Tuberculosis in the Elderly
Abstract
1. Introduction
2. Epidemiology
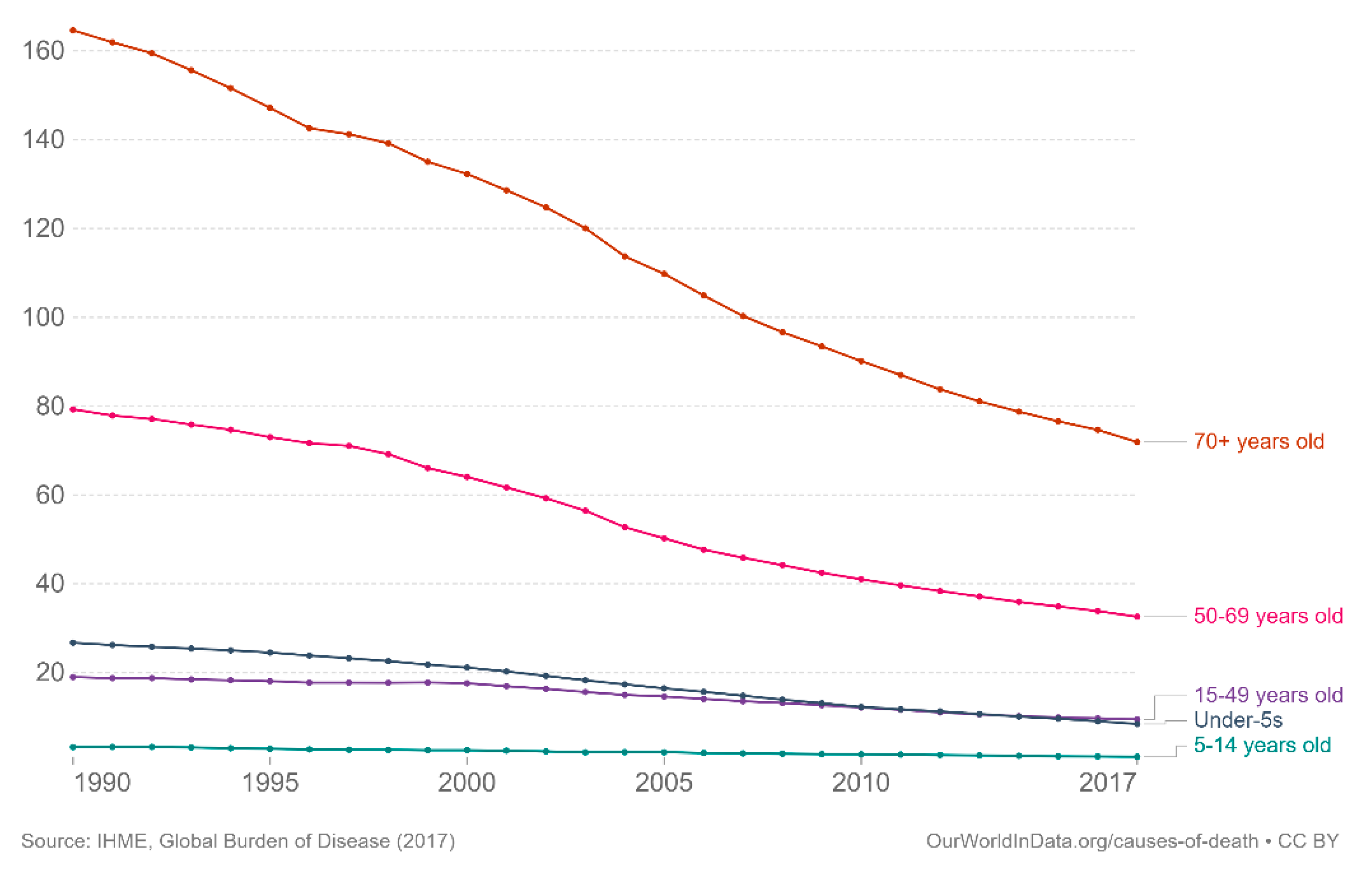
2.1. TB Is a Common Communicable Disease, including in the Elderly, Although It Is Decreasing
2.2. TB Outcome
2.3. Drug-Resistant TB
2.4. HIV and TB in the Elderly
3. Pathophysiology
3.1. Modified Pulmonary Function in the Elderly and Structural Changes in the Senescent Lung Promote Lung Infection
3.2. Risk Factors of TB in the Elderly Are Linked Simultaneously to Various Parameters
4. Diagnosis
4.1. Clinical Findings
4.2. Radiological Findings
4.3. Microbiological Findings
5. Treatment
5.1. Treatment Tolerance
5.2. Pharmacokinetic Issues and Drug-to-Drug Interaction
6. Outcome
6.1. Specific Data in Elderly People
6.2. Paradoxical Reaction during TB Treatment
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- World Health Organization. Available online: www.Who.Int/Tb/Dat (accessed on 18 October 2021).
