Preparation and Characterization of Facilitated Transport Membranes Composed of Chitosan-Styrene and Chitosan-Acrylonitrile Copolymers Modified by Methylimidazolium Based Ionic Liquids for CO2 Separation from CH4 and N2
Abstract
:1. Introduction
2. Results and Discussion
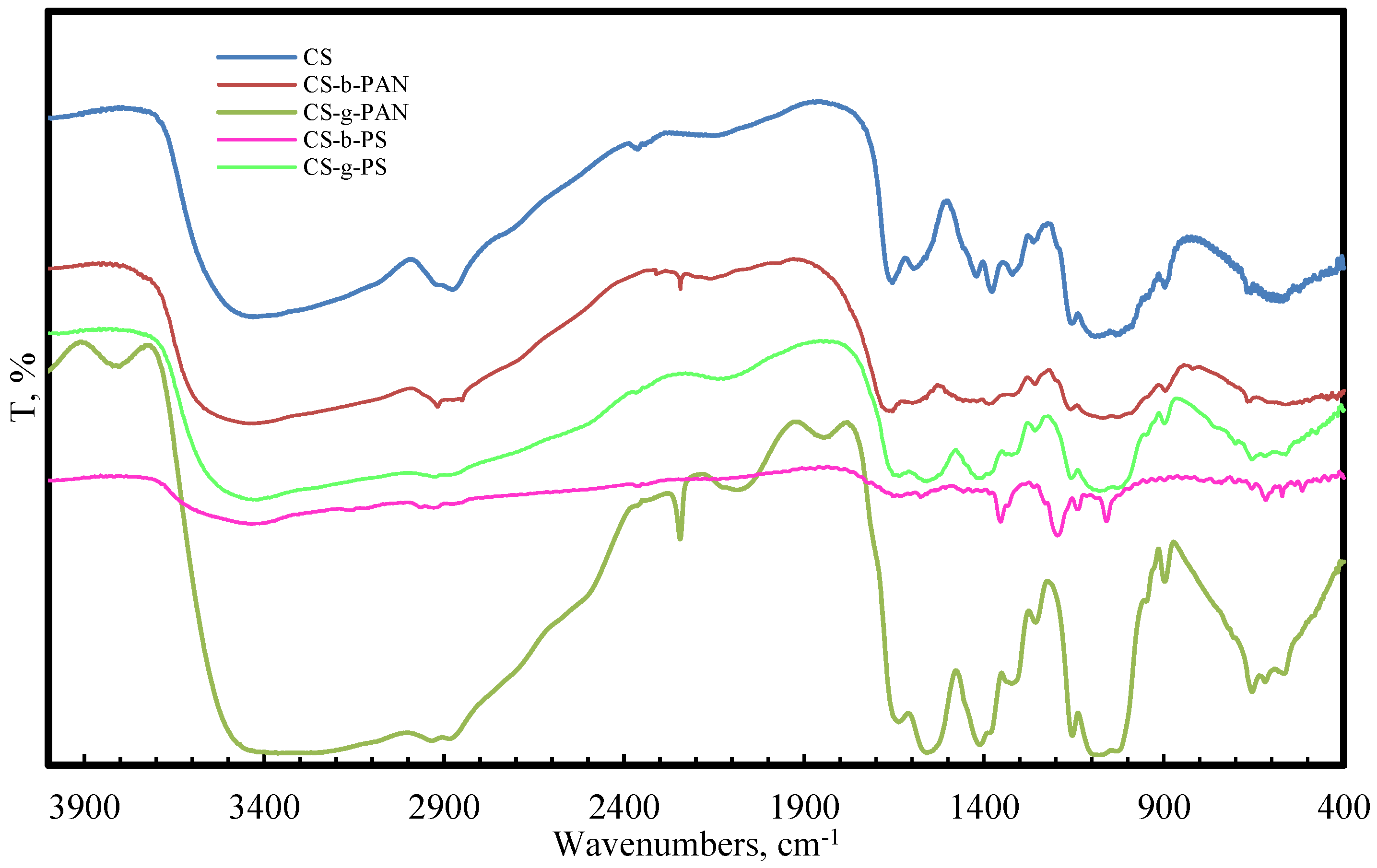
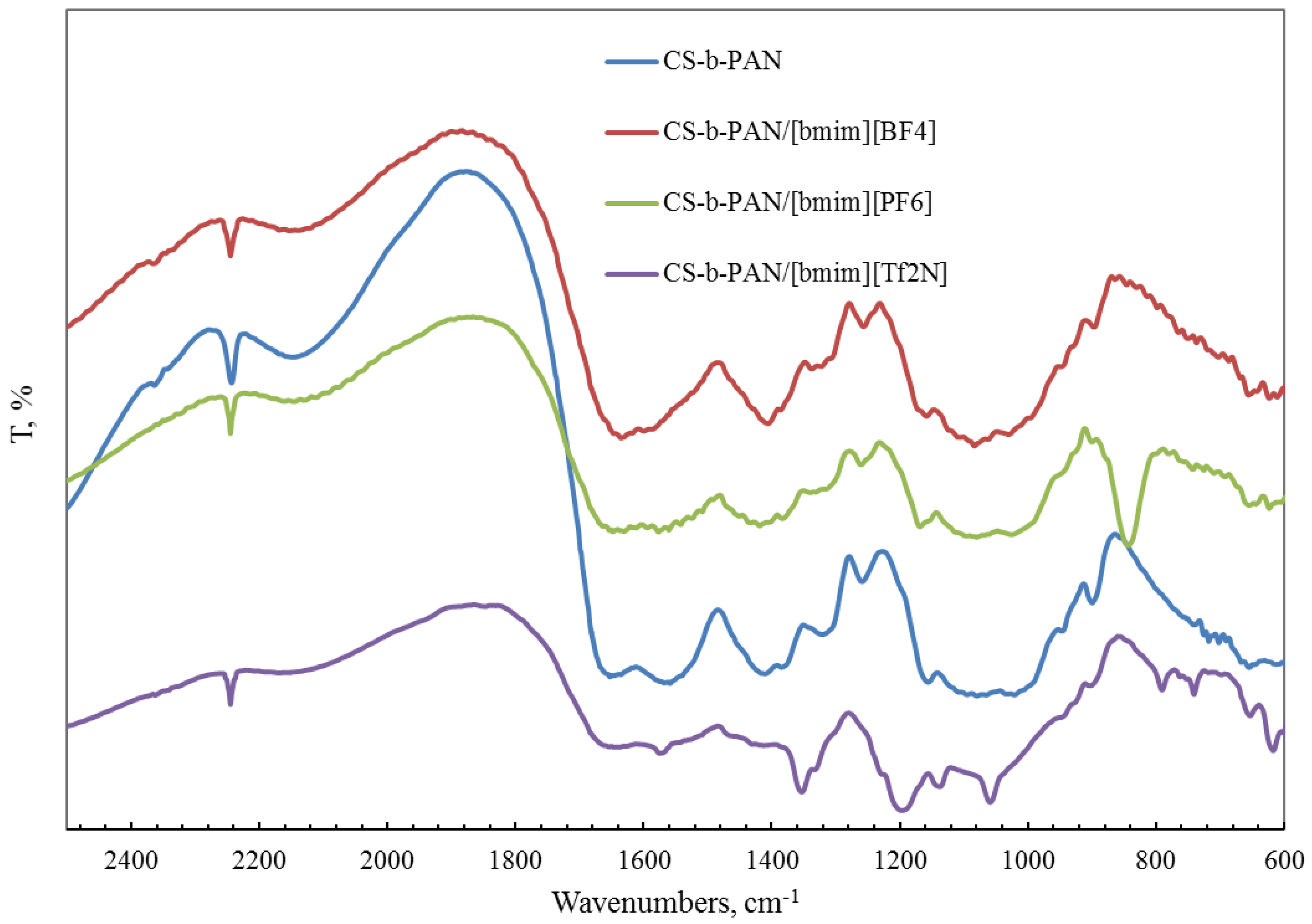
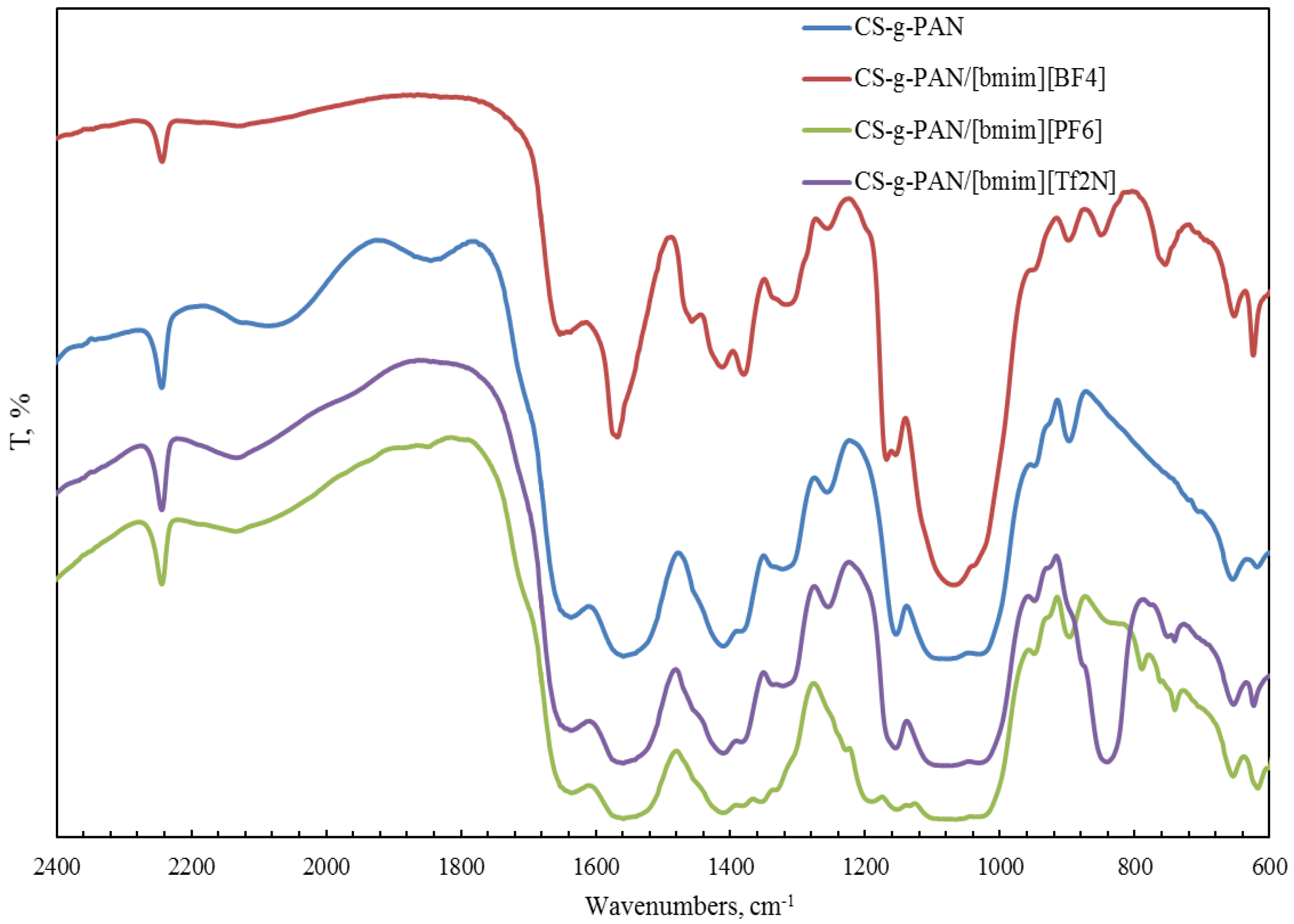
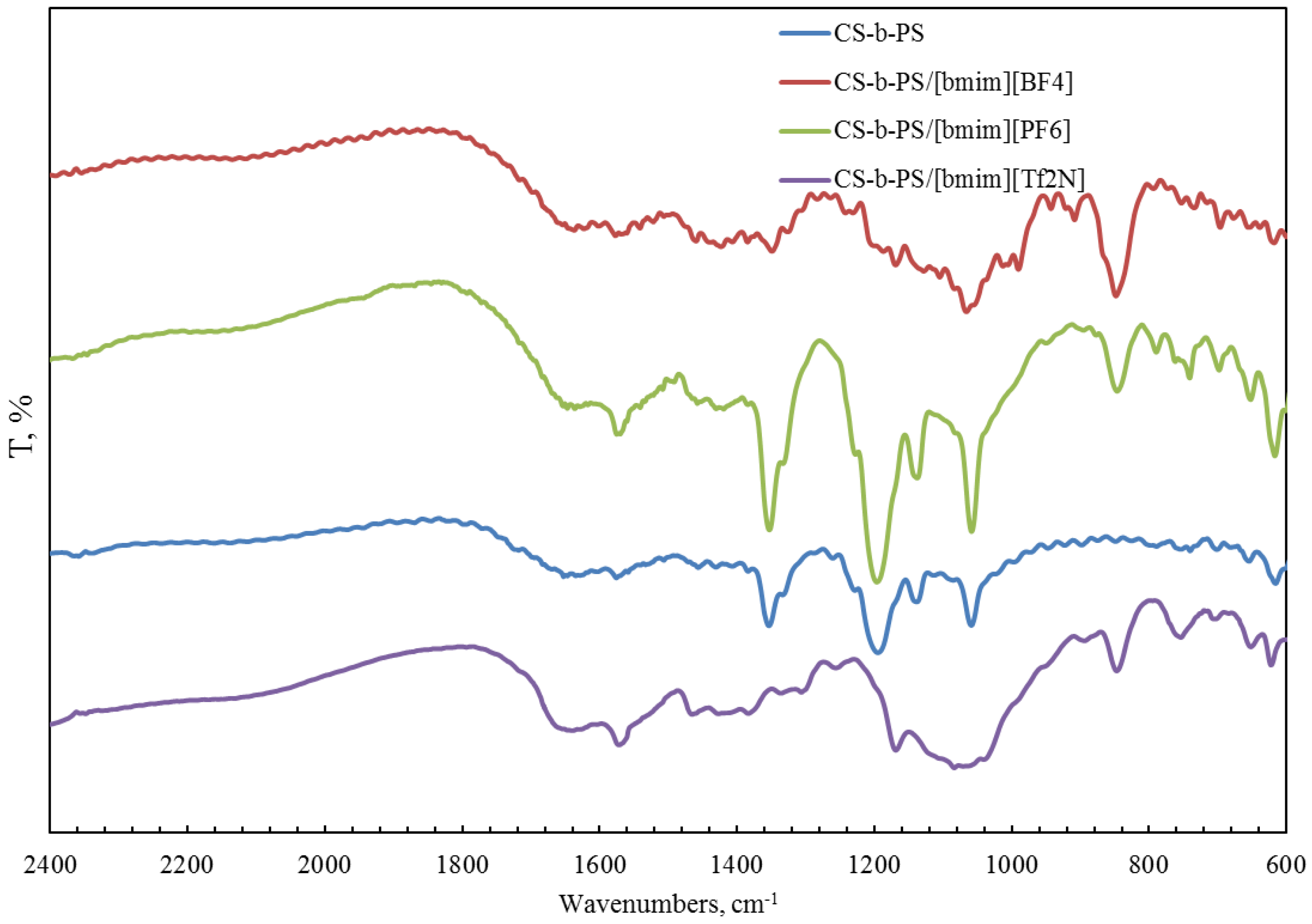
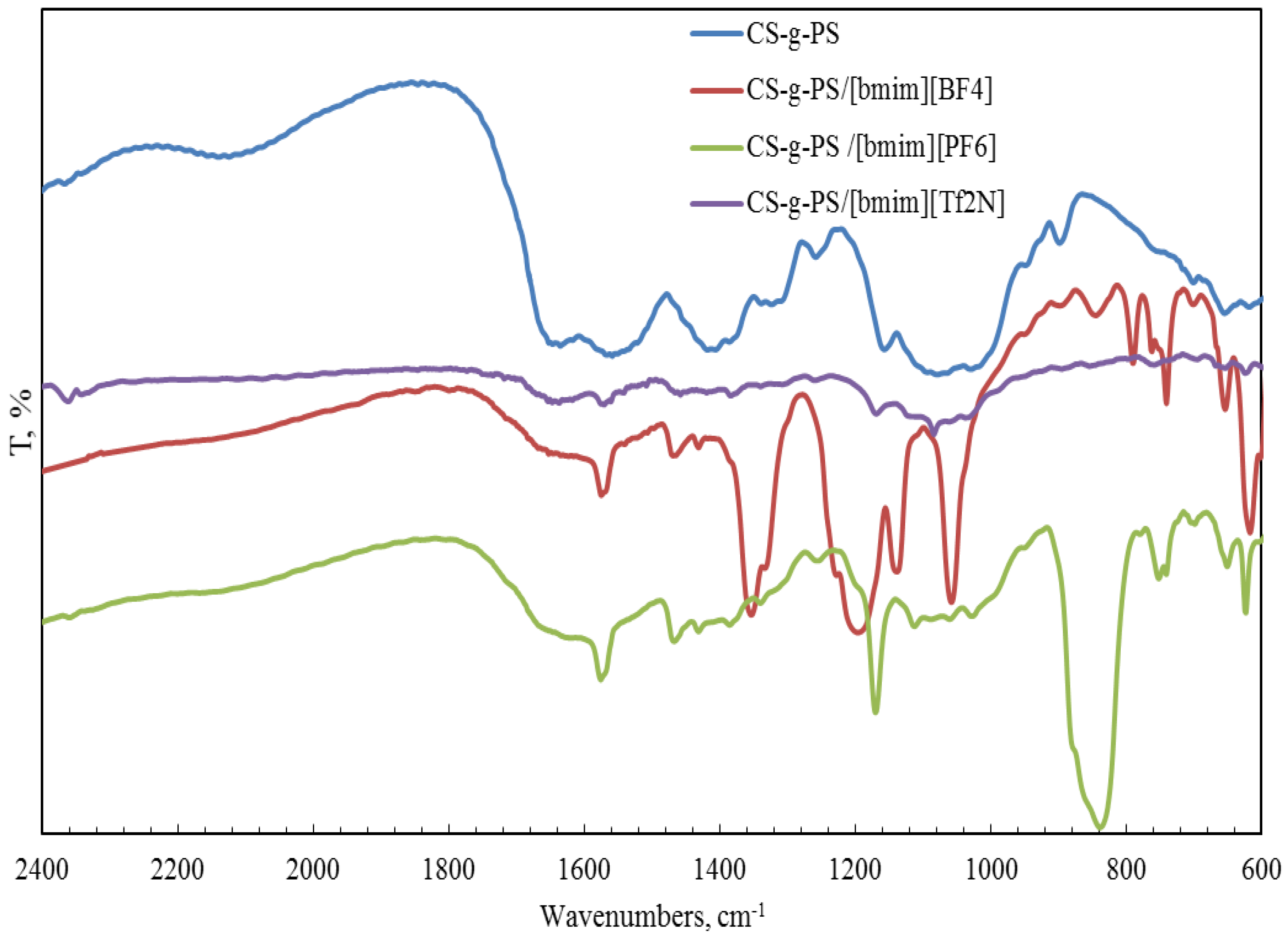
2.1. Structural Properties
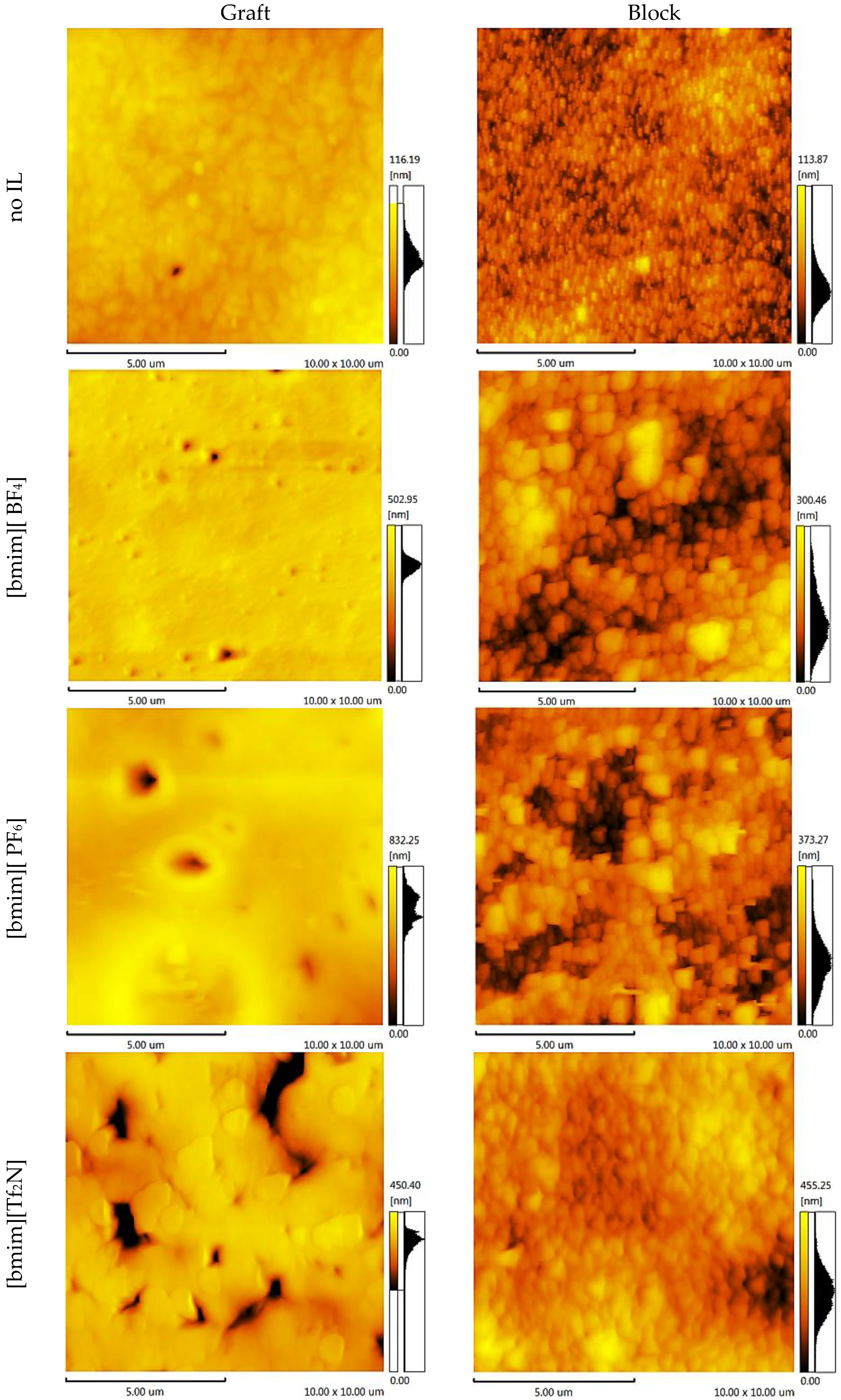
2.2. Surface Properties
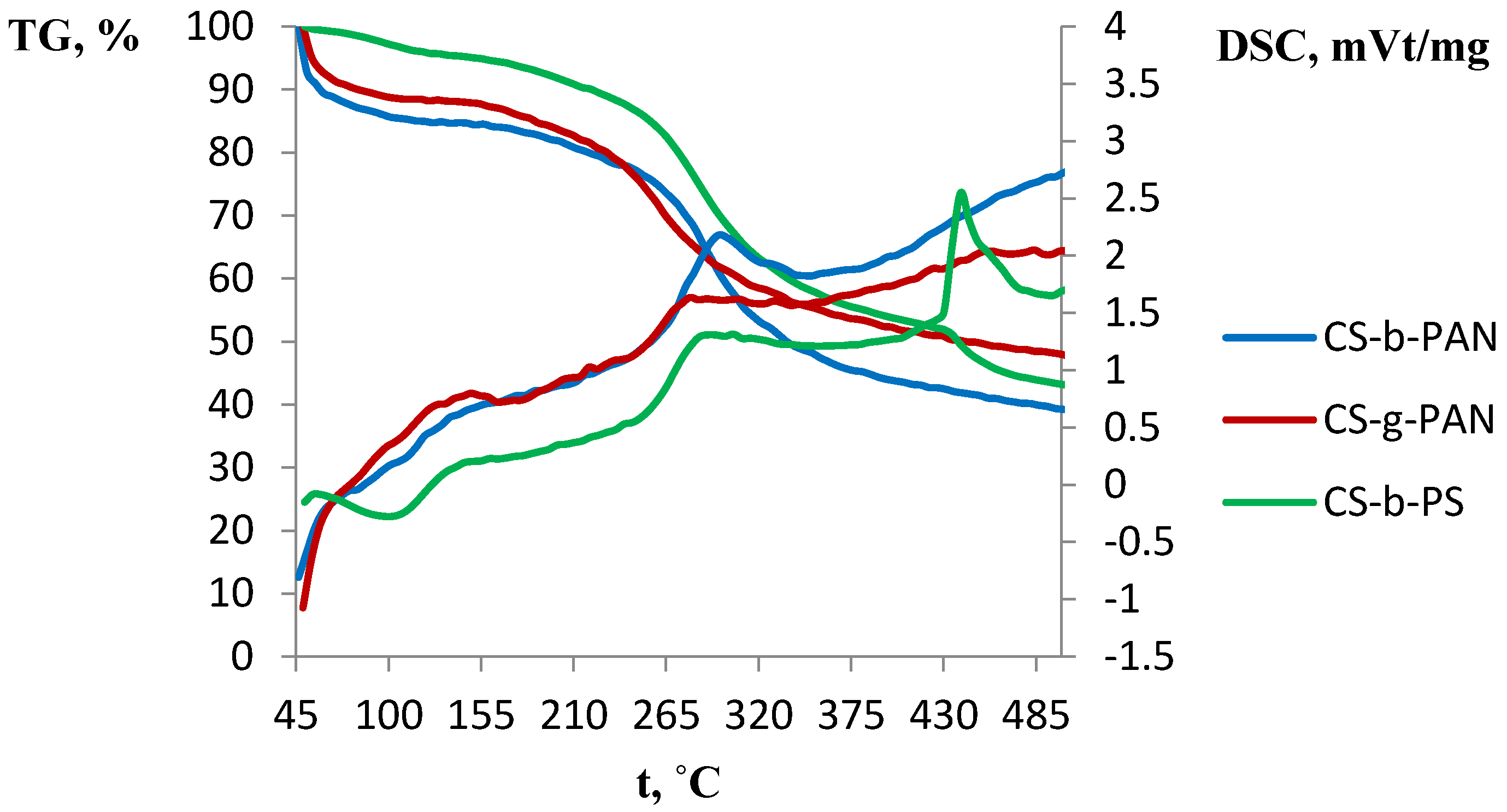
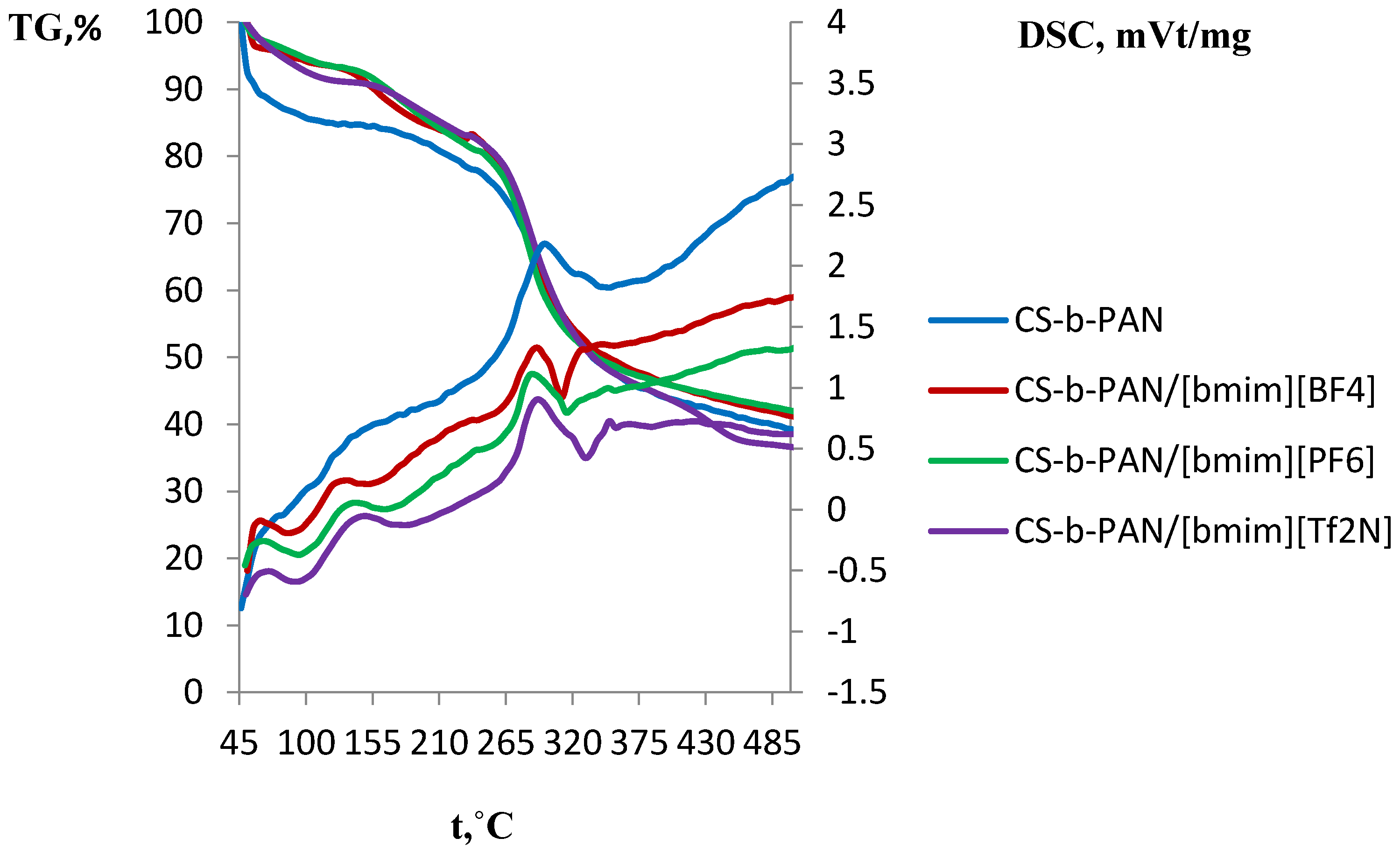
2.3. Thermal Properties
2.4. Mechanical Properties
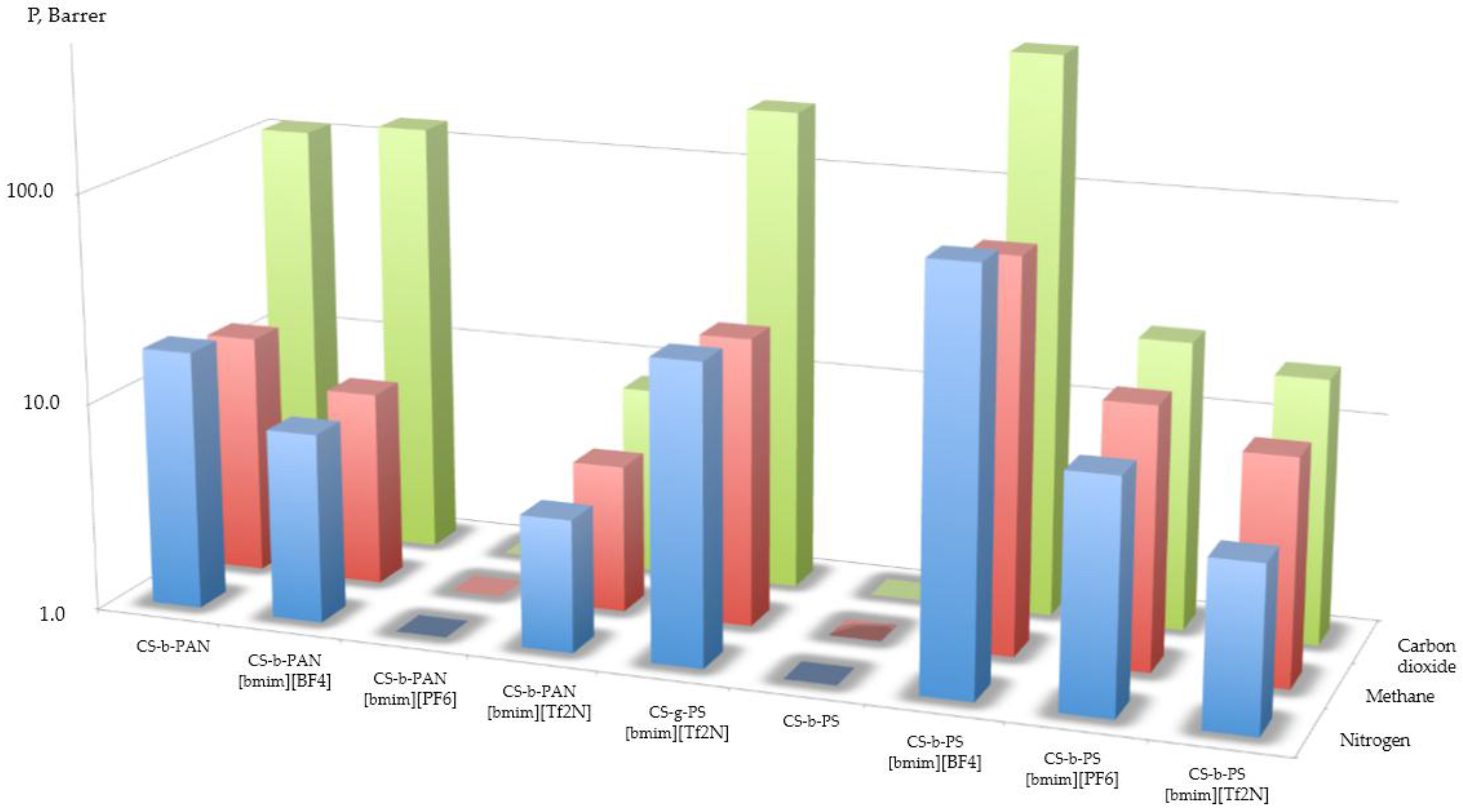
2.5. Gas Separation Properties
3. Materials and Methods
3.1. Materials
3.2. Copolymer Synthesis
3.3. Fourier-Transform Infrared Spectroscopy Analysis
3.4. IL Doping
3.5. Membrane Preparation
3.6. Atomic Force Microscopy Topology Analysis
3.7. Study of the Surface Chemical Nature
3.8. Tension Elongation Measurement
3.9. Thermogravimetric Analysis
3.10. Gas Permeation Tests
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| CS | chitosan |
| PS | poly(styrene) |
| PAN | poly(acrylonitrile) |
| AN | acrylonitrile |
| S | styrene |
| IL | ionic liquid |
| FTIR | Fourier-transform infrared spectroscopy |
| AFM | atomic force micrsocopy |
| TGA | thermal gravimetric analysis |
| DSC | differential scanning calorimetry |
| TMC | trimesoyl chloride |
| PAMMAM | poly(amidoamine) |
| [bmim][BF4] | 1-butyl-3-methylimidazolium tetrafluoroborate |
| [bmim][PF6] | 1-butyl-3-methylimidazolium hexafluorophosphate |
| [bmim][Tf2N] | 1-butyl-3-methylimidazolium bis(trifluoromethylsulfonyl)imide |
References
- Izvozchikova, V.; Pastukhova, N.; Ryabov, S.; Semchikov, Y.; Smirnova, L.; Mochalova, A. Flocculation Power of Chitosan and Its Derivatives in Mixtures with Anionic Flocculants. Russ. J. Appl. Chem. 2003, 76, 1784–1786. [Google Scholar] [CrossRef]
- Younes, I.; Rinaudo, M. Chitin and Chitosan Preparation from Marine Sources. Structure, Properties and Applications. Mar. Drugs 2015, 13, 1133–1174. [Google Scholar] [CrossRef] [PubMed]
- Hamed, I.; Özogul, F.; Regenstin, J.M. Industrial applications of crustacean by-products (chitin, 1 chitosan, and chitooligosaccharides): A review. Trends Food Sci. Tech. 2016, 48, 40–50. [Google Scholar] [CrossRef]
- Rinaudo, M. Chitin and chitosan: Properties and applications. Prog. Polym. Sci. 2006, 31, 603–632. [Google Scholar] [CrossRef]
- Dutta, P.K.; Dutta, J.; Tripathi, V.S. Chitin and chitosan: Chemistry, properties and applications. JSIR 2004, 63, 20–31. [Google Scholar]
