Causes of Exocrine Pancreatic Insufficiency Other Than Chronic Pancreatitis
Abstract
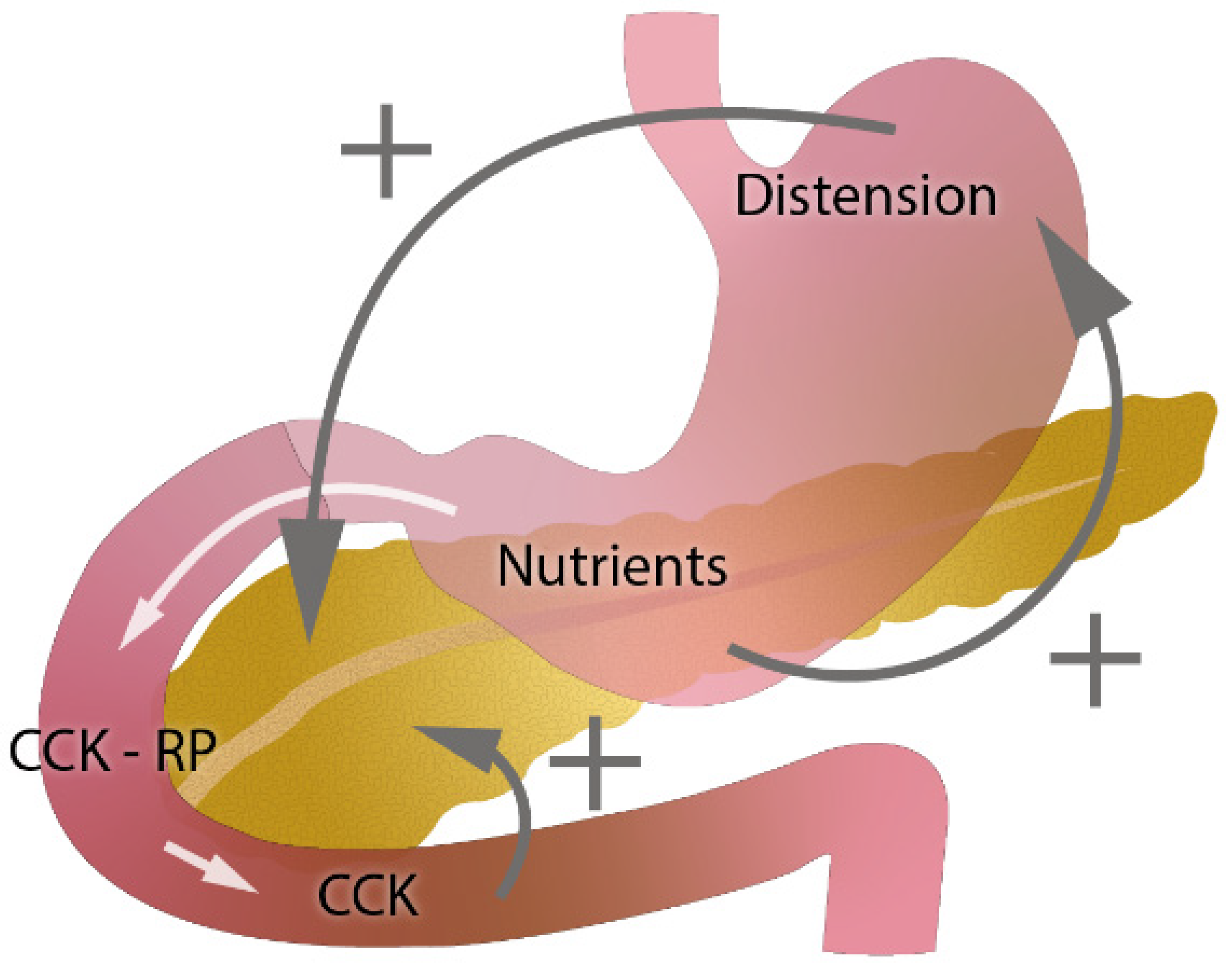
1. Introduction
2. Conditions Associated with EPI
2.1. Pancreatic Adenocarcinoma and Other Malignant Pancreatic Diseases
2.2. Pancreatic Resections
2.3. Obstruction of the Pancreatic Duct
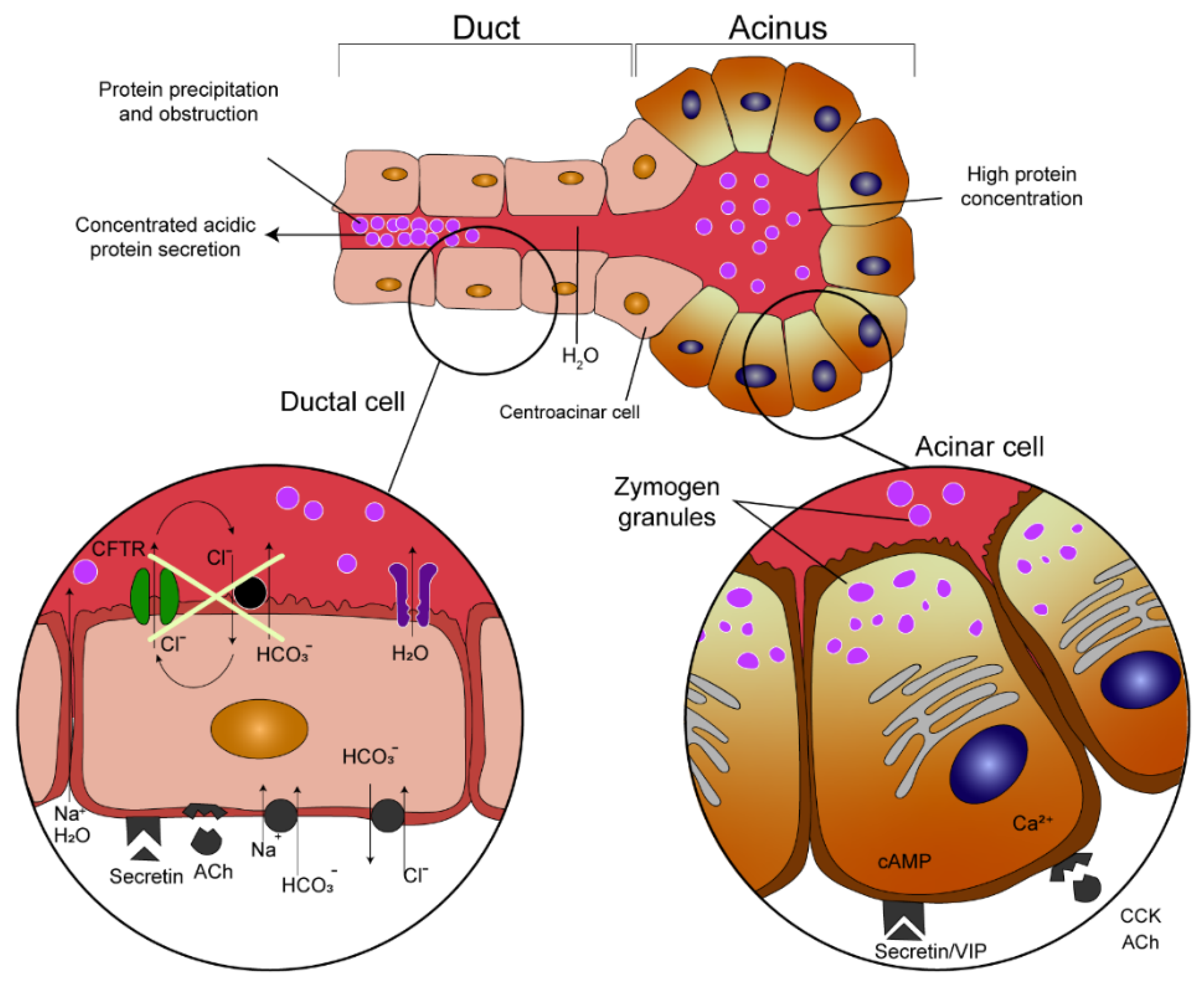
2.4. Cystic Fibrosis
2.5. Exocrine Pancreatic Insufficiency in Children
2.5.1. Shwachman–Diamond Syndrome
2.5.2. Chronic Pancreatitis
2.5.3. Johanson–Blizzard Syndrome
2.5.4. Pearson Syndrome
2.5.5. Other Causes
3. Poorly Clarified Mechanisms Associated with EPI
3.1. Diabetes Mellitus
3.2. Celiac Disease
3.3. Inflammatory Bowel Diseases
3.4. Gastrointestinal Surgeries Other Than Pancreatic Resections
3.5. Zollinger–Ellison Syndrome
3.6. Non-Alcoholic Fatty Pancreas Disease
3.7. Age
3.8. Relationship between Exocrine Pancreatic Secretion and Intestinal Microbiota, Small Intestinal Bacterial Overgrowth (SIBO)
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Singh, V.K.; Haupt, M.E.; Geller, D.E.; Hall, J.A.; Diez, P.M.Q. Less Common Etiologies of Exocrine Pancreatic Insufficiency. World J. Gastroenterol. 2017, 23, 7059–7076. [Google Scholar] [CrossRef] [PubMed]
- Keller, J.; Layer, P. Human Pancreatic Exocrine Response to Nutrients in Health and Disease. Gut 2005, 54 (Suppl. 6), vi1–vi28. [Google Scholar] [CrossRef] [PubMed]
