Extraperitoneal Robotic Laparo-Endoscopic Single-Site Plus1-Port Radical Prostatectomy Using the da Vinci Single-Site Platform
Abstract
1. Introduction
2. Materials and Methods
2.1. Patients
2.2. Device Description
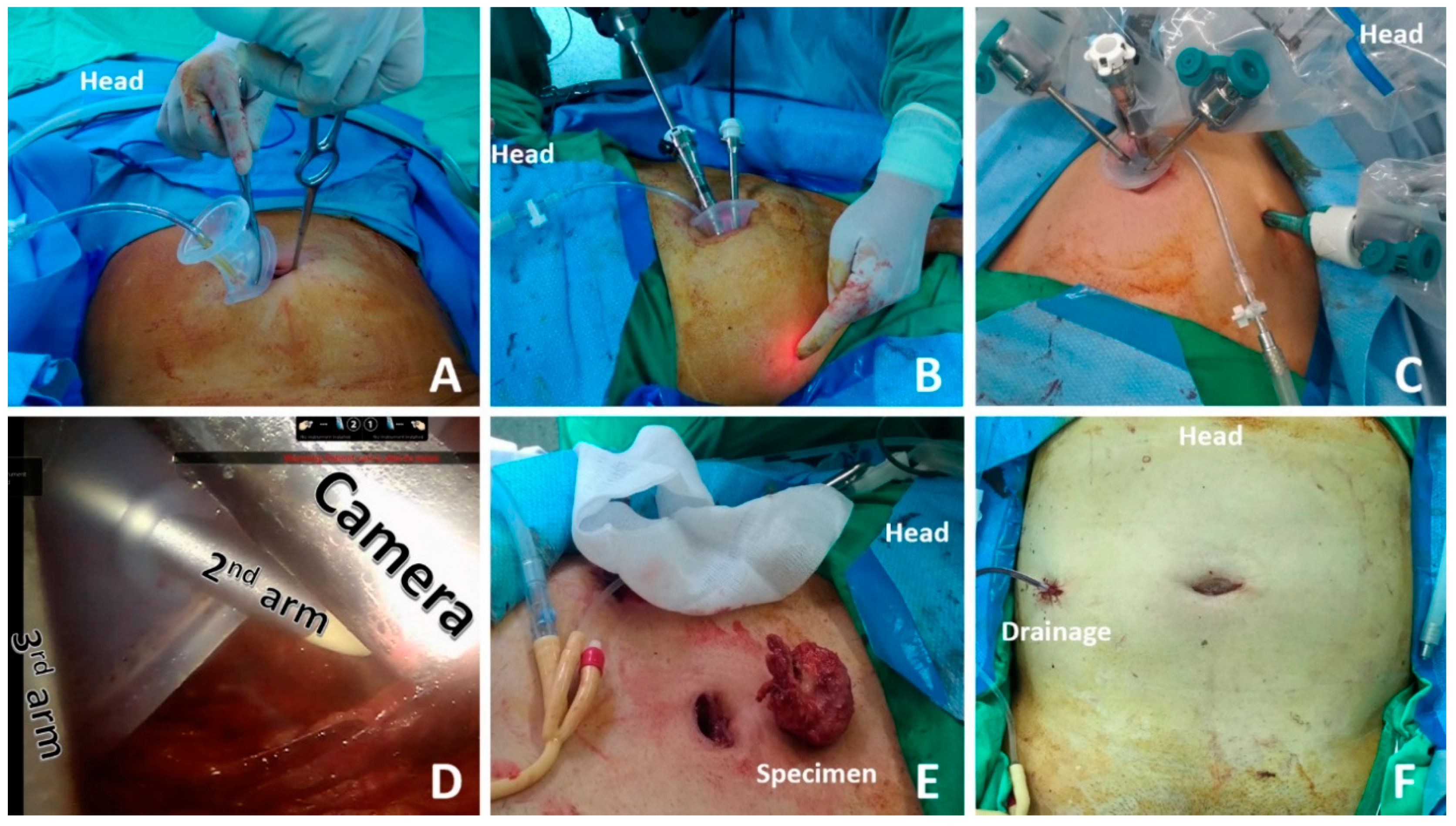
2.3. Surgical Technique
2.4. Statistic Method
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Schuessler, W.W.; Schulam, P.G.; Clayman, R.V.; Kavoussi, L.R. Laparoscopic radical prostatectomy: Initial short-term experience. Urology 1997, 50, 854–857. [Google Scholar] [CrossRef]
- Binder, J.; Kramer, W. Robotically-assisted laparoscopic radical prostatectomy. BJU Int. 2001, 87, 408–410. [Google Scholar] [CrossRef] [PubMed]
- Loulmet, D.; Carpentier, A.; d’Attellis, N.; Berrebi, A.; Cardon, C.; Ponzio, O.; Aupècle, B.; Relland, J.Y. Endoscopic coronary artery bypass grafting with the aid of robotic assisted instruments. J. Thorac. Cardiovasc. Surg. 1999, 118, 4–10. [Google Scholar] [CrossRef]
- Freire, M.P.; Choi, W.W.; Lei, Y.; Carvas, F.; Hu, J.C. Overcoming the learning curve for robotic-assisted laparoscopic radical prostatectomy. Urol. Clin. N. Am. 2010, 37, 37–47. [Google Scholar] [CrossRef] [PubMed]
- Martín, O.D.; Azhar, R.A.; Clavijo, R.; Gidelman, C.; Medina, L.; Troche, N.R.; Brunacci, L.; Sotelo, R. Single port radical prostatectomy: Current status. J. Robot. Surg. 2016, 10, 87–95. [Google Scholar] [CrossRef] [PubMed]
- Kaouk, J.; Bertolo, R.; Eltemamy, M.; Garisto, J. Single-port robot-assisted radical prostatectomy: First clinical experience using the SP surgical system. Urology 2019, 124, 309. [Google Scholar] [CrossRef] [PubMed]
