Purification of Pharmaceutical Solvents by Pervaporation through Hybrid Silica Membranes
Abstract
1. Introduction
2. Theoretical Aspects
3. Materials and Methods
3.1. Chemical Products and Membrane
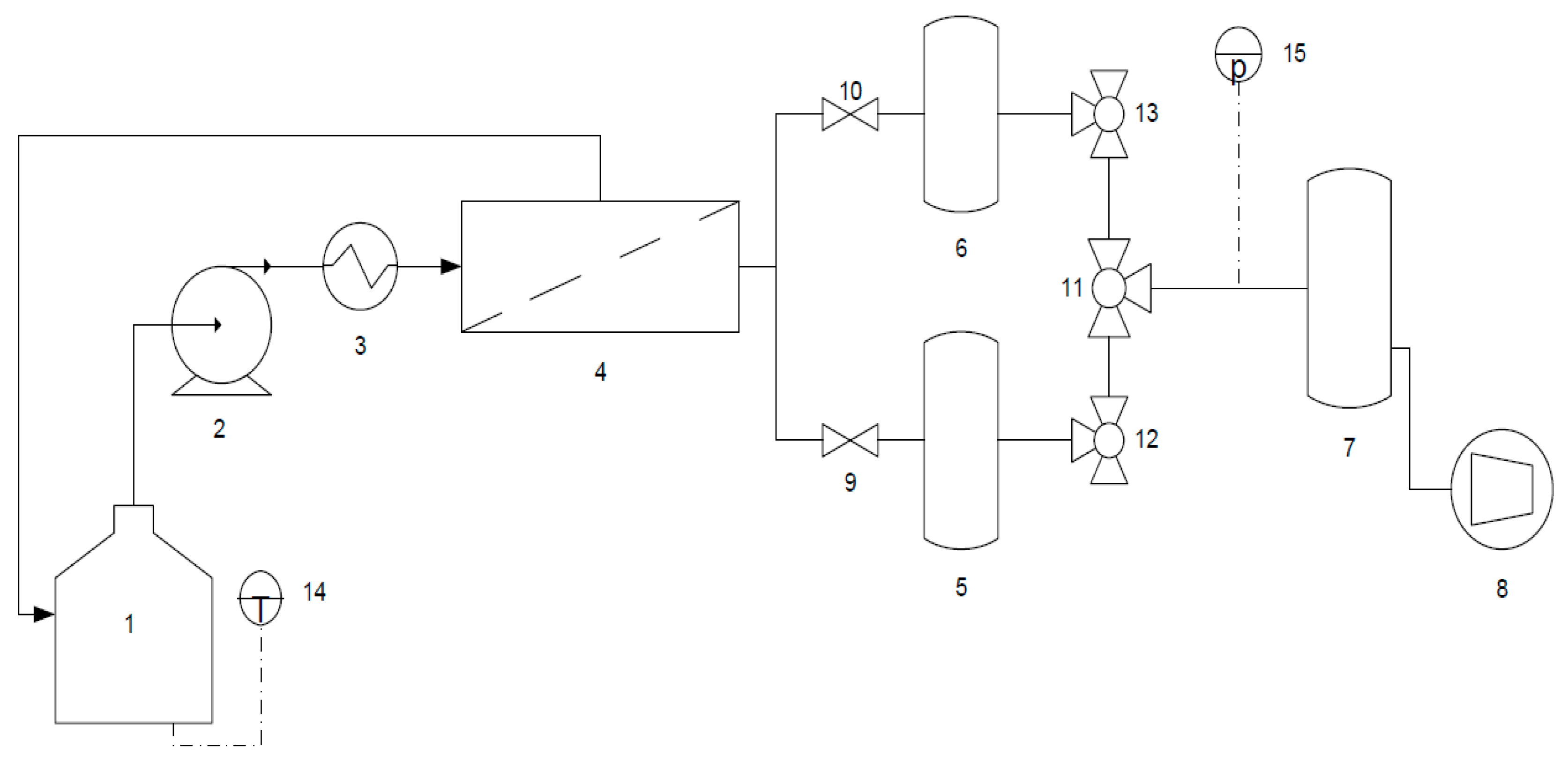
