Cutaneous Adverse Reactions Associated with SARS-CoV-2 Vaccines
Abstract
:1. Introduction
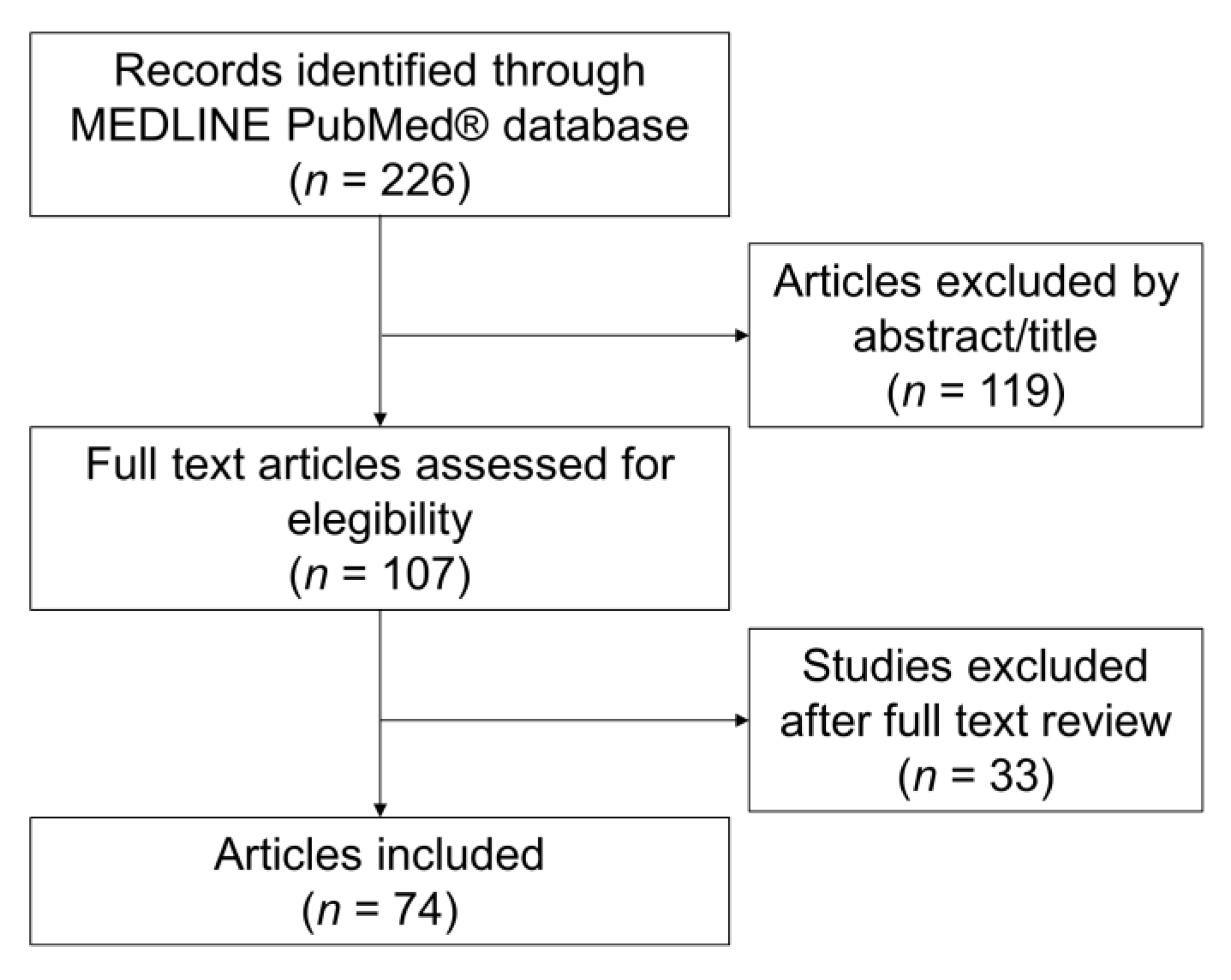
2. Materials and Methods
3. Results
3.1. Characteristics of Subjects Developing Cutaneous Adverse Reactions
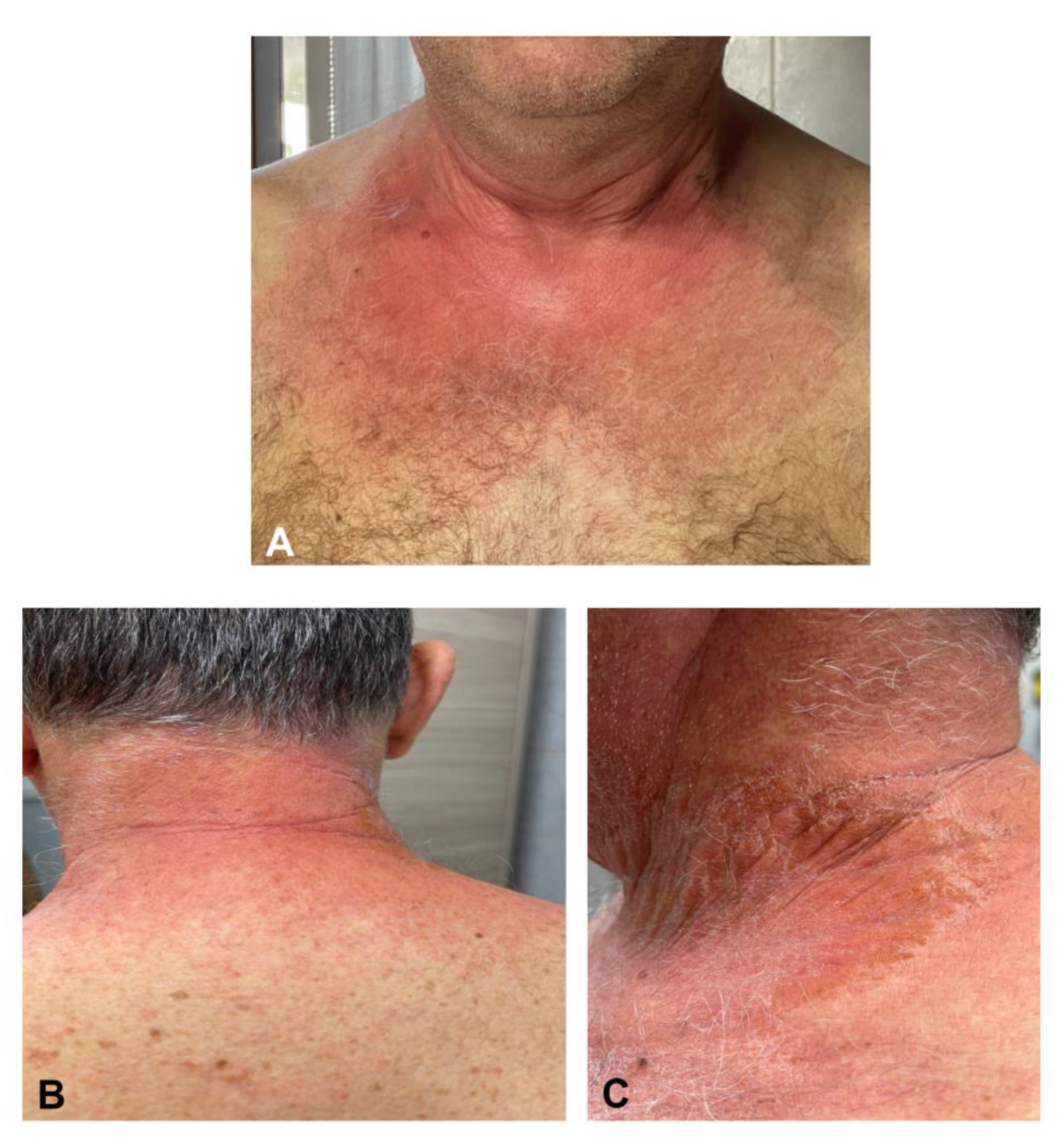
3.2. Injection Site Reactions
3.3. Filler Reactions
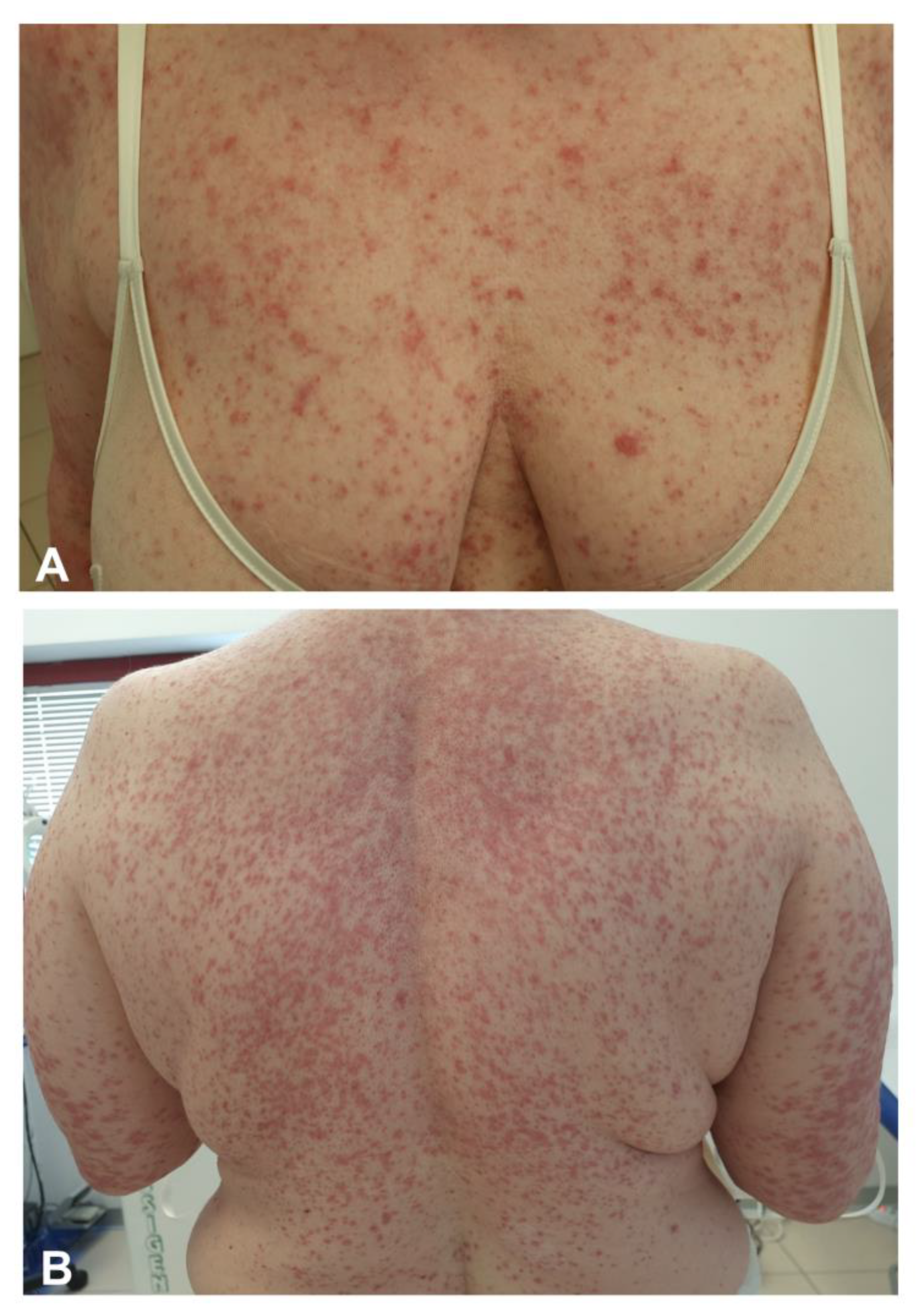
3.4. Exanthemas
3.5. Vascular Lesions
3.6. Urticaria/Angioedema
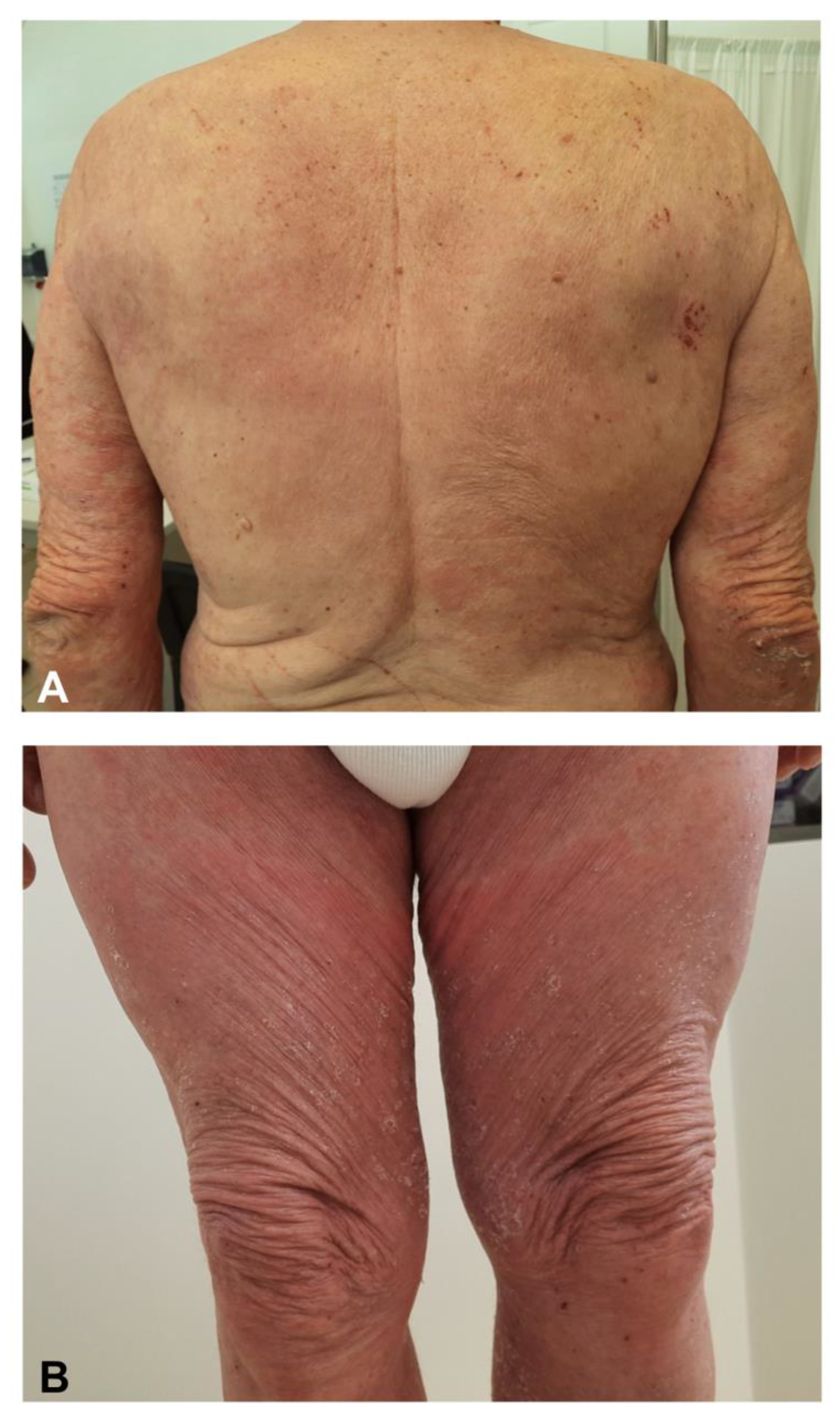
3.7. Eczematous Dermatitis
3.8. Autoimmune Bullous Reactions
3.9. Severe Cutaneous Adverse Reactions (SCARs)
3.10. Other Dermatoses
3.11. Flare of Existing Dermatoses
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Chakraborty, S.; Mallajosyula, V.; Tato, C.M.; Tan, G.S.; Wang, T.T. SARS-CoV-2 vaccines in advanced clinical trials: Where do we stand? Adv. Drug Deliv. Rev. 2021, 172, 314–338. [Google Scholar] [CrossRef] [PubMed]
- Hernández, A.F.; Calina, D.; Poulas, K.; Docea, A.O.; Tsatsakis, A.M. Safety of COVID-19 vaccines administered in the EU: Should we be concerned? Toxicol. Rep. 2021, 8, 871–879. [Google Scholar] [CrossRef] [PubMed]
- Teijaro, J.R.; Farber, D.L. COVID-19 vaccines: Modes of immune activation and future challenges. Nat. Rev. Immunol. 2021, 21, 195–197. [Google Scholar] [CrossRef] [PubMed]
- Castells, M.C.; Phillips, E.J. Maintaining Safety with SARS-CoV-2 Vaccines. N. Engl. J. Med. 2021, 384, 643–649. [Google Scholar] [CrossRef]
- Blumental, S.; Debré, P. Challenges and issues of anti-SARS-CoV-2 vaccines. Front. Med. 2021, 8, 664179. [Google Scholar] [CrossRef]
- Català, A.; Muñoz-Santos, C.; Galván-Casas, C.; Roncero Riesco, M.; Revilla Nebreda, D.; Solá-Truyols, A.; Giavedoni, M.; Llamas-Velasco, C.; González-Cruz, X.; Cubiró, R. Cutaneous reactions after SARS-COV-2 vaccination: A cross-sectional Spanish nationwide study of 405 cases. Br. J. Dermatol. 2021. Epub ahead of print. [Google Scholar] [CrossRef]
- McMahon, D.E.; Amerson, E.; Rosenbach, M.; Lipoff, J.B.; Moustafa, D.; Tyagi, A.; Desai, S.R.; French, L.E.; Lim, H.W.; Thiers, B.H.; et al. Cutaneous reactions reported after Moderna and Pfizer COVID-19 vaccination: A registry-based study of 414 cases. J. Am. Acad. Dermatol. 2021, 85, 46–55. [Google Scholar] [CrossRef]
- Robinson, L.B.; Fu, X.; Hashimoto, D.; Wickner, P.; Shenoy, E.S.; Landman, A.B.; Blumenthal, K.G. Incidence of Cutaneous Reactions After Messenger RNA COVID-19 Vaccines. JAMA Dermatol. 2021, 157, 1000–1002. [Google Scholar] [CrossRef]
- Saifuddin, A.; Koesnoe, S.; Kurniati, N.; Sirait, S.; Arisanty, R.; Yunihastuti, E. COVID Arm After Moderna Booster in Healthcare Worker: A Case Report. Acta Med. Indones. 2021, 53, 326–330. [Google Scholar]
- Samarakoon, U.; Alvarez-Arango, S.; Blumenthal, K.G. Delayed Large Local Reactions to mRNA Covid-19 Vaccines in Blacks, Indigenous Persons, and People of Color. N. Engl. J. Med. 2021, 385, 662–664. [Google Scholar] [CrossRef]