- Global Tuberculosis Report 2020. Available online: https://www.who.int/publications-detail-redirect/9789240013131 (accessed on 18 October 2021).
- Tuberculose. Available online: https://www.santepubliquefrance.fr/maladies-et-traumatismes/maladies-et-infections-respiratoires/tuberculose (accessed on 18 October 2021).
- Stead, W.W. Tuberculosis among elderly persons, as observed among nursing home residents. Int. J. Tuberc. Lung Dis. Off. J. Int. Union Tuberc. Lung Dis. 1998, 2, S64–S70. [Google Scholar]
- Orme, I.M. A Mouse Model of the Recrudescence of Latent Tuberculosis in the Elderly. Am. Rev. Respir. Dis. 1988, 137, 716–718. [Google Scholar] [CrossRef]
- Guthmann, J.-P. Les issues de traitement des tuberculoses sensibles déclarées en France, 2008–2014/Treatment outcome of non-resistant tuberculosis cases reported in France, 2008–2014. Bull. Epidémiol. Hebd. 2018, 10, 6–7. [Google Scholar]
- Guthmann, J.-P.; Léon, L.; Antoine, D.; Lévy-Bruhl, D. Tuberculosis treatment outcomes of notified cases: Trends and determinants of potential unfavourable outcome, France, 2008 to 2014. Eurosurveillance 2020, 25, 1900191. [Google Scholar] [CrossRef]
- Global Burden of Disease Study 2017 (GBD 2017) Population and Fertility 1950–2017|GHDx. Available online: http://ghdx.healthdata.org/record/ihme-data/gbd-2017-population-and-fertility-1950–2017 (accessed on 19 October 2021).
- Hauer, B.; Brodhun, B.; Altmann, D.; Fiebig, L.; Loddenkemper, R.; Haas, W. Tuberculosis in the elderly in Germany. Eur. Respir. J. 2011, 38, 467–470. [Google Scholar] [CrossRef] [PubMed]
- Guaraldi, G.; Zona, S.; Brothers, T.D.; Carli, F.; Stentarelli, C.; Dolci, G.; Santoro, A.; Beghetto, B.; Menozzi, M.; Mussini, C.; et al. Aging with HIV vs. HIV Seroconversion at Older Age: A Diverse Population with Distinct Comorbidity Profiles. PLoS ONE 2015, 10, e0118531. [Google Scholar] [CrossRef] [PubMed]
- Mcnicoll, G. World population ageing, 1950–2050. Choice Rev. Online 2002, 40, 40–1307. [Google Scholar] [CrossRef]
- Smit, M.; Brinkman, K.; Geerlings, S.; Smit, C.; Thyagarajan, K.; van Sighem, A.; de Wolf, F.; Hallett, T. Athena observational cohort Future Challenges for Clinical Care of an Ageing Population Infected with HIV: A Modelling Study. Lancet Infect. Dis. 2015, 15, 810–818. [Google Scholar] [CrossRef]
- Tran, T.-C. Découvertes de séropositivité VIH chez les seniors en France, 2008–2016/New HIV diagnoses in people aged 50 years or older in France, 2008–2016. Bull. Épidémiol. Hebd. 2018, 7, 792–798. [Google Scholar]
- Ford, N.; Matteelli, A.; Shubber, Z.; Hermans, S.; Meintjes, G.; Grinsztejn, B.; Waldrop, G.; Kranzer, K.; Doherty, M.; Getahun, H. TB as a cause of hospitalization and in-hospital mortality among people living with HIV worldwide: A systematic review and meta-analysis. J. Int. AIDS Soc. 2016, 19, 20714. [Google Scholar] [CrossRef]
- Meintjes, G.; Brust, J.C.M.; Nuttall, J.; Maartens, G. Management of active tuberculosis in adults with HIV. Lancet HIV 2019, 6, e463–e474. [Google Scholar] [CrossRef]
