Separation of Soluble Benzene from an Aqueous Solution by Pervaporation Using a Commercial Polydimethylsiloxane Membrane
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Membrane Characterizations
2.2.1. Contact Angle
2.2.2. Scanning Electron Microscopy (SEM)
2.2.3. Fourier-Transform Infrared Spectroscopy (FTIR)
2.2.4. Energy-Dispersive Spectroscopy (EDS)
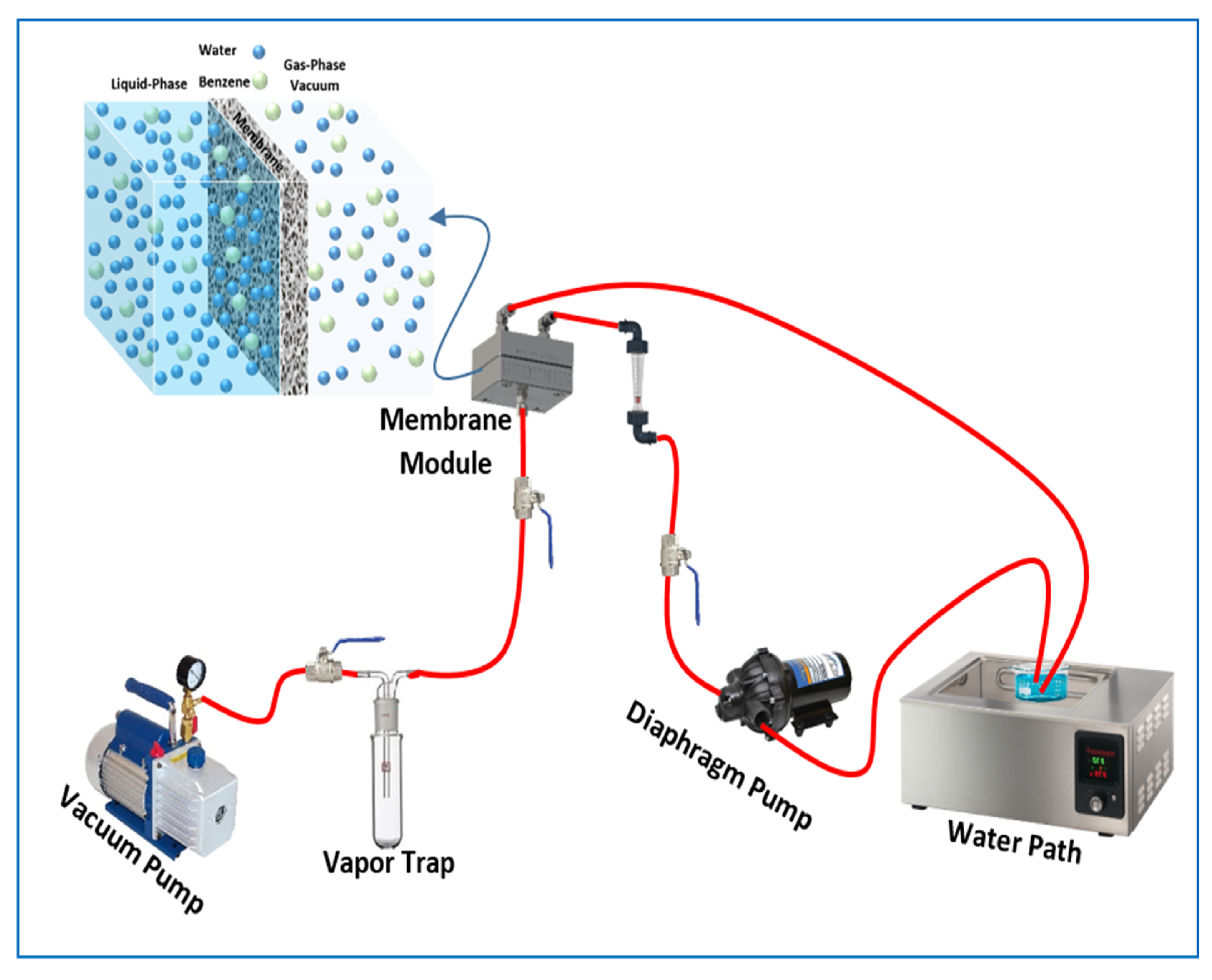
2.3. Pervaporation Process
2.4. Experimental Design
3. Results and Discussion
3.1. Membrane Characterizations
3.1.1. Contact Angle
3.1.2. SEM Analysis
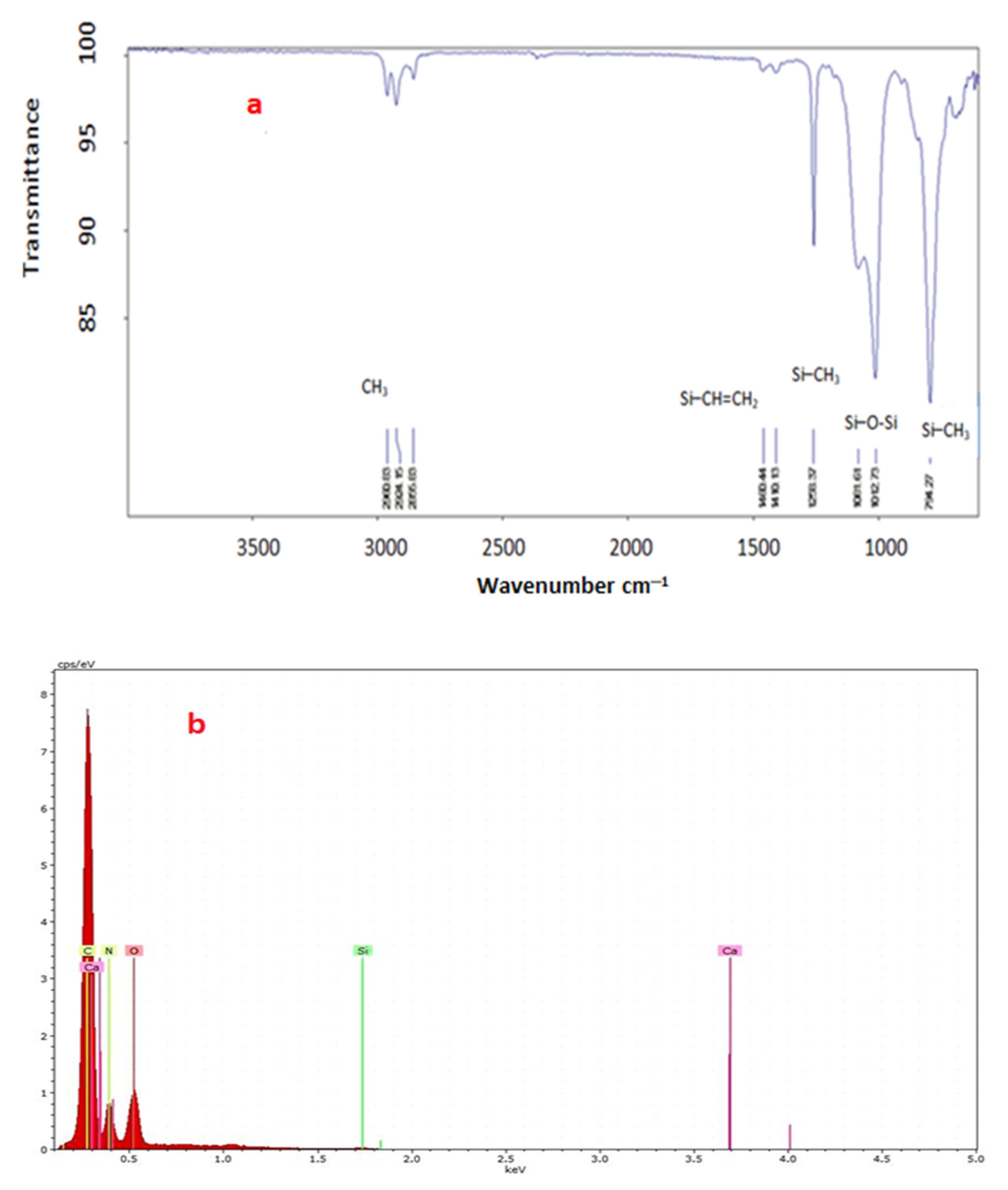
3.1.3. FTIR
3.1.4. Energy-Dispersive Spectroscopy (EDS)
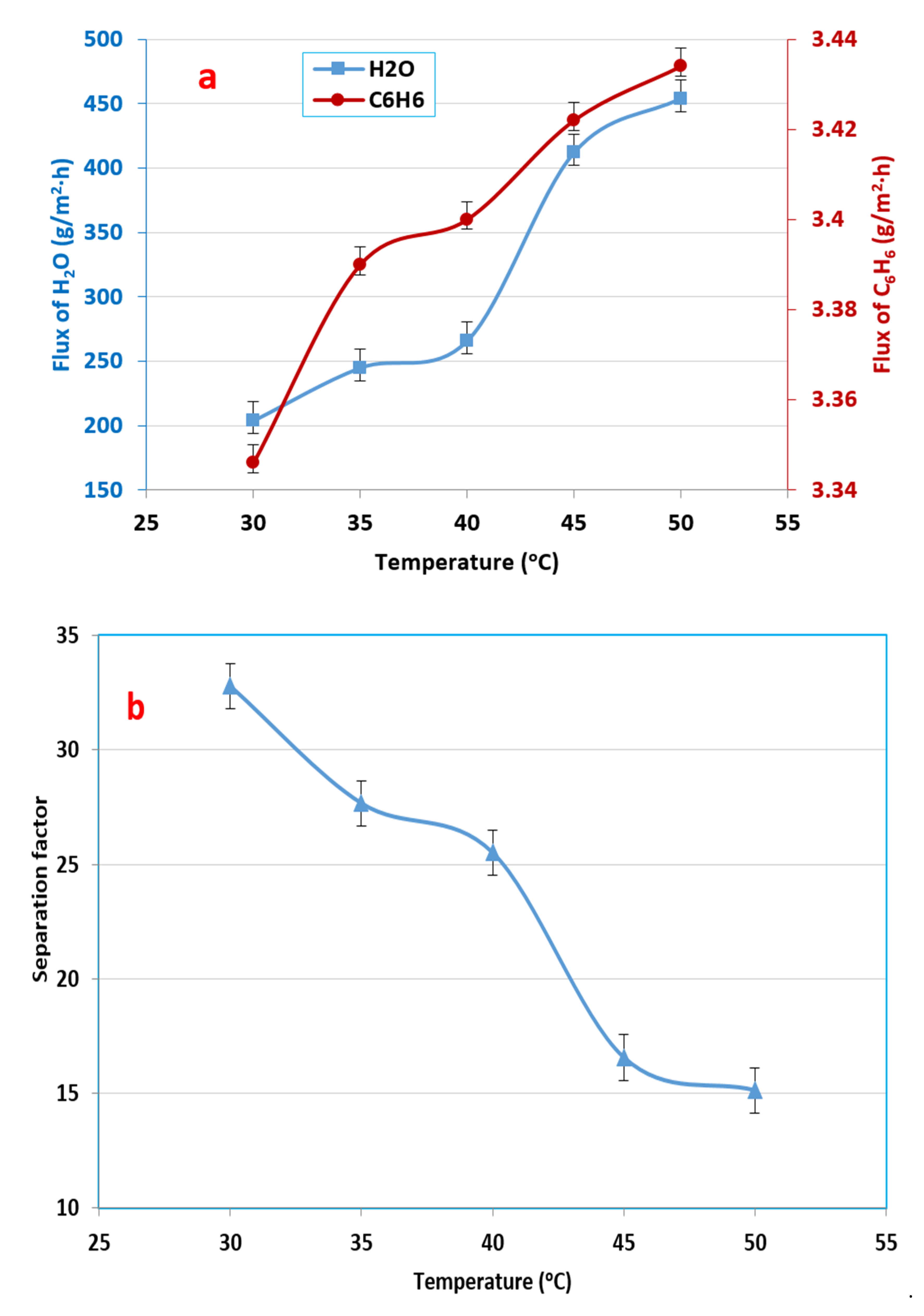
3.2. Influence of Feed Temperature in Benzene–Water Mixture
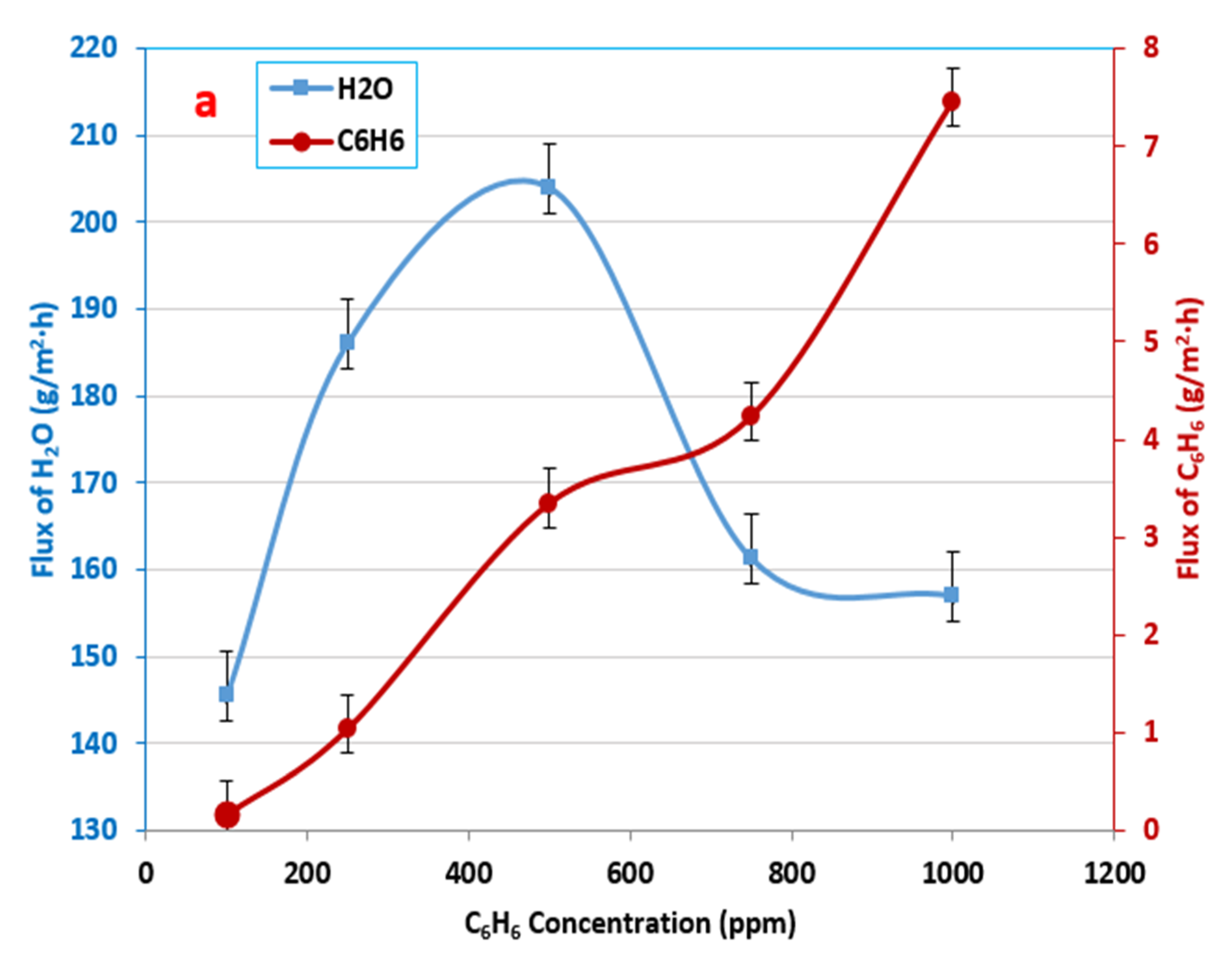
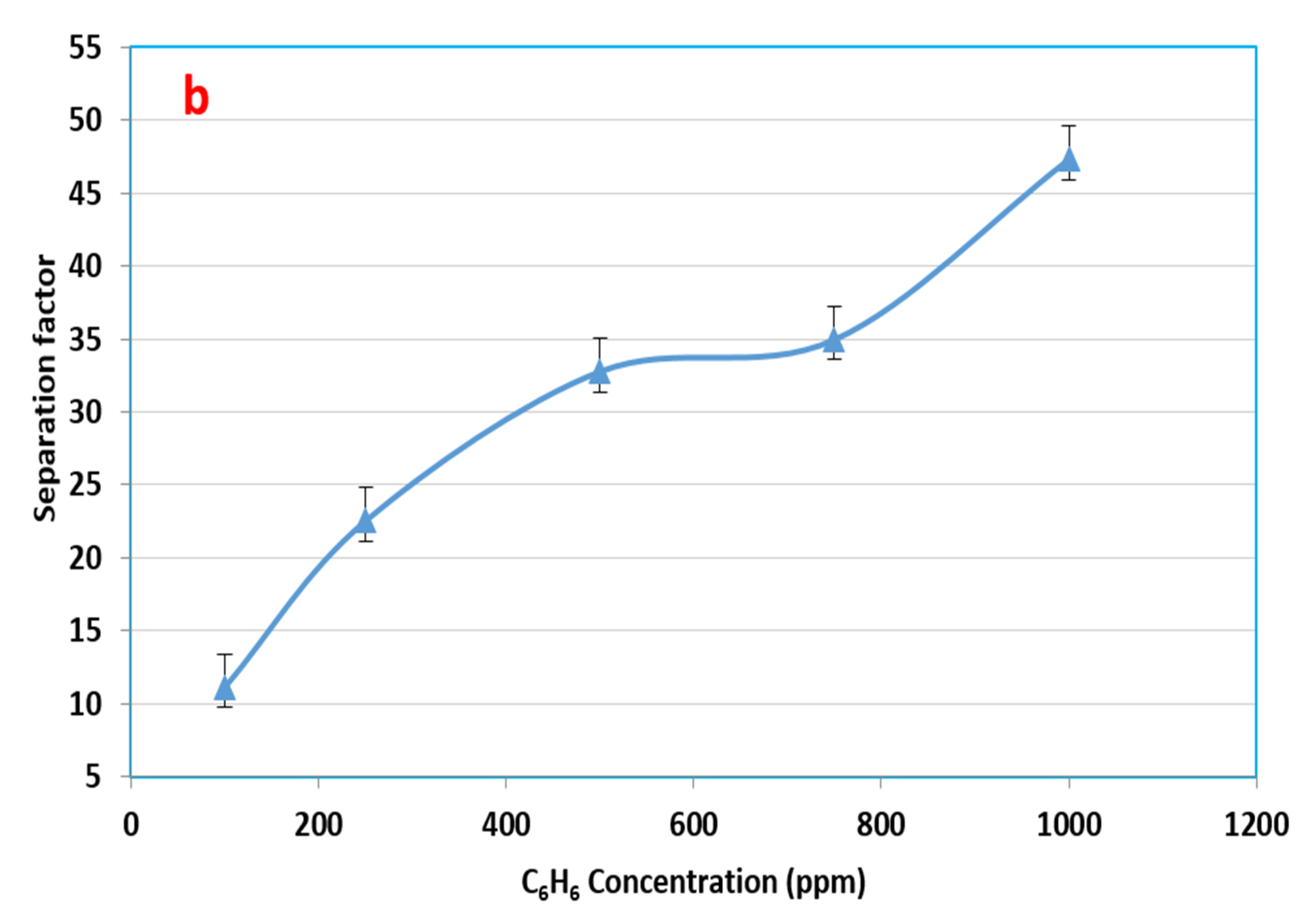
3.3. Influence of the Initial Feed Concentration in Benzene–Water Solution
3.4. Effect of Feed Flow Rate on the Benzene Flux
3.5. Results of the RSM
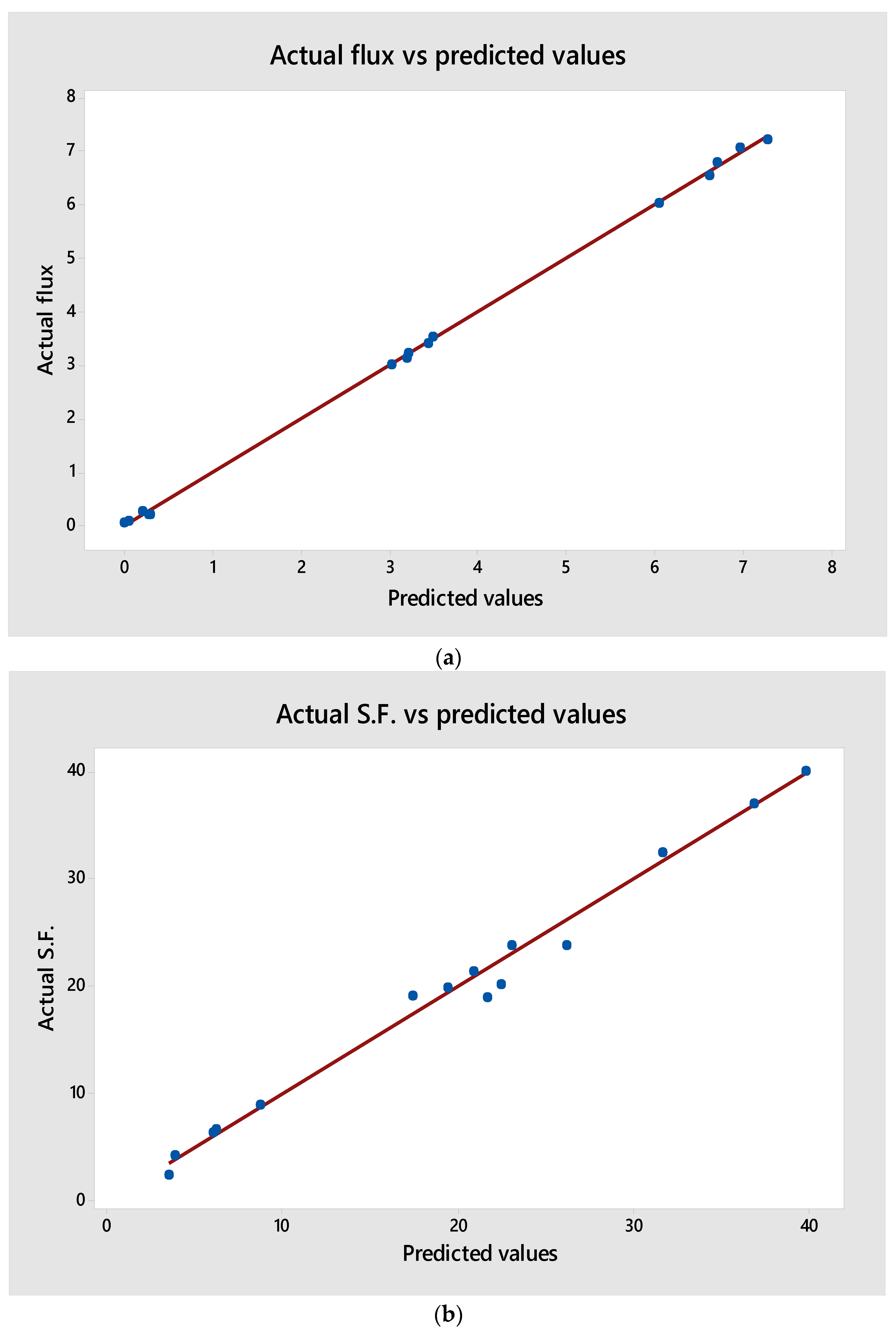
3.5.1. Predicted Model and ANOVA Calculations
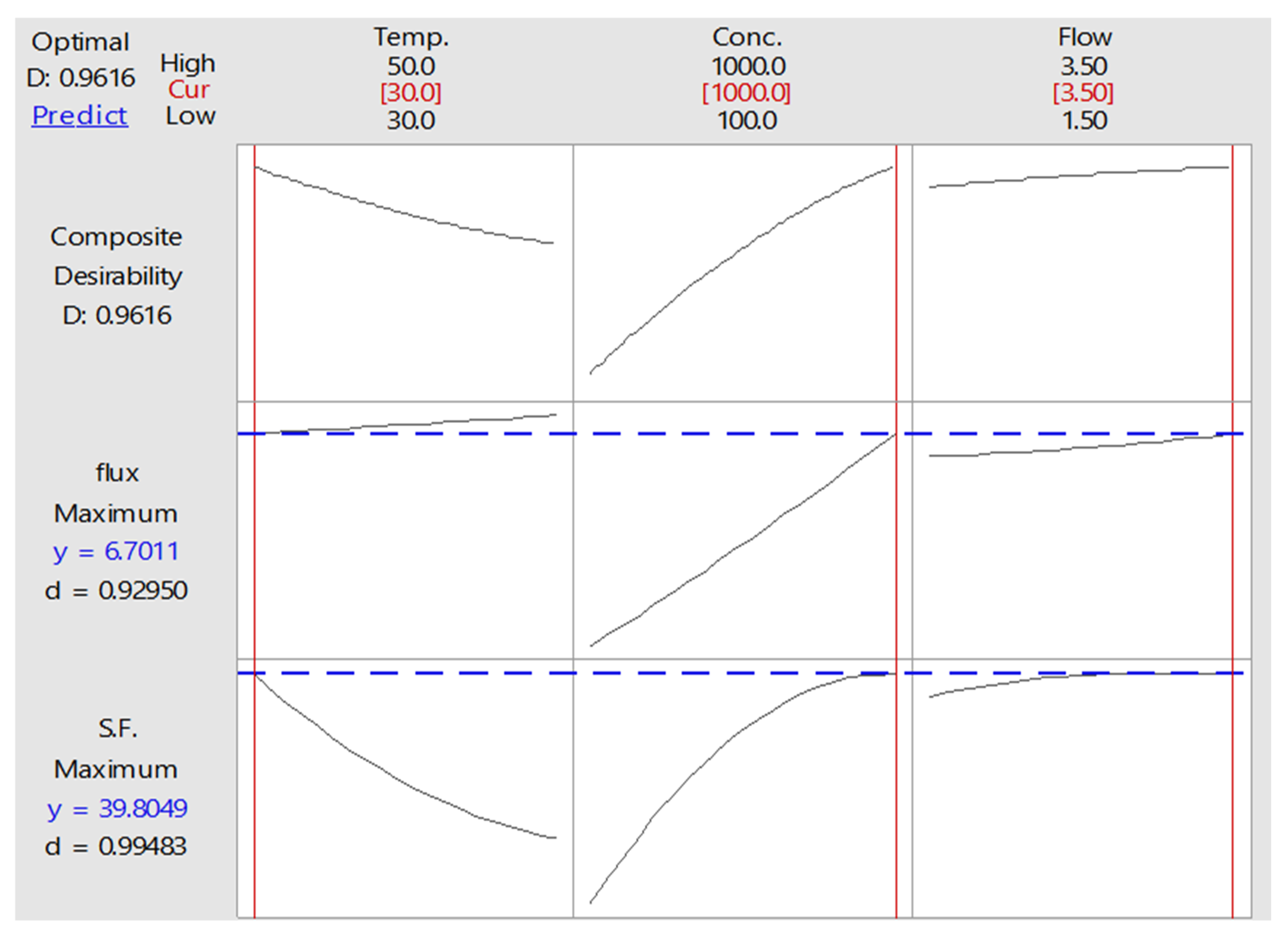
3.5.2. Optimization of PV Process
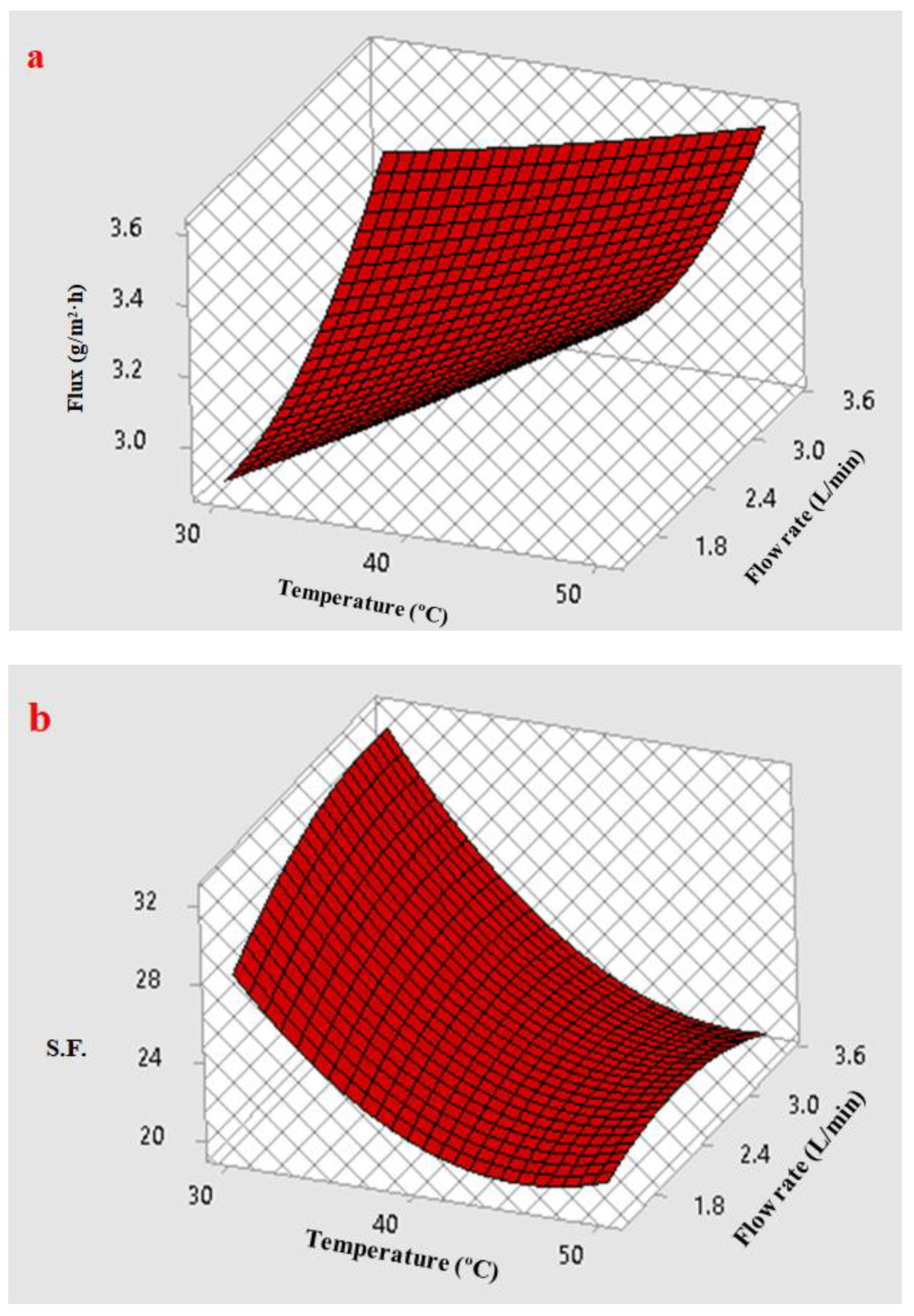
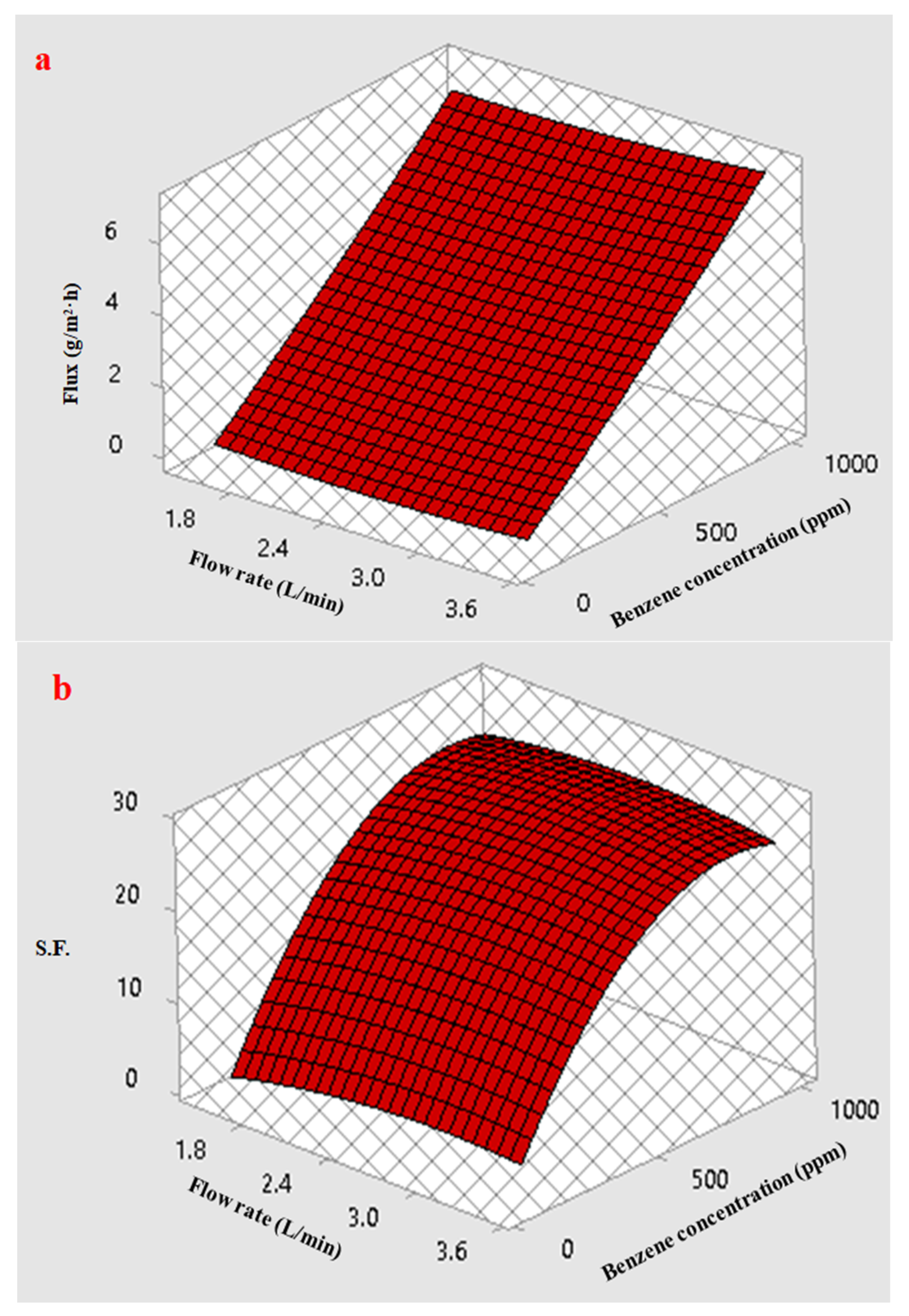
3.5.3. Response Surface Plots of Multiple Effects
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Maddah, H.; Chogle, A. Biofouling in reverse osmosis: Phenomena, monitoring, controlling and remediation. Appl. Water Sci. 2017, 7, 2637–2651. [Google Scholar] [CrossRef]
- Kujawa, J.; Cerneaux, S.; Kujawski, W. Removal of hazardous volatile organic compounds from water by vacuum pervaporation with hydrophobic ceramic membranes. J. Membr. Sci. 2015, 474, 11–19. [Google Scholar] [CrossRef]
- Hömmerich, U.; Rautenbach, R. Design and optimization of combined pervaporation/distillation processes for the production of MTBE. J. Membr. Sci. 1998, 146, 53–64. [Google Scholar] [CrossRef]