- Fernandez-Nieto, D.; Hammerle, J.; Fernandez-Escribano, M.; Moreno-Del Real, C.M.; Garcia-Abellas, P.; Carretero-Barrio, I.; Solano-Solares, E.; de-la-Hoz-Caballer, B.; Jimenez-Cauhe, J.; Ortega-Quijano, D.; et al. Skin manifestations of the BNT162b2 mRNA COVID-19 vaccine in healthcare workers. ‘COVID-arm’: A clinical and histological characterization. J. Eur. Acad. Dermatol. Venereol. 2021, 35, e425-7. [Google Scholar] [CrossRef]
- Lindgren, A.L.; Austin, A.H.; Welsh, K.M. COVID Arm: Delayed Hypersensitivity Reactions to SARS-CoV-2 Vaccines Misdiagnosed as Cellulitis. J. Prim. Care Community Health 2021, 12, 21501327211024431. [Google Scholar] [CrossRef]
- Gruenstein, D.; Levitt, J. Skin necrosis at both COVID-19 vaccine injection sites. JAAD Case Rep. 2021, 15, 67–68. [Google Scholar] [CrossRef]
- Blumenthal, K.G.; Freeman, E.E.; Saff, R.R.; Robinson, L.B.; Wolfson, A.R.; Foreman, R.K.; Hashimoto, D.; Banerji, A.; Li, L.; Anvari, S.; et al. Delayed Large Local Reactions to mRNA-1273 Vaccine against SARS-CoV-2. N. Engl. J. Med. 2021, 384, 1273–1277. [Google Scholar] [CrossRef]
- Maurelli, M.; Bergamaschi, R.; Antonini, A.; Fargnoli, M.C.; Puma, E.; Mallucci, G.; Totaro, R.; Girolomoni, G. Interferon-beta injection site reactions in patients with multiple sclerosis. J. Dermatol. Treat. 2018, 29, 831–834. [Google Scholar] [CrossRef]
- Michon, A. Hyaluronic acid soft tissue filler delayed inflammatory reaction following COVID-19 vaccination—A case report. J. Cosmet. Dermatol. 2021, 20, 2684–2690. [Google Scholar] [CrossRef]
- Munavalli, G.G.; Guthridge, R.; Knutsen-Larson, S.; Brodsky, A.; Matthew, E.; Landau, M. “COVID-19/SARS-CoV-2 virus spike protein-related delayed inflammatory reaction to hyaluronic acid dermal fillers: A challenging clinical conundrum in diagnosis and treatment”. Arch. Dermatol. Res. 2021, 1–15. [Google Scholar] [CrossRef]
- Annabi, E.; Dupin, N.; Sohier, P.; Garel, B.; Franck, N.; Aractingi, S.; Guégan, S.; Oulès, B. Rare cutaneous adverse effects of COVID-19 vaccines: A case series and review of the literature. J. Eur. Acad. Dermatol. Venereol. 2021. [Google Scholar] [CrossRef]
- Farinazzo, E.; Ponis, G.; Zelin, E.; Errichetti, E.; Stinco, G.; Pinzani, C.; Gambelli, A.; De Manzini, N.; Toffoli, L.; Moret, A.; et al. Cutaneous adverse reactions after m-RNA COVID-19 vaccine: Early reports from Northeast Italy. J. Eur. Acad. Dermatol. Venereol. 2021, 35, e548–e551. [Google Scholar] [CrossRef]
- Larson, V.; Seidenberg, R.; Caplan, A.; Brinster, N.K.; Meehan, S.A.; Kim, R.H. Clinical and histopathological spectrum of delayed adverse cutaneous reactions following COVID-19 vaccination. J. Cutan. Pathol. 2021, 1–8. [Google Scholar] [CrossRef]
- Jedlowski, P.M.; Jedlowski, M.F. Morbilliform rash after administration of Pfizer-BioNTech COVID-19 mRNA vaccine. Dermatol. Online J. 2021, 27, 13030. [Google Scholar] [CrossRef]
