Modelling and Comparative Analysis of Different Methods of Liquid Membrane Separations
Abstract
1. Introduction
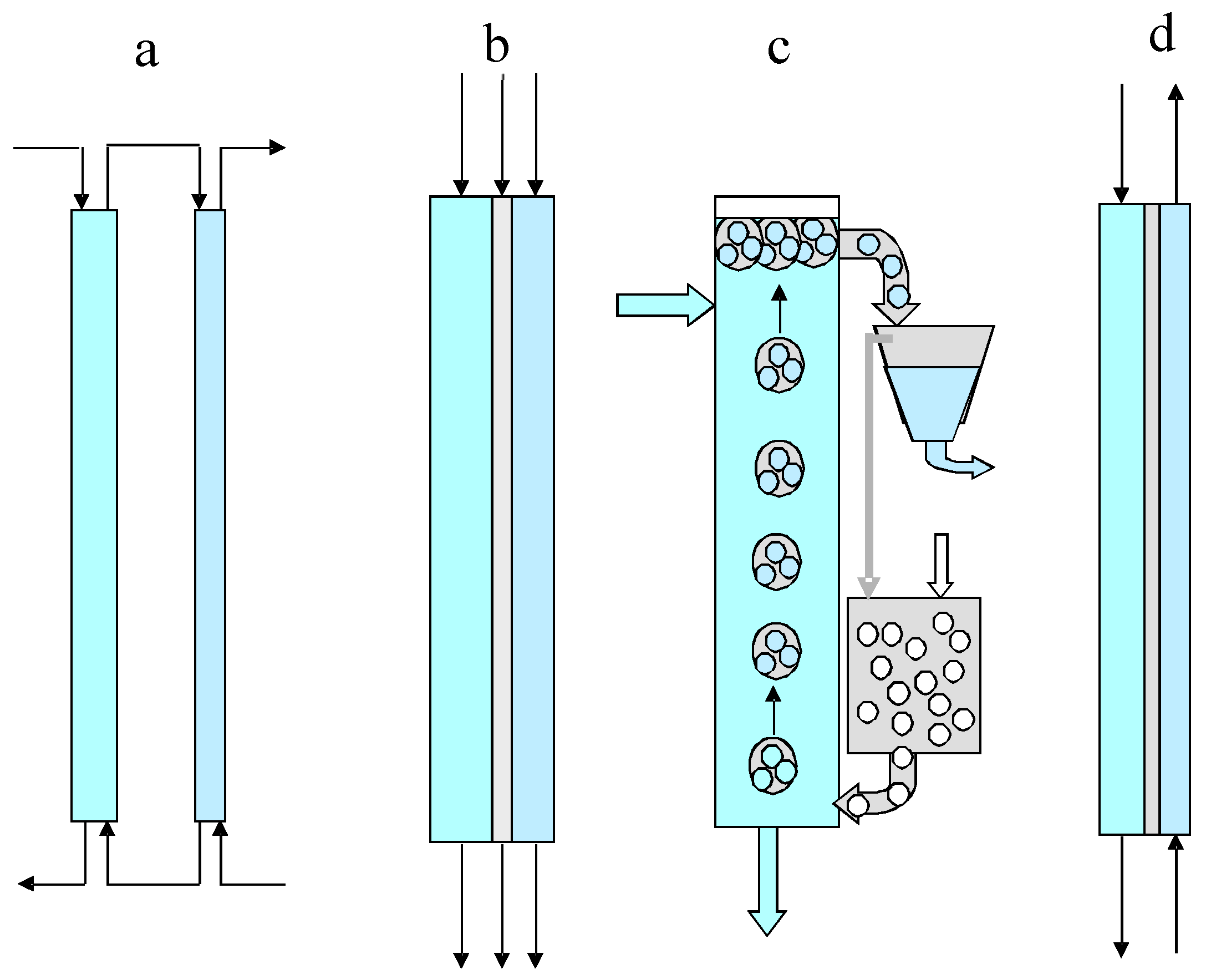
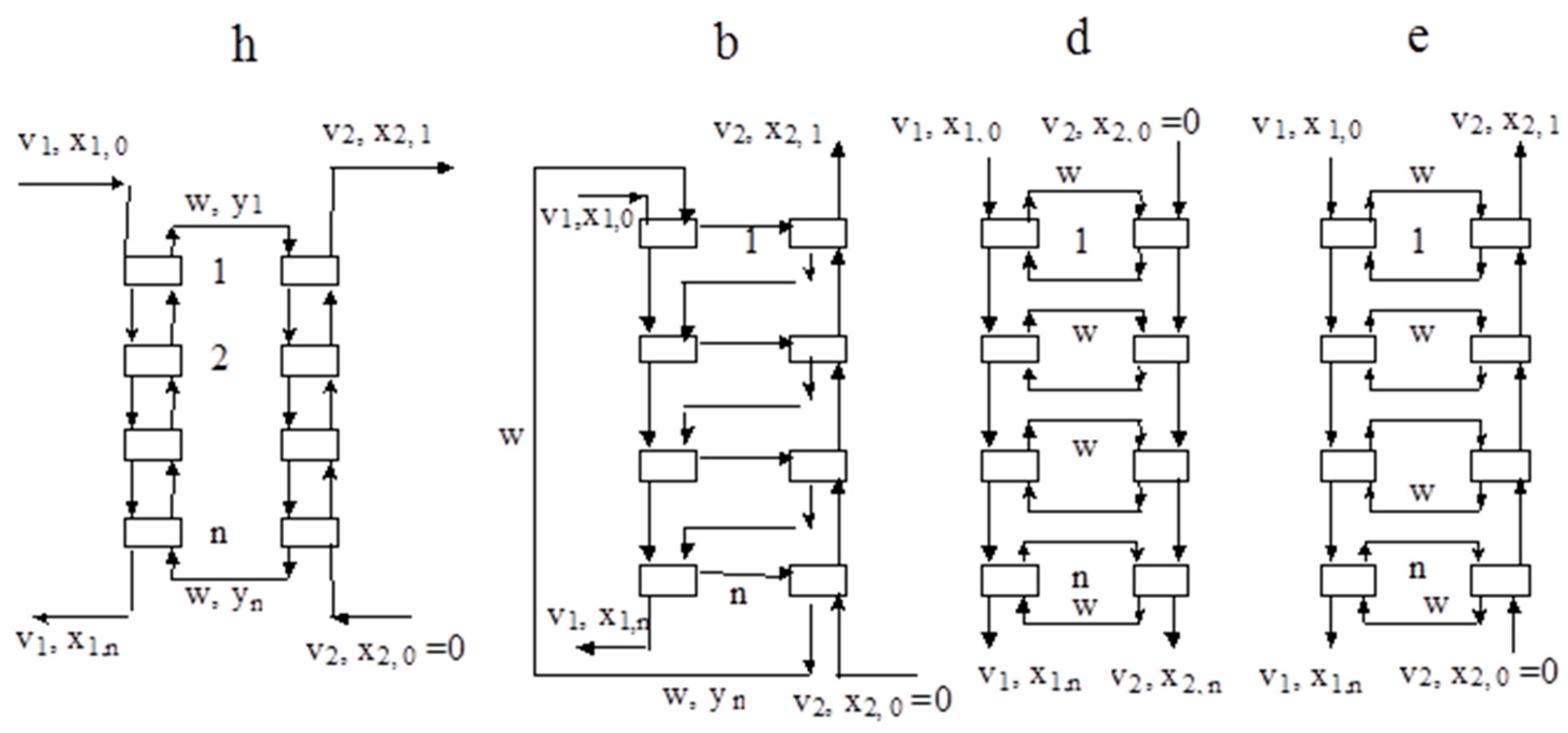
2. Modelling of Continuous Versions of Extraction-Stripping Separation
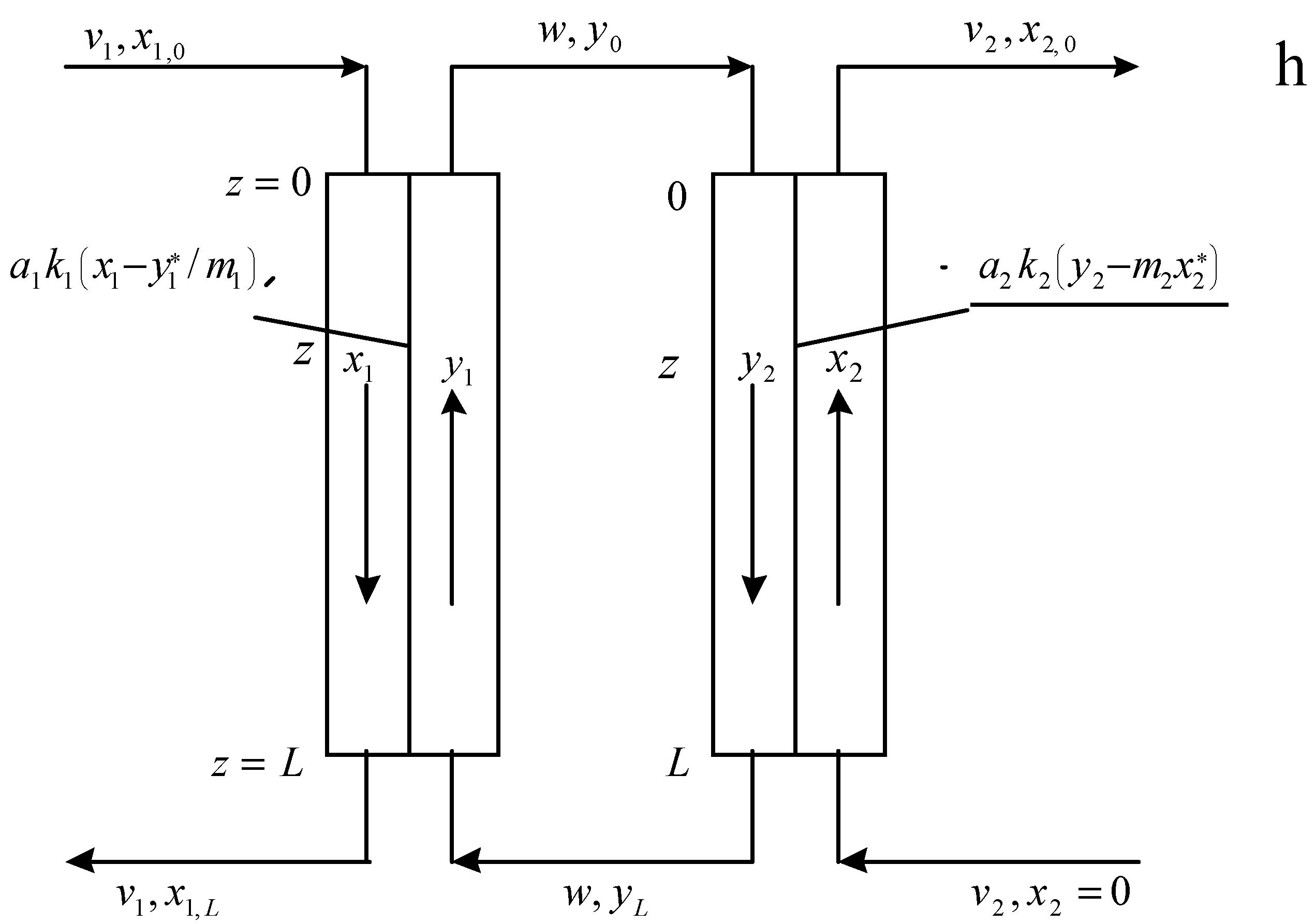
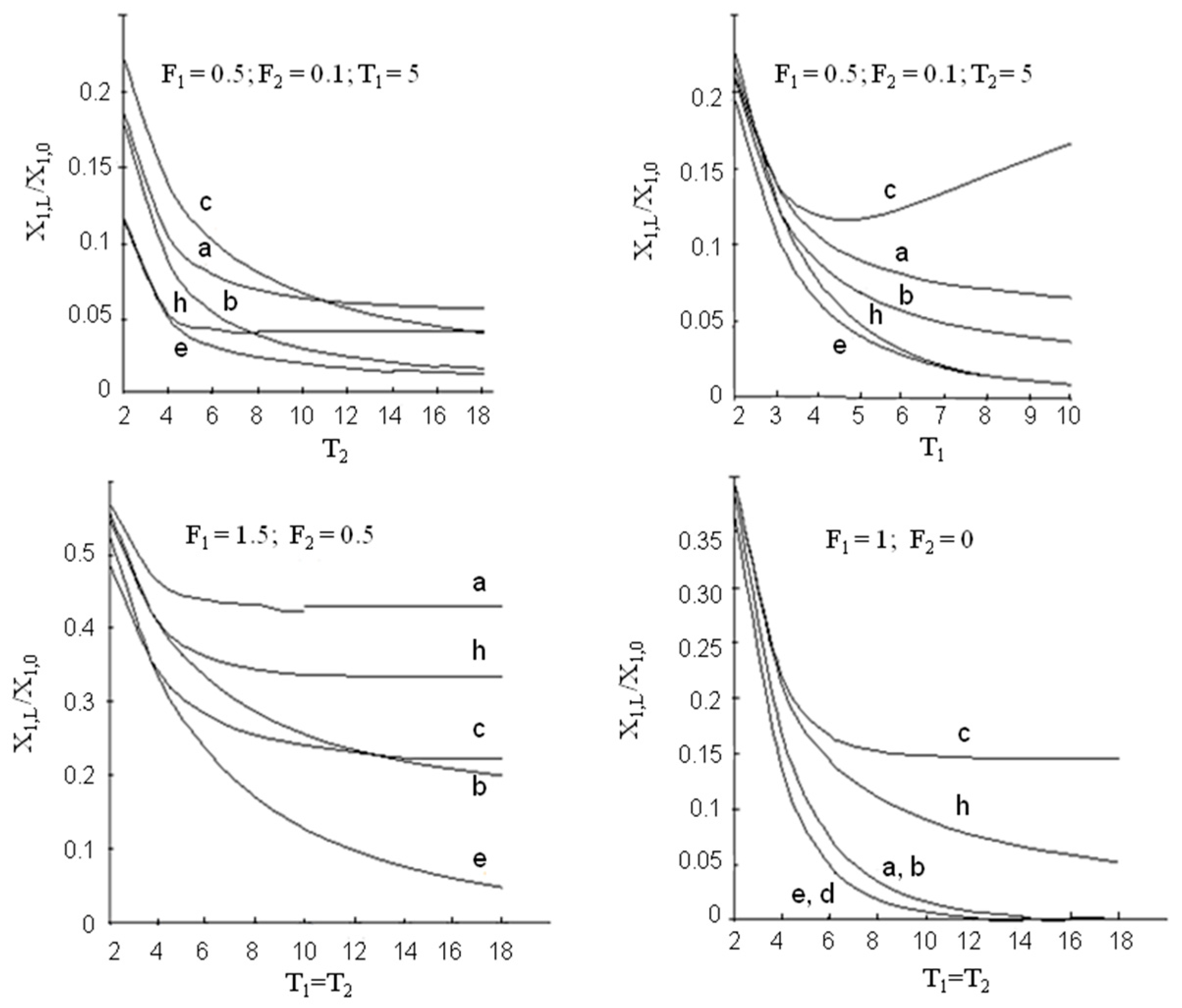
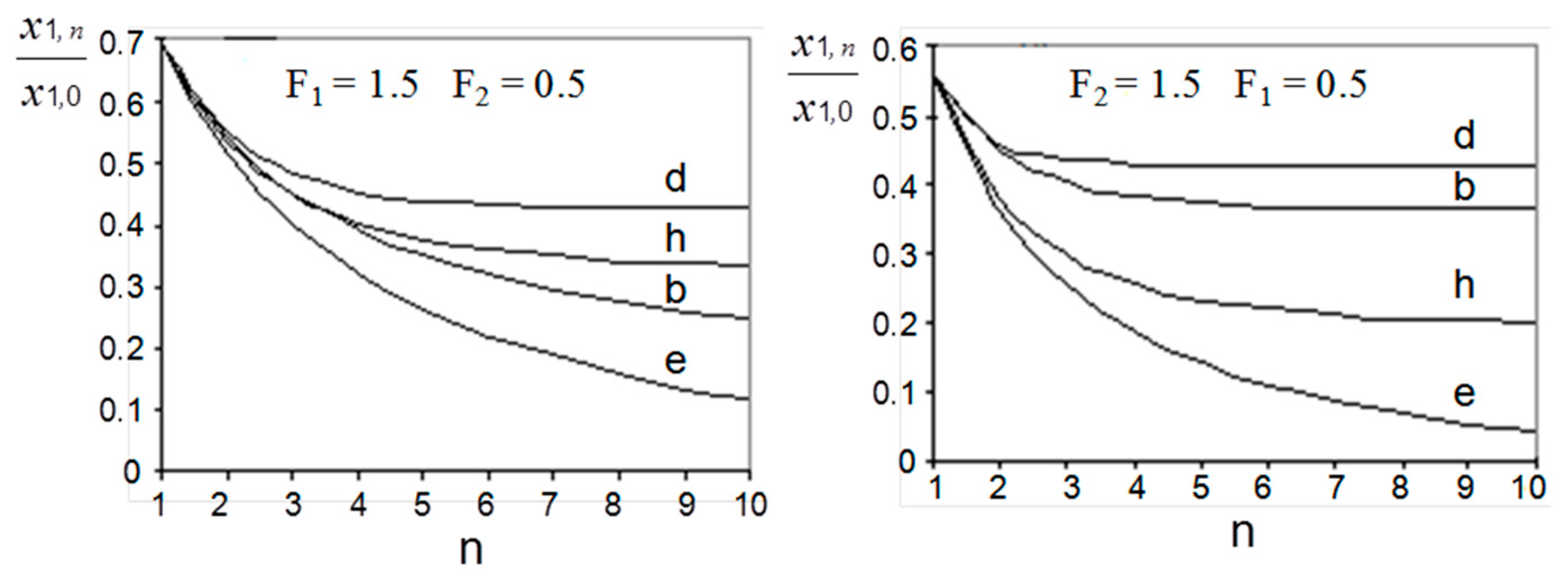
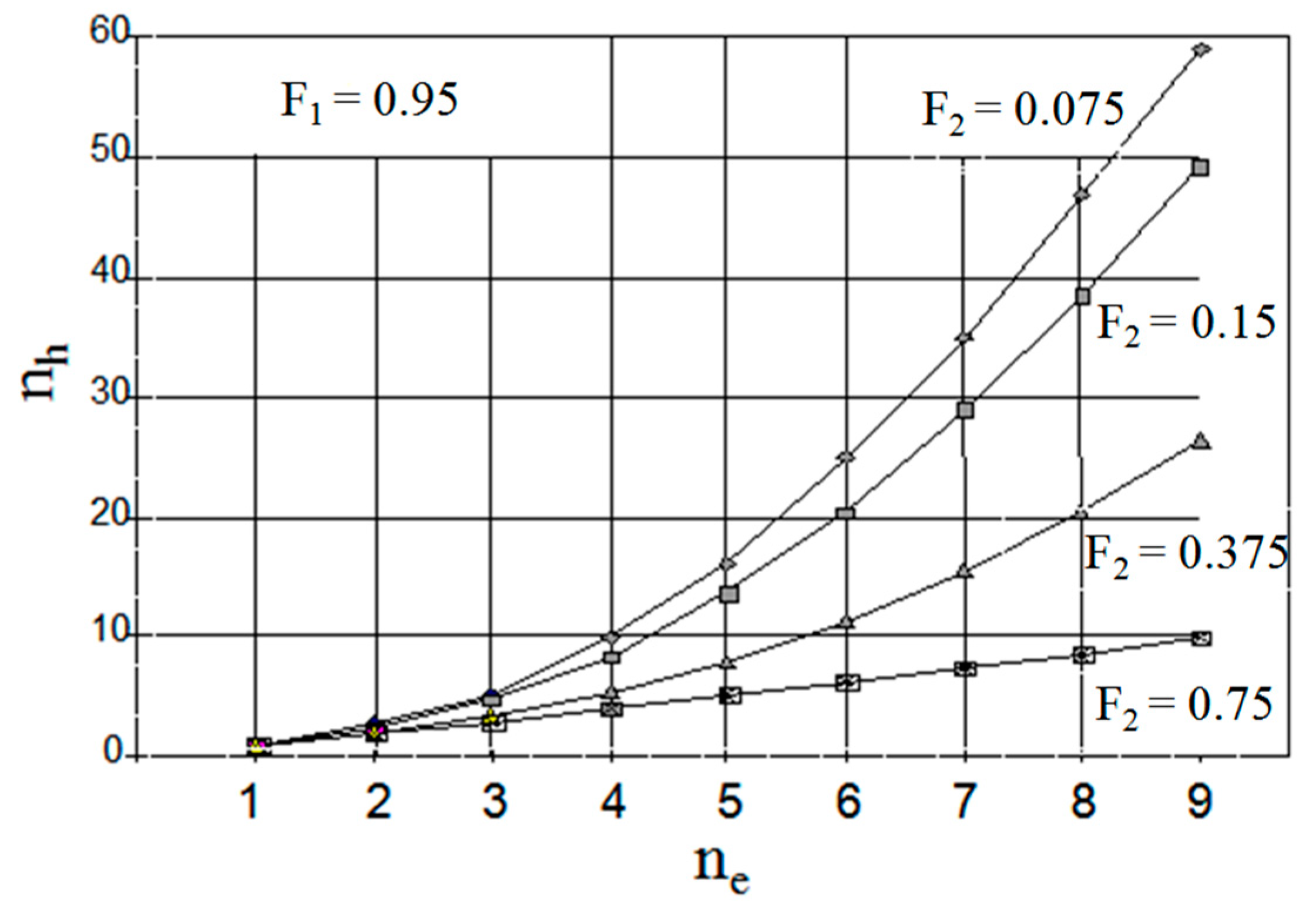
3. Modelling of Staged Versions of Extraction-Stripping Separation
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Lazarova, Z.; Boyadzhiev, L. Liquid film pertraction—A liquid membrane preconcentration technique. Talanta 1992, 39, 931–935. [Google Scholar] [CrossRef] [PubMed]
- Boyadshiev, L. The three-phase liquid extraction or the liquid membranes. Isot. Isot. Environ. Health Stud. 1984, 20, 345–352. [Google Scholar] [CrossRef]
- Boyadzhiev, L.; Atanassova, I. Recovery of L-lysine from dilute water solutions by liquid pertraction. Biotechnol. Bioeng. 1991, 38, 1059–1064. [Google Scholar] [CrossRef] [PubMed]
- Admawi, H.K.; Mohammed, A.A. A comprehensive review of emulsion liquid membrane for toxic contaminants removal: An overview on emulsion stability and extraction efficiency. J. Environ. Chem. Eng. 2023, 11, 109936. [Google Scholar] [CrossRef]