- Sampranpiboon, P.; Jiraratananon, R.; Uttapap, D.; Feng, X.; Huang, R.Y.M. Pervaporation separation of ethyl butyrate and isopropanol with polyether block amide (PEBA) membranes. J. Membr. Sci. 2000, 173, 53–59. [Google Scholar] [CrossRef]
- Yoshida, W.; Cohen, Y. Removal of methyltert-butylether from water by pervaporation using ceramic-supported polymer membranes. J. Membr. Sci. 2004, 229, 27–32. [Google Scholar] [CrossRef]
- Zadaka-Amir, D.; Nasser, A.; Nir, S.; Mishael, Y.G. Removal of methyl tertiary-butyl ether (MTBE) from water by polymer-zeolite composites. Microporous Mesoporous Mater. 2012, 151, 216–222. [Google Scholar] [CrossRef]
- Zhou, K.; Zhang, Q.G.; Han, G.L.; Zhu, A.M.; Liu, Q.L. Pervaporation of water– ethanol and methanol–MTBE mixtures using poly(vinylalcohol)/cellulose acetate blended membranes. J. Membr. Sci. 2013, 448, 93–101. [Google Scholar] [CrossRef]
- Levchuk, I.; Bhatnagar, A.; Sillanpää, M. Overview of technologies for removal of methyl tert-butyl ether (MTBE) from water. Sci. Total Environ. 2014, 476–477, 415–433. [Google Scholar] [CrossRef]
- Rutkiewicz, I.; Kujawski, W.; Namiesnik, J. Pervaporation of volatile organohalogen compounds through polydimethylsiloxane membrane. Desalination 2010, 264, 160–164. [Google Scholar] [CrossRef]
- Perego, C.; Bagatin, R.; Tagliabue, M.; Vignola, R. Zeolites and related mesoporous materials for multi-talented environmental solutions. Microporous Mesoporous Mater. 2013, 166, 37–49. [Google Scholar] [CrossRef]
- Xia, S.; Dong, X.; Zhu, Y.; Wei, W.; Xiangli, F.; Jin, W. Dehydration of ethyl acetate-water mixtures using PVA/ceramic composite pervaporation membrane. Sep. Purif. Technol. 2011, 77, 53–59. [Google Scholar] [CrossRef]
- Peng, M.; Vane, L.M.; Liu, S.X. Recent advances in VOCs removal from water by pervaporation. J. Hazard. Mater. 2003, 98, 69–90. [Google Scholar] [CrossRef]