- Lam, M.; Egail, M.; Bedlow, A.J.; Tso, S. Ribonucleic acid COVID-19 vaccine-associated cutaneous adverse drug events: A case series of two patients. Clin. Exp. Dermatol. 2021, 46, 1131–1134. [Google Scholar] [CrossRef]
- Holmes, G.A.; Desai, M.; Limone, B.; Love, J.; Tawfik, M.; Wong, L.; Furukawa, B. A case series of cutaneous COVID-19 vaccine reactions at Loma Linda University Department of Dermatology. JAAD Case Rep. 2021, 16, 53–57. [Google Scholar] [CrossRef]
- Kadali, R.A.; Janagama, R.; Peruru, S.; Malayala, S.V. Side effects of BNT162b2 mRNA COVID-19 vaccine: A randomized, cross-sectional study with detailed self-reported symptoms from healthcare workers. Int. J. Infect. Dis. 2021, 106, 376–381. [Google Scholar] [CrossRef]
- D’Cruz, A.; Parker, H.; Saha, M. A Bullous Eruption following the Pfizer- BioNTech Covid-19 Vaccination. J. Eur. Acad. Dermatol. Venereol. 2021, 35, e864–e865. [Google Scholar] [CrossRef]
- Cyrenne, B.; Al-Mohammedi, F.; DeKoven, J.; Alhusayen, R. Pityriasis rosea-like eruptions following vaccination with BNT162b2 mRNA COVID-19 Vaccine. J. Eur. Acad. Dermatol. Venereol. 2021, 35, e546–e548. [Google Scholar] [CrossRef]
- Abdullah, L.; Hasbani, D.; Kurban, M.; Abbas, O. Pityriasis rosea after mRNA COVID-19 vaccination. Int. J. Dermatol. 2021, 60, 1150–1151. [Google Scholar] [CrossRef]
- Song, E.J.; Wong, A.J.S. Widespread annular eruption after Ad26.COV2.S COVID-19 vaccine. JAAD Case Rep. 2021, 13, 30–32. [Google Scholar] [CrossRef]
- Guzmán-Pérez, L.; Puerta-Peña, M.; Falkenhain-López, D.; Montero-Menárguez, J.; Gutiérrez-Collar, C.; Rodríguez-Peralto, J.L.; Sanz-Bueno, J. Small-vessel vasculitis following Oxford-AstraZeneca vaccination against SARS-CoV-2. J. Eur. Acad. Dermatol. Venereol. 2021, 35, e741–e743. [Google Scholar]
- Berry, C.T.; Eliliwi, M.; Gallagher, S.; Panaccione, S.; Klein, W.M.; Healy, A.L.; Stoecker, B.; Kallas, R. Cutaneous small vessel vasculitis following single-dose Janssen Ad26.COV2.S vaccination. JAAD Case Rep. 2021, 15, 11–14. [Google Scholar] [CrossRef] [PubMed]
- Felicetti, P.; Trotta, F.; Bonetto, C.; Santuccio, C.; Brauchli Pernus, Y.; Burgner, D.; Chandler, R.; Girolomoni, G.; Hadden, R.D.M.; Kochhar, S.; et al. Spontaneous reports of vasculitis as an adverse event following immunization: A descriptive analysis across three international databases. Vaccine 2016, 34, 6634–6640. [Google Scholar] [CrossRef] [PubMed]
- Guetl, K.; Gary, T.; Raggam, R.B.; Schmid, J.; Wölfler, A.; Brodmann, M. SARS-CoV-2 vaccine-induced immune thrombotic thrombocytopenia treated with immunoglobulin and argatroban. Lancet 2021, 397, e19. [Google Scholar] [CrossRef]
- King, E.R.; Towner, E. A Case of Immune Thrombocytopenia After BNT162b2 mRNA COVID-19 Vaccination. Am. J. Case Rep. 2021, 22, e931478-1. [Google Scholar] [CrossRef] [PubMed]
- Maayan, H.; Kirgner, I.; Gutwein, O.; Herzog-Tzarfati, K.; Rahimi-Levene, N.; Koren-Michowitz, M.; Blickstein, D. Acquired thrombotic thrombocytopenic purpura: A rare disease associated with BNT162b2 vaccine. J. Thromb. Haemost. 2021, 19, 2314–2317. [Google Scholar] [CrossRef]
