Highly Conductive Fe-Doped (La,Sr)(Ga,Mg)O3−δ Solid-State Membranes for Electrochemical Application
Abstract
1. Introduction
2. Materials and Methods
2.1. Sample Preparation and Characterization
2.2. Electrical and Electrochemical Measurements
3. Results
3.1. Samples Characterization
3.2. Total and Partial Conductivity
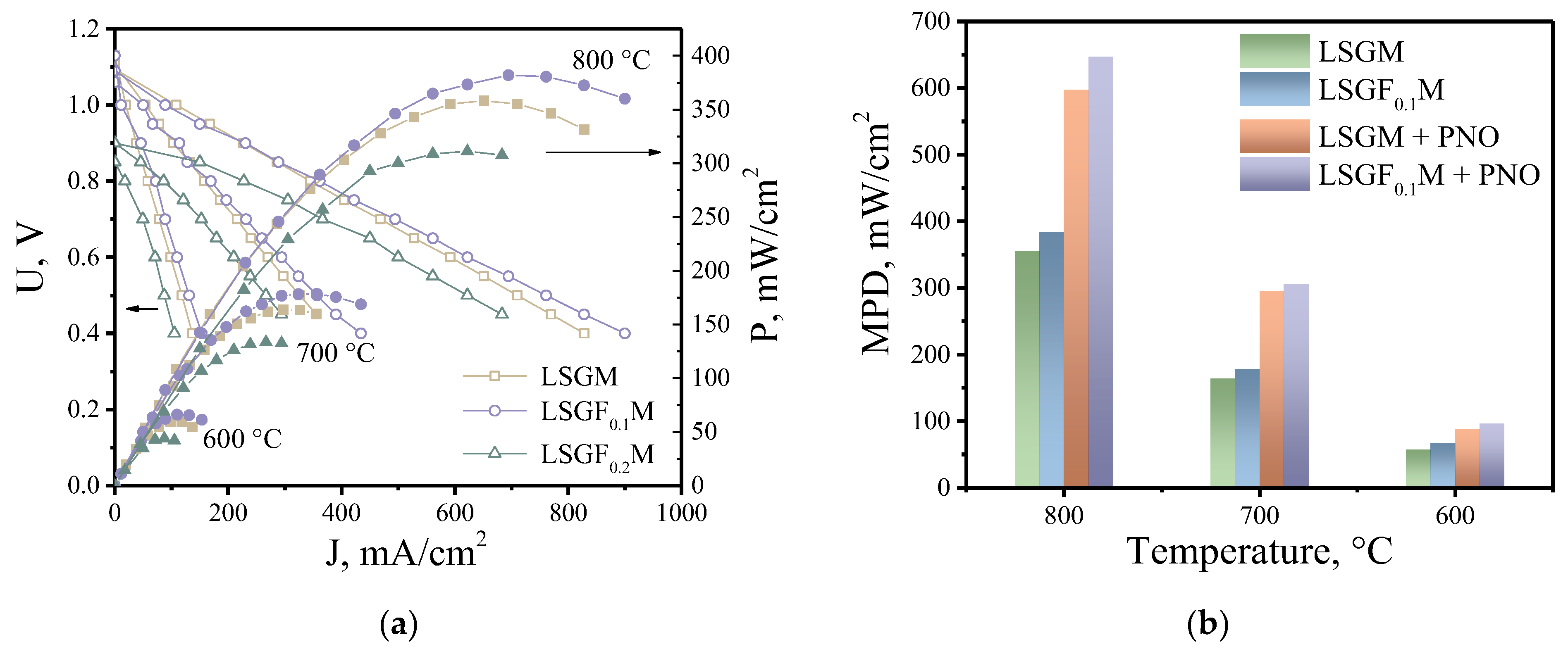
3.3. High-Temperature Electrochemical Study
3.4. SOFCs Testing
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Serga, V.; Burve, R.; Krumina, A.; Pankratova, V.; Popov, A.I.; Pankratov, V. Study of phase composition, photocatalytic activity, and photoluminescence of TiO2 with Eu additive produced by the extraction-pyrolytic method. J. Mater. Res. Technol. 2021, 13, 2350–2360. [Google Scholar] [CrossRef]
- Uklein, A.V.; Multian, V.V.; Kuz’micheva, G.M.; Linnik, R.P.; Lisnyak, V.V.; Popov, A.I.; Gayvoronsky, V.Y. Nonlinear optical response of bulk ZnO crystals with different content of intrinsic defects. Optic. Mater. 2018, 84, 738–747. [Google Scholar] [CrossRef]
- Thirunavukkarasu, G.K.; Hanif, M.B.; Liapun, V.; Hensel, K.; Kupčík, J.; Lorincik, J.; Elantyev, I.; Monfort, O.; Motola, M. Decrypting the growth of anodic TiO2 nanotube layers in eco-friendly fluoride-free nitrate-based electrolyte for enhanced photocatalytic degradation of organic pollutants. Mater. Res. Bull. 2023, 165, 112322. [Google Scholar] [CrossRef]
- Hanif, M.B.; Rauf, S.; Motola, M.; Babar, Z.U.D.; Li, C.-J.; Li, C.-X. Recent progress of perovskite-based electrolyte materials for solid oxide fuel cells and performance optimizing strategies for energy storage applications. Mater. Res. Bull. 2022, 146, 111612. [Google Scholar] [CrossRef]