- Ortiz, I.; Urtiaga, A.; Ibáñez, R.; Gómez, P.; Gorri, D. Laboratory- and pilot plant-scale study on the dehydration of cyclohexane by pervaporation. J. Chem. Technol. Biotechnol. 2006, 81, 48–57. [Google Scholar] [CrossRef]
- Sadrzadeh, M.; Rezakazemi, M.; Mohammadi, T. Fundamentals and Measurement Techniques for Gas Transport in Polymers; Elsevier Inc.: Amsterdam, The Netherlands, 2018; ISBN 9780128098851. [Google Scholar]
- Shahverdi, M.; Baheri, B.; Rezakazemi, M.; Motaee, E.; Mohammadi, T. Pervaporation study of ethylene glycol dehydration through synthesized (PVA–4A)/polypropylene mixed matrix composite membranes. Polym. Eng. Sci. 2013, 53, 1487–1493. [Google Scholar] [CrossRef]
- Pearce, E.M. Kirk-Othmer encyclopedia of chemical technology, 3rd ed., Vol. I, Wiley-Interscience, New York, 1978. J. Polym. Sci. Part C Polym. Lett. 1978, 16, 248. [Google Scholar] [CrossRef]
- Malakar, S.; Saha, P.D. Estimation of VOC Emission in Petroleum Refinery ETP and Comparative Analysis with Measured VOC Emission Rate. Int. J. Eng. Sci. 2015, 4, 20–29. [Google Scholar]
- Maddah, H.A. Optimal operating conditions in designing photocatalytic reactor for removal of phenol from wastewater. ARPN J. Eng. Appl. Sci. 2016, 11, 1799–1802. [Google Scholar]
- Farhan, N.M.; Ibrahim, S.S.; Leva, L.; Yave, W.; Alsalhy, Q.F. The combination of a new PERVAPTM membrane and molecular sieves enhances the ethanol drying process. Chem. Eng. Process. Process Intensif. 2022, 174, 108863. [Google Scholar] [CrossRef]
- He, X.; Wang, T.; Huang, J.; Chen, J.; Li, J. Fabrication and characterization of superhydrophobic PDMS composite membranes for efficient ethanol recovery via pervaporation. Sep. Purif. Technol. 2020, 241, 116675. [Google Scholar] [CrossRef]
- Hamouni, S.; Arous, O.; Abdessemed, D.; Nezzal, G.; Bruggen, B. Van Der Alcohol and Alkane Organic Extraction Using Pervaporation Process. In Macromolecular Symposia; Wiley: Hoboken, NJ, USA, 2019. [Google Scholar] [CrossRef]