- Yu, J.N.; Angeles, C.B.; Lim, H.G.; Chavez, C.; Roxas-Rosete, C. Cutaneous reactions to inactivated SARS-CoV-2 vaccine and ChAdOx1-S (recombinant) vaccine against SARS-CoV-2: A case series from the Philippines. J. Eur. Acad. Dermatol. Venereol. 2021, 35, e841–e845. [Google Scholar] [CrossRef]
- Mazzatenta, C.; Piccolo, V.; Pace, G.; Romano, I.; Argenziano, G.; Bassi, A. Purpuric lesions on the eyelids developed after BNT162b2 mRNA COVID-19 vaccine: Another piece of SARS-CoV-2 skin puzzle? J. Eur. Acad. Dermatol. Venereol. 2021, 35, e543–e545. [Google Scholar] [CrossRef]
- Pasternack, R.; Pohjavaara, S. A skin reaction with rust-like discolouration to mRNA COVID-19 vaccine. J. Eur. Acad. Dermatol. Venereol. 2021, 35, e737–e738. [Google Scholar] [CrossRef]
- Heck, E.; Rankin, B.D.; Schneider, M.; Prajapati, V.H. Flagellate Purpura Associated With COVID-19 Vaccination. J. Eur. Acad. Dermatol. Venereol. 2021. [Google Scholar] [CrossRef]
- Temiz, S.A.; Abdelmaksoud, A.; Dursun, R.; Vestita, M. Acral chilblain-like lesions following inactivated SARS-CoV-2 vaccination. Int. J. Dermatol. 2021, 60, 1152–1153. [Google Scholar] [CrossRef]
- Davido, B.; Mascitti, H.; Fortier-Beaulieu, M.; Jaffal, K.; de Truchis, P. ’Blue toes’ following vaccination with the BNT162b2 mRNA COVID-19 vaccine. J. Travel Med. 2021, 28, taab024. [Google Scholar] [CrossRef]
- Pileri, A.; Guglielmo, A.; Raone, B.; Patrizi, A. Chilblain lesions after COVID-19 mRNA vaccine. Br. J. Dermatol. 2021, 185, e3. [Google Scholar] [CrossRef]
- Corbeddu, M.; Diociaiuti, A.; Vinci, M.R.; Santoro, A.; Camisa, V.; Zaffina, S.; El Hachem, M. Transient cutaneous manifestations after administration of Pfizer-BioNTech COVID-19 Vaccine: An Italian single-centre case series. J. Eur. Acad. Dermatol. Venereol. 2021, 35, e483–e485. [Google Scholar] [CrossRef]
- Al-Ansari, R.Y.; Al-Sharari, M.; Al-Saadi, T. Palms and soles itchiness as a side effect of COVID-19 vaccination. J. Infect. Public Health 2021, 14, 1389–1391. [Google Scholar] [CrossRef]
- Bianchi, L.; Biondi, F.; Hansel, K.; Murgia, N.; Tramontana, M.; Stingeni, L. Skin tests in urticaria/angioedema and flushing to Pfizer-BioNTech SARS-CoV-2 vaccine: Limits of intradermal testing. Allergy 2021, 76, 2605–2607. [Google Scholar] [CrossRef]
- Weinstock-Guttman, B.; Jakimovski, D. Late-onset cutaneous reaction to BNT162b2 mRNA COVID-19 vaccine in an immunocompromised patient. Mult. Scler. 2021. [Google Scholar] [CrossRef]
- Leasure, A.C.; Cowper, S.E.; McNiff, J.; Cohen, J.M. Generalized eczematous reactions to the Pfizer-BioNTech COVID-19 vaccine. J. Eur. Acad. Dermatol. Venereol. 2021. [Google Scholar] [CrossRef]
- Coto-Segura, P.; Fernández-Prada, M.; Mir-Bonafé, M.; García-García, B.; González-Iglesias, I.; Alonso-Penanes, P.; González-Guerrero, M.; Gutiérrez-Palacios, A.; Miranda-Martínez, E.; Martinón-Torres, F. Vesicolous-bullous skin reactions induced by COVID-19 mRNA vaccine: Report of four cases and review of the literature. Clin. Exp. Dermatol. 2021. [Google Scholar] [CrossRef]