- Pikalova, E.Y.; Kalinina, E.G.; Pikalova, N.S.; Filonova, E.A. High-Entropy Materials in SOFC Technology: Theoretical Foundations for Their Creation, Features of Synthesis, and Recent Achievements. Materials 2022, 15, 8783. [Google Scholar] [CrossRef]
- Iwahara, H.; Esaka, T.; Uchida, H.; Maeda, N. Proton conduction in sintered oxides and its application to steam electrolysis for hydrogen production. Solid State Ion. 1981, 3–4, 359–363. [Google Scholar] [CrossRef]
- Yuan, L.; Zeng, S.; Zhang, X.; Ji, X.; Zhang, S. Advances and challenges of electrolyzers for large-scale CO2 electroreduction. Mater. Rep. Energy 2023, 3, 100177. [Google Scholar] [CrossRef]
- Cheng, H.; Wang, X.; Meng, X.; Meng, B.; Sunarso, J.; Tan, X.; Liu, L.; Liu, S. Dual-layer BaCe0.8Y0.2O3−δ-Ce0.8Y0.2O2−δ/BaCe0.8Y0.2O3−δ-Ni hollow fiber membranes for H2 separation. J. Membr. Sci. 2020, 601, 117801. [Google Scholar] [CrossRef]
- Hashim, S.S.; Somalu, M.R.; Loh, K.S.; Liu, S.; Zhou, W.; Sunarso, J. Perovskite-based proton conducting membranes for hydrogen separation: A review. Int. J. Hydrogen Energy 2018, 43, 15281–15305. [Google Scholar] [CrossRef]
- He, J.; Xu, X.; Li, M.; Zhou, S.; Zhou, W. Recent advances in perovskite oxides for non-enzymatic electrochemical sensors: A review. Anal. Chim. Acta 2023, 1251, 341007. [Google Scholar] [CrossRef]
- Zhai, H.; Wu, Z.; Fang, Z. Recent progress of Ga2O3-based gas sensors. Ceram. Int. 2022, 48, 24213–24233. [Google Scholar] [CrossRef]
- Filonova, E.; Medvedev, D. Recent Progress in the Design, Characterisation and Application of LaAlO3—and LaGaO3—Based Solid Oxide Fuel Cell Electrolytes. Nanomaterials 2022, 12, 1991. [Google Scholar] [CrossRef]
