MF–NF Treatment Train for Pig Manure: Nutrient Recovery and Reuse of Product Water
Abstract
:1. Introduction
2. Materials and Methods
2.1. Pig Manure Sampling
2.2. Membrane Characteristics
2.3. Membrane Filtration Processes
2.3.1. Microfiltration
2.3.2. Nanofiltration
2.4. Analytical Process
2.5. Calculated Parameters
3. Results and Discussion
3.1. Chemical Characterization of Manure
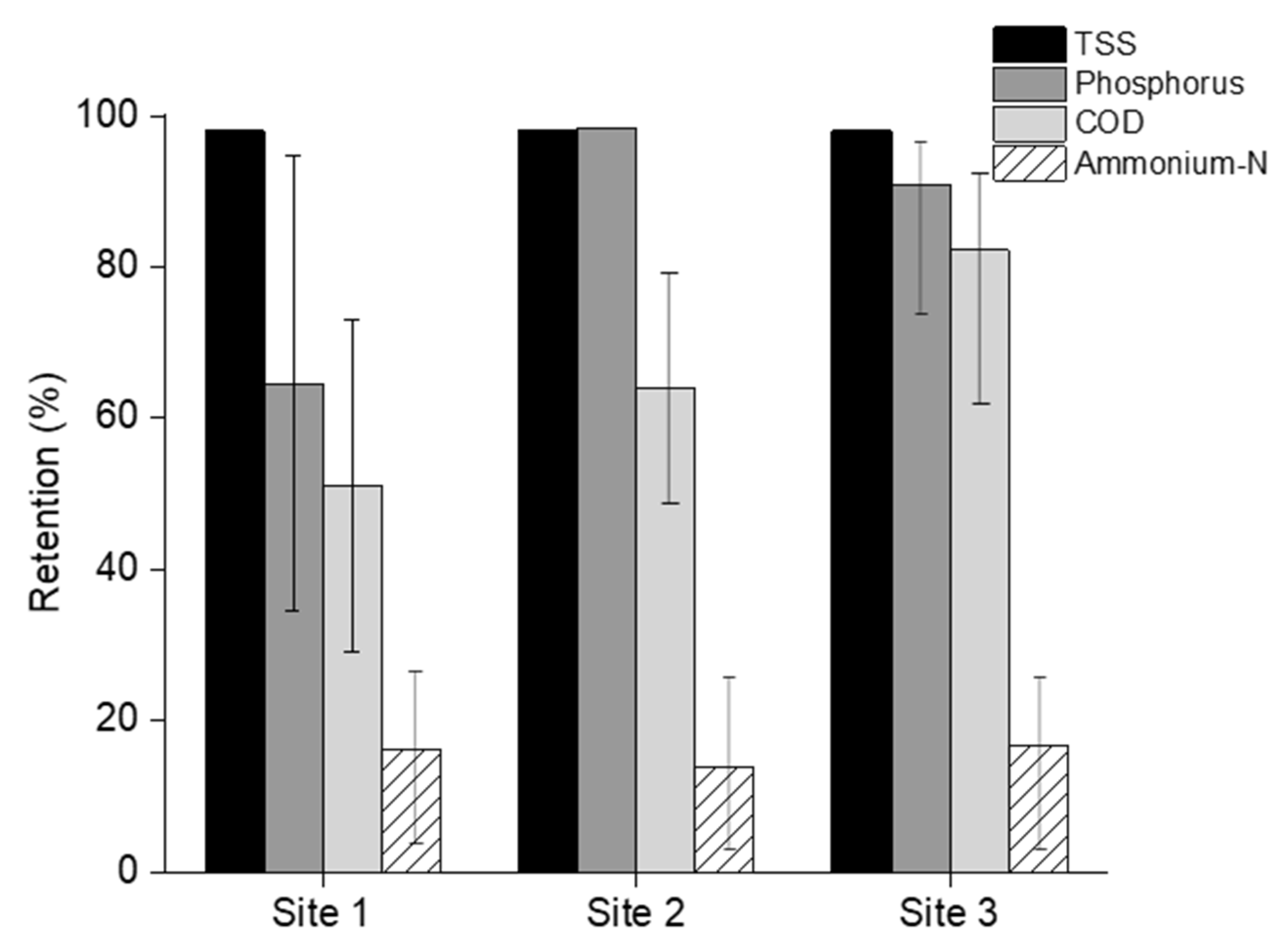
3.2. Microfiltration of Manure
3.3. Nanofiltration of MF Permeate
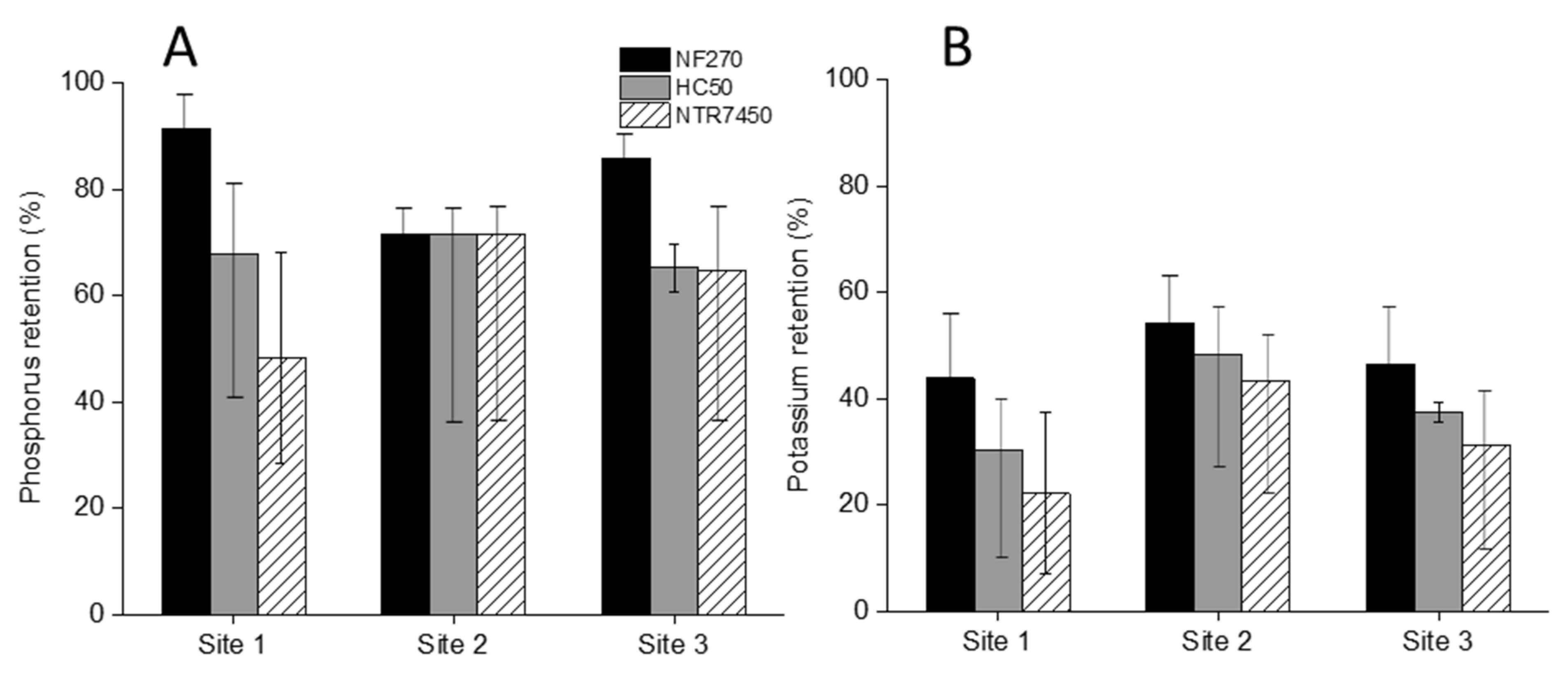
3.3.1. DTN and NH4+-N Retention
3.3.2. Phosphorus and Potassium Retention
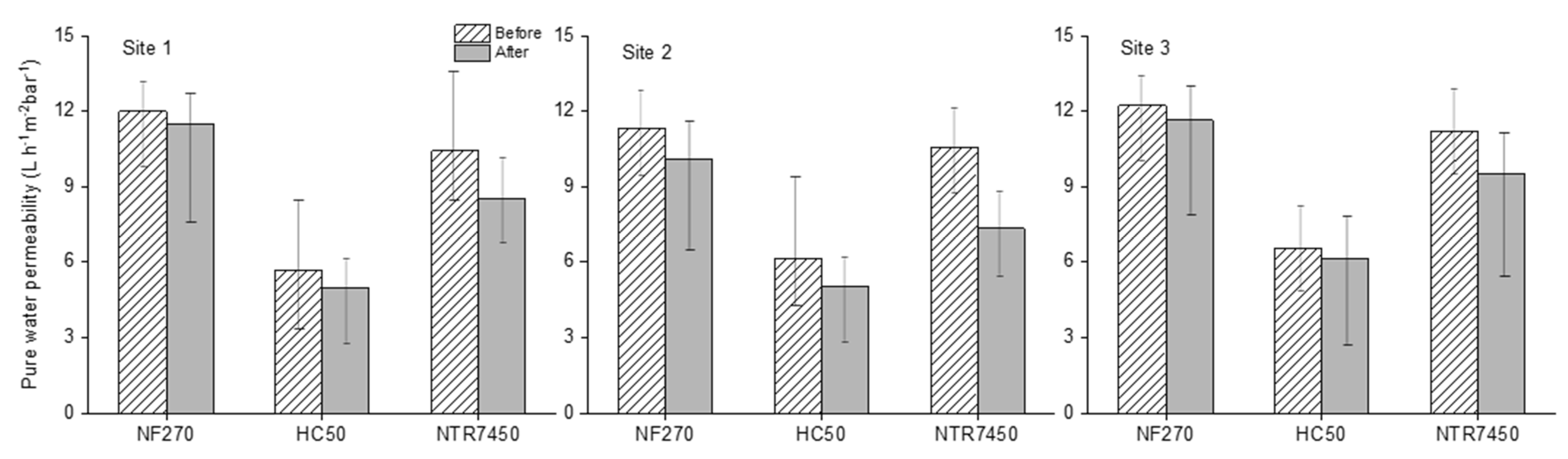
3.4. Membrane Performance
4. Conclusions
- i.
- MF retained phosphorus above 80% within a smaller MF concentrate volume, which accounted for 40% of the initial feed volume. Additionally, the MF permeate contained above 80% of the total nitrogen and most of the dissolve potassium.
- ii.
- NF of the MF permeate by three different NF membranes showed a maximum of 50–70% potassium and NH4+-N retention, respectively, within smaller NF concentrate volumes, which accounted for 30% of the initial feed volume of MF. Among all of the NF membranes, NF270 showed the most promising retention and was found to be the least prone to fouling.
- iii.
- Finally, the MF–NF treatment train was able to produce a particle-free final product water, which accounted for 30% of the initial feed volume of MF. This has the potential to be reused in farms to wash barns, to irrigate nearby cultures, or can be applied to specific fields based on the demand.
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Haneklaus, S.; Schick, J.; Kratz, S.; Rückamp, D.; Schnug, E. Variable rate application of manure—Gain or pain, Landbauforschung. Appl. Agric. For. Res. 2016, 66, 11–20. [Google Scholar]