- Tomayko, M.M.; Damsky, W.; Fathy, R.; McMahon, D.E.; Turner, N.; Valentin, M.N.; Rallis, T.; Aivaz, O.; Fox, L.P.; Freeman, E.E. Subepidermal blistering eruptions, including bullous pemphigoid, following COVID-19 vaccination. J. Allergy Clin. Immunol. 2021, 148, 750–751. [Google Scholar] [CrossRef]
- Solimani, F.; Mansour, Y.; Didona, D.; Dilling, A.; Ghoreschi, K.; Meier, K. Development of severe pemphigus vulgaris following SARS-CoV-2 vaccination with BNT162b2. J. Eur. Acad. Dermatol. Venereol. 2021, 35, e649. [Google Scholar] [CrossRef]
- Lospinoso, K.; Nichols, C.S.; Malachowski, S.J.; Mochel, M.C.; Nutan, F. A case of severe cutaneous adverse reaction following administration of the Janssen Ad26.COV2.S COVID-19 vaccine. JAAD Case Rep. 2021, 13, 134–137. [Google Scholar] [CrossRef]
- Dash, S.; Sirka, C.S.; Mishra, S.; Viswan, P. COVID-19 vaccine-induced Stevens-Johnson syndrome. Clin. Exp. Dermatol. 2021. [Google Scholar] [CrossRef]
- Bakir, M.; Almeshal, H.; Alturki, R.; Obaid, S.; Almazroo, A. Toxic Epidermal Necrolysis Post COVID-19 Vaccination—First Reported Case. Cureus 2021, 13, e17215. [Google Scholar] [CrossRef]
- Khalid, M.; Lipka, O.; Becker, C. Moderna COVID-19 Vaccine Induced Skin Rash. Vis. J. Emerg. Med. 2021, 25, 101108. [Google Scholar] [CrossRef]
- Kong, J.; Cuevas-Castillo, F.; Nassar, M.; Lei, C.M.; Idrees, Z.; Fix, W.C.; Halverstam, C.; Mir, A.; Elbendary, A.; Mathew, A. Bullous drug eruption after second dose of mRNA-1273 (Moderna) COVID-19 vaccine: Case report. J. Infect. Public Health 2021, 14, 1392–1394. [Google Scholar] [CrossRef]
- Torrealba-Acosta, G.; Martin, J.C.; Huttenbach, Y.; Garcia, C.R.; Sohail, M.R.; Agarwal, S.K.; Wasko, C.; Bershad, E.M.; Hirzallah, M.I. Acute encephalitis, myoclonus and Sweet syndrome after mRNA-1273 vaccine. BMJ Case Rep. 2021, 14, e243173. [Google Scholar] [CrossRef]
- Mintoff, D.; Pisani, D.; Betts, A.; Scerri, L. SARS-CoV-2 mRNA vaccine-associated fixed drug eruption. J. Eur. Acad. Dermatol. Venereol. 2021, 35, e560–e563. [Google Scholar] [CrossRef]
- Ciccarese, G.; Drago, F.; Rebora, A.; Parodi, A. Two cases of papulo-pustular rosacea-like eruptions following COVID-19 vaccinations. J. Eur. Acad. Dermatol. Venereol. 2021. [Google Scholar] [CrossRef]
- Sotiriou, E.; Tsentemeidou, A.; Bakirtzi, K.; Lallas, A.; Ioannides, D.; Vakirlis, E. Psoriasis exacerbation after COVID-19 vaccination: A report of 14 cases from a single centre. J. Eur. Acad. Dermatol. Venereol. 2021, 35, e857–e859. [Google Scholar] [CrossRef]
- Krajewski, P.K.; Matusiak, Ł.; Szepietowski, J.C. Psoriasis flare-up associated with second dose of Pfizer-BioNTech BNT16B2b2 COVID-19 mRNA vaccine. J. Eur. Acad. Dermatol. Venereol. 2021, 35, e632–e634. [Google Scholar] [CrossRef]