- Xu, D.; Hein, S.; Wang, K. Chitosan membrane in separation applications. Mater. Sci. Technol. Ser. 2008, 24, 1076–1087. [Google Scholar] [CrossRef]
- Casado-Coterillo, C.; López-Guerrero, M.M.; Irabien, Á. Synthesis and characterization of ETS-10/acetate-based ionic liquid/chitosan mixed matrix membranes for CO2/N2 permeation. Membranes 2014, 4, 287–301. [Google Scholar] [CrossRef] [PubMed]
- Shaposhnik, V.A. Membrane methods for separation of substances. ISSEP 1999, 9, 27–32. [Google Scholar]
- Freitas, F.; Alves, V.D.; Reis, M.A.; Crespo, J.G.; Coelhoso, I.M. Microbial polysaccharide-based membranes: Current and future applications. J. Appl. Polym. Sci. 2013, 131. [Google Scholar] [CrossRef]
- Chakrabarty, T.; Kumar, M.; Vinod, K. Chitosan Based Membranes for Separation, Pervaporation and Fuel Cell Applications: Recent Developments. In Biopolymers; Elnashar, M., Ed.; INTECH Open Access Publisher: Rijeka, Croatia, 2010; pp. 201–226. [Google Scholar]
- El-Azzami, L.A.; Grulke, E.A. Carbon dioxide separation from hydrogen and nitrogen by fixed facilitated transport in swollen chitosan membranes. JMS 2008, 323, 225–234. [Google Scholar] [CrossRef]
- Ito, A.; Sato, M.; Anma, T. Permeability of CO2 through chitosan membrane swollen by water vapor in feed gas. Angew. Makromol. Chem. 1997, 248, 85–94. [Google Scholar] [CrossRef]
- Bae, S.Y.; Lee, K.H.; Yi, S.C.; Kim, H.T.; Kim, Y.H.; Kumazawa, H. CO2, N2 gas sorption and permeation behavior of chitosanmembrane. Korean J. Chem. Eng. 1998, 15, 223–226. [Google Scholar] [CrossRef]
- El-Azzami, L.A.; Grulke, E.A. Carbon dioxide separation from hydrogen and nitrogen. Facilitated transport in arginine salt–chitosan membranes. JMS 2009, 328, 15–22. [Google Scholar] [CrossRef]
- Liao, C.-T.; Ho, M.-H. The fabrication of biomimetic chitosan scaffolds by using SBF treatment with different crosslinking agents. Membranes 2011, 1, 3–12. [Google Scholar] [CrossRef] [PubMed]
- Xiao, S.; Feng, X.; Huang, R.Y.M. Trimesoyl chloride crosslinked chitosan membranes for CO2/N2 separation and pervaporation dehydration of isopropanol. JMS 2007, 306, 36–46. [Google Scholar] [CrossRef]
- Kouketsu, T.; Duan, S.; Kai, T.; Kazama, S.; Yamada, K. PAMAM dendrimer composite membrane for CO2 separation: Formation of a chitosan gutter layer. JMS 2007, 287, 51–59. [Google Scholar] [CrossRef]
- Shen, J.-N.; Yu, C.-C.; Zeng, G.-N.; van der Bruggen, B. Preparation of a facilitated transport membrane composed of carboxymethyl chitosan and polyethylenimine for CO2/N2 separation. Int. J. Mol. Sci. 2013, 14, 3621–3638. [Google Scholar] [CrossRef] [PubMed]