- Janssens, J.-P.; Krause, K.-H. Pneumonia in the very old. Lancet Infect. Dis. 2004, 4, 112–124. [Google Scholar] [CrossRef]
- Akgün, K.M.; Crothers, K.; Pisani, M.A. Epidemiology and Management of Common Pulmonary Diseases in Older Persons. J. Gerontol. Ser. A Boil. Sci. Med. Sci. 2012, 67, 276–291. [Google Scholar] [CrossRef] [PubMed]
- Gavazzi, G.; Krause, K.-H. Ageing and infection. Lancet Infect. Dis. 2002, 2, 659–666. [Google Scholar] [CrossRef]
- Müller, L.; Di Benedetto, S.; Pawelec, G. The Immune System and Its Dysregulation with Aging. Subcell. Biochem. 2019, 91, 21–43. [Google Scholar]
- Solana, R.; Tarazona, R.; Aiello, A.E.; Akbar, A.N.; Appay, V.; Beswick, M.; Bosch, J.A.; Campos, C.; Cantisán, S.; Cicin-Sain, L.; et al. CMV and Immunosenescence: From basics to clinics. Immun. Ageing 2012, 9, 23. [Google Scholar] [CrossRef] [PubMed]
- Franceschi, C.; Capri, M.; Monti, D.; Giunta, S.; Olivieri, F.; Sevini, F.; Panourgia, M.P.; Invidia, L.; Celani, L.; Scurti, M.; et al. Inflammaging and anti-inflammaging: A systemic perspective on aging and longevity emerged from studies in humans. Mech. Ageing Dev. 2007, 128, 92–105. [Google Scholar] [CrossRef]
- Piergallini, T.J.; Turner, J. Tuberculosis in the elderly: Why inflammation matters. Exp. Gerontol. 2018, 105, 32–39. [Google Scholar] [CrossRef] [PubMed]
- Yew, W.W.; Yoshiyama, T.; Leung, C.C.; Chan, D.P. Epidemiological, clinical and mechanistic perspectives of tuberculosis in older people. Respirology 2018, 23, 567–575. [Google Scholar] [CrossRef] [PubMed]
- Davies, P.D.O. Tuberculosis in the elderly. J. Antimicrob. Chemother. 1994, 34 (Suppl. A), 93–100. [Google Scholar] [CrossRef]
- Packham, S. Tuberculosis in the Elderly. Gerontology 2001, 47, 175–179. [Google Scholar] [CrossRef] [PubMed]
- Hatipoglu, O.N.; Osma, E.; Manisali, M.; Ucan, E.S.; Balci, P.; Akkoclu, A.; Akpinar, O.; Karlikaya, C.; Yuksel, C. High resolution computed tomographic findings in pulmonary tuberculosis. Thorax 1996, 51, 397–402. [Google Scholar] [CrossRef]
- Abbara, A.; Collin, S.M.; Kon, O.M.; Buell, K.; Sullivan, A.; Barrett, J.; Corrah, T.; McGregor, A.; Hansel, T.; John, L.; et al. Time to diagnosis of tuberculosis is greater in older patients: A retrospective cohort review. ERJ Open Res. 2019, 5, 00228–02018. [Google Scholar] [CrossRef]
- Teale, C.; Goldman, J.M.; Pearson, S.B. The Association of Age with the Presentation and Outcome of Tuberculosis: A Five-year Survey. Age Ageing 1993, 22, 289–293. [Google Scholar] [CrossRef]
- Pratt, R.H.; Winston, C.A.; Kammerer, J.S.; Armstrong, L.R. Tuberculosis in Older Adults in the United States, 1993–2008. J. Am. Geriatr. Soc. 2011, 59, 851–857. [Google Scholar] [CrossRef]
- Rajaram, M.; Malik, A.; Mohapatra, M.M.; Vijayageetha, M.; Babu, V.M.; Vally, S.; Saka, V.K. Comparison of Clinical, Radiological and Laboratory Parameters between Elderly and Young Patient with Newly Diagnosed Smear Positive Pulmonary Tuberculosis: A Hospital-Based Cross Sectional Study. Cureus 2020, 12, e8319. [Google Scholar] [CrossRef]