- Gaboardi, F.; Pini, G.; Suardi, N.; Montorsi, F.; Passaretti, G.; Smelzo, S. Robotic laparoendoscopic single-site radical prostatectomy (R-LESS-RP) with daVinci Single-Site® platform. Concept and evolution of the technique following an IDEAL phase 1. J. Robot. Surg. 2019, 13, 215–226. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, D.K.; Sharma, V.; Toussi, A.; Viers, B.R.; Tollefson, M.K.; Gettman, M.T.; Frank, I. Initial Experience with da Vinci Single-port Robot-assisted Radical Prostatectomies. Eur. Urol. 2020, 77, 373–379. [Google Scholar] [CrossRef] [PubMed]
- Dobbs, R.W.; Magnan, B.P.; Abhyankar, N.; Hemal, A.K.; Challacombe, B.; Hu, J.; Dasgupta, P.; Porpiglia, F.; Crivellaro, S. Cost effectiveness and robot-assisted urologic surgery: Does it make dollars and sense? Minerva Urol. Nefrol. 2017, 69, 313–323. [Google Scholar] [PubMed]
- Mattevi, D.; Luciani, L.G.; Vattovani, V.; Chiodini, S.; Puglisi, M.; Malossini, G. First case of robotic laparoendoscopic single-site radical prostatectomy with single-site VesPa platform. J. Robot. Surg. 2018, 12, 381–385. [Google Scholar] [CrossRef] [PubMed]
- Freedland, S.J.; Sutter, M.E.; Dorey, F.; Aronson, W.J. Defining the ideal cutpoint for determining PSA recurrence after radical prostatectomy. Prostate-specific antigen. Urology 2003, 61, 365–369. [Google Scholar] [CrossRef]
- Holze, S.; Mende, M.; Healy, K.V.; Koehler, N.; Gansera, L.; Truss, M.C.; Rebmann, U.; Degener, S.; Stolzenburg, J.U. Comparison of various continence definitions in a large group of patients undergoing radical prostatectomy: A multicentre, prospective study. BMC Urol. 2019, 19, 70. [Google Scholar] [CrossRef] [PubMed]