- Guo, T.; Lou, C.; Zhai, W.; Tang, X.; Hashmi, M.Z.; Murtaza, R.; Li, Y.; Liu, X.; Xu, J. Increased occurrence of heavy metals, antibiotics and resistance genes in surface soil after long-term application of manure. Sci. Total Environ. 2018, 635, 995–1003. [Google Scholar] [CrossRef] [PubMed]
- Webb, J.; Pain, B.; Bittman, S.; Morgan, J. The impacts of manure application methods on emissions of ammonia, nitrous oxide and on crop response—A review. Agric. Ecosyst. Environ. 2010, 137, 39–46. [Google Scholar] [CrossRef]
- Ledda, C.; Schievano, A.; Salati, S.; Adani, F. Nitrogen and water recovery from animal slurries by a new integrated ultrafiltration, reverse osmosis and cold stripping process: A case study. Water Res. 2013, 47, 6157–6166. [Google Scholar] [CrossRef] [PubMed]
- Giola, P.; Basso, B.; Pruneddu, G.; Giunta, F.; Jones, J.W. Impact of manure and slurry applications on soil nitrate in a maize–triticale rotation: Field study and long term simulation analysis. Eur. J. Agron. 2012, 38, 43–53. [Google Scholar] [CrossRef]
- Ngatia, L.; Grace, J.M., III; Moriasi, D.; Taylor, R. Nitrogen and phosphorus eutrophication in marine ecosystems. In Monitoring of Marine Pollution; IntechOpen: London, UK, 2019; pp. 1–17. [Google Scholar]
- Wick, K.; Heumesser, C.; Schmid, E. Groundwater nitrate contamination: Factors and indicators. J. Environ. Manag. 2012, 111, 178–186. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Almutairi, F.M.; Williams, P.M.; Lovitt, R.W. Polymer enhanced membrane filtration of metals: Retention of single and mixed species of metal ions based on adsorption isotherms. Desalination Water Treat. 2011, 28, 130–136. [Google Scholar] [CrossRef]