- Unlu, D. Concentration of aroma compounds by pervaporation process using polyvinyl chloride membrane. Flavour Fragr. J. 2019, 34, 493–505. [Google Scholar] [CrossRef]
- Kujawska, A.; Knozowska, K.; Kujawa, J.; Li, G.; Kujawski, W. Fabrication of PDMS based membranes with improved separation efficiency in hydrophobic pervaporation. Sep. Purif. Technol. 2020, 234, 116092. [Google Scholar] [CrossRef]
- Peng, P.; Lan, Y. Modified Silica Incorporating into PDMS Polymeric Membranes for Bioethanol Selection. Adv. Polym. Technol. 2019, 2019, 5610282. [Google Scholar] [CrossRef]
- Wang, Y.M.; Ma, X.; Xue, T.; Wu, C.; Ji, M.; Li, M.Y. Green recovery of hazardous acetonitrile from high-salt chemical wastewater by pervaporation. J. Clean. Prod. 2018, 197, 742–749. [Google Scholar] [CrossRef]
- Ye, H.; Yan, X.; Zhang, X.; Song, W. Pervaporation properties of oleyl alcohol - filled polydimethylsiloxane membranes for the recovery of phenol from wastewater. Iran. Polym. J. 2017, 26, 639–649. [Google Scholar] [CrossRef]
- Wu, Y.; Tan, H.; Li, D.; Jin, Y. Pervaporation of Aqueous Solution of Acetaldehyde Through ZSM-5 Filled PDMS Composite Membrane. Chin. J. Chem. Eng. 2012, 20, 625–632. [Google Scholar] [CrossRef]
- Aliabadi, M.; Aroujalian, A.; Raisi, A. Removal of styrene from petrochemical wastewater using pervaporation process. DES 2012, 284, 116–121. [Google Scholar] [CrossRef]
- Hilmioglu, N.D.; Yildirim, A.E.; Tulbentci, S. A Pervaporation Application for Treating Methyl tert-Butyl Ether (MTBE)-Contaminated Water/Wastewater. In Global Warming; Springer: Boston, MA, USA, 2010. [Google Scholar] [CrossRef]
- Khayet, M.; Cojocaru, C.; Zakrzewska-trznadel, G. Studies on pervaporation separation of acetone, acetonitrile and ethanol from aqueous solutions. Sep. Purif. Technol. 2008, 63, 303–310. [Google Scholar] [CrossRef]