- Morales, M.; Roa, J.J.; Tartaj, J.; Segarra, M. A review of doped lanthanum gallates as electrolytes for intermediate temperature solid oxides fuel cells: From materials processing to electrical and thermo-mechanical properties. J. Europ. Ceram. Soc. 2016, 36, 1–16. [Google Scholar] [CrossRef]
- Huang, K.; Tichy, R.S.; Goodenough, J.B. Superior Perovskite Oxide-Ion Conductor; Strontium- and Magnesium-Doped LaGaO3: I, Phase Relationships and Electrical Properties. J. Am. Ceram. Soc. 1998, 81, 2565–2575. [Google Scholar] [CrossRef]
- Ishihara, T.; Matsuda, H.; Takita, Y. Doped LaGaO3 perovskite type oxide as a new oxide ionic conductor. J. Am. Chem. Soc. 1994, 116, 3801–3803. [Google Scholar] [CrossRef]
- Zamudio-García, J.; Caizan-Juanarena, L.; Porras-Vazquez, J.M.; Losilla, E.R.; Marrero-Lopez, D. A review on recent advances and trends in symmetrical electrodes for solid oxide cells. J. Power Sources 2022, 520, 230852. [Google Scholar] [CrossRef]
- Zhu, K.; Luo, B.; Liu, Z.; Wen, X. Recent advances and prospects of symmetrical solid oxide fuel cells. Ceram. Int. 2022, 48, 8972–8986. [Google Scholar] [CrossRef]
- Zhang, J.; Ricote, S.; Hendriksen, P.V.; Chen, Y. Advanced Materials for Thin-Film Solid Oxide Fuel Cells: Recent Progress and Challenges in Boosting the Device Performance at Low Temperatures. Adv. Funct. Mater. 2022, 32, 2111205. [Google Scholar] [CrossRef]
- Shi, H.; Su, C.; Ran, R.; Cao, J.; Shao, Z. Electrolyte materials for intermediate-temperature solid oxide fuel cells. Prog. Nat. Sci Mater. Int. 2020, 30, 764–774. [Google Scholar] [CrossRef]
- Lybye, D.; Poulsen, F.W.; Mogensen, M. Conductivity of A- and B-site doped LaAlO3, LaGaO3, LaScO3 and LaInO3 perovskites. Solid State Ion. 2000, 128, 91–103. [Google Scholar] [CrossRef]