- Neffati, O.; Gharsalli, H.; Aouadi, S.; Hafaeid, S.; Gheriani, A.; Naji, H.; Maâlej, S.; El Gharbi, L.D. Particularités de la tuberculose pulmonaire chez le sujet âgé. Rev. des Mal. Respir. 2016, 33, A146. [Google Scholar] [CrossRef]
- Bommart, S.; Charriot, J.; Nagot, N.; Vernhet-Kovacsik, H.; Revel, M.P.; Boissin, C.; Bourdin, A.; Tuaillon, E. Differentiating between active and latent tuberculosis with chest computed tomography. Diagn. Interv. Imaging 2021, 102, 605–610. [Google Scholar] [CrossRef]
- Choe, J.; Jung, K.H.; Park, J.-H.; Kim, S.-H.; Kim, M.Y. Clinical and radiologic characteristics of radiologically missed miliary tuberculosis. Medicine 2021, 100, e23833. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.H.; Han, D.H.; Song, J.W.; Chung, H.S. Diagnostic and Therapeutic Problems of Pulmonary Tuberculosis in Elderly Patients. J. Korean Med. Sci. 2005, 20, 784–789. [Google Scholar] [CrossRef]
- Fujimoto, S.; Saito, K.; Matsubayashi, S.; Sato, A.; Yamada, M.; Yamanaka, Y.; Fujisaki, I.; Odashima, K.; Seki, A.; Kazuyori, T.; et al. Clinical utility of thoracoscopy in elderly tuberculous pleurisy patients under local anesthesia. J. Infect. Chemother. Off. J. Jpn. Soc. Chemother. 2021, 27, 40–44. [Google Scholar] [CrossRef]
- Rai, S.; Thomas, W.M. Diagnosis of abdominal tuberculosis: The importance of laparoscopy. J. R. Soc. Med. 2003, 96, 586–588. [Google Scholar] [CrossRef] [PubMed]
- Diacon, A.H.; Van De Wal, B.W.; Wyser, C.; Smedema, J.-P.; Bezuidenhout, J.; Bolliger, C.T.; Walzl, G. Diagnostic tools in tuberculous pleurisy: A direct comparative study. Eur. Respir. J. 2003, 22, 589–591. [Google Scholar] [CrossRef] [PubMed]
- Murray, P.R.; Elmore, C.; Krogstad, N.J. The Acid-Fast Stain: A Specific and Predictive Test for Mycobacterial Disease. Ann. Intern. Med. 1980, 92, 512–513. [Google Scholar] [CrossRef] [PubMed]
- Sharma, S.K.; Mohan, A.; Sharma, A. Challenges in the diagnosis & treatment of miliary tuberculosis. Indian J. Med. Res. 2012, 135, 703–730. [Google Scholar] [PubMed]
- Espasa, M.; Salvado, M.; Vicente, E.; Tudo, G.; Alcaide, F.; Coll, P.; Martin-Casabona, N.; Torra, M.; Fontanals, D.; Gonzalez-Martin, J. Evaluation of the VersaTREK System Compared to the Bactec MGIT 960 System for First-Line Drug Susceptibility Testing of Mycobacterium tuberculosis. J. Clin. Microbiol. 2011, 50, 488–491. [Google Scholar] [CrossRef]
- Yang, X.; Che, N.; Duan, H.; Liu, Z.; Li, K.; Li, H.; Guo, C.; Liang, Q.; Yang, Y.; Wang, Y.; et al. Cell-free Mycobacterium tuberculosis DNA test in pleural effusion for tuberculous pleurisy: A diagnostic accuracy study. Clin. Microbiol. Infect. Off. Publ. Eur. Soc. Clin. Microbiol. Infect. Dis. 2020, 26, 1089.e1–1089.e6. [Google Scholar] [CrossRef]
- Li, X.; Du, W.; Wang, Y.; Liu, Z.; Li, K.; Chen, H.; Liu, R.; Ma, L.; Zhang, L.; Dong, Y.; et al. Rapid Diagnosis of Tuberculosis Meningitis by Detecting Mycobacterium tuberculosis Cell-Free DNA in Cerebrospinal Fluid. Am. J. Clin. Pathol. 2019, 153, 126–130. [Google Scholar] [CrossRef]