- Shen, Y.; Wang, H.; Liu, J.; Zhang, Y. Enhanced Performance of a Novel Polyvinyl Amine/Chitosan/Graphene Oxide Mixed Matrix Membrane for CO2 Capture. ACS Sustain. Chem. Eng. 2015, 3, 1819–1829. [Google Scholar] [CrossRef]
- Assis, O.B.G.; Hotchkiss, J.H. Surface hydrophobic modification of chitosan thin films by hexamethyldisilazane plasma deposition: Effects on water vapour, CO2 and O2 permeabilities. Packag. Technol. Sci. 2007, 20, 293–297. [Google Scholar] [CrossRef]
- Bai, R.-K.; Huang, M.-Y.; Jiang, Y.-Y. Selective permeabilities of chitosan-acetic acid complex membrane and chitosan-polymer complex membranes for oxygen and carbon dioxide. Polym. Bull. 1988, 20. [Google Scholar] [CrossRef]
- Mecerreyes, D. Applications of Ionic Liquids in Polymer Science and Technology; Springer: Berlin, Germany; Heidelberg, Germany, 2015; pp. 1–387. [Google Scholar]
- Babamohammadi, S.; Shamiri, A.; Aroua, M.K. A review of CO2 capture by absorption in ionic liquid-based solvents. Rev. Chem. Eng. 2015, 31, 383–412. [Google Scholar] [CrossRef]
- Soliman, S.M. Theoretical studies on interaction between CO2 gas and imidazolium-type organic ionic liquid using DFT and natural bond orbital calculations. Asian J. Chem. 2016, 28, 116–120. [Google Scholar] [CrossRef]
- Izgorodina, E.I.; Hodgson, J.L.; Weis, D.C.; Pas, S.J.; Macfarlane, D.R. Physical absorption of CO2 in protic and aprotic ionic liquids: A interaction perspective. J. Phys. Chem. B 2015, 119, 11748–11759. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Zhang, L.; Zheng, Y.; Zheng, C. Effect of SO2 on CO2 absorption in flue gas by ionic liquid 1-ethyl-3-methylimidazolium Acetate. Ind. Eng. Chem. Res. 2015, 54, 8569–8578. [Google Scholar] [CrossRef]
- Albo, J.; Luis, P.; Irabien, A. Carbon Dioxide Capture from Flue Gases Using a Cross-Flow Membrane Contactor and the Ionic Liquid 1-Ethyl-3-methylimidazolium Ethylsulfate. Ind. Eng. Chem. Res. 2010, 49, 11045–11051. [Google Scholar] [CrossRef]
- Albo, J.; Luis, P.; Irabien, A. Absorption of coal combustion flue gases in ionic liquids using different membrane contactors. Desalination Water Treat. 2011, 27, 54–59. [Google Scholar] [CrossRef]
- Albo, J.; Irabien, A. Non-dispersive absorption of CO2 in parallel and cross-flow membrane modules using EMISE. J. Chem. Technol. Biotechnol. 2012, 87, 1502–1507. [Google Scholar] [CrossRef]
- Couto, R.; Neves, L.; Simões, P.; Coelhoso, I. Supported ionic liquid membranes and Ion-Jelly® membranes with [BMIM][DCA]: Comparison of its performance for CO2 separation. Membranes 2015, 5, 13–21. [Google Scholar] [CrossRef] [PubMed]