- Zamouche, M.; Tahraoui, H.; Laggoun, Z.; Mechati, S.; Chemchmi, R.; Kanjal, M.I.; Amrane, A.; Hadadi, A.; Mouni, L. Optimization and prediction of stability of emulsified liquid membrane (ELM): Artificial neural network. Processes 2023, 11, 364. [Google Scholar] [CrossRef]
- Karmakar, R.; Singh, P.; Datta, A.; Sen, K. Emulsion liquid membrane in the selective extraction of Dy. Chem. Eng. Res. Des. 2022, 187, 497–506. [Google Scholar] [CrossRef]
- Khan, H.W.; Elgharbawy, A.A.M.; Bustam, M.A.; Goto, M.; Moniruzzaman, M. Ionic liquid-based green emulsion liquid membrane for the extraction of the poorly soluble drug ibuprofen. Molecules 2023, 28, 2345. [Google Scholar] [CrossRef]
- Wang, D.; Wang, Q.; Zhang, X.; Liu, T.; Zhang, H. Conversion of waste oil from oil refinery into emulsion liquid membrane for removal of phenol: Stability evaluation, modeling and optimization. Membranes 2022, 12, 1202. [Google Scholar] [CrossRef]
- Zhu, G.; Wang, Y.; Huang, Q.; Zhang, R.; Chen, D.; Wang, S.; Yang, X. Emulsion liquid membrane for simultaneous extraction and separation of copper from nickel in ammoniacal solutions. Miner. Eng. 2022, 188, 107849. [Google Scholar] [CrossRef]