- Albuquerque, M.; Martino, V.; Pollet, E.; Avérous, L.; Reis, M. Mixed culture polyhydroxyalkanoate (PHA) production from volatile fatty acid (VFA)-rich streams: Effect of substrate composition and feeding regime on PHA productivity, composition and properties. J. Biotechnol. 2011, 151, 66–76. [Google Scholar] [CrossRef]
- Riaño, B.; Molinuevo, B.; García-González, M. Potential for methane production from anaerobic co-digestion of swine manure with winery wastewater. Bioresour. Technol. 2011, 102, 4131–4136. [Google Scholar] [CrossRef]
- Gerardo, M.; Zacharof, M.; Lovitt, R. Strategies for the recovery of nutrients and metals from anaerobically digested dairy farm sludge using cross-flow microfiltration. Water Res. 2013, 47, 4833–4842. [Google Scholar] [CrossRef]
- Kim, A.H.; Anthony, C.Y.; El Abbadi, S.H.; Lu, K.; Chan, D.; Appel, E.A.; Criddle, C.S. More than a fertilizer: Wastewater-derived struvite as a high value, sustainable fire retardant. Green Chem. 2021, 23, 4510–4523. [Google Scholar] [CrossRef]
- Lu, N.C.; Liu, J. Removal of phosphate and fluoride from wastewater by a hybrid precipitation–microfiltration process. Sep. Purif. Technol. 2010, 74, 329–335. [Google Scholar] [CrossRef]
- Quan, X.; Wang, F.; Zhao, Q.; Zhao, T.; Xiang, J. Air stripping of ammonia in a water-sparged aerocyclone reactor. J. Hazard. Mater. 2009, 170, 983–988. [Google Scholar] [CrossRef] [PubMed]
- Uysal, A.; Yilmazel, Y.D.; Demirer, G.N. The determination of fertilizer quality of the formed struvite from effluent of a sewage sludge anaerobic digester. J. Hazard. Mater. 2010, 181, 248–254. [Google Scholar] [CrossRef] [PubMed]
- Hjorth, M.; Christensen, K.V.; Christensen, M.L.; Sommer, S.G. Solid-liquid separation of animal slurry in theory and practice. Sustain. Agric. 2011, 2, 953–986. [Google Scholar]