- Zhu, N.; Zhou, D.; Li, S. Diagnostic Accuracy of Metagenomic Next-Generation Sequencing in Sputum-Scarce or Smear-Negative Cases with Suspected Pulmonary Tuberculosis. BioMed Res. Int. 2021, 2021, 9970817. [Google Scholar] [CrossRef]
- Arora, V.; Chopra, K. Geriatric TB: Needs focussed attention under RNTCP. Indian J. Tuberc. 2019, 66, 516–519. [Google Scholar] [CrossRef]
- Ferrara, G.; Losi, M.; D’Amico, R.; Cagarelli, R.; Pezzi, A.; Meacci, M.; Meccugni, B.; Dori, I.M.; Rumpianesi, F.; Roversi, P.; et al. Interferon-Gamma-Release Assays Detect Recent Tuberculosis Re-Infection in Elderly Contacts. Int. J. Immunopathol. Pharmacol. 2009, 22, 669–677. [Google Scholar] [CrossRef] [PubMed]
- Bae, W.; Park, K.U.; Song, E.Y.; Kim, S.J.; Lee, Y.J.; Park, J.S.; Cho, Y.-J.; Yoon, H.I.; Yim, J.-J.; Lee, C.-T.; et al. Comparison of the Sensitivity of QuantiFERON-TB Gold In-Tube and T-SPOT.TB According to Patient Age. PLoS ONE 2016, 11, e0156917. [Google Scholar] [CrossRef] [PubMed]
- Kobashi, Y.; Mouri, K.; Miyashita, N.; Okimoto, N.; Matsushima, T.; Kageoka, T.; Oka, M. QuantiFERON TB-2G test for patients with active tuberculosis stratified by age groups. Scand. J. Infect. Dis. 2009, 41, 841–846. [Google Scholar] [CrossRef]
- Kobashi, Y.; Sugiu, T.; Mouri, K.; Obase, Y.; Miyashita, N.; Oka, M. Indeterminate results of QuantiFERON TB-2G test performed in routine clinical practice. Eur. Respir. J. 2009, 33, 812–815. [Google Scholar] [CrossRef]
- Kobashi, Y.; Mouri, K.; Yagi, S.; Obase, Y.; Miyashita, N.; Okimoto, N.; Matsushima, T.; Kageoka, T.; Oka, M. Clinical Utility of the QuantiFERON TB-2G Test for Elderly Patients with Active Tuberculosis. Chest 2008, 133, 1196–1202. [Google Scholar] [CrossRef]
- Stead, W.W.; Lofgren, J.P. Does the Risk of Tuberculosis Increase in Old Age? J. Infect. Dis. 1983, 147, 951–955. [Google Scholar] [CrossRef]
- Tebruegge, M.; De Graaf, H.; Sukhtankar, P.; Elkington, P.; Marshall, B.; Schuster, H.; Patel, S.; Faust, S.N. Extremes of Age Are Associated with Indeterminate QuantiFERON-TB Gold Assay Results. J. Clin. Microbiol. 2014, 52, 2694–2697. [Google Scholar] [CrossRef]
- Chee, C.B.E.; Gan, S.H.; KhinMar, K.W.; Barkham, T.M.; Koh, C.K.; Liang, S.; Wang, Y.T. Comparison of Sensitivities of Two Commercial Gamma Interferon Release Assays for Pulmonary Tuberculosis. J. Clin. Microbiol. 2008, 46, 1935–1940. [Google Scholar] [CrossRef]
- Le Hang, N.T.; Lien, L.T.; Kobayashi, N.; Shimbo, T.; Sakurada, S.; Thuong, P.H.; Hong, L.T.; Tam, D.B.; Hijikata, M.; Matsushita, I.; et al. Analysis of Factors Lowering Sensitivity of Interferon-γ Release Assay for Tuberculosis. PLoS ONE 2011, 6, e23806. [Google Scholar] [CrossRef]
- Stead, W.W. Tuberculosis among Elderly Persons: An Outbreak in a Nursing Home. Ann. Intern. Med. 1981, 94, 606–610. [Google Scholar] [CrossRef]