- Lo, I.S.; Lee, H.Y.; Chou, Y.H.; Huang, C.N.; Wu, W.J.; Yeh, H.C.; Yang, K.F.; Lee, C.H.; Li, C.C. Robot-assisted extraperitoneal radical prostatectomy, single site plus two model. J. Laparoendosc. Adv. Surg. Tech. A 2018, 28, 140–144. [Google Scholar] [CrossRef] [PubMed]
- Kaouk, J.; Aminsharifi, A.; Wilson, C.A.; Sawczyn, G.; Garisto, J.; Francavilla, S.; Abern, M.; Crivellaro, S. Extraperitoneal versus Transperitoneal Single Port Robotic Radical Prostatectomy: A Comparative Analysis of Periopera-tive Outcomes. J. Urol. 2020, 203, 1135–1140. [Google Scholar] [CrossRef] [PubMed]
- Weight, C.J.; Reuther, A.M.; Gunn, P.W.; Zippe, C.R.; Dhar, N.B.; Klein, E.A. Limited pelvic lymph node dissection does not improve biochemical relapse-free survival at 10 years after radical prostatectomy in patients with low-risk prostate cancer. Urology 2008, 71, 141–145. [Google Scholar] [CrossRef] [PubMed]
| Parameter | Cohort (n = 120) |
|---|---|
| Age at RP (years), Mean (range) | 67.1 (49.0–78.0) |
| BMI (kg/m2), Mean (range) | 24.9 (18.8–33.8) |
| PSA level before RP (ng/mL), Mean (range) | 15.0 (0.4–95.8) |
| cT stage before RP, n (%) | |
| cT1 | 11 (9%) |
| cT2a/b/c | 91 (76%) |
| cT3 | 18 (15%) |
| pT stage at RP, n (%) | |
| pTx | 3 (3%) |
| pT2 | 68 (57%) |
| pT3 | 49 (41%) |
| pGleason score at RP, n (%) | |
| 6 (grade group 1) | 46 (38%) |
| 7 (grade group 2–3) | 45 (38%) |
| 8–10 (grade group 4–5) | 29 (24%) |
| Lymph node dissection | 61 (51%) |
| Prostate wright (g), Mean (range) | 41 (33–54) |
| Parameter | Cohort (n = 120) |
|---|---|
| Extraperitonium setting time (min), Mean (range) | 29.8 (10.0–120.0) |
| Robotic console time (min), Mean (range) | 140.1 (54.0–270.0) |
| Operation time (min), Mean (range) | 243.1 (115.0–440.0) |
| Length of stay (days), Mean (range) | 9.1 (5.0–17.0) |