- Naim, M.M.; Moneer, A.A.; El-Shafei, A.A.; Elewa, M.M. Automated prototype for desalination by emulsion liquid membrane technique. Desalin. Water Treat. 2017, 73, 164–174. [Google Scholar] [CrossRef]
- Kohli, H.P.; Gupta, S.; Chakraborty, M. Extraction of Ethylparaben by emulsion liquid membrane: Statistical analysis of operating parameters. Colloids Surf. A Physicochem. Eng. Asp. 2018, 539, 371–381. [Google Scholar] [CrossRef]
- León, G.; Gómez, E.; Miguel, B.; Hidalgo, A.M.; Gómez, M.; Murcia, M.D.; Guzmá, M.A. Feasibility of adsorption kinetic models to study carrier-mediated transport of heavy metal ions in emulsion liquid membranes. Membranes 2022, 12, 66. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, A.L.; Shafie, Z.M.; Zaulkiflee, N.D.; Pang, W.Y. Preliminary Study of emulsion liquid membrane formulation on acetaminophen removal from the aqueous phase. Membranes 2019, 9, 133. [Google Scholar] [CrossRef] [PubMed]
- Seifollahi, Z.; Rahbar-Kelishami, A. Diclofenac extraction from aqueous solution by an emulsion liquid membrane: Parameter study and optimization using the response surface methodology. J. Mol. Liq. 2017, 231, 1–10. [Google Scholar] [CrossRef]