- Uragami, T.; Matsuoka, Y.; Miyata, T. Removal of Dilute Benzene in Water through Ionic Liquid/Poly(Vinyl Chloride) Membranes by Pervaporation. 2016. Available online: www.msrjournal.com (accessed on 8 July 2015).
- Ohshima, T.; Matsumoto, M.; Miyata, T.; Uragami, T. Organic-inorganic hybrid membranes for removal of benzene from an aqueous solution by pervaporation. Macromol. Chem. Phys. 2005, 206, 473–483. [Google Scholar] [CrossRef]
- Peng, F.; Jiang, Z.; Hu, C.; Wang, Y.; Xu, H.; Liu, J. Removing benzene from aqueous solution using CMS-filled PDMS pervaporation membranes. Sep. Purif. Technol. 2006, 48, 229–234. [Google Scholar] [CrossRef]
- Salehi Shahrabi, S.; Mortaheb, H.R.; Barzin, J.; Ehsani, M.R. Pervaporative performance of a PDMS/blended PES composite membrane for removal of toluene from water. Desalination 2012, 287, 281–289. [Google Scholar] [CrossRef]
- Zhang, W.D.; Sun, W.; Yang, J.; Ren, Z.Q. The study on pervaporation behaviors of dilute organic solution through PDMS/PTFE composite membrane. Appl. Biochem. Biotechnol. 2010, 160, 156–167. [Google Scholar] [CrossRef]
- Cao, Z.; Xia, C.; Jia, W.; Qing, W.; Zhang, W. Enhancing bioethanol productivity by a yeast-immobilized catalytically active membrane in a fermentation-pervaporation coupling process. J. Membr. Sci. 2020, 595, 117485. [Google Scholar] [CrossRef]
- Li, S.; Qin, F.; Qin, P.; Karim, M.N.; Tan, T. Preparation of PDMS membrane using water as solvent for pervaporation separation of butanol-water mixture. Green Chem. 2013, 15, 2180–2190. [Google Scholar] [CrossRef]
- Wee, S.L.; Tye, C.T.; Bhatia, S. Process optimization studies for the dehydration of alcohol-water system by inorganic membrane based pervaporation separation using design of experiments (DOE). Sep. Purif. Technol. 2010, 71, 192–199. [Google Scholar] [CrossRef]
- Khayet, M.; Cojocaru, C.; Zakrzewska-Trznadel, G. Response surface modelling and optimization in pervaporation. J. Membr. Sci. 2008, 321, 272–283. [Google Scholar] [CrossRef]