- Khan, M.; Kovnat, D.M.; Bachus, B.; Whitcomb, M.E.; Brody, J.S.; Snider, G.L. Clinical and roentgenographic spectrum of pulmonary tuberculosis in the adult. Am. J. Med. 1977, 62, 31–38. [Google Scholar] [CrossRef]
- Morris, C.D. Pulmonary tuberculosis in the elderly: A different disease? Thorax 1990, 45, 912–913. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Oshi, D.C.; Oshi, S.N.; Alobu, I.; Ukwaja, K.N. Profile and Treatment Outcomes of Tuberculosis in the Elderly in Southeastern Nigeria, 2011–2012. PLoS ONE 2014, 9, e111910. [Google Scholar] [CrossRef]
- Nagu, T.; Ray, R.; Munseri, P.; Moshiro, C.; Shayo, G.; Kazema, R.; Mugusi, F.; Pallangyo, K. Tuberculosis among the elderly in Tanzania: Disease presentation and initial response to treatment. Int. J. Tuberc. Lung Dis. Off. J. Int. Union Tuberc. Lung Dis. 2017, 21, 1251–1257. [Google Scholar] [CrossRef]
- Velayutham, B.R.V.; Nair, D.; Chandrasekaran, V.; Raman, B.; Sekar, G.; Watson, B.; Charles, N.; Malaisamy, M.; Thomas, A.; Swaminathan, S. Profile and Response to Anti-Tuberculosis Treatment among Elderly Tuberculosis Patients Treated under the TB Control Programme in South India. PLoS ONE 2014, 9, e88045. [Google Scholar] [CrossRef]
- Stead, A.W.W.; Dutt, A.K. Tuberculosis in Elderly Persons. Annu. Rev. Med. 1991, 42, 267–276. [Google Scholar] [CrossRef]
- Kwon, Y.S.; Chi, S.Y.; Oh, I.J.; Kim, K.S.; Kim, Y.I.; Lim, S.C.; Kim, Y.C. Clinical characteristics and treatment outcomes of tuberculosis in the elderly: A case control study. BMC Infect. Dis. 2013, 13, 121. [Google Scholar] [CrossRef]
- Yee, D.; Valiquette, C.; Pelletier, M.; Parisien, I.; Rocher, I.; Menzies, D. Incidence of Serious Side Effects from First-Line Anti-tuberculosis Drugs among Patients Treated for Active Tuberculosis. Am. J. Respir. Crit. Care Med. 2003, 167, 1472–1477. [Google Scholar] [CrossRef]
- Teleman, M.D.; Chee, C.B.E.; Earnest, A.; Wang, Y.T. Hepatotoxicity of tuberculosis chemotherapy under general programme conditions in Singapore. Int. J. Tuberc. Lung Dis. Off. J. Int. Union Tuberc. Lung Dis. 2002, 6, 699–705. [Google Scholar]
- Saukkonen, J.J.; Cohn, D.L.; Jasmer, R.M.; Schenker, S.; Jereb, J.A.; Nolan, C.M.; Peloquin, C.A.; Gordin, F.M.; Nunes, D.; Strader, D.B.; et al. An Official ATS Statement: Hepatotoxicity of Anti-tuberculosis Therapy. Am. J. Respir. Crit. Care Med. 2006, 174, 935–952. [Google Scholar] [CrossRef]
- Chang, K.C.; Leung, C.C.; Yew, W.W.; Lau, T.Y.; Tam, C.M. Hepatotoxicity of Pyrazinamide: Cohort and Case-Control Analyses. Am. J. Respir. Crit. Care Med. 2008, 177, 1391–1396. [Google Scholar] [CrossRef] [PubMed]
- Toren, K.G.; Spitters, C.; Pecha, M.; Bhattarai, S.; Horne, D.J.; Narita, M. Tuberculosis in Older Adults: Seattle & King County, Washington. Clin. Infect. Dis. Off. Publ. Infect. Dis. Soc. Am. 2019, 70, 1202–1207. [Google Scholar] [CrossRef]