- Akhmetshina, A.I.; Davletbaeva, I.M.; Grebenschikova, E.S.; Sazanova, T.S.; Petukhov, A.N.; Atlaskin, A.A.; Razov, E.N.; Zaripov, I.I.; Martins, C.F.; Neves, L.A.; et al. The effect of microporous polymeric support modification on surface and gas transport properties of supported ionic liquid membranes. Membranes 2016, 6. [Google Scholar] [CrossRef] [PubMed]
- Chapman, P.D.; Oliveira, T.; Livingston, A.G.; Li, K. Membranes for the dehydration of solvents by pervaporation. JMS 2008, 318, 5–37. [Google Scholar] [CrossRef]
- Shao, P.; Huang, R.Y.M. Polymeric membrane pervaporation. JMS 2007, 287, 162–179. [Google Scholar] [CrossRef]
- Bolto, B.; Hoang, M.; Xie, Z. A review of membrane selection for the dehydration of aqueous ethanol by pervaporation. Chem. Eng. Process. 2011, 50, 227–235. [Google Scholar] [CrossRef]
- Pratiwi, M.A.; Sudrajat, R.W.; Sutanti, S.; Susanto, H. Preparation of chitosan-alginate/PES pervaporation membranes for bioethanol dehydration. Adv. Mat. Res. 2015, 1123, 182–186. [Google Scholar] [CrossRef]
- Han, Y.-J.; Wang, K.-H.; Lai, J.-Y.; Liu, Y.-L. Hydrophilic chitosan-modified polybenzoimidazole membranes for pervaporation dehydration of isopropanol aqueous solutions. JMS 2014, 463, 17–23. [Google Scholar] [CrossRef]
- Zhang, Q.G.; Hu, W.W.; Zhu, A.M.; Liu, Q.L. UV-crosslinked chitosan/polyvinylpyrrolidone blended membranes for pervaporation. RSC Adv. 2013, 3, 1855–1861. [Google Scholar] [CrossRef]
- Shen, J.; Chu, Y.; Ruan, H.; Wu, L.; Gao, C.; Bruggen, B.V. Pervaporation of benzene/cyclohexane mixtures through mixed matrix membranes of chitosan and Ag+/carbon nanotubes. JMS 2014, 462, 160–169. [Google Scholar] [CrossRef]
- Casado-Coterillo, C.; Andrés, F.; Téllez, C.; Coronas, J.; Irabien, Á. Synthesis and characterization of ETS-10/chitosan nanocomposite membranes for pervaporation. Sep. Sci. Tech. 2014, 49, 1903–1909. [Google Scholar] [CrossRef]
- Sajjan, A.M.; Premakshi, H.G.; Kariduraganavar, M.Y. Synthesis and characterization of GTMAC grafted chitosan membranes for the dehydration of low water content isopropanol by pervaporation. J. Ind. Eng. Chem. 2015, 25, 151–161. [Google Scholar] [CrossRef]
- Varghese, J.G.; Kittur, A.A.; Rachipudi, P.S.; Kariduraganavar, M.Y. Synthesis, characterization and pervaporation performance of chitosan-g-polyaniline membranes for the dehydration of isopropanol. JMS 2010, 364, 111–121. [Google Scholar] [CrossRef]
- Prashanth, K.V.H.; Tharanathan, R.N. Studies on graft copolymerization of chitosan with synthetic monomers. Carb. Pol. 2003, 54, 343–351. [Google Scholar] [CrossRef]