3.2. Experimental Set-Up
4. Results and Discussion
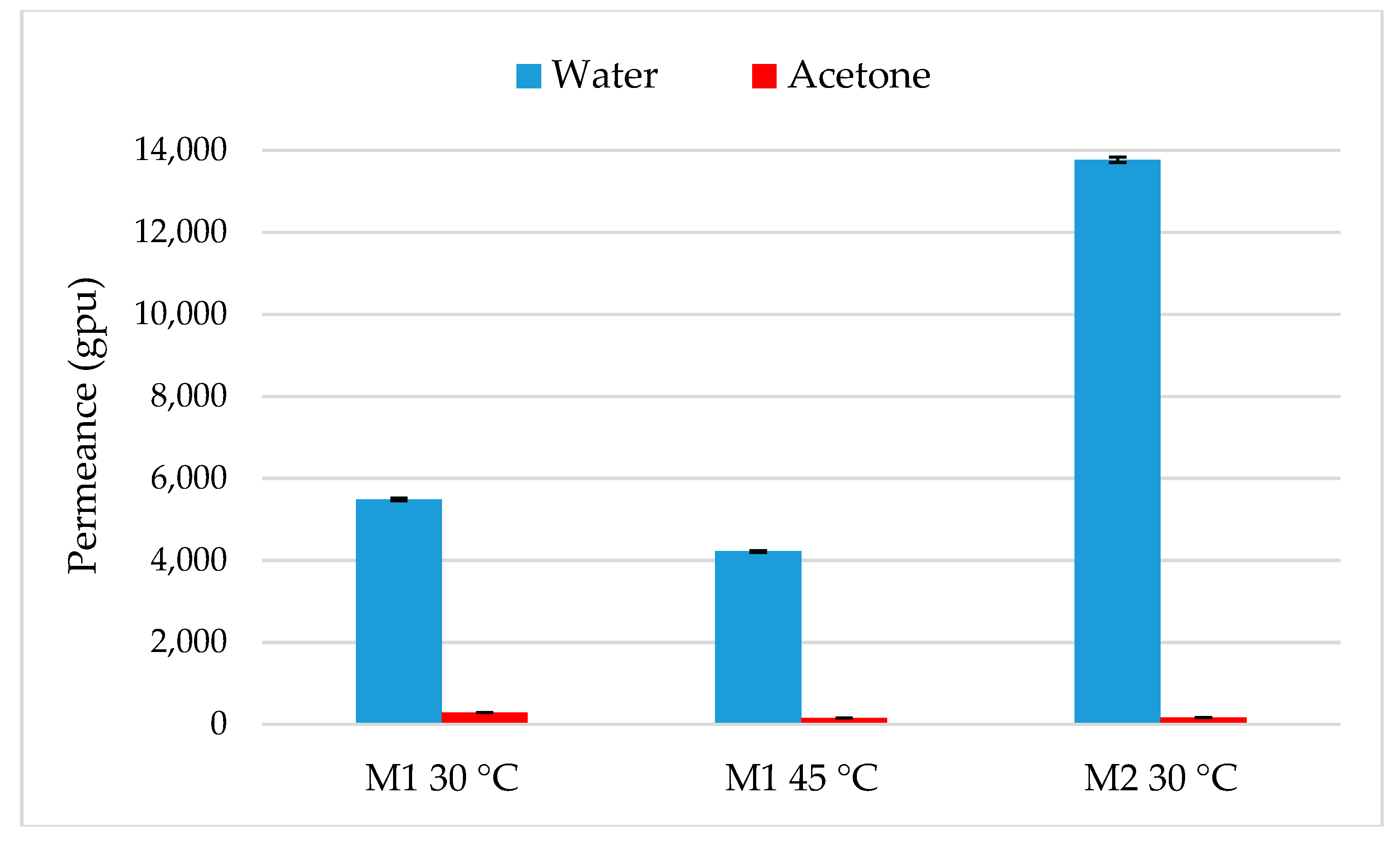
4.1. Pure Solvents Permeation
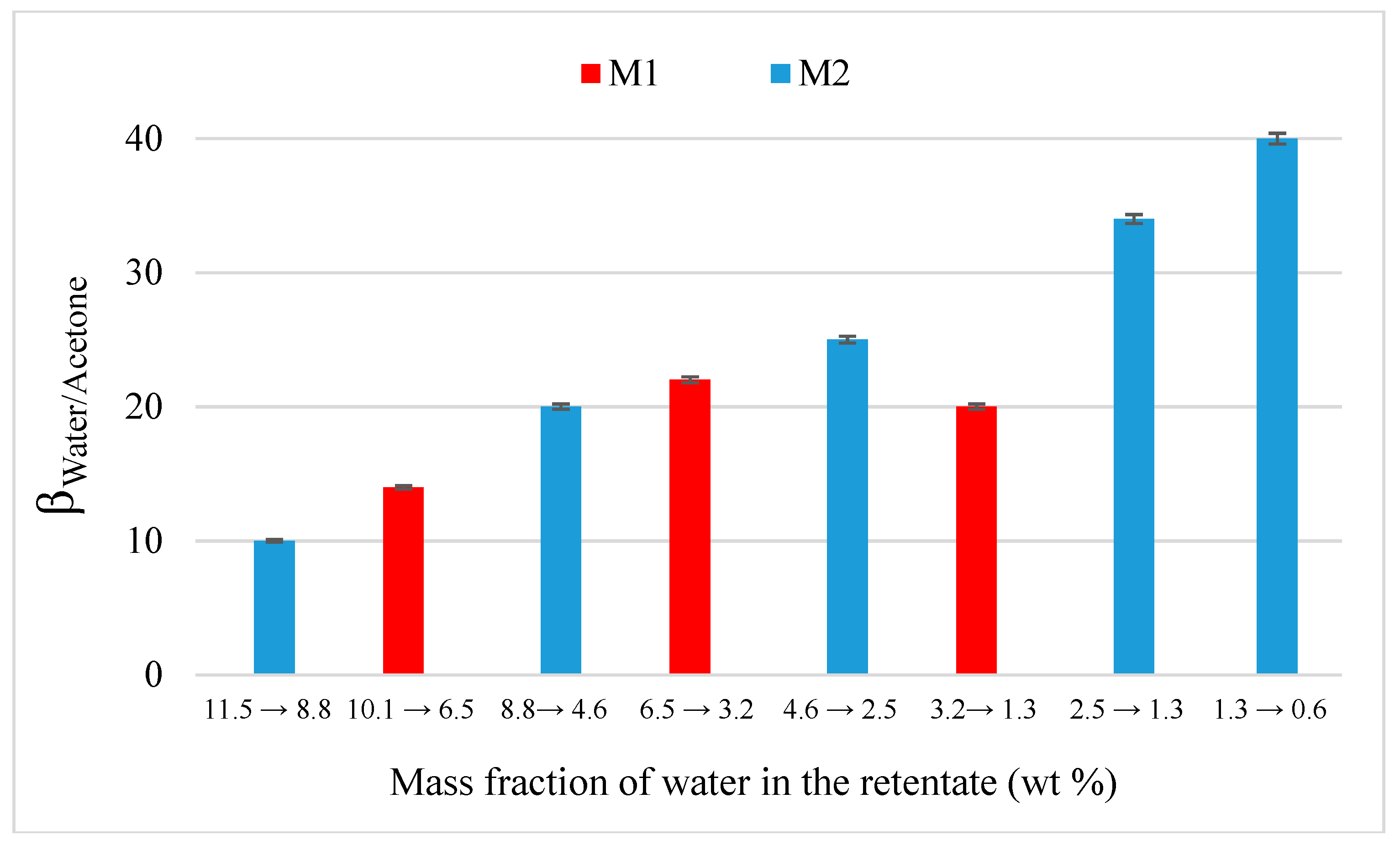
4.2. Acetone Dehydration
4.3. Organic Purification from Multi-Components Mixtures