- Rzelewska-Piekut, M.; Regel-Rosocka, M. Liquid membranes for separation of metal ions from wastewaters. Phys. Sci. Rev. 2021, 20210049. [Google Scholar] [CrossRef]
- Ried, W. Membrane-Supported Liquid-Liquid Extraction—Where do we stand today? ChemBioEng Rev. 2021, 8, 6–14. [Google Scholar] [CrossRef]
- Amini, M.; Rahbar-Kelishami, A.; Alipour, M.; Vahidi, O. Supported liquid membrane in metal ion separation: An overview. J. Membr. Sci. Res. 2018, 4, 121–135. [Google Scholar]
- Farah, M.; Giralt, J.; Stüber, F.; Font, J.; Fabregat, A.; Fortuny, A. Supported liquid membranes for the removal of pharmaceuticals from aqueous solutions. J. Water Process Eng. 2022, 49, 103170. [Google Scholar] [CrossRef]
- Pilli, S.R.; Ali, W.; Motana, S.; Khan, M.E.; Rajesh, Y.; Khan, A.U.; Bashiri, A.H.; Zakri, W. Novel-supported ionic liquid membranes for an effective removal of pentachlorophenol from wastewater. J. Mol. Liq. 2023, 380, 121629. [Google Scholar] [CrossRef]
- Li, L.; Yu, B.; Davis, K.; King, A.; Dal-Cin, M.; Nicalek, A.; Du, N. Separation of neodymium(III) and lanthanum(III) via a flat sheet-supported liquid membrane with different extractant-acid systems. Membranes 2022, 12, 1197. [Google Scholar] [CrossRef]