- Jayakumar, R.; Prabaharan, M.; Reis, R.L.; Mano, J.F. Graft copolymerized chitosan—Present status and applications. Carb Pol. 2005, 62, 142–158. [Google Scholar] [CrossRef]
- Kalia, S.; Sabaa, M.W. Polysaccharide Based Graft Copolymers; Springer: Berlin, Germany; Heidelberg, Germany, 2013; pp. 1–353. [Google Scholar]
- Lv, P.; Bin, Y.; Li, Y.; Chen, R.; Wang, X.; Zhao, B. Studies on graft copolymerization of chitosan with acrylonitrile by the redox system. Polymer 2009, 50, 5675–5680. [Google Scholar] [CrossRef]
- Singh, V.; Tripathi, D.N.; Tiwari, A.; Sanghi, R. Microwave promoted synthesis of chitosan-graft-poly(acrylonitrile). J. App. Pol. Sci. 2005, 95, 820–825. [Google Scholar] [CrossRef]
- Thakur, V.K.; Thakur, M.K. Recent advances in graft copolymerization and applications of chitosan: A review. ACS Sustain. Chem. Eng. 2014, 2, 2637–2652. [Google Scholar] [CrossRef]
- Sadeghi, M.; Mohammadinasab, E.; Shafiei, F.; Mirdarikvande, S.; Sadeghi, H. Modification of chitosan radiation-induced graft copolymerization of acrylonitrile onto chitosan. J. Biol. Environ. Sci. 2014, 4, 227–232. [Google Scholar]
- Sharma, A.K.; Mishra, A.K. Microwave assisted synthesis of chitosan-graft-styrene for efficient Cr(VI) removal. Adv. Mat. Lett. 2010, 1, 59–66. [Google Scholar] [CrossRef]
- Liu, P.; Zhai, M.; Wu, J. Study on radiation-induced grafting of styrene onto chitin and chitosan. Rad. Phys. Chem. 2001, 61, 149–153. [Google Scholar] [CrossRef]
- Garcia-Valdez, O.; Ramirez-Wong, D.G.; Saldivar-Guerra, E.; Luna-Barcenas, G. Grafting of chitosan with styrene and maleic anhydride via nitroxide-mediated radical polymerization in supercritical carbon dioxide. Macromol. Chem. Phys. 2013, 214, 1396–1404. [Google Scholar] [CrossRef]
- Huang, J.; Rüther, T. Why are Ionic Liquids Attractive for CO2 Absorption? An Overview. Aust. J. Chem. 2009, 62, 298. [Google Scholar] [CrossRef]
- Xie, H.; Zhang, S.; Li, S. Chitin and chitosan dissolved in ionic liquids as reversible sorbents of CO2. Green Chem. 2006, 8, 630–633. [Google Scholar] [CrossRef]
- Albo, J.; Tsuru, T. Thin Ionic Liquid Membranes Based on Inorganic Supports with Different Pore Sizes. Ind. Eng. Chem. Res. 2014, 53, 8045–8056. [Google Scholar] [CrossRef]
- Santos, E.; Albo, J.; Irabien, A. Acetate based Supported Ionic Liquid Membranes (SILMs) for CO2 separation: Influence of the temperature. J. Membr. Sci. 2014, 452, 277–283. [Google Scholar] [CrossRef]
- Sun, J.; Wang, J.; Cheng, W.; Zhang, J.; Li, X.; Zhang, S.; Sheb, Y. Chitosan functionalized ionic liquid as a recyclable biopolymer-supported catalyst for cycloaddition of CO2. Green Chem. 2012, 14, 654–660. [Google Scholar] [CrossRef]