- Catarino, M.; Ferreira, A.; Mendes, A. Study and optimization of aroma recovery from beer by pervaporation. J. Membr. Sci. 2009, 341, 51–59. [Google Scholar] [CrossRef]
- Kataoka, T.; Tsuru, T.; Nakao, S.I.; Kimura, S. Permeation equations developed for prediction of membrane performance in pervaporation, vapor permeation and reverse osmosis based on the solution-diffusion model. J. Chem. Eng. Jpn. 1991, 24, 326–333. [Google Scholar] [CrossRef]
- Lipnizki, F.; Trägårdh, G. Modelling of pervaporation: Models to analyze and predict the mass transport in pervaporation. Separation and Purification Methods. Sep. Purif. Methods 2001, 30, 45–125. [Google Scholar] [CrossRef]
- Knozowska, K.; Kujawska, A.; Li, G.; Kujawa, J.; Bryjak, M.; Kujawski, W.; Lipnizki, F.; Ahrn, L.; Petrini, I.; Kujawski, J.K. Membrane assisted processing of acetone, butanol, and ethanol (ABE) aqueous streams. Chem. Eng. Process.-Process Intensif. 2021, 166, 108462. [Google Scholar] [CrossRef]
- Safi, N.N.; Ibrahim, S.S.; Zouli, N.; Majdi, H.S.; Alsalhy, Q.F.; Drioli, E.; Figoli, A. A systematic framework for optimizing a sweeping gas membrane distillation (SGMD). Membranes 2020, 10, 254. [Google Scholar] [CrossRef]
- Rashid, K.T.; Rahman, S.B.A.; Alsalhy, Q.F. Optimum Operating Parameters for Hollow Fiber Membranes in Direct Contact Membrane Distillation. Arab. J. Sci. Eng. 2016, 41, 2647–2658. [Google Scholar] [CrossRef]
- Myers, R.H.; Montgomery, D.C.; Anderson-Cook, C.M. Response Surface Methodology: Process and Product Optimization Using Designed Experiments; John Wiley Sons: New York, NY, USA, 2002. [Google Scholar]
- Fadhil, S.; Alsalhy, Q.F.; Makki, H.F.; Ruby-Figueroa, R.; Marino, T.; Criscuoli, A.; Macedonio, F.; Giorno, L.; Drioli, E.; Figoli, A. Seawater desalination using PVDF-HFP membrane in DCMD process: Assessment of operating condition by response surface method. Chem. Eng. Commun. 2019, 206, 237–246. [Google Scholar] [CrossRef]