- León, G.; Hidalgo, A.M.; Miguel, B.; Guzmán, M.A. Pertraction of Co(II) through novel ultrasound prepared supported liquid membranes containing D2EHPA. Optimization and transport parameters. Membranes 2020, 10, 436. [Google Scholar] [CrossRef] [PubMed]
- Alemrajabi, M.; Ricknell, J.; Samak, S.; Varela, R.R.; Martinez, J.; Hedman, F.; Forsberg, K.; Rasmuson, A.C. Separation of rare-earth elements using supported liquid membrane extraction in pilot scale. Ind. Eng. Chem. Res. 2022, 61, 18475–18491. [Google Scholar] [CrossRef]
- Zante, G.; Boltoeva, M.; Masmoudi, A.; Barillon, R.; Trébouet, D. Supported ionic liquid and polymer inclusion membranes for metal separation. Sep. Purif. Rev. 2022, 51, 100–116. [Google Scholar] [CrossRef]
- Jean, E.; Villemin, D.; Hlaibi, M.; Lebrun, L. Heavy metal ions extraction using new supported liquid membranes containing ionic liquid as carrier. Sep. Purif. Technol. 2018, 201, 1–9. [Google Scholar] [CrossRef]
- Chen, Z.; He, Q.; Deng, X.; Peng, J.; Du, K.; Sun, Y. Engineering solid nanochannels with macrocyclic host–guest chemistry for stimuli responses and molecular separations. Chem. Commun. 2023, 59, 1907–1916. [Google Scholar] [CrossRef]