- Lindkvist, B. Diagnosis and Treatment of Pancreatic Exocrine Insufficiency. World J. Gastroenterol. 2013, 19, 7258–7266. [Google Scholar] [CrossRef]
- Domínguez-Muñoz, J.E. Pancreatic Enzyme Replacement Therapy: Exocrine Pancreatic Insufficiency after Gastrointestinal Surgery. HPB 2009, 11 (Suppl. 3), 3–6. [Google Scholar] [CrossRef]
- Löhr, J.M.; Dominguez-Munoz, E.; Rosendahl, J.; Besselink, M.; Mayerle, J.; Lerch, M.M.; Haas, S.; Akisik, F.; Kartalis, N.; Iglesias-Garcia, J.; et al. United European Gastroenterology Evidence-Based Guidelines for the Diagnosis and Therapy of Chronic Pancrea-titis (HaPanEU). United Eur. Gastroenterol. J. 2017, 5, 153–199. [Google Scholar] [CrossRef] [PubMed]
- Dominguez-Munoz, J.E.; Drewes, A.M.; Lindkvist, B.; Ewald, N.; Czakó, L.; Rosendahl, J.; Löhr, J.M.; HaPanEU/UEG Working Group. Recommendations from the United European Gastroenterology Evidence-Based Guidelines for the Diagnosis and Therapy of Chronic Pancreatitis. Pancreatology 2018, 18, 847–854. [Google Scholar] [CrossRef] [PubMed]
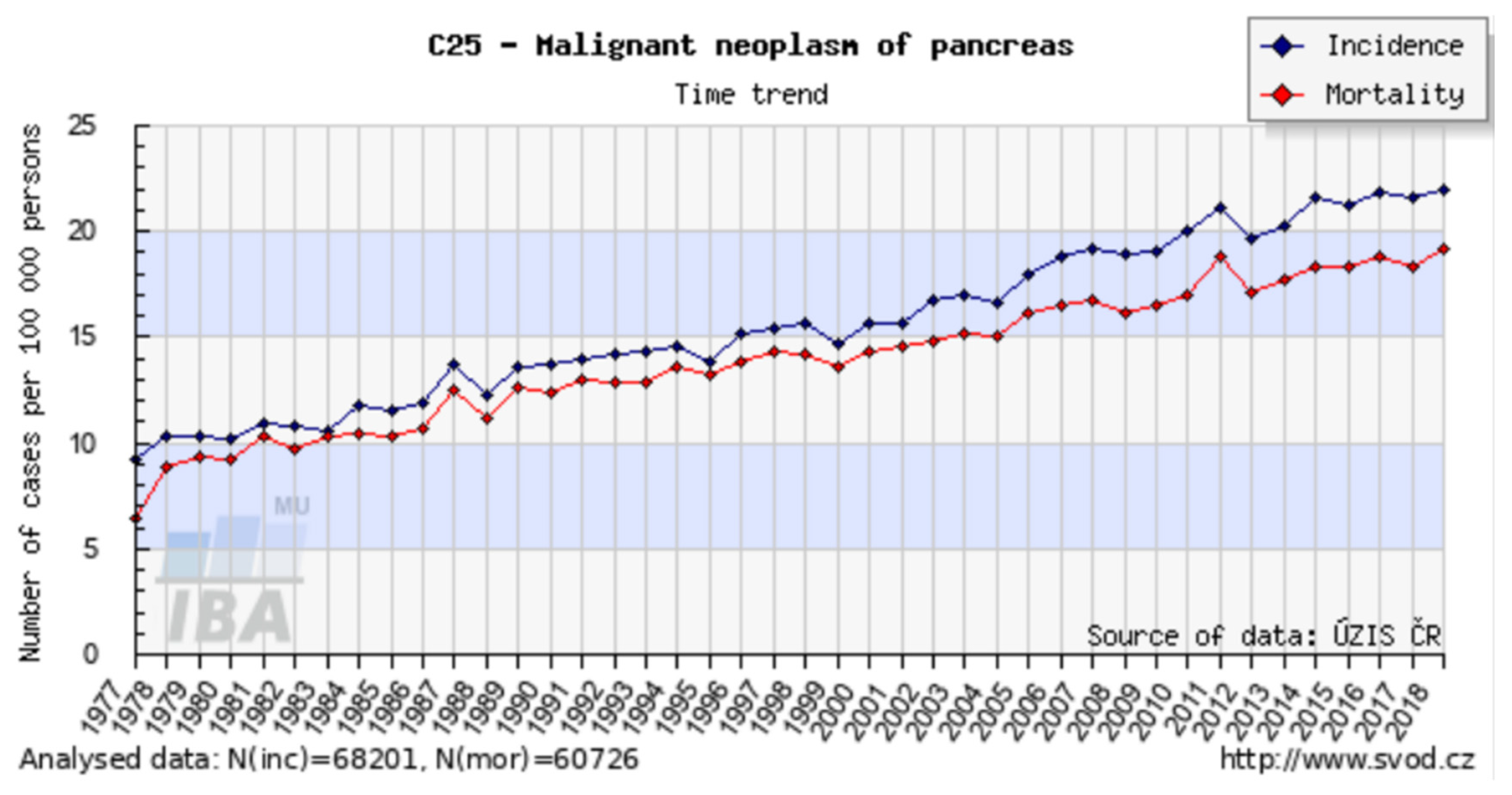
- Dušek, L.; Mužík, J.; Kubásek, M.; Koptíková, J.; Žaloudík, J.; Vyzula, R. Epidemiology of Malignant Tumours in the Czech Republic, Version 7.0; Masaryk University: Brno, Czech Republic, 2005; ISSN 1802–8861. Available online: http://www.svod.cz (accessed on 8 May 2019).