- Masse, L.; Massé, D.; Pellerin, Y. The use of membranes for the treatment of manure: A critical literature review. Biosyst. Eng. 2007, 98, 371–380. [Google Scholar] [CrossRef]
- Samanta, P.; von Ungern-Sternberg Schwark, L.; Horn, H.; Saravia, F. Nutrient recovery and ammonia-water production by MF-vacuum evaporation treatment of pig manure. J. Environ. Chem. Eng. 2022, 10, 106929. [Google Scholar] [CrossRef]
- Ersahin, M.E.; Ozgun, H.; Dereli, R.K.; Ozturk, I.; Roest, K.; van Lier, J.B. A review on dynamic membrane filtration: Materials, applications and future perspectives. Bioresour. Technol. 2012, 122, 196–206. [Google Scholar] [CrossRef]
- Hu, X.; Zhou, Q.; Luo, Y. Occurrence and source analysis of typical veterinary antibiotics in manure, soil, vegetables and groundwater from organic vegetable bases, northern China. Environ. Pollut. 2010, 158, 2992–2998. [Google Scholar] [CrossRef]
- Lamshöft, M.; Sukul, P.; Zühlke, S.; Spiteller, M. Behaviour of 14C-sulfadiazine and 14C-difloxacin during manure storage. Sci. Total Environ. 2010, 408, 1563–1568. [Google Scholar] [CrossRef]
- Wei, C.-H.; Laborie, S.; Aim, R.B.; Amy, G. Full utilization of silt density index (SDI) measurements for seawater pre-treatment. J. Membr. Sci. 2012, 405, 212–218. [Google Scholar] [CrossRef]
- López-Muñoz, M.J.; Sotto, A.; Arsuaga, J.M.; Van der Bruggen, B. Influence of membrane, solute and solution properties on the retention of phenolic compounds in aqueous solution by nanofiltration membranes. Sep. Purif. Technol. 2009, 66, 194–201. [Google Scholar] [CrossRef]
- Nitto, HYDRACoRe10 and 50 LD Series; Hydranautics Nitto Group Company: Oceanside, CA, USA, 2018.