- Yahaya, G.O. Separation of volatile organic compounds (BTEX) from aqueous solutions by a composite organophilic hollow fiber membrane-based pervaporation process. J. Membr. Sci. 2008, 319, 82–90. [Google Scholar] [CrossRef]
- World Health Organization (WHO). Background Document for Development of WHO Guidelines for Drinking-Water Quality, 2nd ed.; WHO: Geneva, Switzerland, 1996. [Google Scholar]
- Basile, A.; Figoli, A.; Khayet, M. Pervaporation, Vapour Permeation and Membrane Distillation: Principles and Applications; Elsevier: Amsterdam, The Netherlands, 2015; ISBN 9781782422563. [Google Scholar]
- Feng, X.; Huang, R.Y.M. Separation of isopropanol from water by pervaporation using silicone-based membranes. J. Membr. Sci. 1992, 74, 171–181. [Google Scholar] [CrossRef]
- Peng, F.; Pan, F.; Li, D.; Jiang, Z. Pervaporation properties of PDMS membranes for removal of benzene from aqueous solution: Experimental and modeling. Chem. Eng. J. 2005, 114, 123–129. [Google Scholar] [CrossRef]
- Wu, H.; Liu, L.; Pan, F.; Hu, C.; Jiang, Z. Pervaporative removal of benzene from aqueous solution through supramolecule calixarene filled PDMS composite membranes. Sep. Purif. Technol. 2006, 51, 352–358. [Google Scholar] [CrossRef]
- Uragami, T.; Ohshima, T.; Miyata, T. Removal of benzene from an aqueous solution of dilute benzene by various cross-linked poly(dimethylsiloxane) membranes during pervaporation. Macromolecules 2003, 36, 9430–9436. [Google Scholar] [CrossRef]
- Kansara, A.M.; Jadav, G.L.; Chaudhri, S.G.; Singh, P.S. Preparation of Poly(dimethylsiloxane)-Polysulfone Composite Membrane by Sequential Absorption-Reaction-Evaporation Process and its Application in Treatment of Aqueous Solution Containing Volatile Organics. Sep. Sci. Technol. 2014, 49, 2834–2846. [Google Scholar] [CrossRef]