- Domínguez-Muñoz, J.E. Pancreatic Enzyme Therapy for Pancreatic Exocrine insufficiency. Curr. Gastroenterol. Rep. 2007, 9, 116–122. [Google Scholar] [CrossRef]
- Domínguez-Munoz, J.E. Pancreatic Exocrine Insufficiency: Diagnosis and Treatment. J. Gastroenterol. Hepatol. 2011, 26 (Suppl. 2), 12–16. [Google Scholar] [CrossRef] [PubMed]
- Pezzilli, R.; Andriulli, A.; Bassi, C.; Balzano, G.; Cantore, M.; Delle Fave, G.; Falconi, M.; Exocrine Pancreatic Insufficiency collaborative (EPIc) Group. Exocrine Pancreatic Insufficiency in Adults: A Shared Position Statement of the Italian Association for the Study of the Pancreas. World J. Gastroenterol. 2013, 19, 7930–7946. [Google Scholar] [CrossRef] [PubMed]
- DiMagno, E.P. Medical Treatment of Pancreatic Insufficiency. Mayo Clin. Proc. 1979, 54, 435–442. [Google Scholar]
- Perez, M.M.; Newcomer, A.D.; Moertel, C.G.; Go, V.L.; Dimagno, E.P. Assessment of Weight Loss, Food intake, Fat Metabolism, Malabsorption, and Treatment of Pancreatic Insufficiency in Pancreatic Cancer. Cancer 1983, 52, 346–352. [Google Scholar] [CrossRef]
- Partelli, S.; Frulloni, L.; Minniti, C.; Bassi, C.; Barugola, G.; D’Onofrio, M.; Crippa, S.; Falconi, M. Faecal elastase-1 Is an Independent Predictor of Survival in Advanced Pancreatic Cancer. Dig. Liver Dis. 2012, 44, 945–951. [Google Scholar] [CrossRef] [PubMed]
- DiMagno, E.P.; Malagelada, J.R.; Go, V.L. The Relationships between Pancreatic Ductal Obstruction and Pancreatic Secretion in Man. Mayo Clin. Proc. 1979, 54, 157–162. [Google Scholar] [PubMed]
- DiMagno, E.P.; Go, V.L.; Summerskill, W.H. Relations between Pancreatic Enzyme Outputs and Malab-sorption in Severe Pancreatic Insufficiency. N. Engl. J. Med. 1973, 288, 813–815. [Google Scholar] [CrossRef]
- Bachmann, J.; Ketterer, K.; Marsch, C.; Fechtner, K.; Krakowski-Roosen, H.; Büchler, M.W.; Friess, H.; Martignoni, M.E. Pancreatic Cancer Related Cachexia: Influence on Metabolism and Correlation to Weight Loss and Pulmonary Function. BMC Cancer 2009, 9, 255. [Google Scholar] [CrossRef]
- Roberts, K.J.; Bannister, C.A.; Schrem, H. Enzyme Replacement Improves Survival among Pati ents with Pancreatic Cancer: Results of a Population Based Study. Pancreatology 2019, 19, 114–121. [Google Scholar] [CrossRef]
- Fieker, A.; Philpott, J.; Armand, M. Enzyme Replacement Therapy for Pancreatic insufficiency: Present and Future. Clin. Exp. Gastroenterol. 2011, 4, 55–73. [Google Scholar] [PubMed]
- Domínguez-Munoz, J.E.; Nieto, L.; Iglesias-García, J. Pancreatic Enzyme Replacement Therapy and Nutritional Advice Are Associated with Longer Survival Inpatients with Unresectable Pancreatic Cancer. Pancreas 2013, 42, 1347. [Google Scholar]
- Bruno, M.J.; Haverkort, E.B.; Tijssen, G.P.; Tytgat, G.N.; van Leeuwen, D.J. Placebo Controlled Trial of Entericcoated Pancreatin Microsphere Treatment in Patients with Unresectable Cancer of the Pancreatic Head Region. Gut 1998, 42, 92–96. [Google Scholar] [CrossRef]
- Woo, S.M.; Joo, J.; Kim, S.Y.; Park, S.J.; Han, S.S.; Kim, T.H.; Koh, Y.H.; Chung, S.H.; Kim, Y.H.; Moon, H.; et al. Efficacy of Pancreatic Exocrine Replacement Therapy for Patients with Unresectable Pancreatic Cancer in a Randomized Trial. Pancreatology 2016, 16, 1099–1105. [Google Scholar] [CrossRef]
- Iglesia, D.; Avci, B.; Kiriukova, M.; Panic, N.; Bozhychko, M.; Sandru, V.; de-Madaria, E.; Capurso, G. Pancreatic Exocrine Insufficiency and Pancreatic Enzyme Replacement Therapy in Patients with Advanced Pancreatic Cancer: A Systematic Review and Meta-Analysis. United Eur. Gastroenterol. J. 2020, 8, 1115–1125. [Google Scholar] [CrossRef] [PubMed]
- Dasari, B.; Farid, S.; Roberts, K.J.; Morris-Stiff, G. Overview of Pancreatic Resections: Indications and Procedures. Br. J. Hosp. Med. 2015, 76, C34–C37. [Google Scholar] [CrossRef] [PubMed]
- Struyvenberg, M.R.; Martin, C.R.; Freedman, S.D. Practical Guide to Exocrine Pancreatic Insufficiency—Breaking the Myths. BMC Med. 2017, 15, 29. [Google Scholar] [CrossRef] [PubMed]
- Jang, J.Y.; Kim, S.W.; Park, S.J.; Park, Y.H. Comparison of the Functional Outcome after Pylorus-preserving Pancreatoduodenectomy: Pancreatogastrostomy and Pancreatojejunostomy. World J. Surg. 2002, 26, 366–371. [Google Scholar]
- Chaudhary, A.; Domínguez-Muñoz, J.E.; Layer, P.; Lerch, M.M. Pancreatic Exocrine Insufficiency as a Complication of Gastrointestinal Surgery and the Impact of Pancreatic Enzyme Replacement Therapy. Dig. Dis. 2020, 38, 53–68. [Google Scholar] [CrossRef] [PubMed]
- Yuasa, Y.; Murakami, Y.; Nakamura, H.; Uemura, K.; Ohge, H.; Sudo, T.; Hashimoto, Y.; Nakashima, A.; Hiyama, E.; Sueda, T. Histological Loss of Pancreatic Exocrine Cells Correlates with Pancreatic Exocrine Function after Pancreatic Surgery. Pancreas 2012, 41, 928–933. [Google Scholar] [CrossRef]
- Löser, C.H.R.; Brauer, C.; Aygen, S.; Hennemann, O.; Fölsch, U.R. Comparative Clinical Evaluation of the 13C-Mixed Triglyceride Breath Test as an Indirect Pancreatic Function Test. Scand. J. Gastroenterol. 1998, 33, 327–334. [Google Scholar] [PubMed]
- Roeyen, G.; Jansen, M.; Hartman, V.; Chapelle, T.; Bracke, B.; Ysebaert, D.; De Block, C. The Impact of Pancreaticoduodenectomy on Endocrine and Exocrine Pancreatic Function: A Prospective Cohort Study Based on Pre- and Postoperative Function Tests. Pancreatology 2017, 17, 974–982. [Google Scholar] [CrossRef] [PubMed]
- Lim, P.W.; Dinh, K.H.; Sullivan, M.; Wassef, W.Y.; Zivny, J.; Whalen, G.F.; LaFemina, J. Thirty-Day Outcomes Underestimate Endocrine and Exocrine Insufficiency after Pancreatic Resection. HPB 2016, 18, 360–366. [Google Scholar] [CrossRef]