- Li, R.; Lin, Q.; Li, S.; Sun, Y.; Liu, Y. MXenes functionalized with macrocyclic hosts: From molecular design to applications. ChemPlusChem 2023, 88, e202200423. [Google Scholar] [CrossRef]
- Kostanyan, A.E.; Belova, V.V.; Voshkin, A.A. Three- and multi-phase extraction as a tool for the implementation of liquid membrane separation methods in practice. Membranes 2022, 12, 926. [Google Scholar] [CrossRef]
- Kostanyan, A.E. Comparison between various schemes of three-phase extraction: Mass transfer between two liquid phases through an exchange medium. Theor. Found. Chem. Eng. 1999, 33, 584–592. [Google Scholar]
- Kostanyan, A.E.; Safiulina, A.M.; Tananaev, I.T.; Myasoedov, B.F. Multiphase extraction: Design of single-and multistage separation using liquid pseudomembranes. Dokl. Chem. 2005, 404, 203–205. [Google Scholar] [CrossRef]
- Kostanyan, A.E.; Safiulina, A.M.; Tananaev, I.G. Linear models of three-phase extraction processes. Theor. Found. Chem. Eng. 2007, 41, 755–759. [Google Scholar] [CrossRef]
- Belova, V.V.; Kostanyan, A.E.; Zakhodyaeva, Y.A.; Kholkin, A.I.; Logutenko, O.A. On the application of bulk-supported liquid membrane techniques in hydrometallurgy. Hydrometallurgy 2014, 150, 144–152. [Google Scholar] [CrossRef]
