ | 326 (45) | 262 (42) | 438 (44) | ||
| SP | 271 (37) | 195 (31) | 262 (26) | ||
| Variable | Study Outcome | ||||
|---|---|---|---|---|---|
| Hospital Admissions during Pregnancy n = 2352 | Outpatient Visits during Pregnancy n = 2352 | ||||
| Univariable analyses | |||||
| IRR (95% CI) | p-value | IRR (95% CI) | p-value | ||
| HIV status 1 | 1.39 (1.04–1.87) | 0.03 | 0.91 (0.77–1.08) | 0.28 | |
| Age in years | ≤19 | 1 | <0.01 | 1 | <0.001 |
| 20–24 | 0.60 (0.41–0.87) | 0.86 (0.71–1.05) | |||
| ≥25 | 0.67 (0.49–0.92) | 0.75 (0.63–0.90) | |||
| Multivariable analysis | |||||
| IRR (95% CI) | p-value | IRR (95% CI) | p-value | ||
| HIV status 1 | 1.28 (0.90–1.81) | 0.17 | 0.83 (0.69–1.00) | <0.05 | |
| Age in years | ≤19 | 1 | <0.01 | 1 | 0.02 |
| 20–24 | 0.55 (0.37–0.80) | 0.86 (0.71–1.04) | |||
| ≥25 | 0.60 (0.42–0.85) | 0.76 (0.63–0.92) | |||
| Variable | Study Outcome | ||||
|---|---|---|---|---|---|
| Miscarriages n = 2352 | Stillbirths n = 2352 | ||||
| Univariable analyses | |||||
| OR (95% CI) | p-value | OR (95% CI) | p-value | ||
| HIV status 1 | 1.38 (0.70–2.71) | 0.35 | 1.84 (1.14–2.97) | 0.01 | |
| Age in years | ≤19 | 1 | 0.30 | 1 | 0.73 |
| 20–24 | 1.16 (0.45–3.03) | 0.99 (0.52–1.89) | |||
| ≥25 | 1.78 (0.80–3.99) | 1.20 (0.69–2.10) | |||
| Multivariable analysis | |||||
| OR (95% CI) | p-value | OR (95% CI) | p-value | ||
| HIV status 1 | 0.93 (0.39–2.25) | 0.87 | 1.74 (0.99–3.04) | 0.05 | |
| Age in years | ≤19 | 1 | 0.25 | 1 | 0.78 |
| 20–24 | 1.10 (0.40–3.03) | 0.92 (0.47–1.79) | |||
| ≥25 | 1.91 (0.78–4.67) | 1.13 (0.62–2.06) | |||
| Variable | Study Outcome | ||||||
|---|---|---|---|---|---|---|---|
| Low Birth Weight n = 2299 | Small for Gestational Age n = 2045 | Premature Births n = 2045 | |||||
| Univariable analyses | |||||||
| OR (95% CI) | p-value | OR (95% CI) | p-value | OR (95% CI) | p-value | ||
| HIV status 1 | 1.09 (0.80–1.48) | 0.58 | 0.93 (0.74–1.16) | 0.50 | 0.99 (0.64–1.51) | 0.95 | |
| Age in years | ≤19 | 1 | <0.001 | 1 | <0.001 | 1 | <0.001 |
| 20–24 | 0.42 (0.29–0.61) | 0.52 (0.40–0.68) | 0.42 (0.24–0.71) | ||||
| ≥25 | 0.39 (0.28–0.55) | 0.46 (0.37–0.59) | 0.52 (0.34–0.79) | ||||
| Multivariable analysis | |||||||
| OR (95% CI) | p-value | OR (95% CI) | p-value | OR (95% CI) | p-value | ||
| HIV status 1 | 1.52 (1.07–2.16) | 0.02 | 1.10 (0.85–1.42) | 0.47 | 1.34 (0.83–2.16) | 0.23 | |
| Age in years | ≤19 | 1 | <0.001 | 1 | <0.001 | 1 | <0.01 |
| 20–24 | 0.40 (0.27–0.59) | 0.50 (0.38–0.65) | 0.42 (0.24–0.72) | ||||
| ≥25 | 0.37 (0.26–0.53) | 0.43 (0.34–0.56) | 0.51 (0.32–0.82) | ||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pons-Duran, C.; Casellas, A.; Bardají, A.; Valá, A.; Sevene, E.; Quintó, L.; Macete, E.; Menéndez, C.; González, R. Adolescent, Pregnant, and HIV-Infected: Risk of Adverse Pregnancy and Perinatal Outcomes in Young Women from Southern Mozambique. J. Clin. Med. 2021, 10, 1564. https://doi.org/10.3390/jcm10081564
Pons-Duran C, Casellas A, Bardají A, Valá A, Sevene E, Quintó L, Macete E, Menéndez C, González R. Adolescent, Pregnant, and HIV-Infected: Risk of Adverse Pregnancy and Perinatal Outcomes in Young Women from Southern Mozambique. Journal of Clinical Medicine. 2021; 10(8):1564. https://doi.org/10.3390/jcm10081564
Chicago/Turabian StylePons-Duran, Clara, Aina Casellas, Azucena Bardají, Anifa Valá, Esperança Sevene, Llorenç Quintó, Eusebio Macete, Clara Menéndez, and Raquel González. 2021. "Adolescent, Pregnant, and HIV-Infected: Risk of Adverse Pregnancy and Perinatal Outcomes in Young Women from Southern Mozambique" Journal of Clinical Medicine 10, no. 8: 1564. https://doi.org/10.3390/jcm10081564
APA StylePons-Duran, C., Casellas, A., Bardají, A., Valá, A., Sevene, E., Quintó, L., Macete, E., Menéndez, C., & González, R. (2021). Adolescent, Pregnant, and HIV-Infected: Risk of Adverse Pregnancy and Perinatal Outcomes in Young Women from Southern Mozambique. Journal of Clinical Medicine, 10(8), 1564. https://doi.org/10.3390/jcm10081564