- Dervenis, C. Exocrine Pancreatic Insufficiency and Malnutrition after Gastrointestinal Surgery. HPB 2009, 11 (Suppl. 3), 1–2. [Google Scholar] [CrossRef][Green Version]
- Domínguez-Muñoz, J.E. Pancreatic Enzyme Therapy for Pancreatic Exocrine Insufficiency. Gastroenterol. Hepatol. 2011, 7, 401–403. [Google Scholar] [CrossRef]
- Kunovský, L.; Kala, Z.; Procházka, V.; Potrusil, M.; Dastych, M.; Novotný, I.; Andrasina, T.; Pavlovský, Z.; Eid, M.; Moravcik, P. Surgical Treatment of Ampullary Adenocarcinoma—Single Center Experience and a Review of Literature. Klin. Onkol. 2018, 31, 46–52. [Google Scholar] [CrossRef] [PubMed]
- Kunovský, L.; Kala, Z.; Procházka, V.; Dastych, M.; Novotný, I.; Trna, J.; Mazanec, J.; Andrašina, T.; Eid, M.; Potrusil, M.; et al. Laparoscopic Pancreaticoduodenectomy for Ampullary Adenocarcinoma: A Case Report. Gastroent. Hepatol. 2018, 72, 401–407. [Google Scholar] [CrossRef]
- Rosenstein, B.J.; Cutting, G.R. The Diagnosis of Cystic Fibrosis: A Consensus Statement. Cystic Fibrosis Foundation Consensus Panel. J. Pediatr. 1998, 132, 589–595. [Google Scholar] [CrossRef]
- Kopelman, H.; Corey, M.; Gaskin, K.; Durie, P.; Weizman, Z.; Forstner, G. Impaired Chloride Secretion, as Well as Bicarbonate Secretion, Underlies the Fluid Secretory Defect in the Cystic Fibrosis Pancreas. Gas-Troenterology 1988, 95, 349–355. [Google Scholar] [CrossRef]
- Wilschanski, M.; Durie, P.R. Patterns of GI Disease in Adulthood Associated with Mutations in the CFTR Gene. Gut 2007, 56, 1153–1163. [Google Scholar] [CrossRef]
- Wilschanski, M.; Novak, I. The Cystic Fibrosis of Exocrine Pancreas. Cold Spring Harb. Perspect. Med. 2013, 3, a009746. [Google Scholar] [CrossRef]
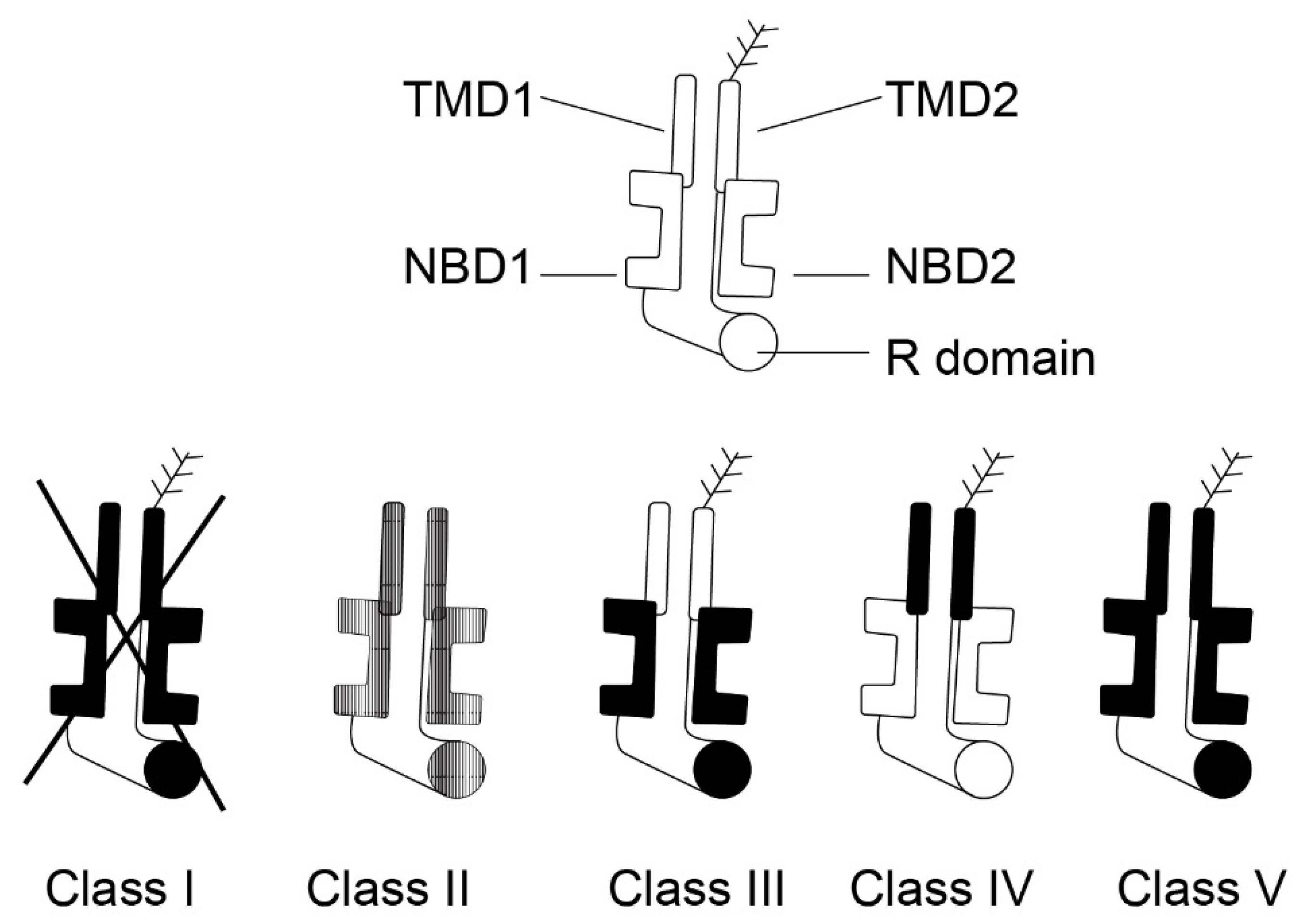
- Mickle, J.E.; Cutting, G.R. Genotype-Phenotype Relationships in Cystic Fibrosis. Med. Clin. N. Am. 2000, 84, 597–607. [Google Scholar] [CrossRef]
- Welsh, M.J.; Smith, A.E. Molecular Mechanisms of CFTR Chloride Channel Dysfunction in Cystic Fibrosis. Cell 1993, 73, 1251–1254. [Google Scholar] [CrossRef]
- Cuppens, H. What Is Clinically Relevant about the Genetics of Cystic Fibrosis? In Clinical Pancreatology for Practicing Gastroenterologists and Surgeons; Dominguez-Munoz, J.E., Ed.; Blackwell Publishing: Cornwall, ON, Canada, 2005; pp. 214–219. [Google Scholar]
- Rowntree, R.K.; Harris, A. The Phenotypic Consequences of CFTR Mutations. Ann. Hum. Genet. 2003, 67, 471–485. [Google Scholar] [CrossRef]
- Singh, V.K.; Schwarzenberg, S.J. Pancreatic Insufficiency in Cystic Fibrosis. J. Cyst. Fibros. 2017, 16 (Suppl. 2), S70–S78. [Google Scholar] [CrossRef] [PubMed]
- De Boeck, K.; Weren, M.; Proesmans, M.; Kerem, E. Pancreatitis among Patients with Cystic Fibrosis: Correlation with Pancreatic Status and Genotype. Pediatrics 2005, 115, e463–e469. [Google Scholar] [CrossRef]
- Van de Vijver, E.; Desager, K.; Mulberg, A.E.; Staelens, S.; Verkade, H.J.; Bodewes, F.A.; Malfroot, A.; Hauser, B.; Sinaasappel, M.; Van Biervliet, S.; et al. Treatment of Infants and Toddlers with Cystic Fibrosis-Related Pancreatic Insufficiency and Fat Mal-absorption with Pancrelipase MT. J. Pediatr. Gastroenterol. Nutr. 2011, 53, 61–64. [Google Scholar] [CrossRef] [PubMed]
- Heijerman, H.G.; Lamers, C.B.; Bakker, W. Omeprazole Enhances the Efficacy of Pancreatin (Pancrease) in Cystic Fibrosis. Ann. Intern. Med. 1991, 114, 200–201. [Google Scholar] [CrossRef] [PubMed]
- Maqbool, A.; Stallings, V.A. Update on Fat-Soluble Vitamins in Cystic Fibrosis. Curr. Opin. Pulm. Med. 2008, 14, 574–581. [Google Scholar] [CrossRef]
- Tesaříková, P.; Kunovský, L.; Trna, J.; Dítě, P.; Vaculová, J.; Kala, Z. Cystic Fibrosis and Exocrine Pan-Creatic Insufficiency. Gastroent. Hepatol. 2019, 73, 303–307. [Google Scholar] [CrossRef]
- Condren, M.E.; Bradshaw, M.D. Ivacaftor: A Novel Gene-Based Therapeutic Approach for Cystic Fibrosis. J. Pediatr. Pharmacol. Ther. 2013, 18, 8–13. [Google Scholar] [CrossRef]
- Dřevínek, P. Kauzální terapie cystické fibrózy. Postgrad. Med. 2014, 16, 21–22. [Google Scholar]
- Brewington, J.J.; McPhail, G.L.; Clancy, J.P. Lumacaftor Alone and Combined with Ivacaftor: Preclinical and Clinical Trial Experience of F508del CFTR Cor Rection. Expert Rev. Respir. Med. 2016, 10, 5–17. [Google Scholar] [CrossRef] [PubMed]
- Walker, S.; Flume, P.; McNamara, J.; Solomon, M.; Chilvers, M.; Chmiel, J.; Harris, R.S.; Haseltine, E.; Stiles, D.; Li, C.; et al. VX15-661-113 Investigator Group. A phase 3 Study of Tezacaftor in Combination with Ivacaftor in Children Aged 6 through 11 Years with Cystic Fibrosis. J. Cyst. Fibros. 2019, 18, 708–713. [Google Scholar] [CrossRef]
- Uc, A.; Fishman, D.S. Pancreatic Disorders. Pediatr. Clin. N. Am. 2017, 64, 685–706. [Google Scholar] [CrossRef] [PubMed]
- Widodo, A.D.; Setiabudy, R.; Timan, I.S.; Bardosono, S.; Winarta, W.; Firmansyah, A. Pancreatic Enzyme Replacement Therapy (PERT) in Children with Persistent Diarrhea: Avoidance of Elemental Diet Need, Accessibility and Costs. Asia Pac. J. Clin. Nutr. 2018, 27, 512–518. [Google Scholar]
- Myers, K.C.; Bolyard, A.A.; Otto, B.; Wong, T.E.; Jones, A.T.; Harris, R.E.; Davies, S.M.; Dale, D.C.; Shimamura, A. Variable Clinical Presentation of Shwachman–Diamond Syndrome: Update from the North American Shwachman–Diamond Syndrome Registry. J. Pediatr. 2014, 164, 866–870. [Google Scholar] [CrossRef] [PubMed]
- Dror, Y.; Donadieu, J.; Koglmeier, J.; Dodge, J.; Toiviainen-Salo, S.; Makitie, O.; Kerr, E.; Zeidler, C.; Shimamura, A.; Shah, N.; et al. Draft Consensus Guidelines for Diagnosis and Treatment of Shwachman–Diamond Syndrome. Ann. N. Y. Acad. Sci. 2011, 1242, 40–55. [Google Scholar] [CrossRef] [PubMed]
