Conversion of Waste Oil from Oil Refinery into Emulsion Liquid Membrane for Removal of Phenol: Stability Evaluation, Modeling and Optimization
Abstract
:1. Introduction
2. Materials and Methods
2.1. Experimental Procedure
2.2. RSM Model Development and Analysis
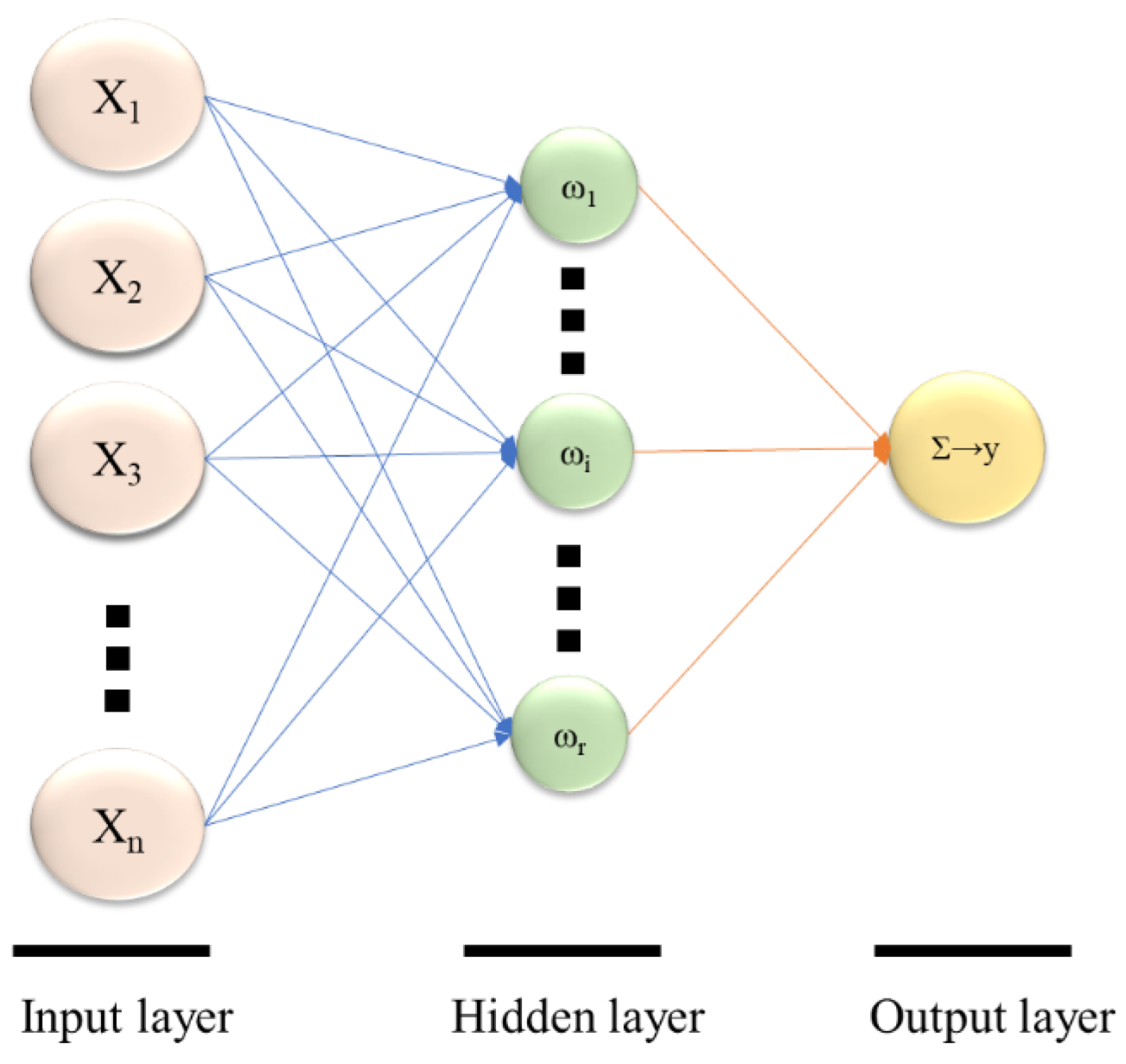
2.3. ANN Model Design and Analysis
3. Results and Discussion
3.1. RSM Modeling and Forecasting Performance Evaluating
3.2. Effects of Different Variables on the Stability of WELM
3.3. ANN Modeling and Assessment
4. Comparison of RSM and ANN Models
- Before establishing RSM model, experiments must be designed and analyzed to obtain the model. The ANN model can be established based on the data that is already available.