| Catheter duration (days), Mean (range) | 6.7 (4.0–14.0) |
| EBL (mL), Mean (range) | 467.1 (20.0–1600.0) |
| Positive margin, n (%) | 43.0 (36%) |
| Complications | |
| Clavien I | 0 |
| Clavien II | 11 (9.2%) |
| Clavien III | 0 |
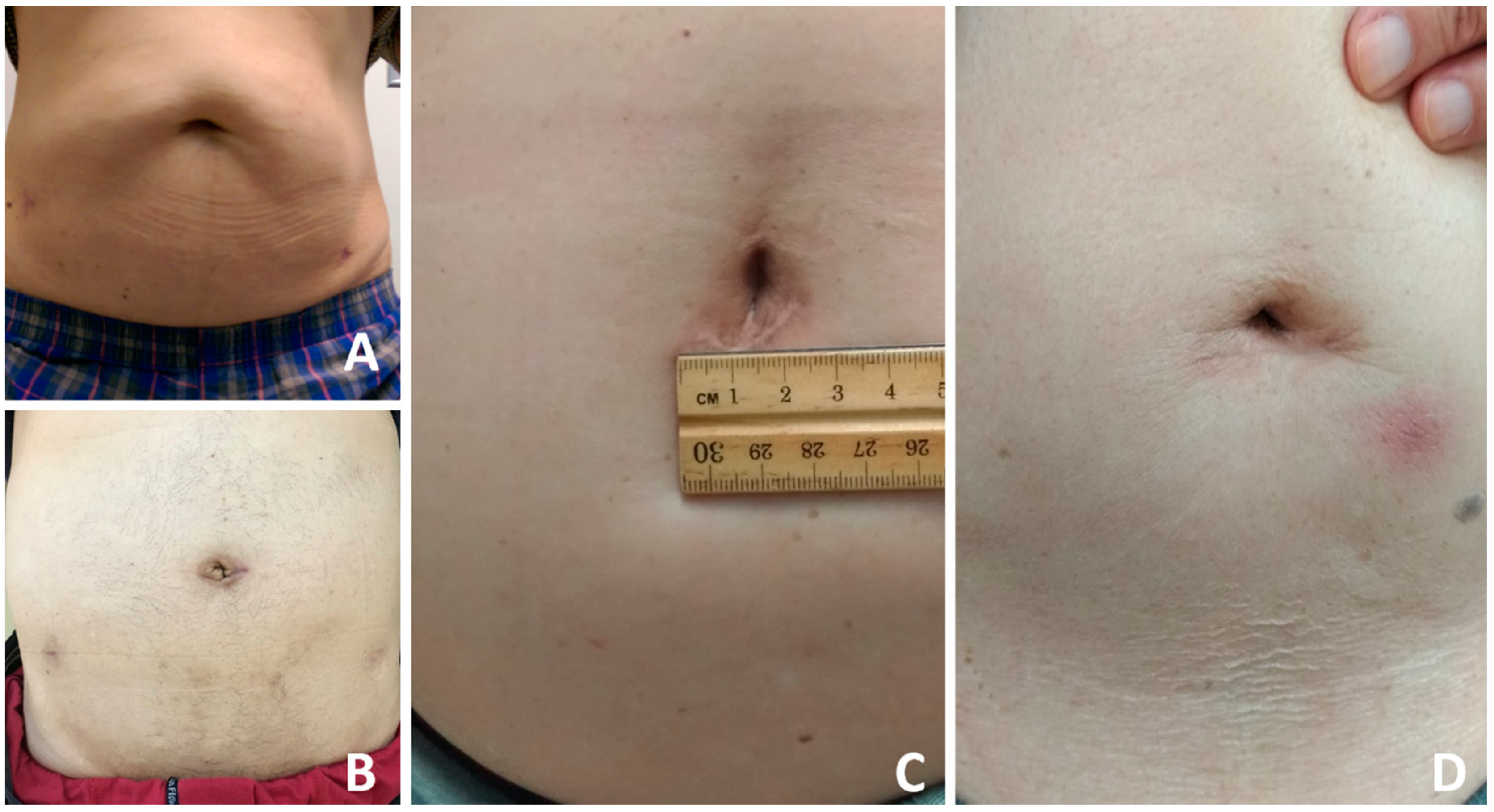
| Umbilical wound size (cm), Mean (range) | 3.1 (2.6–4.1) |
| Detectable PSA after RP, n (%) | 17 (14%) |
| Incontinence rate, n (%) | 2 (1%) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, C.-C.; Chien, T.-M.; Lee, M.-R.; Lee, H.-Y.; Ke, H.-L.; Wen, S.-C.; Chou, Y.-H.; Wu, W.-J. Extraperitoneal Robotic Laparo-Endoscopic Single-Site Plus1-Port Radical Prostatectomy Using the da Vinci Single-Site Platform. J. Clin. Med. 2021, 10, 1563. https://doi.org/10.3390/jcm10081563
Li C-C, Chien T-M, Lee M-R, Lee H-Y, Ke H-L, Wen S-C, Chou Y-H, Wu W-J. Extraperitoneal Robotic Laparo-Endoscopic Single-Site Plus1-Port Radical Prostatectomy Using the da Vinci Single-Site Platform. Journal of Clinical Medicine. 2021; 10(8):1563. https://doi.org/10.3390/jcm10081563
Chicago/Turabian StyleLi, Ching-Chia, Tsu-Ming Chien, Ming-Ru Lee, Hsiang-Ying Lee, Hung-Lung Ke, Sheng-Chen Wen, Yii-Her Chou, and Wen-Jeng Wu. 2021. "Extraperitoneal Robotic Laparo-Endoscopic Single-Site Plus1-Port Radical Prostatectomy Using the da Vinci Single-Site Platform" Journal of Clinical Medicine 10, no. 8: 1563. https://doi.org/10.3390/jcm10081563
APA StyleLi, C.-C., Chien, T.-M., Lee, M.-R., Lee, H.-Y., Ke, H.-L., Wen, S.-C., Chou, Y.-H., & Wu, W.-J. (2021). Extraperitoneal Robotic Laparo-Endoscopic Single-Site Plus1-Port Radical Prostatectomy Using the da Vinci Single-Site Platform. Journal of Clinical Medicine, 10(8), 1563. https://doi.org/10.3390/jcm10081563