- Muzzarelli, R.A.A. Biomedical exploitation of chitin and chitosan via mechano-chemical disassembly, electrospinning, dissolutionin imidazoliumiIonic liquids, and supercritical drying. Mar. Drugs 2011, 9, 1510–1533. [Google Scholar] [CrossRef] [PubMed]
- Baranov, I.A.; Andriyanova, N.A.; Mochalova, A.E.; Sibirkin, A.A.; Baten’kin, M.A.; Smirnova, L.A. Grafting polymerization of acrylonitrile and methyl acrylate on chitosan in the presence of cobalt (III) complexes. Polym. Sci. Ser. B 2012, 54, 167–174. [Google Scholar] [CrossRef]
- Mochalova, A.E.; Kruglova, E.N.; Yunin, P.A.; Apryatina, K.V.; Smirnova, O.N.; Smirnova, L.A. Graft and block copolymers of chitosan with vinyl monomers: Synthesis, structure, and properties. Polym. Sci. Ser. B 2015, 57, 93–105. [Google Scholar] [CrossRef]
- Rumengan, I.; Suryanto, E.; Modaso, R.; Wullur, S.; Tallei, T.; Limbong, D. Structural Characteristics of Chitin y Chitosan Isolated from the Biomass of Cultivated Rotifer, Brachionus rotundiformis. Int. J. Fish. Aquat. Sci. 2014, 3, 12–18. [Google Scholar]
- Ramya, R.; Sudha, P.N.; Mahalakshmi, J. Preparation and characterization of chitosan binary blend. Int. J. Sci. Res. Publ. 2012, 2, 1–9. [Google Scholar]
- Beyler, C.L.; Hirschler, M.M. Thermal decomposition of polymers. In SFPE Handbook of Fire Protection Engineering; Beyler, C., Ed.; Springer: New York, NY, USA, 2002; Volume 2, pp. 110–131. [Google Scholar]
- Mark, J.E. Polymer Data Handbook; Oxford University Press: New York, NY, USA, 2009; pp. 1–1250. [Google Scholar]
- Powel, C.E.; QIao, G.G. Polymeric CO2/N2 gas separation membranes for the capture of carbon dioxide from power plant flue gases. JMS 2006, 279, 1–49. [Google Scholar] [CrossRef]
- Kohl, A.L.; Nielsen, L.B. Gas Purification, 5th ed.; Gulf Professional Publishing: Houston, TX, USA, 1997; pp. 154–196. [Google Scholar]
- Dai, Z.; Noble, R.D.; Gin, D.L.; Zhang, X.; Deng, L. Combination of ionic liquids with membrane technology: A new approach for CO2 separation. J. Membr. Sci. 2016, 497, 1–20. [Google Scholar] [CrossRef]
- Bazhenov, S.; Ramdin, M.; Volkov, A.; Volkov, V.; Vlugt, T.J.H.; Loos, T.W. CO2 Solubility in Biodegradable Hydroxylammonium-Based Ionic Liquids. J. Chem. Eng. Data 2014, 59, 703–708. [Google Scholar] [CrossRef]
- Hanioka, S.; Maruyama, T.; Sotani, T.; Teramoto, M.; Matsuyama, H.; Nakashima, K.; Hanaki, M.; Kubota, F.; Goto, M. CO2 separation facilitated by task-specific ionic liquids using a supported liquid membrane. J. Membr. Sci. 2008, 314, 1–4. [Google Scholar] [CrossRef]
- Noble, R.D.; Gin, D.L. Perspective on ionic liquids and ionic liquid membranes. J. Membr. Sci. 2011, 369, 1–4. [Google Scholar] [CrossRef]