- The interaction effects and degrees of multiple variable factors can be obtained through the establishment of the RSM model. However, the establishment of the ANN model is a “black box” model establishment process, and it is difficult to compare and analyze the effect of variables on the results intuitively.
- The RSM model can be obtained with fewer experimental data. The predictability of the model established by the ANN method will be significantly improved when the input experimental data increased.
5. Conclusions
- The concentration of Span-80, liquid paraffin, internal-oil ratio, and the rotational speed of homogenizer have significant effect on the demulsification rate of the WELM.
- The removal rate of phenol from purified water of sour water stripper by WELM were higher than 85%.
- The 3D response surface figures of the variables interactive effect on the demulsification rate were obtained by response surface analysis, and combined with the p value in the analysis of variance table, it can be found that the interaction effect between the concentration of Span-80 and the rotational speed of homogenizer, the concentration of liquid paraffin and the internal-oil ratio on demulsification rate are significant, and other interaction effects are negligible.
- The RSM and ANN models have predictability for the demulsification rate of the ELM, and the RSM model is better. Under the optimal conditions, the demulsification rate is only 0.481%, which shows high stability and provides the best conditions for subsequent WELM recycling.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Qi, Y.B.; Luo, C.; Zhang, S.J. Research progress on dephenolization technologies of wastewater containing phenolic compounds by liquid membrane. Chem. Ind. Eng. Prog. 2021, 40, 348–355. [Google Scholar]
- Ng, Y.S.; Jayakumar, N.S.; Hashim, M.A. Performance evaluation of organic emulsion liquid membrane on phenol removal. J. Hazard. Mater. 2010, 184, 255–260. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yan, B.-C.; Zhang, Y.-Y.; Jiao, X.-S.; Chen, K.; Huang, X.-J.; Yin, J.-C.; Wang, L. Advances in research on structural characteristics and stability of emulsion membranes. Appl. Chem. Ind. 2020, 49, 1231–1235. [Google Scholar]
- Ahmad, A.L.; Kusumastuti, A.; Derek, C.; Ooi, B.S. Emulsion liquid membrane for cadmium removal: Studies on emulsion diameter and stability. Desalination 2012, 287, 30–34. [Google Scholar] [CrossRef]
- Sulaiman, R.; Othman, N.; Amin, N.; Othman, N.; Amin, N. Emulsion liquid membrane stability in the extraction of ionized nanosilver from wash water. J. Ind. Eng. Chem. 2014, 20, 3243–3250. [Google Scholar] [CrossRef]
- Abbassian, K.; Kargari, A. Effect of polymer addition to membrane phase to improve the stability of emulsion liquid membrane for phenol pertraction. Desalin. Water Treat. 2014, 57, 2942–2951. [Google Scholar] [CrossRef]
- Sujatha, S.; Rajamohan, N.; Vasseghian, Y.; Rajasimman, M. Conversion of waste cooking oil into value-added emulsion liquid membrane for enhanced extraction of lead: Performance evaluation and optimization. Chemosphere 2021, 284, 131385. [Google Scholar] [CrossRef]