- Nyström, M.; Kaipia, L.; Luque, S. Fouling and retention of nanofiltration membranes. J. Membr. Sci. 1995, 98, 249–262. [Google Scholar] [CrossRef]
- Tsuru, T.; Shutou, T.; Nakao, S.-I.; Kimura, S. Peptide and amino acid separation with nanofiltration membranes. Sep. Sci. Technol. 1994, 29, 971–984. [Google Scholar] [CrossRef]
- Daphne, L.H.X.; Utomo, H.D.; Kenneth, L.Z.H. Correlation between turbidity and total suspended solids in Singapore rivers. J. Water Sustain. 2011, 1, 313–322. [Google Scholar]
- Tittonell, P.; Rufino, M.C.; Janssen, B.H.; Giller, K.E. Carbon and nutrient losses during manure storage under traditional and improved practices in smallholder crop-livestock systems—Evidence from Kenya. Plant Soil 2010, 328, 253–269. [Google Scholar] [CrossRef] [Green Version]
- Chelme-Ayala, P.; El-Din, M.G.; Smith, R.; Code, K.R.; Leonard, J. Advanced treatment of liquid swine manure using physico-chemical treatment. J. Hazard. Mater. 2011, 186, 1632–1638. [Google Scholar] [CrossRef]
- Garzón-Zúñiga, M.A.; Lessard, P.; Aubry, G.; Buelna, G. Aeration effect on the efficiency of swine manure treatment in a trickling filter packed with organic materials. Water Sci. Technol. 2007, 55, 135–143. [Google Scholar] [CrossRef]
- Pérez-Sangrador, M.P.; León-Cófreces, M.C.; Acítores-Benavente, M.; García-González, M.C. Solids and nutrient removal from flushed swine manure using polyacrylamides. J. Environ. Manag. 2012, 93, 67–70. [Google Scholar] [CrossRef]
- Tao, W.; Ukwuani, A.T. Coupling thermal stripping and acid absorption for ammonia recovery from dairy manure: Ammonia volatilization kinetics and effects of temperature, pH and dissolved solids content. Chem. Eng. J. 2015, 280, 188–196. [Google Scholar] [CrossRef]
- Dourmad, J.-Y.; Jondreville, C. Impact of nutrition on nitrogen, phosphorus, Cu and Zn in pig manure, and on emissions of ammonia and odours. Livest. Sci. 2007, 112, 192–198. [Google Scholar] [CrossRef] [Green Version]
- Forge, T.; Kenney, E.; Hashimoto, N.; Neilsen, D.; Zebarth, B. Compost and poultry manure as preplant soil amendments for red raspberry: Comparative effects on root lesion nematodes, soil quality and risk of nitrate leaching. Agric. Ecosyst. Environ. 2016, 223, 48–58. [Google Scholar] [CrossRef]
- Zhang, Z.; Liu, D.; Qiao, Y.; Li, S.; Chen, Y.; Hu, C. Mitigation of carbon and nitrogen losses during pig manure composting: A meta-analysis. Sci. Total Environ. 2021, 783, 147103. [Google Scholar] [CrossRef] [PubMed]
- Zacharof, M.-P.; Lovitt, R. Recovery of volatile fatty acids (VFA) from complex waste effluents using membranes. Water Sci. Technol. 2014, 69, 495–503. [Google Scholar] [CrossRef] [PubMed]
- Gao, W.; Han, M.; Qu, X.; Xu, C.; Liao, B. Characteristics of wastewater and mixed liquor and their role in membrane fouling. Bioresour. Technol. 2013, 128, 207–214. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.-C.; Choi, W.J.; Chae, A.N.; Park, J.; Kim, H.J.; Song, K.G. Evaluating integrated strategies for robust treatment of high saline piggery wastewater. Water Res. 2016, 89, 222–231. [Google Scholar] [CrossRef]
- Zielińska, M.; Cydzik-Kwiatkowska, A.; Bułkowska, K.; Bernat, K.; Wojnowska-Baryła, I. Treatment of bisphenol A-containing effluents from aerobic granular sludge reactors with the use of microfiltration and ultrafiltration ceramic membranes. Water Air Soil Pollut. 2017, 228, 282. [Google Scholar] [CrossRef] [Green Version]