- Quattrini, L.; Verardi, L.; Caldarola, G.; Peluso, G.; De Simone, C.; D’Agostino, M. New onset of remitting seronegative symmetrical synovitis with pitting oedema and palmoplantar psoriasis flare-up after Sars-Cov-2 vaccination. J. Eur. Acad. Dermatol. Venereol. 2021, 35, e727–e729. [Google Scholar] [CrossRef]
- Lehmann, M.; Schorno, P.; Hunger, R.E.; Heidemeyer, K.; Feldmeyer, L.; Yawalkar, N. New onset of mainly guttate psoriasis after COVID-19 vaccination: A case report. J. Eur. Acad. Dermatol. Venereol. 2021, 35, e752–e755. [Google Scholar] [CrossRef] [PubMed]
- Elamin, S.; Hinds, F.; Tolland, J. A case of de novo generalised pustular psoriasis following Oxford-AstraZeneca COVID-19 Vaccine. Clin. Exp. Dermatol. 2021. [Google Scholar] [CrossRef]
- Hunjan, M.K.; Roberts, C.; Karim, S.; Hague, J. Pityriasis rubra pilaris-like eruption following administration of the BNT163b2 (Pfizer-BioNTech) mRNA COVID-19 vaccine. Clin. Exp. Dermatol. 2021. [Google Scholar] [CrossRef] [PubMed]
- Alflen, C.; Birch, K.; Shilian, R.; Wu, S.S.; Hostoffer, R., Jr. Two Cases of Well Controlled Chronic Spontaneous Urticaria Triggered by the Moderna COVID-19 Vaccine. Allergy Rhinol. 2021, 12, 21526567211026271. [Google Scholar] [CrossRef] [PubMed]
- Damiani, G.; Pacifico, A.; Pelloni, F.; Iorizzo, M. The first dose of COVID-19 vaccine may trigger pemphigus and bullous pemphigoid flares: Is the second dose therefore contraindicated? J. Eur. Acad. Dermatol. Venereol. 2021, 35, e645. [Google Scholar] [CrossRef] [PubMed]
- Sahni, M.K.; Roy, K.; Asati, D.P.; Khurana, U. An old entity, a new trigger: Post COVID-19 vaccine Pityriasis rubra pilaris. Int. J. Risk Saf. Med. 2021, 1–4, preprint. [Google Scholar] [CrossRef] [PubMed]
- Cohen, S.R.; Prussick, L.; Kahn, J.S.; Gao, D.X.; Radfar, A.; Rosmarin, D. Leukocytoclastic vasculitis flare following the COVID-19 vaccine. Int. J. Dermatol. 2021, 60, 1032–1033. [Google Scholar] [CrossRef]
- Lavery, M.J.; Nawimana, S.; Parslew, R.; Stewart, L. A flare of pre-existing erythema multiforme following BNT162b2 (Pfizer-BioNTech) COVID-19 vaccine. Clin. Exp. Dermatol. 2021, 46, 1325–1327. [Google Scholar] [CrossRef]
- Elbæk, M.V.; Vinding, G.R.; Jemec, G.B.E. Darier’s Disease Flare following COVID-19 Vaccine. Case Rep. Dermatol. 2021, 13, 432–436. [Google Scholar] [CrossRef]
- Kreuter, A.; Burmann, S.N.; Burkert, B.; Oellig, F.; Michalowitz, A.L. Transition of cutaneous into systemic lupus erythematosus following adenoviral vector-based SARS-CoV-2 vaccination. J. Eur. Acad. Dermatol. Venereol. 2021, 35, e733–e735. [Google Scholar] [CrossRef]
- Soyfer, V.; Gutfeld, O.; Shamai, S.; Schlocker, A.; Merimsky, O. COVID-19 Vaccine-Induced Radiation Recall Phenomenon. Int. J. Radiat. Oncol. Biol. Phys. 2021, 110, 957–961. [Google Scholar] [CrossRef]
- Stewart, R.; McDowell, L. Radiation Recall Phenomenon Following COVID-19 Vaccination. Int. J. Radiat. Oncol. Biol. Phys. 2021, 111, 835–836. [Google Scholar] [CrossRef]