- Othman, N.; Shu, L.Y.; Ooi, Z.Y.; Jusoh, N.; Idroas, M.; Goto, M. Easy removing of phenol from wastewater using vegetable oil-based organic solvent in emulsion liquid membrane process. Chin. J. Chem. Eng. 2017, 25, 45–52. [Google Scholar] [CrossRef]
- Tahmasebizadeh, P.; Javanshir, S.; Ahmadi, A. Zinc extraction from a bioleaching solution by emulsion liquid membrane technique. Sep. Purif. Technol. 2021, 276, 119394. [Google Scholar] [CrossRef]
- Arun, V.; Jalaludeen, S.S.; Jayakumar, S.; Swaminathan, S. Effect of contacting pattern and various surfactants on phenol extraction efficiency using emulsion liquid membrane. Int. J. Chem. React. Eng. 2021, 19, 739–747. [Google Scholar] [CrossRef]
- Djenouhat, M.; Hamdaoui, O.; Chiha, M.; Samar, M.H. Ultrasonication-assisted preparation of water-in-oil emulsions and application to the removal of cationic dyes from water by emulsion liquid membrane. Sep. Purif. Technol. 2008, 63, 231–238. [Google Scholar] [CrossRef]
- Mortaheb, H.R.; Amini, M.H.; Sadeghian, F.; Mokhtarani, B.; Daneshyar, H. Study on a new surfactant for removal of phenol from wastewater by emulsion liquid membrane. J. Hazard. Mater. 2008, 160, 582–588. [Google Scholar] [CrossRef] [PubMed]
- Salman, H.M.; Mohammed, A.A. Extraction of lead ions from aqueous solution by co-stabilization mechanisms of magnetic Fe2O3 particles and nonionic surfactants in emulsion liquid membrane. Colloids Surf. A Physicochem. Eng. Asp. 2019, 568, 301–310. [Google Scholar] [CrossRef]
- Kumar, A.; Avinash, T.; Panesar, P.S. Recent developments on sustainable solvents for emulsion liquid membrane processes. J. Clean. Prod. 2019, 240, 118250. [Google Scholar] [CrossRef]
- Shokri, A.; Daraei, P.; Zereshki, S. Water decolorization using waste cooking oil: An optimized green emulsion liquid membrane by RSM. J. Water Process Eng. 2020, 33, 101021. [Google Scholar] [CrossRef]
- Mohammadi, F.; Samaei, M.R.; Azhdarpoor, A.; Teiri, H.; Badeenezhad, A.; Rostami, S. Modelling and optimizing pyrene removal from the soil by phytoremediation using response surface methodology, artificial neural networks, and genetic algorithm. Chemosphere 2019, 237, 124486. [Google Scholar] [CrossRef] [PubMed]
- Daas, A.; Hamdaoui, O. Extraction of bisphenol A from aqueous solutions by emulsion liquid membrane. J. Hazard. Mater. 2010, 348, 360–368. [Google Scholar] [CrossRef]
- Mohammed, S.A.M.; Zouli, N.; Al-Dahhan, M.M.; Zouli, N.; Al-Dahhan, M. Removal of phenolic compounds from synthesized produced water by emulsion liquid membrane stabilized by the combination of surfactant and ionic liquid. Desalin. Water Treat. 2018, 110, 168–179. [Google Scholar] [CrossRef]
- Lin, Z.; Zhang, Z.; Li, Y.; Deng, Y. Magnetic nano-Fe3O4 stabilized Pickering emulsion liquid membrane for selective extraction and separation. Chem. Eng. J. 2016, 288, 305–311. [Google Scholar] [CrossRef]
- Reis, M.T.A.; Freitas, O.M.; Agarwal, S.; Ferreira, L.M.; Ismael, M.R.C.; Machado, R.; Carvalho, J.M. Removal of phenols from aqueous solutions by emulsion liquid membranes. J. Hazard. Mater. 2011, 192, 986–994. [Google Scholar] [CrossRef]
- Hussein, M.A.; Mohammed, A.A.; Atiya, M.A.; Mohammed, A.A.; Atiya, M.A. Application of emulsion and Pickering emulsion liquid membrane technique for wastewater treatment: An overview. Environ. Sci. Pollut. Res. 2019, 26, 36184–36204. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Thakur, A.; Panesar, P.S. A review on emulsion liquid membrane (ELM) for the treatment of various industrial effluent streams. Rev. Environ. Sci. Biotechnol. 2019, 18, 153–182. [Google Scholar] [CrossRef]