- Kostanian, A.E. Staged versions of liquid membrane extraction processes. Solvent Extr. Ion Exch. 2013, 31, 297–305. [Google Scholar] [CrossRef]
- Kostanyan, A.E. On the application of liquid-membrane principle in a system of mixing-settling extractors. Theor. Found. Chem. Eng. 2008, 42, 718–723. [Google Scholar] [CrossRef]
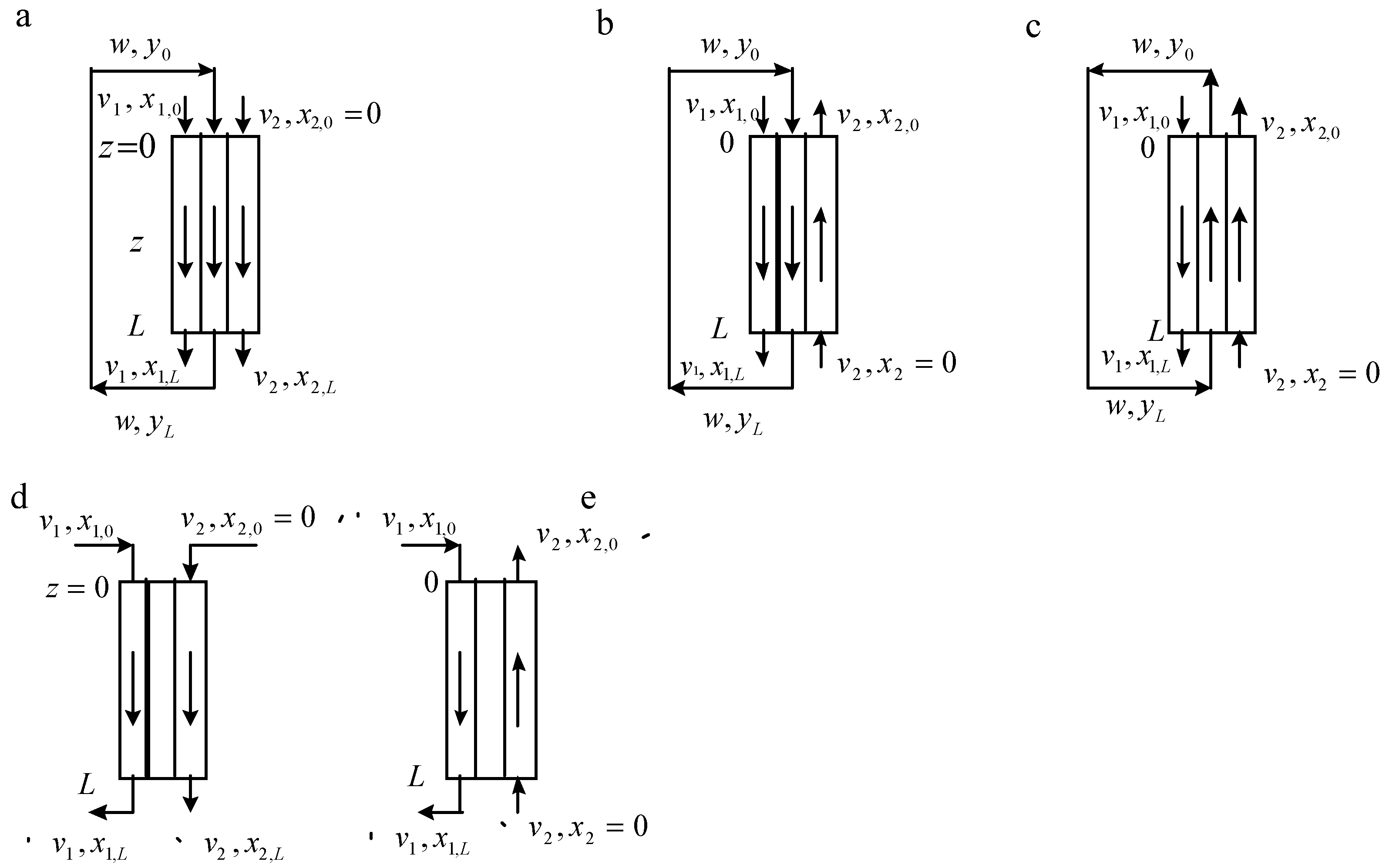
| Number of Stages | x1 (%) Experiment | x1 (%) Theory | x2 (%) Experiment | x2 (%) Theory |
|---|---|---|---|---|
| v1 = 1.12 L/h, x1,0 = 4.8%, v2 = 2.36 L/h, w = 3.0 L/h | ||||
| 1 | 3.49 | 3.00 | 2.00 | 2.04 |
| 2 | 2.20 | 1.78 | 1.36 | 1.18 |
| 3 | 1.27 | 1.01 | 0.72 | 0.61 |
| 4 | 0.70 | 0.50 | 0.26 | 0.24 |
| v1 = 1.48 L/h, x1,0 = 4.7%, v2 = 2.64 L/h, w = 6.0 L/h | ||||
| 1 | 2.94 | 2.96 | 2.44 | 2.37 |
| 2 | 1.83 | 1.79 | 1.40 | 1.40 |
| 3 | 0.98 | 1.00 | 0.74 | 0.74 |
| 4 | 0.49 | 0.47 | 0.23 | 0.30 |
| v1 = 1.15 L/h, x1,0 = 4.8%, v2 = 2.16 L/h, w = 10.0 L/h | ||||
| 1 | 2.77 | 2.72 | 2.34 | 2.40 |
| 2 | 1.56 | 1.48 | 1.28 | 0.29 |
| 3 | 0.76 | 0.74 | 0.62 | 0.63 |
| 4 | 0.34 | 0.30 | 0.23 | 0.23 |
| Number of Stages | v1 = 1.15 L/h, x1,0 = 4.8%, v2 = 2.16 L/h, w = 10.0 L/h | |||
|---|---|---|---|---|