- Lopatynsky-Reyes, E.Z.; Acosta-Lazo, H.; Ulloa-Gutierrez, R.; Ávila-Aguero, M.L.; Chacon-Cruz, E. BCG Scar Local Skin Inflammation as a Novel Reaction Following mRNA COVID-19 Vaccines in Two International Healthcare Workers. Cureus 2021, 13, e14453. [Google Scholar] [CrossRef]
- Vastarella, M.; Picone, V.; Martora, F.; Fabbrocini, G. Herpes zoster after ChAdOx1 nCoV-19 vaccine: A case series. J. Eur. Acad. Dermatol. Venereol. 2021, 35, e845–e846. [Google Scholar] [CrossRef]
- Özdemir, A.K.; Kayhan, S.; Çakmak, S.K. Herpes zoster after inactivated SARS-CoV-2 vaccine in two healthy young adults. J. Eur. Acad. Dermatol. Venereol. 2021, 35, e846–e847. [Google Scholar] [CrossRef]
- David, E.; Landriscina, A. Herpes Zoster Following COVID-19 Vaccination. J. Drugs Dermatol. 2021, 20, 898–900. [Google Scholar]
- Santovito, L.S.; Pinna, G. A case of reactivation of varicella-zoster virus after BNT162b2 vaccine second dose? Inflamm. Res. 2021, 70, 935–937. [Google Scholar] [CrossRef]
- Sprute, R.; Schumacher, S.; Pauls, M.; Pauls, W.; Cornely, O.A. Delayed Cutaneous Hypersensitivity Reaction to Vaxzevria (ChAdOx1-S) Vaccine against SARS-CoV-2. Drugs R D 2021, 1–4. [Google Scholar] [CrossRef]
- Gisondi, P.; PIaserico, S.; Bordin, C.; Alaibac, M.; Girolomoni, G.; Naldi, L. Cutaneous manifestations of SARS-CoV-2 infection: A clinical update. J. Eur. Acad. Dermatol. Venereol. 2020, 34, 2499–2504. [Google Scholar] [CrossRef]
- Caubet, J.C.; Ponvert, C. Vaccine allergy. Immunol. Allergy Clin. N. Am. 2014, 34, 597–613. [Google Scholar] [CrossRef]
- Bonadonna, P.; Brockow, K.; Niedoszytko, M.; Elberink, H.O.; Akin, C.; Nedoszytko, B.; Butterfield, J.H.; Alvarez-Twose, I.; Sotlar, K.; Schwaab, J.; et al. COVID-19 Vaccination in Mastocytosis: Recommendations of the European Competence Network on Mastocytosis (ECNM) and American Initiative in Mast Cell Diseases (AIM). J. Allergy Clin. Immunol. Pract. 2021, 9, 2139–2144. [Google Scholar] [CrossRef]
- Ring, J.; Worm, M.; Wollenberg, A.; Thyssen, J.P.; Jakob, T.; Klimek, L.; Bangert, C.; Barbarot, S.; Bieber, T.; de Bruin-Weller, M.S.; et al. Risk of severe allergic reactions to COVID-19 vaccines among patients with allergic skin diseases—Practical recommendations. A position statement of ETFAD with external experts. J. Eur. Acad. Dermatol. Venereol. 2021, 35, e362–e365. [Google Scholar] [CrossRef]
- Kim, M.A.; Lee, Y.W.; Kim, S.R.; Kim, J.H.; Min, T.K.; Park, H.S.; Shin, M.; Ye, Y.M.; Lee, S.; Lee, J.; et al. COVID-19 Vaccine-associated Anaphylaxis and Allergic Reactions: Consensus Statements of the KAAACI Urticaria/Angioedema/Anaphylaxis Working Group. Allergy Asthma Immunol. Res. 2021, 13, 526–544. [Google Scholar] [CrossRef]
- Centers for Disease Control and Prevention. What to Do If You Have an Allergic Reaction After Getting a COVID-19 Vaccine. 2021. Available online: https://www.cdc.gov/coronavirus/2019-ncov/vaccines/safety/allergic-reaction.html (accessed on 27 October 2021).