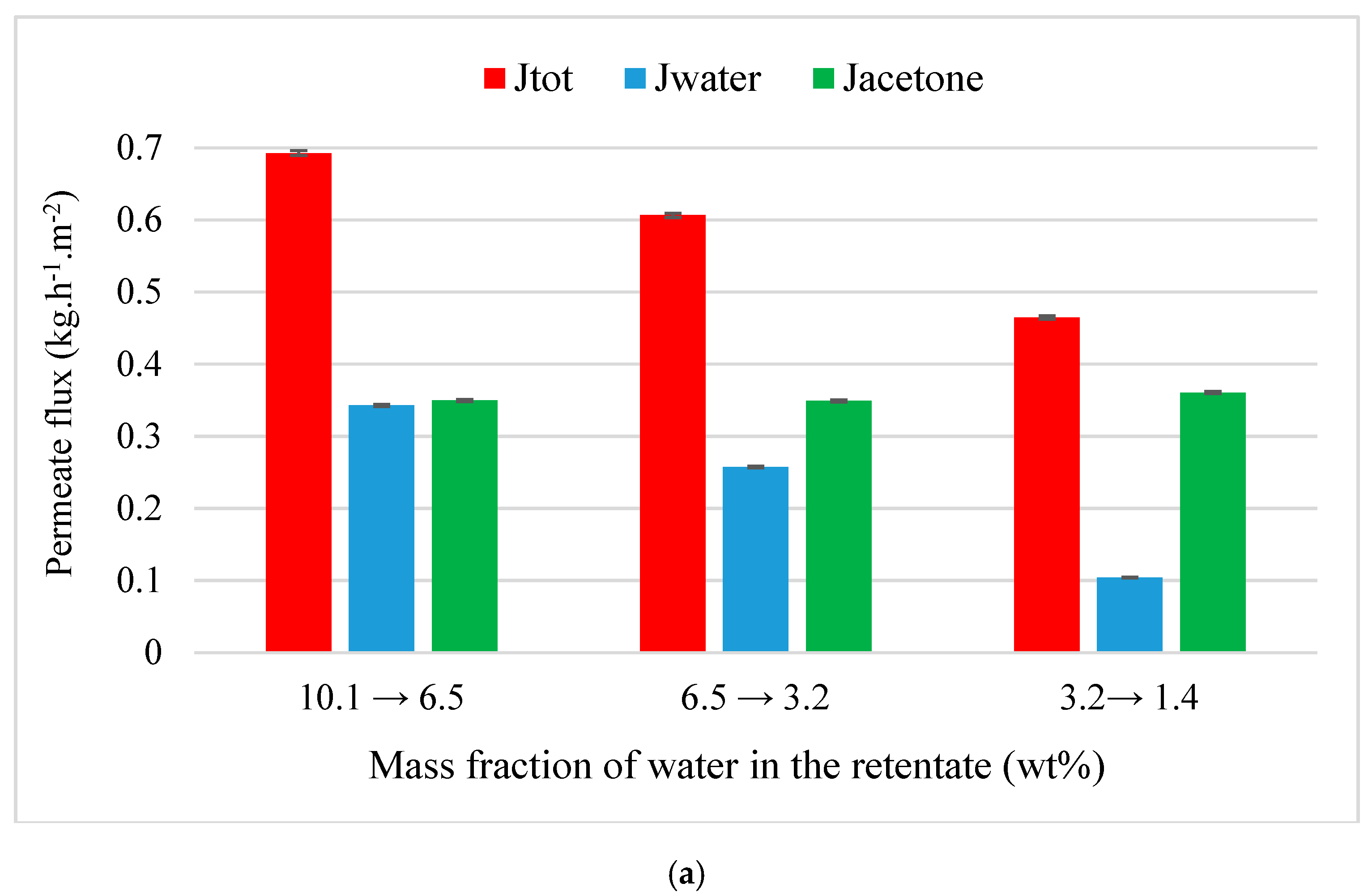
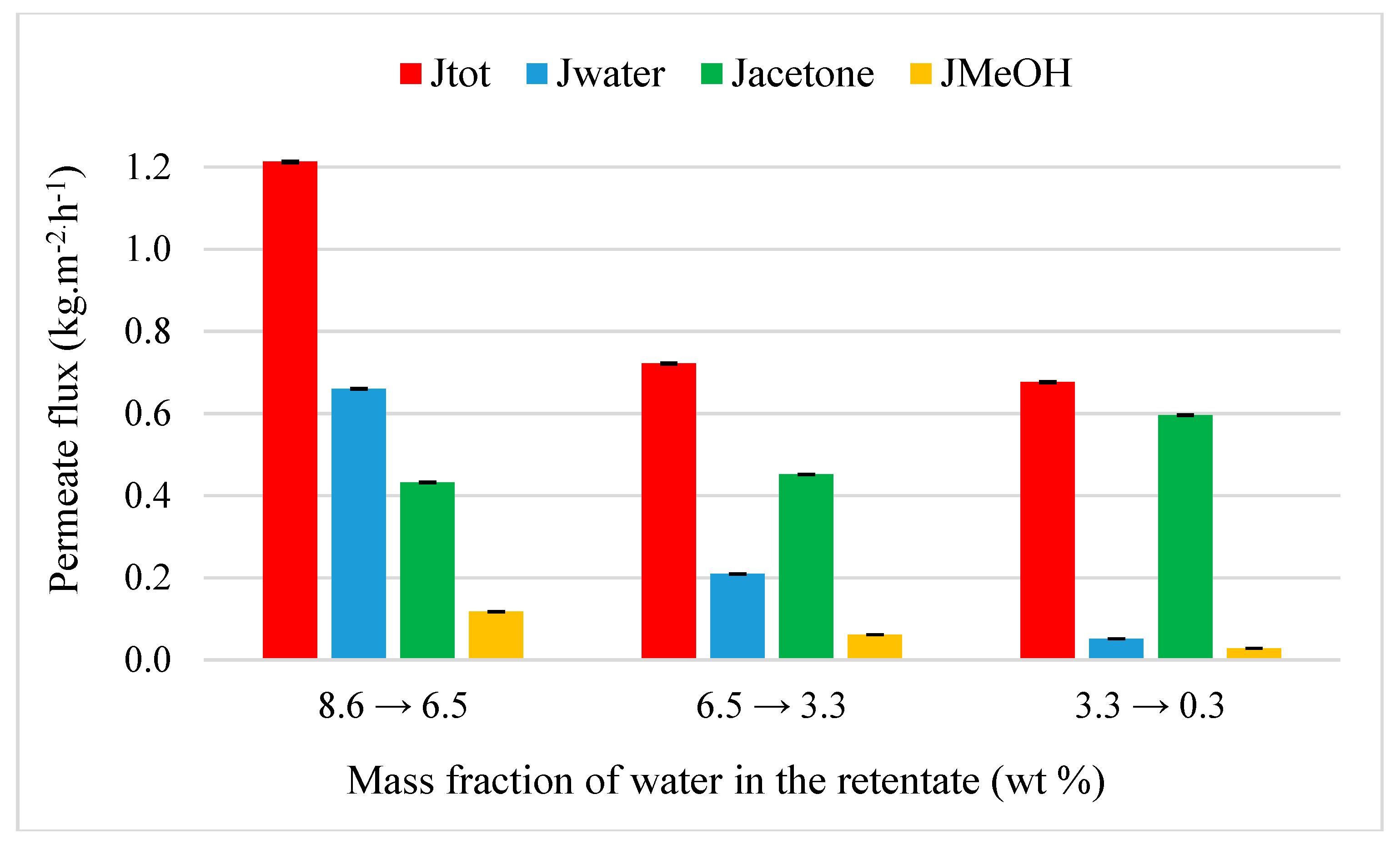
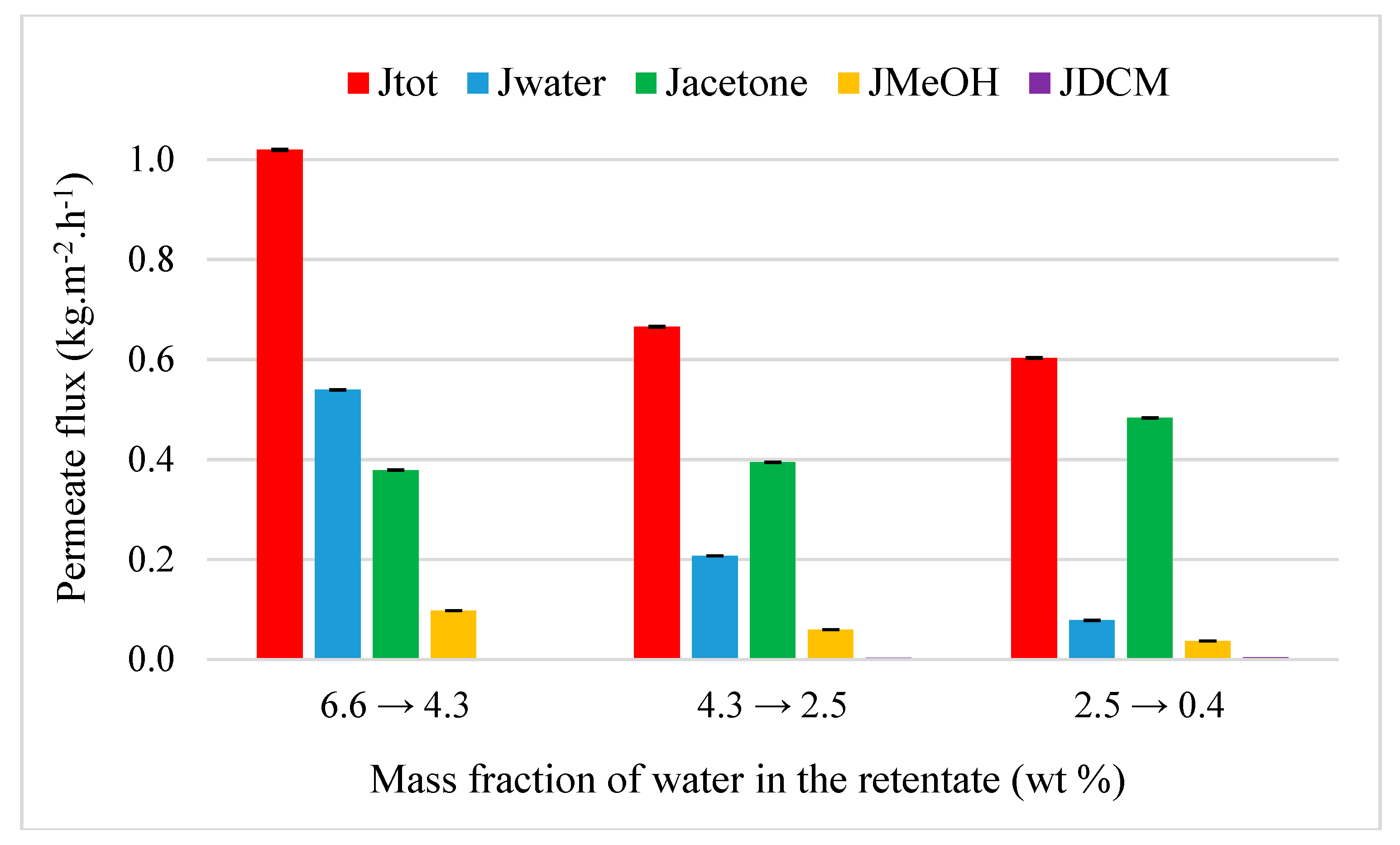
4.3.1. Acetone Purification from Ternary Mixtures
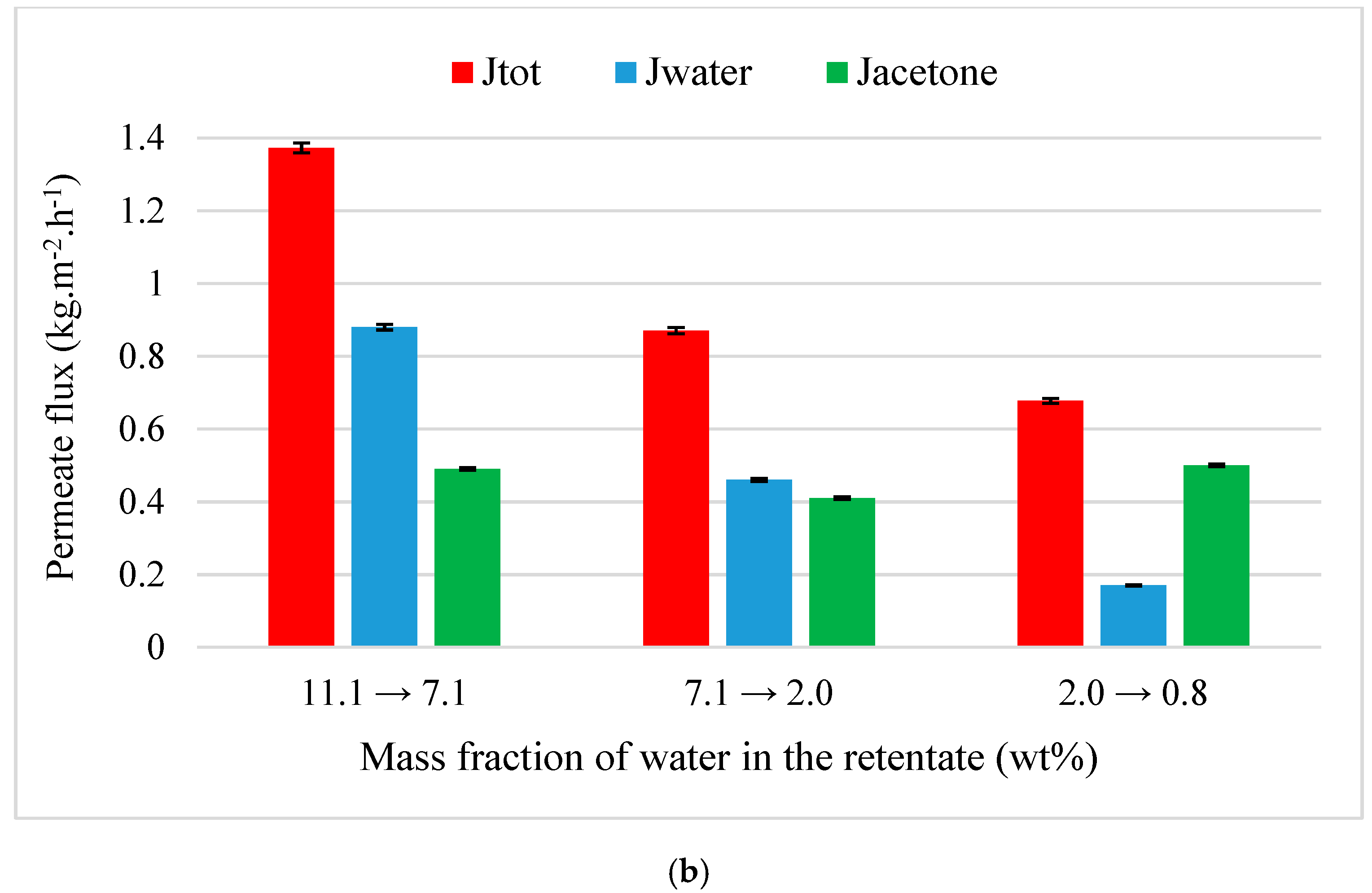