| x1 (%) Experiment | x1 (%) Theory | x2 (%) Experiment | x2 (%) Theory | |
| 1 | 1.84 | 1.86 | 1.83 | 1.85 |
| 2 | 1.43 | 1.40 | 1.81 | 1.82 |
| 3 | 1.37 | 1.33 | 1.71 | 1.71 |
| 4 | 1.36 | 1.32 | 1.40 | 1.32 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kostanyan, A.E.; Voshkin, A.A.; Belova, V.V.; Zakhodyaeva, Y.A. Modelling and Comparative Analysis of Different Methods of Liquid Membrane Separations. Membranes 2023, 13, 554. https://doi.org/10.3390/membranes13060554
Kostanyan AE, Voshkin AA, Belova VV, Zakhodyaeva YA. Modelling and Comparative Analysis of Different Methods of Liquid Membrane Separations. Membranes. 2023; 13(6):554. https://doi.org/10.3390/membranes13060554
Chicago/Turabian StyleKostanyan, Artak E., Andrey A. Voshkin, Vera V. Belova, and Yulia A. Zakhodyaeva. 2023. "Modelling and Comparative Analysis of Different Methods of Liquid Membrane Separations" Membranes 13, no. 6: 554. https://doi.org/10.3390/membranes13060554
APA StyleKostanyan, A. E., Voshkin, A. A., Belova, V. V., & Zakhodyaeva, Y. A. (2023). Modelling and Comparative Analysis of Different Methods of Liquid Membrane Separations. Membranes, 13(6), 554. https://doi.org/10.3390/membranes13060554