- Huang, P.; Petric, A. Superior Oxygen Ion Conductivity of Lanthanum Gallate Doped with Strontium and Magnesium. J. Electrochem. Soc. 1996, 143, 1644–1648. [Google Scholar] [CrossRef]
- Khorkounov, B.A.; Nafe, H.; Aldinger, F. Relationship between the ionic and electronic partial conductivities of co-doped LSGM ceramics from oxygen partial pressure dependence of the total conductivity. J. Solid State Electrochem. 2006, 10, 479–487. [Google Scholar] [CrossRef]
- Trofimenko, N.; Ullmann, H. Transition metal doped lanthanum gallates. Solid State Ion. 1999, 118, 215–227. [Google Scholar] [CrossRef]
- Istomin, S.Y.; Lyskov, N.V.; Mazo, G.N.; Antipov, E.V. Electrode materials based on complex d-metal oxides for symmetrical solid oxide fuel cells. Russ. Chem. Rev. 2021, 90, 644–676. [Google Scholar] [CrossRef]
- Pelosato, R.; Cordaro, G.; Stucchi, D.; Cristiani, C.; Dotelli, G. Cobalt based layered perovskites as cathode material for intermediate temperature Solid Oxide Fuel Cells: A brief review. J. Power Sources 2015, 298, 46–67. [Google Scholar] [CrossRef]
- Gao, Z.; Mogni, L.V.; Miller, E.C.; Railsback, J.G.; Barnett, S.A. A perspective on low-temperature solid oxide fuel cells. Energy Env. Sci. 2016, 9, 1602–1644. [Google Scholar] [CrossRef]
- Osinkin, D.A.; Khodimchuk, A.V.; Antonova, E.P.; Bogdanovich, N.M. Understanding the oxygen reduction kinetics on Sr2-xFe1.5Mo0.5O6−δ: Influence of strontium defciency and correlation with the oxygen isotopic exchange data. Solid State Ion. 2022, 374, 115818. [Google Scholar] [CrossRef]
- Osinkin, D.A. The parallel pathways of hydrogen oxidation reaction on high active decorated Ni–YSZ electrode in electrochemical cell with GDC protective layer. J. Electroanal. Chem. 2022, 927, 116999. [Google Scholar] [CrossRef]