- Dall’oca, C.; Bondi, M.; Merlini, M.; Cipolli, M.; Lavini, F.; Bartolozzi, P. Shwachman–Diamond Syndrome. Musculoskelet. Surg. 2012, 96, 81–88. [Google Scholar] [CrossRef] [PubMed]
- Schwarzenberg, S.J.; Bellin, M.; Husain, S.Z.; Ahuja, M.; Barth, B.; Davis, H.; Durie, P.R.; Fishman, D.S.; Freedman, S.D.; Gariepy, C.E.; et al. Pediatric Chronic Pancreatitis Is Associated with Genetic Risk Factors and Substantial Disease Burden. J. Pediatr. 2015, 166, 890–896.e1. [Google Scholar] [CrossRef] [PubMed]
- Atik, T.; Karakoyun, M.; Sukalo, M.; Zenker, M.; Ozkinay, F.; Aydoğdu, S. Two Novel UBR1 Gene Mutations in a Patient with Johanson Blizzard Syndrome: A Mild Phenotype without Mental Retardation. Gene 2015, 570, 153–155. [Google Scholar] [CrossRef]
- Ellery, K.M.; Erdman, S.H. Johanson–Blizzard Syndrome: Expanding the Phenotype of Exocrine Pancreatic Insufficiency. JOP 2014, 15, 388–390. [Google Scholar]
- Tumino, M.; Meli, C.; Farruggia, P.; La Spina, M.; Faraci, M.; Castana, C.; Di Raimondo, V.; Alfano, M.; Pittalà, A.; Lo Nigro, L.; et al. Clinical Manifestations and Management of Four Children with Pearson Syndrome. Am. J. Med. Genet. A 2011, 155, 3063–3066. [Google Scholar] [CrossRef]
- Hardt, P.D.; Ewald, N. Exocrine Pancreatic Insufficiency in Diabetes Mellitus: A Complication of Diabetic Neuropathy or a Different Type of Diabetes? Exp. Diabetes Res. 2011, 2011, 761950. [Google Scholar] [CrossRef] [PubMed]
- Lankisch, P.G.; Manthey, G.; Otto, J.; Koop, H.; Talaulicar, M.; Willms, B.; Creutzfeldt, W. Exocrine Pancreatic Function in Insulin-Dependent Diabetes Mellitus. Digestion 1982, 25, 211–216. [Google Scholar] [CrossRef]
- Gröger, G.; Layer, P. Exocrine Pancreatic Function in Diabetes Mellitus. Eur. J. Gastroenterol. Hepatol. 1995, 7, 740–746. [Google Scholar]
- Yu, T.Y.; Wang, C.Y. Impact of Non-Alcoholic Fatty Pancreas Disease on Glucose Metabolism. J. Diabetes Investig. 2017, 8, 735–747. [Google Scholar] [CrossRef] [PubMed]
- Smits, M.M.; van Geenen, E.J. The Clinical Significance of Pancreatic Steatosis. Hepatol. Nat. Rev. Gastroenterol. 2011, 8, 169–177. [Google Scholar] [CrossRef] [PubMed]
- Ou, H.Y.; Wang, C.Y.; Yang, Y.C.; Chen, M.F.; Chang, C.J. The association between nonalcoholic fatty pancreas disease and diabetes. PLoS ONE 2013, 8, e62561. [Google Scholar]
- American Diabetes Association. (2) Classification and Diagnosis of Diabetes. Diabetes Care 2015, 38 (Suppl. 1), S8–S16. [Google Scholar] [CrossRef] [PubMed]
- Larger, E.; Philippe, M.F.; Barbot-Trystram, L.; Radu, A.; Rotariu, M.; Nobécourt, E.; Boitard, C. Pancreatic Exocrine Function in Patients with Diabetes. Diabet. Med. 2012, 29, 1047–1054. [Google Scholar] [CrossRef] [PubMed]
- Frier, B.M.; Saunders, J.H.; Wormsley, K.G.; Bouchier, I.A. Exocrine Pancreatic Function in Juvenile-Onset Diabetes Mellitus. Gut 1976, 17, 685–691. [Google Scholar] [CrossRef]
- Ferrer, R.; Medrano, J.; Diego, M.; Calpena, R.; Graells, L.; Moltó, M.; Pérez, T.; Pérez, F.; Salido, G. Effect of Exogenous Insulin and Glucagon on Exocrine Pancreatic Secretion in Rats In Vivo. Int. J. Pancreatol. 2000, 28, 67–75. [Google Scholar] [CrossRef]
- Gyr, K.; Beglinger, C.; Köhler, E.; Trautzl, U.; Keller, U.; Bloom, S.R. Circulating Somatostatin. Physio-logical Regulator of Pancreatic Function? J. Clin. Investig. 1987, 79, 1595–1600. [Google Scholar] [CrossRef]
- Piciucchi, M.; Capurso, G.; Archibugi, L.; Delle Fave, M.M.; Capasso, M.; Delle Fave, G. Exocrine Pancreatic Insufficiency in Diabetic Patients: Prevalence, Mechanisms, and Treatment. Int. J. Endocrinol. 2015, 2015, 595649. [Google Scholar] [CrossRef] [PubMed]
- Nakajima, K.; Oshida, H.; Muneyuki, T.; Kakei, M. Pancrelipase: An Evidence-Based Review of Its Use for Treating Pancreatic Exocrine Insufficiency. Core Evid. 2012, 7, 77–91. [Google Scholar] [CrossRef]
- Cui, Y.; Andersen, D.K. Pancreatogenic Diabetes: Special Considerations for Management. Pancreatology 2011, 11, 279–294. [Google Scholar] [CrossRef] [PubMed]
- Ewald, N.; Kaufmann, C.; Raspe, A.; Kloer, H.U.; Bretzel, R.G.; Hardt, P.D. Prevalence of Diabetes Mellitus Secondary to Pancreatic Diseases (type 3c). Diabetes Metab. Res. Rev. 2012, 28, 338–342. [Google Scholar] [CrossRef] [PubMed]
- Andersen, D.K.; Andren-Sandberg, Å.; Duell, E.J.; Goggins, M.; Korc, M.; Petersen, G.M.; Smith, J.P.; Whitcomb, D.C. Pancreatitis-Diabetes-Pancreatic Cancer: Summary of an NIDDK-NCI Workshop. Pancreas 2013, 42, 1227–1237. [Google Scholar] [CrossRef] [PubMed]
- Parzanese, I.; Qehajaj, D.; Patrinicola, F.; Aralica, M.; Chiriva-Internati, M.; Stifter, S.; Elli, L.; Grizzi, F. Celiac Disease: From Pathophysiology to Treatment. World J. Gastrointest. Pathophysiol. 2017, 8, 27–38. [Google Scholar] [CrossRef] [PubMed]
- Singh, P.; Arora, A.; Strand, T.A.; Leffler, D.A.; Catassi, C.; Green, P.H.; Kelly, C.P.; Ahuja, V.; Makharia, G.K. Global Prevalence of Celiac Disease: Systematic Review and Meta-Analysis. Clin. Gastroenterol. Hepatol. 2018, 16, 823–836.e2. [Google Scholar] [CrossRef]
- Cárdenas, A.; Kelly, C.P. Celiac Sprue. Semin. Gastrointest. Dis. 2002, 13, 232–244. [Google Scholar]
- Capurso, G.; Traini, M.; Piciucchi, M.; Signoretti, M.; Arcidiacono, P.G. Exocrine Pancreatic Insufficiency: Prevalence, Diagnosis and Management. Clin. Exp. Gastroenetrol. 2019, 12, 129–139. [Google Scholar] [CrossRef]
- Evans, K.E.; Leeds, J.S.; Morley, S.; Sanders, D.S. Pancreatic Insufficiency in Adult Celiac Disease: Do Patients Require Long-Term Enzyme Supplementation? Dig. Dis. Sci. 2010, 55, 2999–3004. [Google Scholar] [CrossRef] [PubMed]
- Rana, S.S.; Dambalkar, A.; Chhabra, R.; Sharma, R.; Nada, R.; Sharma, V.; Rana, S.; Bhasin, D.K. Is Pancreatic Exocrine Insufficiency in Celiac Disease Related to Structural Alterations in Pancreatic Parenchyma? Ann. Gastroenterol. 2016, 19, 363–366. [Google Scholar] [CrossRef] [PubMed]