- Davletbaeva, I.M.; Emelina, O.Y.; Vorotyntsev, I.V.; Davletbaev, R.S.; Grebennikova, E.S.; Petukhov, A.N.; Akhmetshina, A.I.; Sazanova, T.S.; Loskutov, V.V. Synthesis and properties of novel polyurethanes based on amino ethers of boric acid for gas separation membranes. RSC Adv. 2015, 5, 65674–65683. [Google Scholar] [CrossRef]
- Aki, S.N.V.K.; Mellein, B.R.; Saurer, E.M.; Brennecke, J.F. High-Pressure Phase Behavior of Carbon Dioxide with Imidazolium-Based Ionic Liquids. J. Phys. Chem. B 2004, 108, 20355–20365. [Google Scholar] [CrossRef]
- Huang, X.; Margulis, C.J.; Li, Y.; Berne, B.J. Why Is the Partial Molar Volume of CO2 So Small When Dissolved in a Room Temperature Ionic Liquid? Structure and Dynamics of CO2 Dissolved in [bmim+] [PF6−]. J. Am. Chem. Soc. 2005, 127, 17842–17851. [Google Scholar] [CrossRef] [PubMed]
- Drozdov, P.N.; Kirillov, Y.P.; Kolotilov, E.Y.; Vorotyntsev, I.V. High purification of gases in radial membrane element. Desalination 2002, 146, 249–254. [Google Scholar] [CrossRef]
- Vorotyntsev, V.M.; Drozdov, P.N.; Vorotyntsev, I.V.; Murav’ev, D.V. Fine gas purification to remove slightly penetrating impurities using a membrane module with a feed reservoir. Dokl. Chem. 2006, 411, 243–245. [Google Scholar] [CrossRef]
- Vorotyntsev, V.M.; Drozdov, P.N.; Vorotyntsev, I.V.; Belyaev, E.S. Deep gas cleaning of highly permeating impurities using a membrane module with a feed tank. Pet. Chem. 2011, 51, 595–600. [Google Scholar] [CrossRef]
- Fedoseeva, E.N.; Smirnova, L.A.; Sorokina, M.A.; Pastukhov, M.O. Radical degradation of chitosan under the action of a redox system. Russ. J. App. Chem. 2006, 79, 845–849. [Google Scholar] [CrossRef]
- Neves, L.A.; Crespo, J.G.; Coelhoso, I.M. Gas permeation studies in supported ionic liquid membranes. J. Membr. Sci. 2010, 357, 160–170. [Google Scholar] [CrossRef]
- Neves, L.A.; Afonso, C.; Coelhoso, I.M.; Crespo, J.G. Integrated CO2 capture and enzymatic bioconvension in supported ionic liquid membranes. Sep. Purif. Technol. 2012, 97, 34–41. [Google Scholar] [CrossRef]

| IL | CS-PAN | CS-PS | ||||||
|---|---|---|---|---|---|---|---|---|
| Graft | Block | Graft | Block | |||||
| Ra, nm | Rz, nm | Ra, nm | Rz, nm | Ra, nm | Rz, nm | Ra, nm | Rz, nm | |
| no | 16.03 | 88.06 | 13.11 | 60.14 | 8.22 | 45.57 | 9.81 | 53.29 |
| [bmim] [BF4] | 106.07 | 470.57 | 7.80 | 35.12 | 19.80 | 230.69 | 41.36 | 149.08 |
| [bmim] [PF6] | 112.28 | 325.72 | 23.29 | 86.97 | 72.03 | 393.60 | 42.34 | 168.30 |
| [bmim] [Tf2N] | 228.53 | 546.18 | 141.58 | 707.65 | 55.33 | 435.08 | 50.27 | 233.43 |