- Gavrilyuk, A.L.; Osinkin, D.A.; Bronin, D.I. On a variation of the Tikhonov regularization method for calculating the distribution function of relaxation times in impedance spectroscopy. Electrochim. Acta 2020, 354, 136683. [Google Scholar] [CrossRef]
- Sepúlveda, E.; Mangalaraja, R.V.; Troncoso, L.; Jiménez, J.; Salvo, C.; Sanhueza, F. Effect of barium on LSGM electrolyte prepared by fast combustion method for solid oxide fuel cells (SOFC). MRS Advances 2022, 7, 1167–1174. [Google Scholar] [CrossRef]
- Garcia-Garcia, F.J.; Tang, Y.; Gotor, Y.T.; Sayagués, M.J. Development by Mechanochemistry of La0.8Sr0.2Ga0.8Mg0.2O2.8 Electrolyte for SOFCs. Materials 2020, 13, 1366. [Google Scholar] [CrossRef]
- Yoo, J.S.; Lee, S.; Yu, J.H.; Woo, S.K.; Park, H.; Kim, H.G. Fe doping effects on phase stability and conductivity of La0.75Sr0.25Ga0.8Mg0.2O3−δ. J. Power Sources 2009, 193, 593–597. [Google Scholar] [CrossRef]
- Jacob, K.T.; Jain, S.; Saji, V.S.; Srikanth, P.V.K. Thermal expansion of doped lanthanum gallates. Bull. Mater. Sci. 2010, 33, 407–411. [Google Scholar] [CrossRef]
- Inaba, H.; Hayashi, H.; Suzuki, M. Structural phase transition of perovskite oxides LaMO3 and La0.9Sr0.1MO3 with different size of B-site ions. Solid State Ion. 2001, 144, 99–108. [Google Scholar] [CrossRef]
- Slater, P.R.; Irvine, J.T.S.; Ishihara, T.; Takita, Y. High-Temperature Powder Neutron Diffraction Study of the Oxide Ion Conductor La0.9Sr0.1Ga0.8Mg0.2O2. J. Solid State Chem. 1998, 139, 135–143. [Google Scholar] [CrossRef]
- Shkerin, S.N.; Bronin, D.I.; Kovyazina, S.A.; Gorelov, V.P.; Kuzmin, A.V.; Martemyanova, Z.S.; Beresnev, S.M. Structure and phase transitions of (La,Sr)(Ga,Mg)O3-a solid electrolyte. Solid State Ion. 2001, 171, 129–134. [Google Scholar] [CrossRef]