- Buchan, A.M.; Grant, S.; Brown, J.C.; Freeman, H.J. A Quantitative Study of Enteric Endocrine Cells in Celiac Sprue. J. Pediatr. Gastroenterol. Nutr. 1984, 3, 665–671. [Google Scholar] [CrossRef]
- Leeds, J.S.; Hopper, A.D.; Hurlstone, D.P.; Edwards, S.J.; McAlindon, M.E.; Lobo, A.J.; Donnelly, M.T.; Morley, S.; Sanders, D.S. Is Exocrine Pancreatic Insufficiency in Adult Coeliac Disease a Cause of Persisting Symptoms? Aliment. Pharmacol. Ther. 2007, 25, 265–271. [Google Scholar] [CrossRef] [PubMed]
- Carroccio, A.; Iacono, G.; Montalto, G.; Cavataio, F.; Lorello, D.; Greco, L.; Soresi, M.; Notarbartolo, A. Pancreatic Enzyme Therapy in Childhood Celiac Disease. A Double-Blind Prospective Randomized Study. Dig. Dis. Sci. 1995, 40, 2555–2560. [Google Scholar] [CrossRef] [PubMed]
- Gomollón, F.; Dignass, A.; Annese, V.; Tilg, H.; Van Assche, G.; Lindsay, J.O.; Peyrin-Biroulet, L.; Cullen, G.J.; Daperno, M.; Kucharzik, T.; et al. 3rd European Evidence-Based Consensus on the Diagnosis and Management of Crohn’s Disease 2016: Part 1: Diagnosis and Medical Management. J. Crohns Colitis 2017, 11, 3–25. [Google Scholar] [CrossRef] [PubMed]
- Xavier, R.J.; Podolsky, D.K. Unravelling the Pathogenesis of Inflammatory Bowel Disease. Nature 2007, 448, 427–434. [Google Scholar] [CrossRef]
- Rothfuss, K.S.; Stange, E.F.; Herrlinger, K.R. Extraintestinal Manifestations and Complications in Inflam-Matory Bowel Diseases. World J. Gastroenterol. 2006, 12, 4819–4831. [Google Scholar] [CrossRef]
- Antonini, F.; Pezzilli, R.; Angelelli, L.; Macarri, G. Pancreatic Disorders in Inflammatory Bowel Disease. World J. Gastrointest. Pathophysiol. 2016, 7, 276–282. [Google Scholar] [CrossRef]
- Navaneethan, U.; Shen, B. Hepatopancreatobiliary Manifestations and Complications Associated with Inflammatory Bowel Disease. Inflamm. Bowel Dis. 2010, 16, 1598–1619. [Google Scholar] [CrossRef]
- Ramos, L.R.; Sachar, D.B.; DiMaio, C.J.; Colombel, J.F.; Torres, J. Inflammatory Bowel Disease and Pancreatitis: A Review. J. Crohns Colitis 2016, 10, 95–104. [Google Scholar] [CrossRef]
- Maconi, G.; Dominici, R.; Molteni, M.; Ardizzone, S.; Bosani, M.; Ferrara, E.; Gallus, S.; Panteghini, M.; Bianchi Porro, G. Prevalence of Pancreatic Insufficiency in Inflammatory Bowel Diseases. Assessment by Fecal elastase-1. Dig. Dis. Sci. 2008, 53, 262–270. [Google Scholar] [CrossRef] [PubMed]
- Ball, W.P.; Baggenstoss, A.H.; Bargen, J.A. Pancreatic Lesions Associated with Chronic Ulcerative Colitis. Arch. Pathol. 1950, 50, 347–358. [Google Scholar]
- Barthet, M.; Lesavre, N.; Desplats, S.; Panuel, M.; Gasmi, M.; Bernard, J.P.; Dagorn, J.C.; Grimaud, J.C. Frequency and Characteristics of Pancreatitis in Patients with Inflammatory Bowel Disease. Pancreatology 2006, 6, 464–471. [Google Scholar] [CrossRef] [PubMed]
- Colombel, J.F.; Shin, A.; Gibson, P.R. AGA Clinical Practice Update on Functional Gastrointestinal Symptoms in Patients with Inflammatory Bowel Disease: Expert Review. Clin. Gastroenterol. Hepatol. 2019, 17, 380–390.e1. [Google Scholar] [CrossRef] [PubMed]
- Braganza, J.M.; Lee, S.H.; McCloy, R.F.; McMahon, M.J. Chronic Pancreatitis. Lancet 2011, 377, 1184–1197. [Google Scholar] [CrossRef]
- Garrido Gómez, E.; Lopez San Román, A.; Bermejo San José, F. Idiopathic Pancreatitis in Inflammatory Bowel Disease. J. Crohns Colitis 2008, 2, 237–240. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Hegnhøj, J.; Hansen, C.P.; Rannem, T.; Søbirk, H.; Andersen, L.B.; Andersen, J.R. Pancreatic Function in Crohn’s Disease. Gut 1990, 31, 1076–1079. [Google Scholar] [CrossRef]
- Masoero, G.; Zaffino, C.; Laudi, C.; Lombardo, L.; Rocca, R.; Gallo, L.; Della Monica, P.; Pera, A. Fecal Pancreatic Elastase 1 in the Work up of Patients with Chronic Diarrhea. Int. J. Pancreatol. 2000, 28, 175–179. [Google Scholar] [CrossRef]
- Komorowski, L.; Teegen, B.; Probst, C.; Aulinger-Stöcker, K.; Sina, C.; Fellermann, K.; Stöcker, W. Autoantibodies against Exocrine Pancreas in Crohn’s Disease Are Directed against Two Antigens: The Glycoproteins CUZD1 and GP2. J. Crohns Colitis 2013, 7, 780–790. [Google Scholar] [CrossRef]
- Piontek, M.; Hengels, K.J.; Strohmeyer, G. Crohn’s Disease: What about the Pancreas? J. Clin. Gastroenterol. 1990, 12, 491–493. [Google Scholar]
- Iida, T.; Wagatsuma, K.; Hirayama, D.; Yokoyama, Y.; Nakase, H. The Etiology of Pancreatic Manifestations in Patients with Inflammatory Bowel Disease. J. Clin. Med. 2019, 8, 916. [Google Scholar] [CrossRef]
- Chen, C.H.; Lin, C.L.; Kao, C.H. Association between Inflammatory Bowel Disease and Cholelithiasis: A Nationwide Population-Based Cohort Study. Int. J. Environ. Res. Public Health 2018, 15, 513. [Google Scholar] [CrossRef]
- Friess, H.; Böhm, J.; Müller, M.W.; Glasbrenner, B.; Riepl, R.L.; Malfertheiner, P.; Büchler, M.W. Maldigestion after Total Gastrectomy Is Associated with Pancreatic Insufficiency. Am. J. Gastroenterol. 1996, 91, 341–347. [Google Scholar] [PubMed]
- Hillman, H.S. Postgastrectomy Malnutrition. Gut 1968, 9, 576–584. [Google Scholar] [CrossRef] [PubMed]
- Gullo, L.; Costa, P.L.; Ventrucci, M.; Mattioli, S.; Viti, G.; Labò, G. Exocrine Pancreatic Function after Total Gastrectomy. Scand. J. Gastroenterol. 1979, 14, 401–407. [Google Scholar]
- Mikhailidis, D.P.; Foo, Y.; Ramdial, L.; Kirk, R.M.; Rosalki, S.B.; Dandona, P. Pancreatic Exocrine Function after Truncal and Highly Selective Vagotomy. J. Clin. Pathol. 1981, 34, 963–964. [Google Scholar] [CrossRef]