- Rousset, S.; Lafaurie, M.; Guet-Revillet, H.; Protin, C.; Le Grusse, J.; Derumeaux, H.; Gandia, P.; Nourhashemi, F.; Sailler, L.; Sommet, A.; et al. Safety of Pyrazinamide for the Treatment of Tuberculosis in Older Patients Over 75 Years of Age: A Retrospective Monocentric Cohort Study. Drugs Aging 2020, 38, 43–52. [Google Scholar] [CrossRef] [PubMed]
- Hase, I.; Toren, K.G.; Hirano, H.; Sakurai, K.; Horne, D.J.; Saito, T.; Narita, M. Pulmonary Tuberculosis in Older Adults: Increased Mortality Related to Tuberculosis within Two Months of Treatment Initiation. Drugs Aging 2021, 38, 807–815. [Google Scholar] [CrossRef]
- Nahid, P.; Dorman, S.E.; Alipanah, N.; Barry, P.M.; Brozek, J.L.; Cattamanchi, A.; Chaisson, L.H.; Chaisson, R.E.; Daley, C.L.; Grzemska, M.; et al. Official American Thoracic Society/Centers for Disease Control and Prevention/Infectious Diseases Society of America Clinical Practice Guidelines: Treatment of Drug-Susceptible Tuberculosis. Clin. Infect. Dis. Off. Publ. Infect. Dis. Soc. Am. 2016, 63, e147–e195. [Google Scholar] [CrossRef]
- Hagiwara, E.; Suido, Y.; Asaoka, M.; Katano, T.; Okuda, R.; Sekine, A.; Kitamura, H.; Baba, T.; Komatsu, S.; Ogura, T. Safety of pyrazinamide-including regimen in late elderly patients with pulmonary tuberculosis: A prospective randomized open-label study. J. Infect. Chemother. Off. J. Jpn. Soc. Chemother. 2019, 25, 1026–1030. [Google Scholar] [CrossRef]
- Desta, Z.; Soukhova, N.V.; Flockhart, D.A. Inhibition of Cytochrome P450 (CYP450) Isoforms by Isoniazid: Potent Inhibition of CYP2C19 and CYP3A. Antimicrob. Agents Chemother. 2001, 45, 382–392. [Google Scholar] [CrossRef]
- Alsultan, A.; Peloquin, C.A. Therapeutic Drug Monitoring in the Treatment of Tuberculosis: An Update. Drugs 2014, 74, 839–854. [Google Scholar] [CrossRef]
- Fried, L.P.; Tangen, C.M.; Walston, J.; Newman, A.B.; Hirsch, C.; Gottdiener, J.; Seeman, T.; Tracy, R.; Kop, W.J.; Burke, G.; et al. Frailty in Older Adults: Evidence for a Phenotype. J. Gerontol. A. Biol. Sci. Med. Sci. 2001, 56, M146–M157. [Google Scholar] [CrossRef]
- Turner, G.; Clegg, A. Best Practice Guidelines for the Management of Frailty: A British Geriatrics Society, Age UK and Royal College of General Practitioners Report. Age Ageing 2014, 43, 744–747. [Google Scholar] [CrossRef] [PubMed]
- Rolfson, D.B.; Majumdar, S.R.; Tsuyuki, R.T.; Tahir, A.; Rockwood, K. Validity and Reliability of the Edmonton Frail Scale. Age Ageing 2006, 35, 526–529. [Google Scholar] [CrossRef]
- Grobler, L.; Nagpal, S.; Sudarsanam, T.D.; Sinclair, D. Nutritional Supplements for People Being Treated for Active Tuberculosis. Cochrane Database Syst. Rev. 2016, CD006086. [Google Scholar] [CrossRef]
- Visser, M.E.; Texeira-Swiegelaar, C.; Maartens, G. The Short-Term Effects of Anti-Tuberculosis Therapy on Plasma Pyridoxine Levels in Patients with Pulmonary Tuberculosis. Int. J. Tuberc. Lung Dis. Off. J. Int. Union Tuberc. Lung Dis. 2004, 8, 260–262. [Google Scholar]
- Furin, J.; Cox, H.; Pai, M. Tuberculosis. Lancet Lond. Engl. 2019, 393, 1642–1656. [Google Scholar] [CrossRef]