- Osinkin, D.A.; Khodimchuk, A.V.; Porotnikova, N.M.; Bogdanovich, N.M.; Fetisov, A.V.; Ananyev, M.V. Rate-Determining Steps of Oxygen Surface Exchange Kinetics on Sr2Fe1.5Mo0.5O6−δ. Energies 2020, 13, 250. [Google Scholar] [CrossRef]
- Osinkin, D.A. Hydrogen oxidation kinetics on a redox stable electrode for reversible solid-state electrochemical devices: The critical influence of hydrogen dissociation on the electrode surface. Electrochim. Acta 2021, 389, 138792. [Google Scholar] [CrossRef]
- Kurumchin, E.K.; Ananev, M.V.; Bronin, D.I.; Vdovin, G.K.; Osinkin, D.A. Conductivity, oxygen interfacial exchange and diffusion in oxides based on lanthanum gallate. Rus. J. Electrochem. 2010, 46, 774–779. [Google Scholar] [CrossRef]
- Osinkin, D.A. Precursor of Pr2NiO4−δ as a highly effective catalyst for the simultaneous promotion of oxygen reduction and hydrogen oxidation reactions in solid oxide electrochemical devices. Int. J. Hydrogen Energy 2021, 46, 24546–24554. [Google Scholar] [CrossRef]
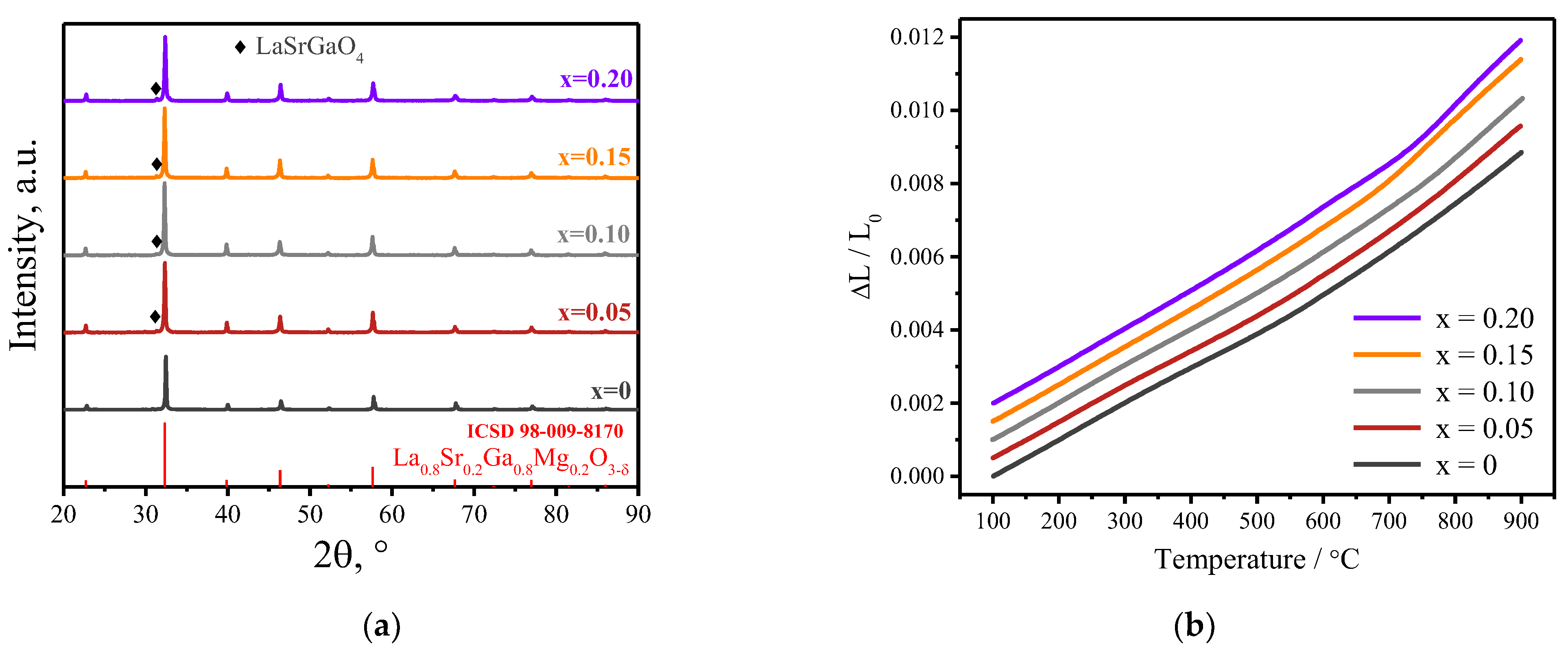
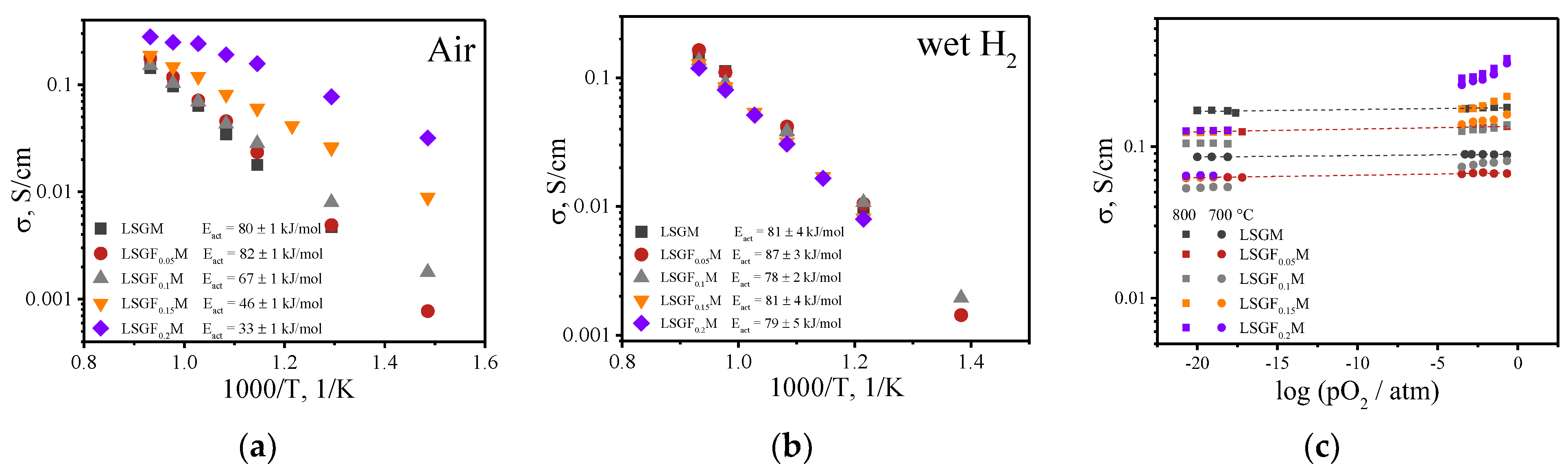


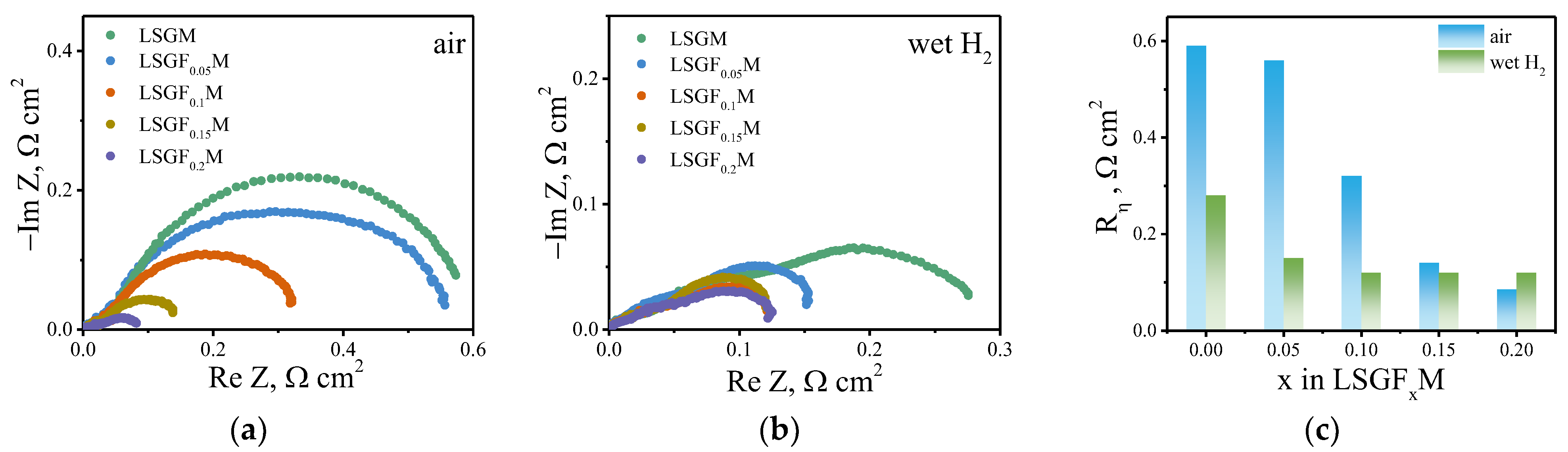
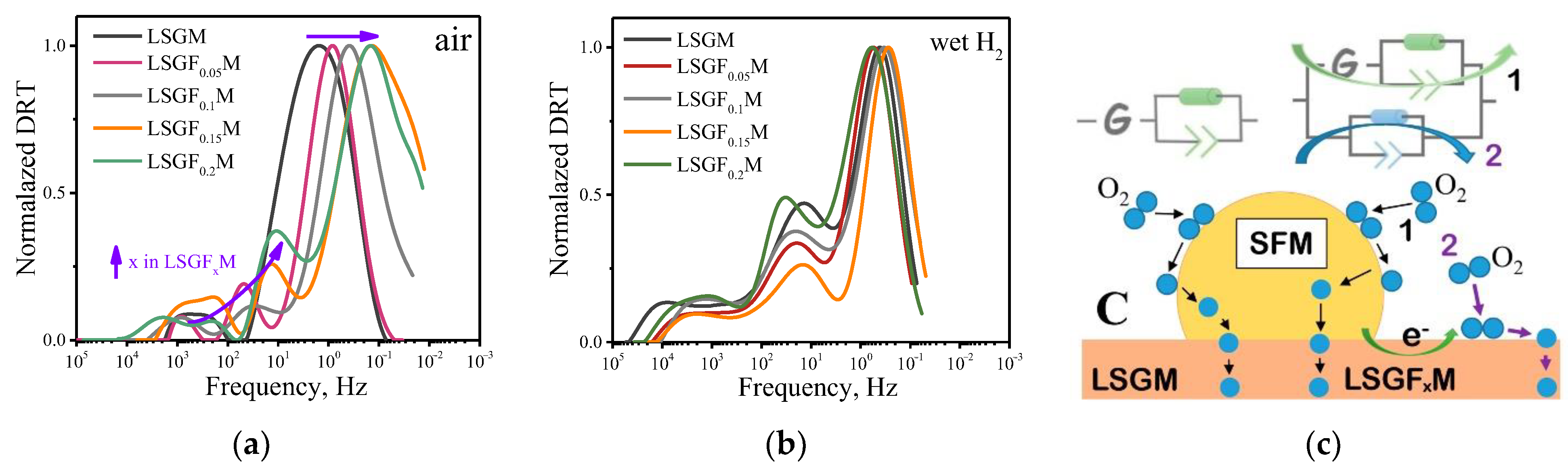
| Given Composition | Concentration of the Cations in the Analyzed Sample/wt.% | Composition Calculated Based on the AES Analysis | ||||
|---|---|---|---|---|---|---|
| La | Sr | Ga | Mg | Fe | ||
| La0.8Sr0.2Ga0.8Mg0.2O3−δ | 46.96 | 8.14 | 23.66 | 1.82 | 0.01 | La0.78Sr0.22Ga0.82Mg0.18O3−δ |
| La0.8Sr0.2Ga0.75Fe0.05Mg0.2O3−δ | 46.88 | 8.13 | 22.60 | 2.00 | 1.20 | La0.78Sr0.22Ga0.76Fe0.05Mg0.19O3−δ |