| Pattern | Subtype | Reported Associated Vaccines |
|---|---|---|
| Injection site reactions | Immediate local reaction | All |
| Delayed local reaction | ||
| Filler reactions | - | Pfizer/BioNTech, Moderna |
| Exanthemas | Erythematous confluent | All |
| Maculopapular | ||
| Papulovesicular | ||
| Pityriasis rosea like | Pfizer/BioNTech, Moderna, AstraZeneca/Oxford | |
| SDRIFE | Pfizer/BioNTech | |
| Annular | Johnson & Johnson/Janssen | |
| Vascular lesions | Leucocytoclasic vasculitis | All |
| Urticarial vasculitis | Pfizer/BioNTech | |
| Purpura/petechiae | All | |
| Livedo racemosa | Pfizer/BioNTech | |
| Chilblain-like | Pfizer/BioNTech, Moderna | |
| Erythromelalgia | Pfizer/BioNTech, Moderna | |
| Urticaria | Wheals and/or angioedema | Pfizer/BioNTech, Moderna, AstraZeneca/Oxford |
| Eczematous dermatitis | - | Pfizer/BioNTech, Moderna |
| Autoimmune bullous reactions | Subepidermal bullous diseases | Pfizer/BioNTech, Moderna |
| Pemphigus | Pfizer/BioNTech | |
| SCARs | AGEP | AstraZeneca/Oxford |
| DRESS | Johnson & Johnson/Janssen | |
| SJS/TEN | Pfizer/BioNTech, AstraZeneca/Oxford | |
| EM major | Moderna | |
| Extensive FDE | Moderna | |
| Other dermatoses | Sweet syndrome | Moderna |
| Rosacea-like eruption | Pfizer/BioNTech, Johnson & Johnson/Janssen | |
| Psoriasis (plaque, guttate, palmoplantar, and generalized pustular) | Pfizer/BioNTech, Moderna, Johnson & Johnson/Janssen | |
| Pityriasis rubra pilaris-like eruption | Pfizer/BioNTech |
| Type | Subtype | Reported Associated Vaccines |
|---|---|---|
| Immuno-mediated dermatoses | Chronic plaque psoriasis | Pfizer/BioNTech, Moderna, Johnson & Johnson/Janssen |
| Atopic dermatitis | Pfizer/BioNTech, Moderna | |
| Lichen ruber planus | Pfizer/BioNTech, Moderna | |
| Chronic spontaneous urticaria | Moderna | |
| Bullous pemphigoid | Pfizer/BioNTech, Moderna | |
| Pemphigus vulgaris | Pfizer/BioNTech, Moderna | |
| Pityriasis rubra pilaris | AstraZeneca/Oxford | |
| Cutaneous small-vessel vasculitis | Pfizer/BioNTech | |
| Erythema multiforme | Pfizer/BioNTech | |
| Darier’s disease | AstraZeneca/Oxford | |
| Systemic lupus erythematosus | AstraZeneca/Oxford | |
| Radiation recall phenomenon | Pfizer/BioNTech, AstraZeneca/Oxford | |
| BCG inflammation | Pfizer/BioNTech, Moderna | |
| Infectious dermatoses | HSV reactivation | All |
| VZV reactivation |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bellinato, F.; Maurelli, M.; Gisondi, P.; Girolomoni, G. Cutaneous Adverse Reactions Associated with SARS-CoV-2 Vaccines. J. Clin. Med. 2021, 10, 5344. https://doi.org/10.3390/jcm10225344
Bellinato F, Maurelli M, Gisondi P, Girolomoni G. Cutaneous Adverse Reactions Associated with SARS-CoV-2 Vaccines. Journal of Clinical Medicine. 2021; 10(22):5344. https://doi.org/10.3390/jcm10225344
Chicago/Turabian StyleBellinato, Francesco, Martina Maurelli, Paolo Gisondi, and Giampiero Girolomoni. 2021. "Cutaneous Adverse Reactions Associated with SARS-CoV-2 Vaccines" Journal of Clinical Medicine 10, no. 22: 5344. https://doi.org/10.3390/jcm10225344
APA StyleBellinato, F., Maurelli, M., Gisondi, P., & Girolomoni, G. (2021). Cutaneous Adverse Reactions Associated with SARS-CoV-2 Vaccines. Journal of Clinical Medicine, 10(22), 5344. https://doi.org/10.3390/jcm10225344