- Malagelada, J.R.; Go, V.L.; Summerskill, W.H. Altered Pancreatic and Biliary Function after Va-gotomy and Pyloroplasty. Gastroenterology 1974, 66, 22–27. [Google Scholar] [CrossRef]
- Wormsley, K.G. The Effect of Vagotomy on the Human Pancreatic Response to Direct and Indirect Stimulation. Scand. J. Gastroenterol. 1972, 7, 85–91. [Google Scholar]
- Beger, H.G.; Mayer, B.; Poch, B. Die Duodenumerhaltende Pankreaskopfresektion. Chirurg 2019, 90, 736–743. [Google Scholar] [CrossRef] [PubMed]
- Huddy, J.R.; Macharg, F.M.S.; Lawn, A.M.; Preston, S.R. Exocrine Pancreatic Insufficiency Following Esophagectomy. Dis. Esophagus 2013, 26, 594–597. [Google Scholar] [CrossRef]
- Lankisch, P.G. Appropriate Pancreatic Function Tests And indication for Pancreatic Enzyme Therapy Following Surgical Procedures on the Pancreas. Pancreatology 2001, 1, 14–26. [Google Scholar] [CrossRef]
- Epelboym, I.; Mazeh, H. Zollinger–Ellison Syndrome: Classical Considerations and Current Contro-Versies. Oncologist 2014, 19, 44–50. [Google Scholar] [CrossRef] [PubMed]
- Campana, D.; Piscitelli, L.; Mazzotta, E.; Bonora, M.; Serra, C.; Salomone, L.; Corinaldesi, R.; Tomassetti, P. Zollinger–Ellison Syndrome. Diagnosis and Therapy. Minerva Med. 2005, 96, 187–206. [Google Scholar] [PubMed]
- Banasch, M.; Schmitz, F. Diagnosis and treatment of gastrinoma in the era of proton pump inhibitors. Wien. Klin Wochenschr. 2007, 119, 573–578. [Google Scholar] [CrossRef]
- Othman, M.O.; Harb, D.; Barkin, J.A. Introduction and practical approach to exocrine pancreatic insuffi-ciency for the practicing clinician. Int. J. Clin. Pract. 2018, 72, e13066. [Google Scholar] [CrossRef] [PubMed]
- Tomassetti, P.; Campana, D.; Piscitelli, L.; Mazzotta, E.; Brocchi, E.; Pezzilli, R.; Corinaldesi, R. Treatment of Zollinger-Ellison syndrome. World J. Gastro-Enterol. 2005, 11, 5423–5432. [Google Scholar] [CrossRef] [PubMed]
- Hauser, S. Mayo Clinic Gastroenterology and Hepatology Board Review; Oxford University Press: Oxford, UK, 2011; p. 102. [Google Scholar]
- Blaho, M.; Dítě, P.; Kunovský, L.; Martínek, A. Fatty pancreas disease: Clinical impact. Vnitrni Lek. 2018, 64, 949–952. [Google Scholar] [CrossRef]
- Lozano, M.; Navarro, S.; Perez-Ayuso, R.; Llach, J.; Ayuso, C.; Guevara, M.C.; Ros, E. Lipomatosis of the pancreas: An unusual cause of massive steatorrhea. Pancreas 1988, 3, 580–582. [Google Scholar] [CrossRef] [PubMed]
- Tomita, Y.; Azuma, K.; Nonaka, Y.; Kamada, Y.; Tomoeda, M.; Kishida, M.; Masahiro, T.; Miyoshi, E. Pancreatic fatty degeneration and fibrosis as predisposing factors for the devel-opment of pancreatic ductal adenocarcinoma. Pancreas 2014, 43, 1032–1041. [Google Scholar] [CrossRef]
- Lesmana, C.R.; Gani, R.A.; Laurentius, A. Non-alcoholic fatty pancreas disease as a risk factor for pancreatic cancer based on endoscopic ultrasound examination among pancreatic cancer patients: A single-center experience. JGH Open 2018, 2, 4–7. [Google Scholar] [CrossRef]
- Lohr, J.-M.; Panic, N.; Vujasinovic, M.; Verbeke, C.S. The ageing pancreas: A systematic review of the evidence and analysis of the consequences (Review). J. Intern. Med. 2018, 283, 446–460. [Google Scholar] [CrossRef] [PubMed]
- Laugier, R.; Bernard, J.P.; Berthezene, P.; Dupuy, P. Changes in Pancreatic Exocrine Secretion with Age: Pancreatic Exocrine Secretion Does Decrease in the Elderly. Digestion 1991, 50, 202–211. [Google Scholar] [CrossRef]
- Saisho, Y.; Butler, A.E.; Meier, J.J.; Monchamp, T.; Allen-Auerbach, M.; Rizza, R.A.; Butler, P.C. Pancreas volumes in humans from birth to age one hundred taking into account sex, obesity, and presence of type-2 diabetes. Clin. Anat. 2007, 20, 933–942. [Google Scholar] [CrossRef]
- Anand, B.S.; Vij, J.C.; Mac, H.S.; Chowdhury, V.; Kumar, A. Effect of aging on the pancreatic ducts: A study based on endoscopic retrograde pancreatography. Gastrointest. Endosc. 1989, 35, 210–213. [Google Scholar] [CrossRef]
- Kreel, L.; Sandin, B. Changes in pancreatic morphology associated with aging. Gut 1973, 14, 962–970. [Google Scholar] [CrossRef] [PubMed]
- Petrone, M.C.; Arcidiacono, P.G.; Perri, F.; Carrara, S.; Boemo, C.; Testoni, P.A. Chronic pancreatitis-like changes detected by endoscopic ultrasound in subjects without signs of pancreatic disease: Do these indicate age-related changes, effects of xenobiotics, or early chronic pancreatitis? Pancreatology 2010, 10, 597–602. [Google Scholar] [CrossRef] [PubMed]
- Ikeda, M.; Sato, T.; Morozumi, A.; Fujino, M.A.; Yoda, Y.; Ochiai, M.; Kobayashi, K. Morphologic changes in the pancreas detected by screening ultraso-nography in a mass survey, with special reference to main duct dilatation, cyst formation, and calcification. Pancreas 1994, 9, 508–512. [Google Scholar] [CrossRef] [PubMed]
- Heuck, A.; Maubach, P.A.; Reiser, M.; Feuerbach, S.; Allgayer, B.; Lukas, P.; Kahn, T. Age-related morphology of the normal pancreas on computed tomography. Gastrointest. Radiol. 1987, 12, 18–22. [Google Scholar] [CrossRef]
- Sato, T.; Ito, K.; Tamada, T.; Sone, T.; Noda, Y.; Higaki, A.; Kanki, A.; Tanimoto, D.; Higashi, H. Age-related changes in normal adult pancreas: MR imaging evaluation. Eur. J. Radiol. 2012, 81, 2093–2098. [Google Scholar] [CrossRef]
- Li, J.; Xie, Y.; Yuan, F.; Song, B.; Tang, C. Noninvasive quantification of pancreatic fat in healthy male popu-lation using chemical shift magnetic resonance imaging: Effect of aging on pancreatic fat content. Pancreas 2011, 40, 295–299. [Google Scholar] [CrossRef]
- Janssen, J.; Papavassiliou, I. Effect of aging and diffuse chronic pancreatitis on pancreas elasticity evaluated using semiquantitative EUS elastography. Ultraschall Med. 2014, 35, 253–258. [Google Scholar] [CrossRef] [PubMed]