- Masse, L.; Massé, D.; Beaudette, V.; Muir, M. Size distribution and composition of particles in raw and anaerobically digested swine manure. Trans. ASAE 2005, 48, 1943–1949. [Google Scholar] [CrossRef]
- Christensen, M.L.; Hjorth, M.; Keiding, K. Characterization of pig slurry with reference to flocculation and separation. Water Res. 2009, 43, 773–783. [Google Scholar] [CrossRef]
- González-Fernández, C.; Nieto-Diez, P.P.; León-Cofreces, C.; García-Encina, P.A. Solids and nutrients removals from the liquid fraction of swine slurry through screening and flocculation treatment and influence of these processes on anaerobic biodegradability. Bioresour. Technol. 2008, 9, 6233–6239. [Google Scholar] [CrossRef] [PubMed]
- Mondor, M.; Masse, L.; Ippersiel, D.; Lamarche, F.; Masse, D. Use of electrodialysis and reverse osmosis for the recovery and concentration of ammonia from swine manure. Bioresour. Technol. 2008, 9, 7363–7368. [Google Scholar] [CrossRef] [PubMed]
- Poulsen, H.; Børsting, C.; Rom, H.; Sommer, S. Nitrogen, Phosphorus and Potassium in Animal Manure: Norm Values 2000; Ministry of Food, Agriculture and Fisheries, Danish Institute of Agricultural Sciences: Tjele, Denmark, 2001; p. 152. [Google Scholar]
- Li, L.-L.; Li, S.-T. Nitrogen mineralization from animal manures and its relation to organic N fractions. J. Integr. Agric. 2014, 13, 2040–2048. [Google Scholar] [CrossRef]
- Cheng, H.; Hong, P.-Y. Removal of antibiotic-resistant bacteria and antibiotic resistance genes affected by varying degrees of fouling on anaerobic microfiltration membranes. Environ. Sci. Technol. 2017, 51, 12200–12209. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kwarciak-Kozlowska, A.; Wlodarczyk, R. Treatment of waterborne pathogens by microfiltration. In Waterborne Pathogens; Elsevier: Amsterdam, The Netherlands, 2020; pp. 81–103. [Google Scholar]
- Ko, Y.W.; Chen, R.M. Ion rejection in single and binary mixed electrolyte systems by nanofiltration: Effect of feed concentration. Sep. Sci. Technol. 2007, 42, 3071–3084. [Google Scholar] [CrossRef]
- Cancino-Madariaga, B.; Hurtado, C.F.; Ruby, R. Effect of pressure and pH in ammonium retention for nanofiltration and reverse osmosis membranes to be used in recirculation aquaculture systems (RAS). Aquac. Eng. 2011, 4, 103–108. [Google Scholar] [CrossRef]
- Hurtado, C.F.; Cancino-Madariaga, B. Ammonia retention capacity of nanofiltration and reverse osmosis membranes in a non steady state system, to be use in recirculation aquaculture systems (RAS). Aquac. Eng. 2014, 58, 29–34. [Google Scholar] [CrossRef]
- Pratofiorito, G.; Horn, H.; Saravia, F. Impact of the Recovery on Concentrating Acetic Acid with Low-Pressure Reverse-Osmosis Membranes. Membranes 2021, 11, 742. [Google Scholar] [CrossRef]
- Bartels, C.; Franks, R.; Rybar, S.; Schierach, M.; Wilf, M. The effect of feed ionic strength on salt passage through reverse osmosis membranes. Desalination 2005, 184, 185–195. [Google Scholar] [CrossRef]
- Gerardo, M.L.; Aljohani, N.H.; Oatley-Radcliffe, D.L.; Lovitt, R.W. Moving towards sustainable resources: Recovery and fractionation of nutrients from dairy manure digestate using membranes. Water Res. 2015, 80, 80–89. [Google Scholar] [CrossRef]