- Bensalah, N.; Midassi, S.; Ahmad, M.I.; Bedoui, A. Degradation of hydroxychloroquine by electrochemical advanced oxidation processes. Chem. Eng. J. 2020, 402, 126279. [Google Scholar] [CrossRef] [PubMed]
- Jusoh, N.; Othman, N.; Nasruddin, N.A. Emulsion liquid membrane technology in organic acid purification. Malays. Soc. Anal. Sci. 2016, 20, 436–443. [Google Scholar] [CrossRef]
- Kohli, H.P.; Gupta, S.; Chakraborty, M. Extraction of Ethylparaben by emulsion liquid membrane: Statistical analysis of operating parameters. Colloids Surf. A Physicochem. Eng. Asp. 2018, 539, 371–381. [Google Scholar] [CrossRef]
- Mohamed Noah, N.F.; Jusoh, N.; Othman, N.; Raja Sulaiman, R.N.; Parker, N.A.M.K. Development of stable green emulsion liquid membrane process via liquid–liquid extraction to treat real chromium from rinse electroplating wastewater. J. Ind. Eng. Chem. 2018, 66, 231–241. [Google Scholar] [CrossRef]
- Kumar, A.; Thakur, A.; Panesar, P.S. A comparative study on experimental and response surface optimization of lactic acid synergistic extraction using green emulsion liquid membrane. Sep. Purif. Technol. 2019, 211, 54–62. [Google Scholar] [CrossRef]
- Raval, A.R.; Kohli, H.P.; Mahadwad, O.K. A Comprehensive Review on Green Emulsion Liquid Membrane and Its Applicability Towards the Removal of Contaminants from the Aquatic Streams. Water Air Soil Pollut. 2022, 9, 233. [Google Scholar] [CrossRef]
- Ahmad, A.L.; Zaulkiflee, N.D.; Kusumastuti, A.; Shah Buddin, M.M.H. Removal of Acetaminophen from Aqueous Solution by Emulsion Liquid Membrane: Emulsion Stability Study. Ind. Eng. Chem. Res. 2019, 58, 713–719. [Google Scholar] [CrossRef]
- Al-Obaidi, O.; Alabdulmuhsin, M.; Tolstik, A.; Trautman, J.G.; Al-Dahhan, M. Removal of hydrocarbons of 4-Nitrophenol by emulsion liquid membrane (ELM) using magnetic Fe2O3 nanoparticles and ionic liquid. J. Water Process Eng. 2021, 39, 101729. [Google Scholar] [CrossRef]
- Zeng, L.; Liu, Y.; Yang, T.; Yang, Y.; Tang, K. Simultaneously enhanced ELM selectivity and stability by difunctional additives for batch and continuous separation of Cd(II)/Cu(II). Chem. Eng. Res. Des. 2018, 140, 261–272. [Google Scholar] [CrossRef]






| Properties | Water Content (%) | Flash Point (°C) | Ignition Temperature (°C) | Condensation Point (°C) | Mechanical Impurities (%) |
|---|---|---|---|---|---|
| Value | 0.76 | 94 | 98 | 18 | 0.47 |
| Variables | Code | Levels | ||
|---|---|---|---|---|
| −1 | 0 | 1 | ||
| Span-80 (wt.%) | Z1 | 1 | 5.5 | 10 |
| liquid paraffin ((%(v/v)) | Z2 | 3 | 9 | 15 |
| internal-oil ratio | Z3 | 2 | 6 | 10 |
| rotational speed of homogenizer (rpm) | Z4 | 1000 | 5500 | 10,000 |
| Source | Sum of Squares | f | Mean Square | F-Value | p-Value |
|---|---|---|---|---|---|
| Model | 69.28 | 4 | 4.95 | 201.69 | <0.0001 |
| A-span80 | 22.56 | 1 | 22.56 | 919.45 | <0.0001 |
| B-liquid paraffin | 0.7201 | 1 | 0.7201 | 29.35 | <0.0001 |
| C-internal-oil ratio | 17.05 | 1 | 17.05 | 695.08 | <0.0001 |
| D-speed | 4.81 | 1 | 4.81 | 196.18 | <0.0001 |
| AB | 0.0613 | 1 | 0.0613 | 2.50 | 0.1361 |