- Tsushima, Y.; Kusano, S. Age-dependent decline in parenchymal perfusion in the normal human pancreas: Measurement by dynamic computed tomography. Pancreas 1998, 17, 148–152. [Google Scholar] [CrossRef]
- Franken, F.H. Geriatric pancreatitis. Z. Gerontol. 1982, 15, 118–121. [Google Scholar] [PubMed]
- Rothenbacher, D.; Low, M.; Hardt, P.D.; Klor, H.U.; Ziegler, H.; Brenner, H. Prevalence and determinants of exocrine pancreatic insufficiency among older adults: Results of a population-based study. Scand. J. Gastroenterol. 2005, 40, 697–704. [Google Scholar] [CrossRef] [PubMed]
- Sakurai, T.; Kudo, M.; Fukuta, N.; Nakatani, T.; Kimura, M.; Park, A.M.; Munakata, H. Involvement of angiotensin II and reactive oxygen species in pancre-atic fibrosis. Pancreatology 2011, 11 (Suppl. 2), 7–13. [Google Scholar] [CrossRef] [PubMed]
- Ishibashi, T.; Matsumoto, S.; Harada, H.; Ochi, K.; Tanaka, J.; Seno, T.; Oka, H.; Miyake, H.; Kimura, I. Aging and exocrine pancreatic function evaluated by the recently standardized secretin test. Nihon Ronen Igakkai Zasshi Jpn. J. Geriatr. 1991, 28, 599–605. [Google Scholar] [CrossRef]
- Hedström, A.; Haas, S.; Berger, B.; Carlsson, J.; Löhr, J. Frequency of exocrine pancreatic insufficiency in 1105 patients with gastrointestinal symptoms. Pancreatology 2015, 15, S74. [Google Scholar] [CrossRef]
- Capurso, G.; Signoretti, M.; Archibugi, L.; Stigliano, S.; Delle Fave, G. Systematic review and meta-analysis: Small intestinal bacterial overgrowth in chronic pancreatitis. United Eur. Gastroenterol. J. 2016, 4, 697–705. [Google Scholar] [CrossRef]
- Frost, F.; Kacprowski, T.; Rühlemann, M.; Bülow, R.; Kühn, J.P.; Franke, A.; Heinsen, F.-A.; Pietzner, M.; Nauck, M.; Völker, U.; et al. Impaired Exocrine Pancreatic Function Associates With Changes in Intestinal Microbiota Composition and Diversity. Gastroenterology 2019, 156, 1010–1015. [Google Scholar] [CrossRef]
- Larsen, J.M. The immune response to Prevotella bacteria in chronic inflammatory disease. Immunology 2017, 151, 363–374. [Google Scholar] [CrossRef] [PubMed]
- Cao, Y.; Adhikari, S.; Ang, A.D.; Moore, P.K.; Bhatia, M. Mechanism of induction of pancreatic acinar cell apoptosis by hydrogen sulfide. Am. J. Physiol. Cell Physiol. 2006, 291, C503–C510. [Google Scholar] [CrossRef]
- Sanjeevi, R.; Jamwal, K.D.; Chowdhury, S.D.; Ramadass, B.; Gayathri, R.; Dutta, A.K.; Joseph, A.J.; Ramakrishna, B.S.; Chacko, A. Assessment of small intestinal bacterial overgrowth in chronic pancreatitis patients using jejunal aspirate culture and glucose hydrogen breath test. Scand. J. Gastroenterol. 2021, 56, 588–593. [Google Scholar] [CrossRef] [PubMed]
- Chonchubhair, H.M.N.; Bashir, Y.; Dobson, M.; Ryan, B.M.; Duggan, S.N.; Conlon, K.C. The prevalence of small intestinal bacterial overgrowth in non-surgical patients with chronic pancreatitis and pancreatic exocrine insufficiency (PEI). Pancreatology 2018, 18, 379–385. [Google Scholar] [CrossRef] [PubMed]
- Akshintala, V.S.; Talukdar, R.; Singh, V.K.; Goggins, M. The Gut Microbiome in Pancreatic Disease. Clin. Gastroenterol. Hepatol. 2019, 17, 290–295. [Google Scholar] [CrossRef] [PubMed]
- Ritz, S.; Hahn, D.; Wami, H.T.; Tegelkamp, K.; Dobrindt, U.; Schnekenburger, J. Gut microbiome as a response marker for pancreatic enzyme replacement therapy in a porcine model of exocrine pancreas insufficiency. Microb. Cell Fact. 2020, 19, 221. [Google Scholar] [CrossRef]
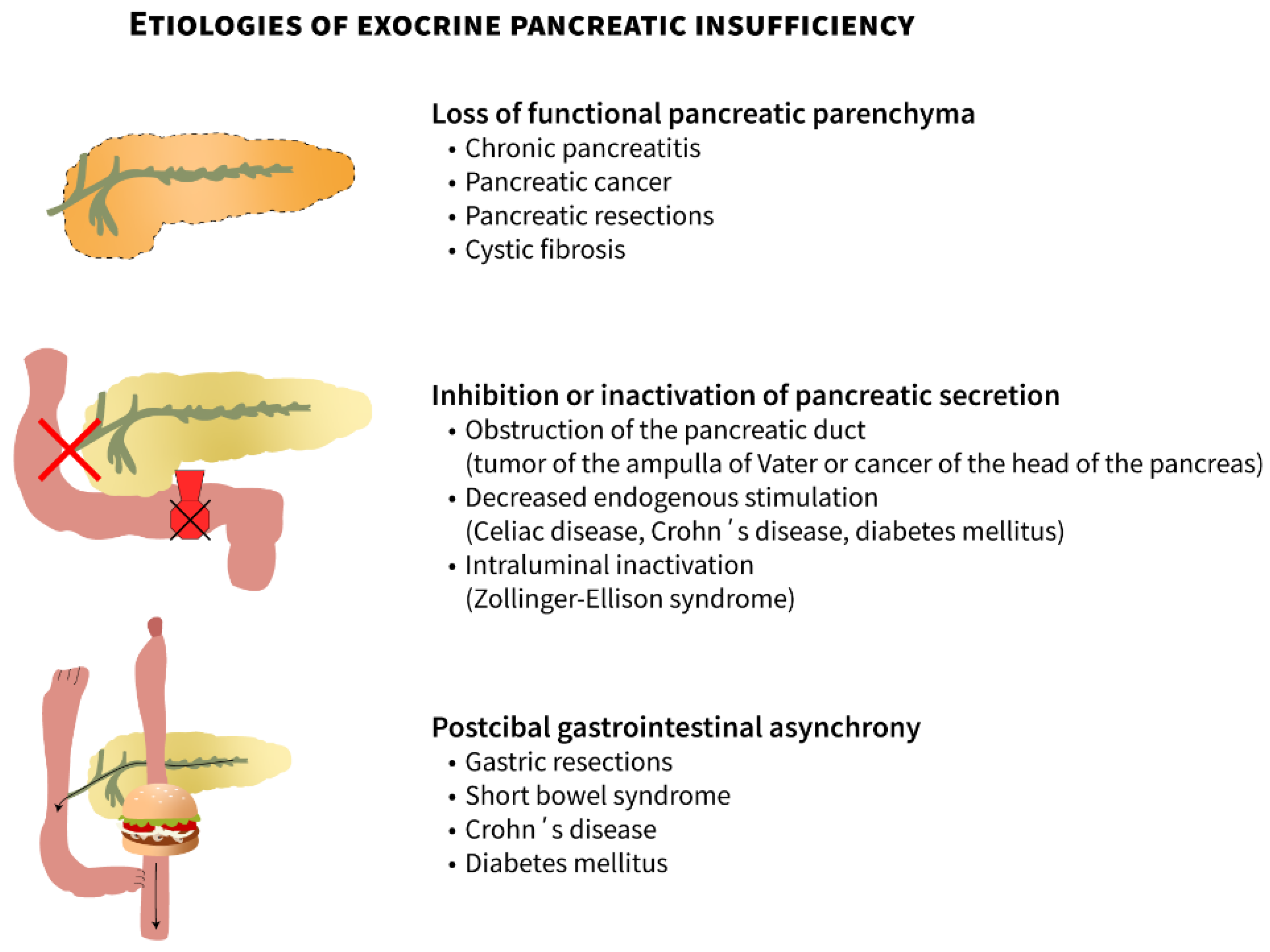
| Obvious association with EPI |
| Chronic pancreatitis (most common) |
| Pancreatic cancer |
| Pancreatic resections |
| Obstruction of the pancreatic duct (periampullary tumors) |
| Cystic fibrosis |
| Rare inherited syndromes (Shwachman–Diamond syndrome, etc.) |
| Not fully clarified mechanisms associated with EPI |
| Diabetes mellitus |
| Celiac disease |
| Inflammatory bowel disease (Crohn’s disease, ulcerative colitis) |
| Gastrointestinal surgeries other than pancreatic resection (e.g., gastric resection) |
| Zollinger–Ellison syndrome |
| Nonalcoholic fatty pancreas disease |
| Age |
| Composition and diversity of the intestinal microbiome, SIBO |
| Cystic fibrosis (most common) |
| Shwachman–Diamond syndrome |
| Chronic pancreatitis |
| Johanson–Blizzard syndrome |
| Pearson syndrome |
| Jeune syndrome |
| Pancreatic hypoplasia |
| Pancreatic aplasia |
| Isolated enzyme deficiencies |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kunovský, L.; Dítě, P.; Jabandžiev, P.; Eid, M.; Poredská, K.; Vaculová, J.; Sochorová, D.; Janeček, P.; Tesaříková, P.; Blaho, M.; et al. Causes of Exocrine Pancreatic Insufficiency Other Than Chronic Pancreatitis. J. Clin. Med. 2021, 10, 5779. https://doi.org/10.3390/jcm10245779
Kunovský L, Dítě P, Jabandžiev P, Eid M, Poredská K, Vaculová J, Sochorová D, Janeček P, Tesaříková P, Blaho M, et al. Causes of Exocrine Pancreatic Insufficiency Other Than Chronic Pancreatitis. Journal of Clinical Medicine. 2021; 10(24):5779. https://doi.org/10.3390/jcm10245779
Chicago/Turabian StyleKunovský, Lumír, Petr Dítě, Petr Jabandžiev, Michal Eid, Karolina Poredská, Jitka Vaculová, Dana Sochorová, Pavel Janeček, Pavla Tesaříková, Martin Blaho, and et al. 2021. "Causes of Exocrine Pancreatic Insufficiency Other Than Chronic Pancreatitis" Journal of Clinical Medicine 10, no. 24: 5779. https://doi.org/10.3390/jcm10245779
APA StyleKunovský, L., Dítě, P., Jabandžiev, P., Eid, M., Poredská, K., Vaculová, J., Sochorová, D., Janeček, P., Tesaříková, P., Blaho, M., Trna, J., Hlavsa, J., & Kala, Z. (2021). Causes of Exocrine Pancreatic Insufficiency Other Than Chronic Pancreatitis. Journal of Clinical Medicine, 10(24), 5779. https://doi.org/10.3390/jcm10245779