| IL | CS-PAN | CS-PS | ||||||
|---|---|---|---|---|---|---|---|---|
| Graft | Block | Graft | Block | |||||
| σ, MPa | ε, % | σ, MPa | ε, % | σ, MPa | ε, % | σ, MPa | ε, % | |
| no | 75.57 | 1.49 | 104.29 | 2.80 | 68.84 | 0.43 | 75.15 | 2.06 |
| [bmim] [BF4] | 12.71 | 0.74 | 78.91 | 2.30 | 8.36 | 0.31 | 42.54 | 2.03 |
| [bmim] [PF6] | 28.29 | 1.41 | 80.19 | 2.37 | 6.62 | 0.39 | 53.19 | 2.36 |
| [bmim] [Tf2N] | 17.35 | 0.79 | 68.97 | 2.39 | 18.96 | 1.06 | 59.72 | 2.36 |
| IL | CS-b-PAN | CS-g-PS | CS-b-PS | |||
|---|---|---|---|---|---|---|
| CO2/CH4 | CO2/N2 | CO2/CH4 | CO2/N2 | CO2/CH4 | CO2/N2 | |
| no | 7.8 | 6.3 | n/c | n/c | n/c | n/c |
| [bmim] [BF4] | 14.6 | 15.5 | n/c | n/c | 6.2 | 4.8 |
| [bmim] [PF6] | n/c * | n/c | n/c | n/c | 1.3 | 1.9 |
| [bmim] [Tf2N] | 1.6 | 1.9 | 8.6 | 7.6 | 1.5 | 3.0 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Otvagina, K.V.; Mochalova, A.E.; Sazanova, T.S.; Petukhov, A.N.; Moskvichev, A.A.; Vorotyntsev, A.V.; Afonso, C.A.M.; Vorotyntsev, I.V. Preparation and Characterization of Facilitated Transport Membranes Composed of Chitosan-Styrene and Chitosan-Acrylonitrile Copolymers Modified by Methylimidazolium Based Ionic Liquids for CO2 Separation from CH4 and N2. Membranes 2016, 6, 31. https://doi.org/10.3390/membranes6020031
Otvagina KV, Mochalova AE, Sazanova TS, Petukhov AN, Moskvichev AA, Vorotyntsev AV, Afonso CAM, Vorotyntsev IV. Preparation and Characterization of Facilitated Transport Membranes Composed of Chitosan-Styrene and Chitosan-Acrylonitrile Copolymers Modified by Methylimidazolium Based Ionic Liquids for CO2 Separation from CH4 and N2. Membranes. 2016; 6(2):31. https://doi.org/10.3390/membranes6020031
Chicago/Turabian StyleOtvagina, Ksenia V., Alla E. Mochalova, Tatyana S. Sazanova, Anton N. Petukhov, Alexandr A. Moskvichev, Andrey V. Vorotyntsev, Carlos A. M. Afonso, and Ilya V. Vorotyntsev. 2016. "Preparation and Characterization of Facilitated Transport Membranes Composed of Chitosan-Styrene and Chitosan-Acrylonitrile Copolymers Modified by Methylimidazolium Based Ionic Liquids for CO2 Separation from CH4 and N2" Membranes 6, no. 2: 31. https://doi.org/10.3390/membranes6020031
APA StyleOtvagina, K. V., Mochalova, A. E., Sazanova, T. S., Petukhov, A. N., Moskvichev, A. A., Vorotyntsev, A. V., Afonso, C. A. M., & Vorotyntsev, I. V. (2016). Preparation and Characterization of Facilitated Transport Membranes Composed of Chitosan-Styrene and Chitosan-Acrylonitrile Copolymers Modified by Methylimidazolium Based Ionic Liquids for CO2 Separation from CH4 and N2. Membranes, 6(2), 31. https://doi.org/10.3390/membranes6020031