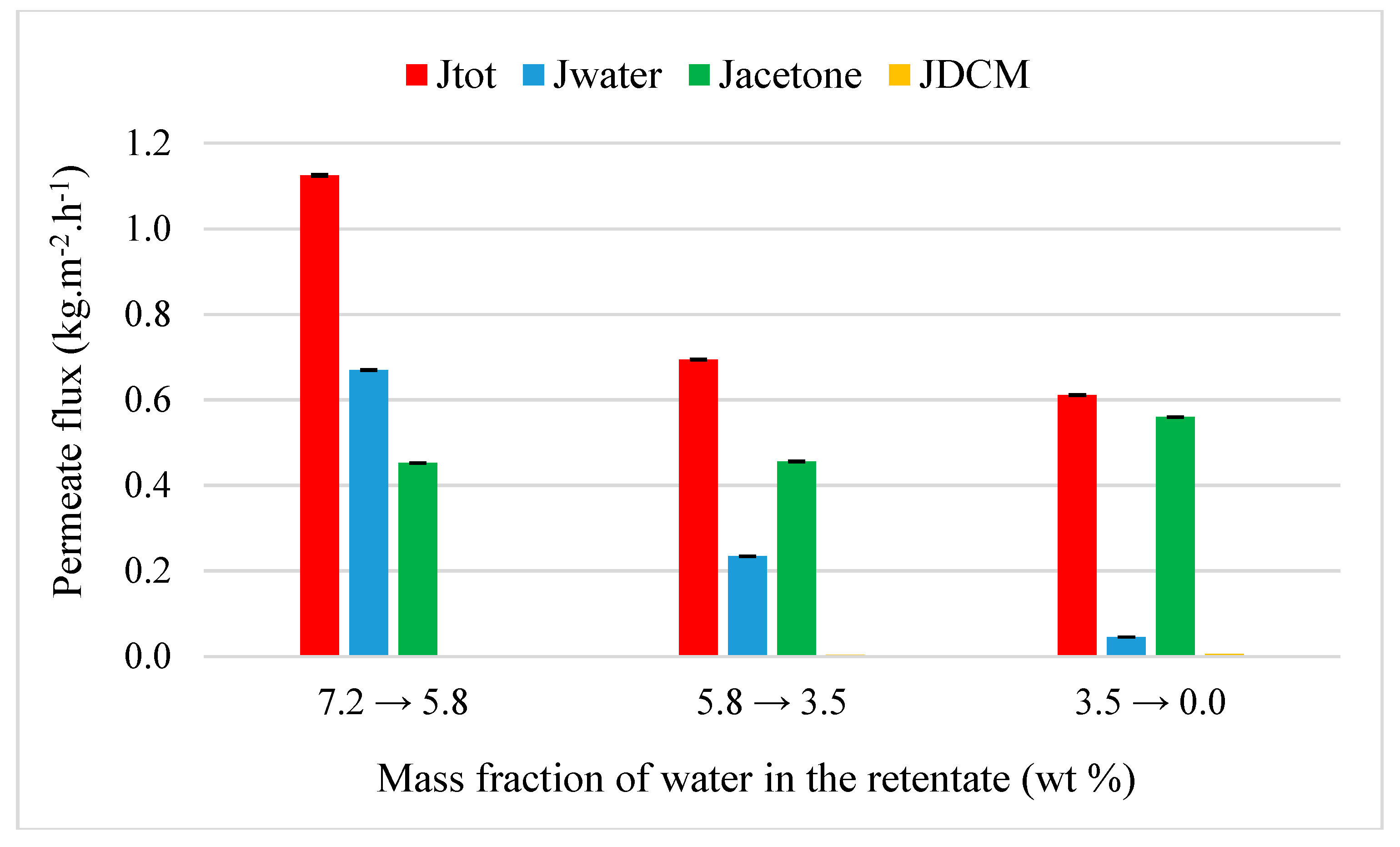
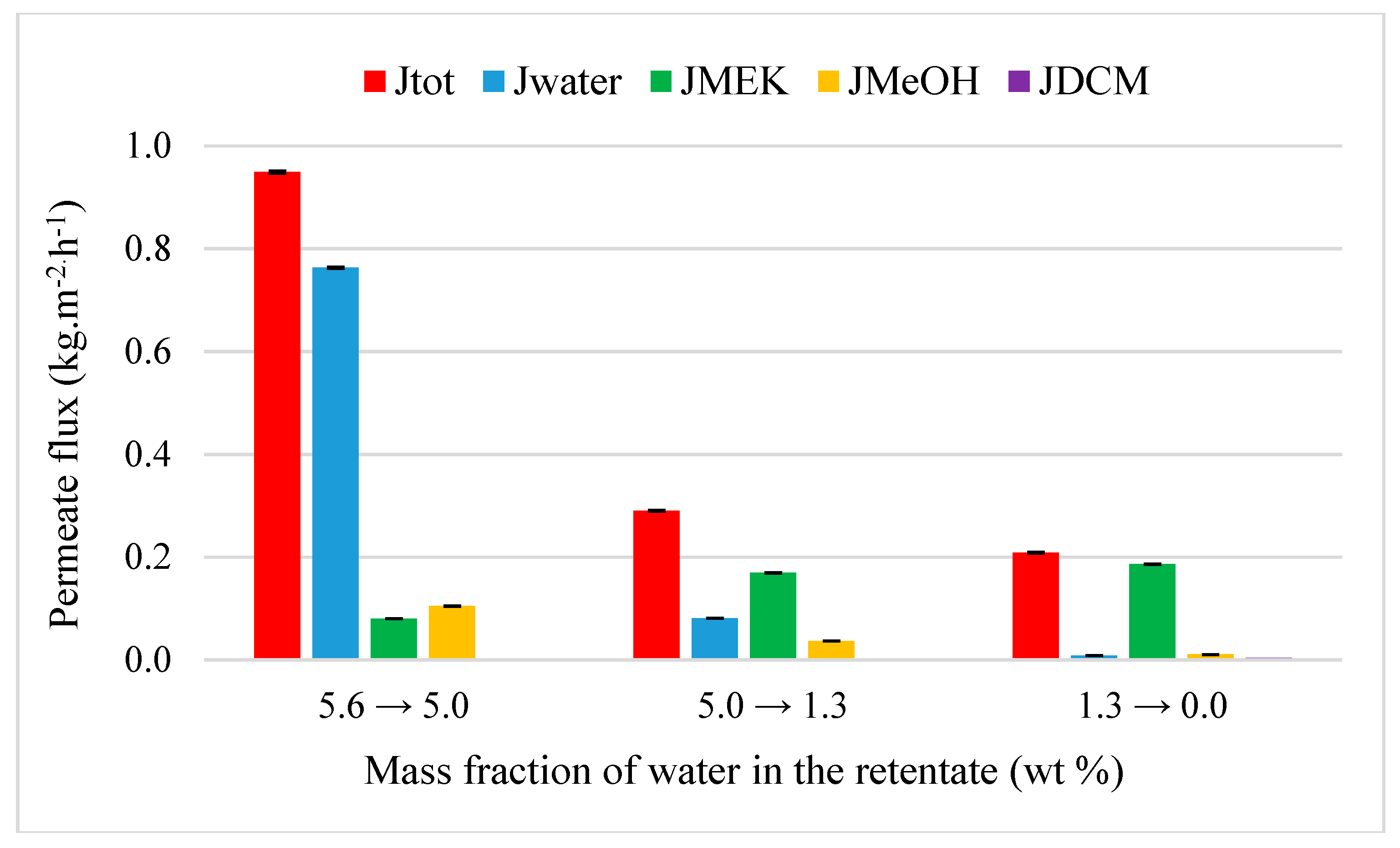
4.3.2. Acetone and MEK Purification from Quaternary Mixtures
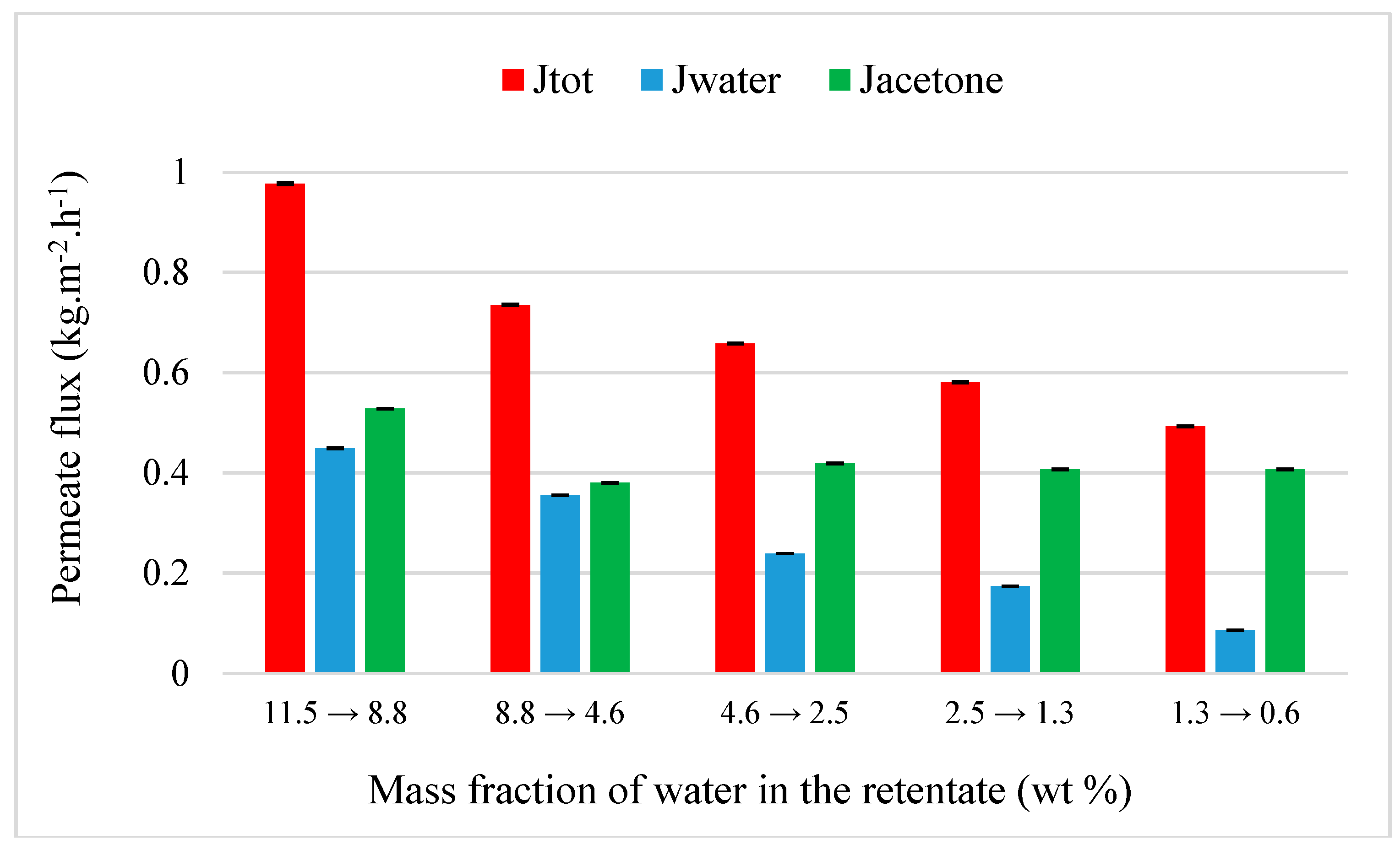
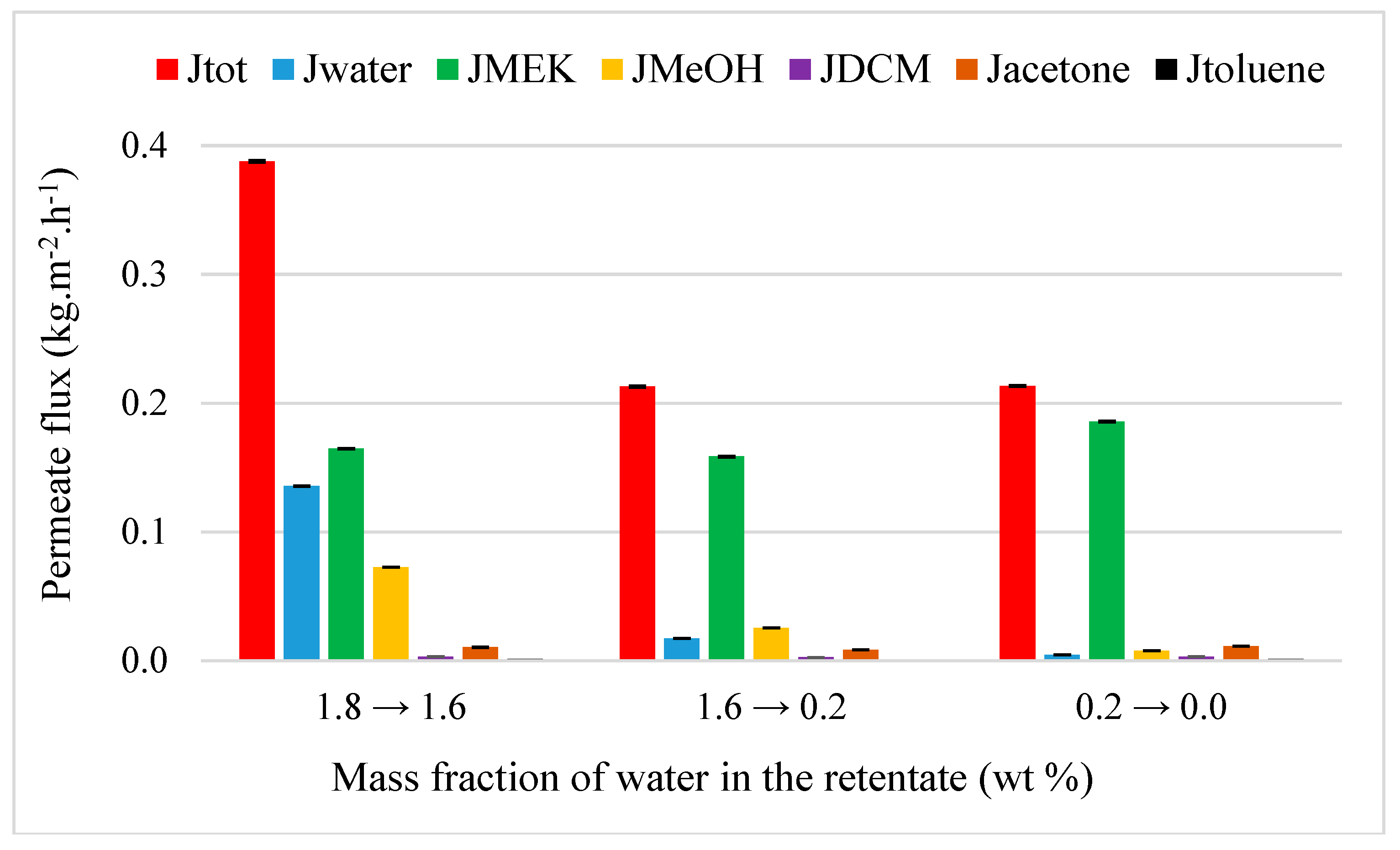
4.3.3. MEK Purification from MEK-Water-MeOH-DCM-Acetone-Toluene Mixture
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Cetin, E.; Odabasi, M.; Seyfioglu, R. Ambient volatile organic compound (VOC) concentrations around a petrochemical complex and a petroleum refinery. Sci. Total. Environ. 2003, 312, 103–112. [Google Scholar] [CrossRef]
- Contact Lenses Market Size, Share, Industry Report, 2020; Radiant Insights Inc.: San Francisco, CA, USA, 2015.