- Ballet, G.T.; Hafiane, A.; Dhahbi, M. Influence of operating conditions on the retention of phosphate in water by nanofiltration. J. Membr. Sci. 2007, 290, 164–172. [Google Scholar] [CrossRef]
- Al-Zoubi, H.; Hilal, N.; Darwish, N.; Mohammad, A. Rejection and modelling of sulphate and potassium salts by nanofiltration membranes: Neural network and Spiegler–Kedem model. Desalination 2007, 206, 42–60. [Google Scholar] [CrossRef]
- Slipko, K.; Reif, D.; Woegerbauer, M.; Hufnagl, P.; Krampe, J.; Kreuzinger, N. Removal of extracellular free DNA and antibiotic resistance genes from water and wastewater by membranes ranging from microfiltration to reverse osmosis. Water Res. 2019, 164, 114916. [Google Scholar] [CrossRef] [PubMed]
- Lan, L.; Kong, X.; Sun, H.; Li, C.; Liu, D. High removal efficiency of antibiotic resistance genes in swine wastewater via nanofiltration and reverse osmosis processes. J. Environ. Manag. 2019, 231, 439–445. [Google Scholar] [CrossRef] [PubMed]
- Van der Bruggen, B.; Vandecasteele, C.; Van Gestel, T.; Doyen, W.; Leysen, R. A review of pressure-driven membrane processes in wastewater treatment and drinking water production. Environ. Prog. 2003, 22, 46–56. [Google Scholar] [CrossRef]
- Winter, J.; Barbeau, B.; Bérubé, P. Nanofiltration and Tight Ultrafiltration Membranes for Natural Organic Matter Removal—Contribution of Fouling and Concentration Polarization to Filtration Resistance. Membranes 2017, 7, 34. [Google Scholar] [CrossRef] [Green Version]



| NF Membrane | Supplier | Surface Layer | pH Tolerance | MWCO *” | Water Permeability |
|---|---|---|---|---|---|
| [Da] | [L h−1 m−2bar−1] | ||||
| NF270 | DuPont | Polyamide | 3 to 11 | 300 [23] | 13.5 [23] |
| HC50 | Nitto | SPES * | 3 to 11 | 1000 [24] | 7.5 [24] |
| NTR7450 | Nitto-Denko | SPES * | 3 to 11 | 2000–3000 [25,26] | 9.2 [23] |
| Parameters | Site 1 | Site 2 | Site 3 |
|---|---|---|---|
| TSS (g L−1) | 3 | 4.9 | 4.7 |
| VSS (% of TSS) | 83 | 78 | 78 |
| tCOD (g L−1) | 11.3 | 11.8 | 10.7 |
| NH4+-N (g L−1) | 4.4 | 2.9 | 4.4 |
| Tot-P (mg L−1) | 185 | 103 | 283 |
| pH | 7.8 | 7.8 | 7.9 |
| DOC (g L−1) | 3.3 | 3 | 2.2 |
| DTN (g L−1) | 3 | 2 | 2.5 |
| Dissolved-P (mg L−1) | 103 | 19.2 | 105 |
| Chloride (mg L−1) | 1745 | 1083 | 1674 |
| Sulphate (mg L−1) | 39 | 86 | 108 |
| Acetic acid (mg L−1) | 3637 | 2211 | 1317 |
| Calcium (mg L−1) | 156 | 138 | 175 |
| K (mg L−1) | 1793 | 1697 | 2737 |
| Sodium (mg L−1) | 895 | 409 | 850 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Samanta, P.; Schönettin, H.M.; Horn, H.; Saravia, F. MF–NF Treatment Train for Pig Manure: Nutrient Recovery and Reuse of Product Water. Membranes 2022, 12, 165. https://doi.org/10.3390/membranes12020165
Samanta P, Schönettin HM, Horn H, Saravia F. MF–NF Treatment Train for Pig Manure: Nutrient Recovery and Reuse of Product Water. Membranes. 2022; 12(2):165. https://doi.org/10.3390/membranes12020165
Chicago/Turabian StyleSamanta, Prantik, Hannah Marie Schönettin, Harald Horn, and Florencia Saravia. 2022. "MF–NF Treatment Train for Pig Manure: Nutrient Recovery and Reuse of Product Water" Membranes 12, no. 2: 165. https://doi.org/10.3390/membranes12020165
APA StyleSamanta, P., Schönettin, H. M., Horn, H., & Saravia, F. (2022). MF–NF Treatment Train for Pig Manure: Nutrient Recovery and Reuse of Product Water. Membranes, 12(2), 165. https://doi.org/10.3390/membranes12020165