- Odone, A.; Roberts, B.; Dara, M.; van den Boom, M.; Kluge, H.; McKee, M. People- and Patient-Centred Care for Tuberculosis: Models of Care for Tuberculosis. Int. J. Tuberc. Lung Dis. Off. J. Int. Union Tuberc. Lung Dis. 2018, 22, 133–138. [Google Scholar] [CrossRef]
- Story, A.; Aldridge, R.W.; Smith, C.M.; Garber, E.; Hall, J.; Ferenando, G.; Possas, L.; Hemming, S.; Wurie, F.; Luchenski, S.; et al. Smartphone-Enabled Video-Observed versus Directly Observed Treatment for Tuberculosis: A Multicentre, Analyst-Blinded, Randomised, Controlled Superiority Trial. Lancet Lond. Engl. 2019, 393, 1216–1224. [Google Scholar] [CrossRef]
- Dooley, D.P.; Carpenter, J.L.; Rademacher, S. Adjunctive Corticosteroid Therapy for Tuberculosis: A Critical Reappraisal of the Literature. Clin. Infect. Dis. Off. Publ. Infect. Dis. Soc. Am. 1997, 25, 872–887. [Google Scholar] [CrossRef] [PubMed]
- Blanc, L.; Gilleron, M.; Prandi, J.; Song, O.-R.; Jang, M.-S.; Gicquel, B.; Drocourt, D.; Neyrolles, O.; Brodin, P.; Tiraby, G.; et al. Mycobacterium Tuberculosis Inhibits Human Innate Immune Responses via the Production of TLR2 Antagonist Glycolipids. Proc. Natl. Acad. Sci. USA 2017, 114, 11205–11210. [Google Scholar] [CrossRef]
- Zhai, W.; Wu, F.; Zhang, Y.; Fu, Y.; Liu, Z. The Immune Escape Mechanisms of Mycobacterium Tuberculosis. Int. J. Mol. Sci. 2019, 20, 340. [Google Scholar] [CrossRef]
- Sueki, H.; Mizukawa, Y.; Aoyama, Y. Immune Reconstitution Inflammatory Syndrome in Non-HIV Immunosuppressed Patients. J. Dermatol. 2018, 45, 3–9. [Google Scholar] [CrossRef]
- Campbell, I.A.; Dyson, A.J. Lymph Node Tuberculosis: A Comparison of Various Methods of Treatment. Tubercle 1977, 58, 171–179. [Google Scholar] [CrossRef]
- Hawkey, C.R.; Yap, T.; Pereira, J.; Moore, D.A.J.; Davidson, R.N.; Pasvol, G.; Kon, O.M.; Wall, R.A.; Wilkinson, R.J. Characterization and Management of Paradoxical Upgrading Reactions in HIV-Uninfected Patients with Lymph Node Tuberculosis. Clin. Infect. Dis. Off. Publ. Infect. Dis. Soc. Am. 2005, 40, 1368–1371. [Google Scholar] [CrossRef] [PubMed]
- Vanhaecke Collard, C.; Dramé, M.; Novella, J.-L.; Blanchard, F.; Pennaforte, J.-L.; Mahmoudi, R. Functional manifestations associated to corticosteroid therapy among the elderly. Rev. Med. Interne 2012, 33, 358–363. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Caraux-Paz, P.; Diamantis, S.; de Wazières, B.; Gallien, S. Tuberculosis in the Elderly. J. Clin. Med. 2021, 10, 5888. https://doi.org/10.3390/jcm10245888
Caraux-Paz P, Diamantis S, de Wazières B, Gallien S. Tuberculosis in the Elderly. Journal of Clinical Medicine. 2021; 10(24):5888. https://doi.org/10.3390/jcm10245888
Chicago/Turabian StyleCaraux-Paz, Pauline, Sylvain Diamantis, Benoit de Wazières, and Sébastien Gallien. 2021. "Tuberculosis in the Elderly" Journal of Clinical Medicine 10, no. 24: 5888. https://doi.org/10.3390/jcm10245888
APA StyleCaraux-Paz, P., Diamantis, S., de Wazières, B., & Gallien, S. (2021). Tuberculosis in the Elderly. Journal of Clinical Medicine, 10(24), 5888. https://doi.org/10.3390/jcm10245888