- Salar-García, M.; Ortiz-Martínez, V.; Hernández-Fernández, F.; Ríos, A.D.L.; Quesada-Medina, J. Ionic liquid technology to recover volatile organic compounds (VOCs). J. Hazard. Mater. 2017, 321, 484–499. [Google Scholar] [CrossRef] [PubMed]
- Geldermann, J.; Treitz, M.; Schollenberger, H.; Rentz, O. Evaluation of VOC recovery strategies. OR Spectrum. 2006, 28, 3–20. [Google Scholar] [CrossRef]
- Kraemer, K.; Harwardt, A.; Bronneberg, R.; Marquardt, W. Separation of butanol from acetone–butanol–ethanol fermentation by a hybrid extraction–distillation process. Comput. Chem. Eng. 2011, 35, 949–963. [Google Scholar] [CrossRef]
- Harvianto, G.R.; Ahmad, F.; Nhien, L.C.; Lee, M. Vapor permeation–distillation hybrid processes for cost-effective isopropanol dehydration: Modeling, simulation and optimization. J. Membr. Sci. 2016, 497, 108–119. [Google Scholar] [CrossRef]
- Crespo, J.; Brazinha, C. 1—Fundamentals of pervaporation. In Pervaporation, Vapour Permeation and Membrane Distillation: Principles and Applications; Woodhead Publishing: Oxford, UK, 2015; pp. 3–15. [Google Scholar]
- Roizard, D.; Favre, E. Trends in design and preparation of polymeric membranes for pervaporation. In Advanced Materials for Membrane Preparation; Bentham Science Publishers: Bussum, The Netherlands, 2012; pp. 163–204. [Google Scholar]
- Klinov, A.V.; Akberov, R.R.; Fazlyev, A.R.; Farakhov, M.I. Experimental investigation and modeling through using the solution-diffusion concept of pervaporation dehydration of ethanol and isopropanol by ceramic membranes HybSi. J. Membr. Sci. 2017, 524, 321–333. [Google Scholar] [CrossRef]
- Sun, H.; Lu, L.; Peng, F.; Wu, H.; Jiang, Z. Pervaporation of benzene/cyclohexane mixtures through CMS-filled poly (vinyl alcohol) membranes. Sep. Purif. Technol. 2006, 52, 203–208. [Google Scholar] [CrossRef]
- Bettens, B.; Verhoef, A.; van Veen, H.M.; Vandecasteele, C.; Degrève, J.; van der Bruggen, B. Pervaporation of binary water–alcohol and methanol–alcohol mixtures through microporous methylated silica membranes: Maxwell–Stefan modeling. Comput. Chem. Eng. 2010, 34, 1775–1788. [Google Scholar] [CrossRef]
- Sampranpiboon, P.; Jiraratananon, R.; Uttapap, D.; Feng, X.; Huang, R. Pervaporation separation of ethyl butyrate and isopropanol with polyether block amide (PEBA) membranes. J. Membr. Sci. 2000, 173, 53–59. [Google Scholar] [CrossRef]
- Babalou, A.; Rafia, N.; Ghasemzadeh, K. 3—Integrated systems involving pervaporation and applications. In Pervaporation, Vapour Permeation and Membrane Distillation: Principles and Applications; Woodhead Publishing: Oxford, UK, 2015; pp. 65–86. [Google Scholar]
- Peng, M.; Vane, L.M.; Liu, S.X. Recent advances in VOCs removal from water by pervaporation. J. Hazard. Mater. 2003, 98, 69–90. [Google Scholar] [CrossRef]
- Figoli, A.; Santoro, S.; Galiano, F.; Basile, A. 2—Pervaporation membranes: Preparation, characterization, and application. In Pervaporation, Vapour Permeation and Membrane Distillation: Principles and Applications; Woodhead Publishing: Oxford, UK, 2015; pp. 19–63. [Google Scholar]
- Garcia, V.; Pongrácz, E.; Muurinen, E.; Keiski, R.L. Pervaporation of dichloromethane from multicomponent aqueous systems containing n-butanol and sodium chloride. J. Membr. Sci. 2009, 326, 92–102. [Google Scholar] [CrossRef]
- Ramaiah, K.P.; Satyasri, D.; Sridhar, S.; Krishnaiah, A. Removal of hazardous chlorinated VOCs from aqueous solutions using novel ZSM-5 loaded PDMS/PVDF composite membrane consisting of three hydrophobic layers. J. Hazard. Mater. 2013, 261, 362–371. [Google Scholar] [CrossRef]
- Konieczny, K.; Bodzek, M.; Panek, D. Removal of volatile compounds from the wastewaters by use of pervaporation. Desalination 2008, 223, 344–348. [Google Scholar] [CrossRef]
- Peng, F.; Jiang, Z.; Hu, C.; Wang, Y.; Xu, H.; Liu, J. Removing benzene from aqueous solution using CMS-filled PDMS pervaporation membranes. Sep. Purif. Technol. 2006, 48, 229–234. [Google Scholar] [CrossRef]
- Smitha, B.; Suhanya, D.; Sridhar, S.; Ramakrishna, M. Separation or organic-organic mixtures by pervaporation—A review. J. Membr. Sci. 2004, 241, 1–21. [Google Scholar] [CrossRef]
- Zhang, W.; Li, G.; Fang, Y.; Wang, X. Maleic anhydride surface-modification of crosslinked chitosan membrane and its pervaporation performance. J. Membr. Sci. 2007, 295, 130–138. [Google Scholar] [CrossRef]
- Ray, S. Effect of copolymer type and composition on separation characteristics of pervaporation membranes—A case study with separation of acetone–water mixtures. J. Membr. Sci. 2006, 270, 73–87. [Google Scholar] [CrossRef]
- Tago, T.; Nakasaka, Y.; Kayoda, A.; Masuda, T. Preparation of hydrophilic silicalite-1 nanocrystal-layered membranes and their application to separating water from water–acetone solution. Microporous Mesoporous Mater. 2008, 115, 176–183. [Google Scholar] [CrossRef][Green Version]
- Wen, H.; Gao, H.; Zhang, T.; Wu, Z.; Gong, P.; Li, Z.; Chen, H.; Cai, D.; Qin, P.; Tan, T. Hybrid pervaporation and salting-out for effective acetone-butanol-ethanol separation from fermentation broth. Bioresour. Technol. Rep. 2018, 2, 45–52. [Google Scholar] [CrossRef]
- Cai, D.; Hu, S.; Miao, Q.; Chen, C.; Chen, H.; Zhang, C.; Li, P.; Qin, P.; Tan, T. Two-stage pervaporation process for effective in situ removal acetone-butanol-ethanol from fermentation broth. Bioresour. Technol. 2017, 224, 380–388. [Google Scholar] [CrossRef]
- Kumar, P.A.; Anilkumar, S.; Varughese, K.; Thomas, S. Separation of n-hexane/acetone mixtures by pervaporation using high density polyethylene/ethylene propylene diene terpolymer rubber blend membranes. J. Hazard. Mater. 2012, 199, 336–342. [Google Scholar] [CrossRef]
- Chen, X.; Lin, X.; Chen, P.; Kita, H. Pervaporation of ketone/water mixtures through silicalite membrane. Desalination 2008, 234, 286–292. [Google Scholar] [CrossRef]
- Smetana, J.F.; Falconer, J.L.; Noble, R.D. Separation of methyl ethyl ketone from water by pervaporation using a silicalite membrane. J. Membr. Sci. 1996, 114, 127–130. [Google Scholar] [CrossRef]
- Akberov, R.R.; Fazlyev, A.R.; Klinov, A.V.; Malygin, A.V.; Farakhov, M.I.; Maryakhina, V.A. Pervaporation technology for regeneration of diethylene glycol at Russian complex gas treatment plants with the use of ceramic membranes HybSi. J. Nat. Gas Sci. Eng. 2015, 26, 670–682. [Google Scholar] [CrossRef]