| AC | 0.0005 | 1 | 0.0005 | 0.0220 | 0.8842 |
| AD | 0.9652 | 1 | 0.9652 | 39.34 | <0.0001 |
| BC | 0.9196 | 1 | 0.9196 | 37.48 | <0.0001 |
| BD | 0.0507 | 1 | 0.0507 | 2.06 | 0.1727 |
| CD | 0.0249 | 1 | 0.0249 | 1.01 | 0.3311 |
| A2 | 12.56 | 1 | 12.56 | 511.81 | <0.0001 |
| B2 | 4.20 | 1 | 4.20 | 171.36 | <0.0001 |
| C2 | 0.0009 | 1 | 0.0009 | 0.0373 | 0.8496 |
| D2 | 10.53 | 1 | 10.53 | 429.26 | <0.0001 |
| Residual | 0.3435 | 1 | 0.0245 | - | - |
| Lack of Fit | 0.3148 | 0 | 0.0315 | 4.40 | 0.0832 |
| Pure Error | 0.0286 | 4 | 0.0072 | - | - |
| Cor Total | 69.62 | 8 | - | - | - |
| Number of Hidden Layer Neurons | MSE (The Best Validation Performance) | R (All Data) |
|---|---|---|
| 4 | 0.0252 | 0.97041 |
| 5 | 0.0000846 | 0.96366 |
| 6 | 0.0000580 | 0.98383 |
| 7 | 0.0000632 | 0.98697 |
| 8 | 0.0000632 | 0.97247 |
| 9 | 0.0000845 | 0.9765 |
| 10 | 0.0000309 | 0.9941 |
| Operation | A | B | C | D |
|---|---|---|---|---|
| Span-80 (wt%) | Liquid Paraffin (%v/v) | Internal-Oil Ratio | Rotational Speed of Homogenizer (rpm) | |
| 1 | 5.5 | 9 | 10 | 1000 |
| 2 | 5.5 | 9 | 6 | 5500 |
| 3 | 5.5 | 9 | 6 | 5500 |
| 4 | 1 | 15 | 6 | 5500 |
| 5 | 5.5 | 3 | 6 | 1000 |
| 6 | 5.5 | 15 | 2 | 5500 |
| 7 | 5.5 | 15 | 6 | 10,000 |
| 8 | 5.5 | 9 | 6 | 5500 |
| 9 | 5.5 | 9 | 10 | 10,000 |
| 10 | 5.5 | 3 | 6 | 10,000 |
| 11 | 5.5 | 9 | 2 | 10,000 |
| 12 | 5.5 | 15 | 10 | 5500 |
| 13 | 1 | 9 | 10 | 5500 |
| 14 | 5.5 | 9 | 2 | 1000 |
| 15 | 1 | 9 | 6 | 10,000 |
| 16 | 1 | 9 | 6 | 1000 |
| 17 | 10 | 9 | 2 | 5500 |
| 18 | 10 | 15 | 6 | 5500 |
| 19 | 10 | 9 | 6 | 1000 |
| 20 | 5.5 | 9 | 6 | 5500 |
| 21 | 5.5 | 9 | 6 | 5500 |
| 22 | 10 | 3 | 6 | 5500 |
| 23 | 10 | 9 | 6 | 10,000 |
| 24 | 5.5 | 15 | 6 | 1000 |
| 25 | 1 | 3 | 6 | 5500 |
| 26 | 5.5 | 3 | 2 | 5500 |
| 27 | 5.5 | 3 | 10 | 5500 |
| 28 | 1 | 9 | 2 | 5500 |
| 29 | 10 | 9 | 10 | 5500 |
| Model | RSM | ANN |
|---|---|---|
| R2 | 0.9951 | 0.9882 |
| RMSE | 0.1050 | 0.1694 |
| MAE | 0.08178 | 0.06847 |
| Model | S(%) | Error(%) |
|---|---|---|
| RSM | 0.536 | 11.4% |
| ANN | 0.545 | 13.3% |
| Experimental value | 0.481 | - |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, D.; Wang, Q.; Zhang, X.; Liu, T.; Zhang, H. Conversion of Waste Oil from Oil Refinery into Emulsion Liquid Membrane for Removal of Phenol: Stability Evaluation, Modeling and Optimization. Membranes 2022, 12, 1202. https://doi.org/10.3390/membranes12121202
Wang D, Wang Q, Zhang X, Liu T, Zhang H. Conversion of Waste Oil from Oil Refinery into Emulsion Liquid Membrane for Removal of Phenol: Stability Evaluation, Modeling and Optimization. Membranes. 2022; 12(12):1202. https://doi.org/10.3390/membranes12121202
Chicago/Turabian StyleWang, Dan, Qingji Wang, Xiaofei Zhang, Taoran Liu, and Hua Zhang. 2022. "Conversion of Waste Oil from Oil Refinery into Emulsion Liquid Membrane for Removal of Phenol: Stability Evaluation, Modeling and Optimization" Membranes 12, no. 12: 1202. https://doi.org/10.3390/membranes12121202
APA StyleWang, D., Wang, Q., Zhang, X., Liu, T., & Zhang, H. (2022). Conversion of Waste Oil from Oil Refinery into Emulsion Liquid Membrane for Removal of Phenol: Stability Evaluation, Modeling and Optimization. Membranes, 12(12), 1202. https://doi.org/10.3390/membranes12121202