| La0.8Sr0.2Ga0.7Fe0.1Mg0.2O3−δ | 48.41 | 7.74 | 20.37 | 1.92 | 2.41 | La0.8Sr0.2Ga0.71Fe0.1Mg0.19O3−δ |
| La0.8Sr0.2Ga0.65Fe0.15Mg0.2O3−δ | 47.38 | 8.02 | 19.77 | 1.96 | 3.56 | La0.79Sr0.21Ga0.66Fe0.15Mg0.19O3−δ |
| La0.8Sr0.2Ga0.6Fe0.2Mg0.2O3−δ | 48.42 | 7.98 | 17.75 | 1.94 | 4.66 | La0.79Sr0.21Ga0.61Fe0.2Mg0.19O3−δ |
| Sample | 100–600 °C | 650–900 °C |
|---|---|---|
| LSGM | 9.75(3) | 12.99(6) |
| LSGF0.05M | 9.79(3) | 13.71(9) |
| LSGF0.1M | 10.09(3) | 13.90(12) |
| LSGF0.15M | 10.63(4) | 16.44(8) |
| LSGF0.2M | 10.76(4) | 17.52(8) |
| Sample | Rbulk, Ω cm | Rgb, Ω cm | Cgb, F/cm | fmax, Hz |
|---|---|---|---|---|
| LSGM | 2340 | 340 | 2.6 × 10−9 | 1.1 × 105 |
| LSGF0.05M | 1469 | 166 | 4 × 10−9 | 2.3 × 105 |
| LSGF0.1M | 206 | 32.6 | 1 × 10−8 | 5 × 105 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gordeev, E.; Belyakov, S.; Antonova, E.; Osinkin, D. Highly Conductive Fe-Doped (La,Sr)(Ga,Mg)O3−δ Solid-State Membranes for Electrochemical Application. Membranes 2023, 13, 502. https://doi.org/10.3390/membranes13050502
Gordeev E, Belyakov S, Antonova E, Osinkin D. Highly Conductive Fe-Doped (La,Sr)(Ga,Mg)O3−δ Solid-State Membranes for Electrochemical Application. Membranes. 2023; 13(5):502. https://doi.org/10.3390/membranes13050502
Chicago/Turabian StyleGordeev, Egor, Semyon Belyakov, Ekaterina Antonova, and Denis Osinkin. 2023. "Highly Conductive Fe-Doped (La,Sr)(Ga,Mg)O3−δ Solid-State Membranes for Electrochemical Application" Membranes 13, no. 5: 502. https://doi.org/10.3390/membranes13050502
APA StyleGordeev, E., Belyakov, S., Antonova, E., & Osinkin, D. (2023). Highly Conductive Fe-Doped (La,Sr)(Ga,Mg)O3−δ Solid-State Membranes for Electrochemical Application. Membranes, 13(5), 502. https://doi.org/10.3390/membranes13050502