- Mah, S.-K.; Chai, S.-P.; Wu, T.Y. Dehydration of glycerin solution using pervaporation: HybSi and polydimethylsiloxane membranes. J. Membr. Sci. 2014, 450, 440–446. [Google Scholar] [CrossRef]
- Wijmans, J.G.; Baker, R.W. The solution-diffusion model: A review. J. Membr. Sci. 1994, 107, 1–21. [Google Scholar] [CrossRef]
- Baker, R.W.; Wijmans, J.; Huang, Y. Permeability, permeance and selectivity: A preferred way of reporting pervaporation performance data. J. Membr. Sci. 2010, 348, 346–352. [Google Scholar] [CrossRef]
- Renon, H.; Prausnitz, J.M. Liquid-Liquid and Vapor-Liquid Equilibria for Binary and Ternary Systems with Dibutyl Ketone, Dimethyl Sulfoxide, n -Hexane, and 1-Hexene. Ind. Eng. Chem. Process. Des. Dev. 1968, 7, 220–225. [Google Scholar] [CrossRef]
- Aroujalian, A.; Raisi, A. Recovery of volatile aroma components from orange juice by pervaporation. J. Membr. Sci. 2007, 303, 154–161. [Google Scholar] [CrossRef]
- Yi, S.; Wan, Y. Volatile organic compounds (VOCs) recovery from aqueous solutions via pervaporation with vinyltriethoxysilane-grafted-silicalite-1/polydimethylsiloxane mixed matrix membrane. Chem. Eng. J. 2017, 313, 1639–1646. [Google Scholar] [CrossRef]
- Luis, P.; van der Bruggen, B. Separation and Purification Technology, the driving force as key element to evaluate the pervaporation performance of multicomponent mixtures. Sep. Purif. Technol. 2015, 148, 94–102. [Google Scholar] [CrossRef]
- Lipnizki, F.; Hausmanns, S. Hydrophobic Pervaporation of Binary and Ternary Solutions: Evaluation of Fluxes, Selectivities, and Coupling Effects. Sep. Sci. Technol. 2005, 39, 2235–2259. [Google Scholar] [CrossRef]
- Van Veen, H.M.; Rietkerk, M.D.; Shanahan, D.P.; Van Tuel, M.M.; Kreiter, R.; Castricum, H.L.; Elshof, J.E.T.; Vente, J.F. Pushing membrane stability boundaries with HybSi® pervaporation membranes. J. Membr. Sci. 2011, 380, 124–131. [Google Scholar] [CrossRef]
- Paradis, G.G.; Shanahan, D.P.; Kreiter, R.; Van Veen, H.M.; Castricum, H.L.; Nijmeijer, A.; Vente, J.F. From hydrophilic to hydrophobic HybSi® membranes: A change of affinity and applicability. J. Membr. Sci. 2013, 428, 157–162. [Google Scholar] [CrossRef]
- Jin, H.; Mo, K.; Wen, F.; Li, Y. Preparation and pervaporation performance of CAU-10-H MOF membranes. J. Membr. Sci. 2019, 577, 129–136. [Google Scholar] [CrossRef]
- Liu, K.; Fang, C.-J.; Li, Z.-Q.; Young, M. Separation of thiophene/n-heptane mixtures using PEBAX/PVDF-composited membranes via pervaporation. J. Membr. Sci. 2014, 451, 24–31. [Google Scholar] [CrossRef]
- Tochigi, K.; Rarey, J.; Gmehling, J. Recommended NRTL Model Parameters by Simultaneous Correlation of VLE, Infinite Dilution Activity Coefficients and Excess Enthalpy Data. J. Chem. Eng. Jpn. 2009, 42, 376–380. [Google Scholar] [CrossRef]
- Yeang, Q.W.; Zein, S.H.S.; Sulong, A.B.; Tan, S.H. Comparison of the pervaporation performance of various types of carbon nanotube-based nanocomposites in the dehydration of acetone. Sep. Purif. Technol. 2013, 107, 252–263. [Google Scholar] [CrossRef]
- Koch, K.; Górak, A. Pervaporation of binary and ternary mixtures of acetone, isopropyl alcohol and water using polymeric membranes: Experimental characterisation and modelling. Chem. Eng. Sci. 2014, 115, 95–114. [Google Scholar] [CrossRef]
- La Rocca, T.; Carretier, E.; Clair, T.; Etienne, M.; Moulin, P. On-Line NIR to Regulate Pervaporation Process: Application for Dehydration. Membranes 2018, 8, 74. [Google Scholar] [CrossRef]


| Compound | Log Kow (-) | Saturated Vapor Pressure at 30 °C (kPa) * | Kinetic Diameter (Å) |
|---|---|---|---|
| Water | - | 4.2 | 2.96 |
| Acetone | −0.24 | 37.5 | 4.69 |
| DCM | 1.25 | 70.5 | 4.60 |
| Ethanol | −0.31 | 10.4 | 4.30 |
| MEK | 0.29 | 15.0 | 5.04 |
| Methanol (MeoH) | −0.77 | 21.9 | 3.80 |
| Toluene | 2.73 | 4.9 | 5.68 |
| Mixture | Temperature (°C) | Membrane | Time (min) |
|---|---|---|---|
| Acetone/Water | 30 | M1 | 295 |
| Acetone/Water | 45 | M1 | 90 |
| Acetone/Water | 30 | M2 | 150 |
| Acetone/Water/MeOH | 50 | M1 | 120 |
| Acetone/Water/DCM | 50 | M1 | 145 |
| Acetone/Water/MeOH/DCM | 50 | M1 | 120 |
| MEK/Water/MeOH/DCM | 50 | M1 | 180 |
| MEK/Water/MeOH/DCM/Acetone/Toluene | 50 | M1 | 210 |
| Compound | Membrane | Temperature | Flux | Permeance (gpu) |
|---|---|---|---|---|
| (°C) | (kg·h−1·m−2) | |||
| Water | M1 | 30 | 1.01 | 11,950 |
| M1 | 45 | 2.63 | 13,019 | |
| M2 | 30 | 1.39 | 16,866 | |
| Ethanol | M1 | 30 | 0.71 | 1262 |
| M2 | 30 | 0.47 | 836 | |
| Acetone | M1 | 30 | 0.76 | 291 |
| M2 | 30 | 0.49 | 188 | |
| MEK | M1 | 30 | 0.27 | 212 |
| M2 | 30 | - | - |
| Membrane | Temperature (°C) | Water Permeance (GPU) | References |
|---|---|---|---|
| PVA-MWCNT/CS | 30 | 1910 | [43] |
| crosslinked PVA-MWCNT/CS | 30 | 1100 | [43] |
| crosslinked PVA-MWCNT/CS | 45 | 300 | [43] |
| PervapTM 1210 (PVA/PAN) | 50 | 3319 | [44] |
| PervapTM 1210 (PVA/PAN) | 60 | 4150 | [44] |
| BTESE hybrid silica | 30 | 5488 | This study |
| BTESE hybrid silica | 45 | 4220 | This study |
| Zr doped BTESM hybrid silica | 30 | 13,770 | This study |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
La Rocca, T.; Carretier, E.; Dhaler, D.; Louradour, E.; Truong, T.; Moulin, P. Purification of Pharmaceutical Solvents by Pervaporation through Hybrid Silica Membranes. Membranes 2019, 9, 76. https://doi.org/10.3390/membranes9070076
La Rocca T, Carretier E, Dhaler D, Louradour E, Truong T, Moulin P. Purification of Pharmaceutical Solvents by Pervaporation through Hybrid Silica Membranes. Membranes. 2019; 9(7):76. https://doi.org/10.3390/membranes9070076
Chicago/Turabian StyleLa Rocca, Thomas, Emilie Carretier, Didier Dhaler, Eric Louradour, Thien Truong, and Philippe Moulin. 2019. "Purification of Pharmaceutical Solvents by Pervaporation through Hybrid Silica Membranes" Membranes 9, no. 7: 76. https://doi.org/10.3390/membranes9070076
APA StyleLa Rocca, T., Carretier, E., Dhaler, D., Louradour, E., Truong, T., & Moulin, P. (2019). Purification of Pharmaceutical Solvents by Pervaporation through Hybrid Silica Membranes. Membranes, 9(7), 76. https://doi.org/10.3390/membranes9070076