- Jadav, G.L.; Aswal, V.K.; Bhatt, H.; Chaudhari, J.C.; Singh, P.S. Influence of film thickness on the structure and properties of PDMS membrane. J. Membr. Sci. 2012, 415–416, 624–634. [Google Scholar] [CrossRef]
- Akhnazarova, S.; Kafarov, V. Experiment Optimization in Chemistry and Chemical Engineering; Mir publishers: Moscow, Russia, 1982. [Google Scholar]
- Chakraborty, S.; Bordoloi, R. Concurrent optimisation of a computer vision system’s multiple responses. Int. J. Adv. Manuf. Technol. 2006, 28, 577–583. [Google Scholar] [CrossRef]
- Hsu, C.M.; Su, C.T.; Liao, D. Simultaneous optimisation of the broadband tap coupler optical performance based on neural networks and exponential desirability functions. Int. J. Adv. Manuf. Technol. 2004, 23, 896–902. [Google Scholar] [CrossRef]
- Cuetos, M.J.; Gómez, X.; Escapa, A.; Morán, A. Evaluation and simultaneous optimization of bio-hydrogen production using 32 factorial design and the desirability function. J. Power Sources 2007, 169, 131–139. [Google Scholar] [CrossRef]
- Pasandideh, S.H.R.; Niaki, S.T.A. Multi-response simulation optimization using genetic algorithm within desirability function framework. Appl. Math. Comput. 2006, 175, 366–382. [Google Scholar] [CrossRef]
- Ferreira, S.L.C.; Bruns, R.E.; Ferreira, H.S.; Matos, G.D.; David, J.M.; Brandão, G.C.; da Silva, E.G.P.; Portugal, L.A.; dos Reis, P.S.; Souza, A.S.; et al. Box-Behnken design: An alternative for the optimization of analytical methods. Anal. Chim. Acta 2007, 597, 179–186. [Google Scholar] [CrossRef]
- Pasamontes, A.; Callao, P. Fractional factorial design and simplex algorithm for optimizing sequential injection analysis (SIA) and second order calibration. Chemom. Intell. Lab. Syst. 2006, 83, 127–132. [Google Scholar] [CrossRef]




| Membrane | ThAL (µm) | ThSL (µm) | VOCs | T (°C) | Con. | Pressure | VOCs Flux (g/m2 h) | S.F. | Ref. |
|---|---|---|---|---|---|---|---|---|---|
| PDMS | 87.5 ± 10.3 | None | Ethanol | 30 | 5 %wt. | @ | 11 | 10 | [23] |
| PDMS_plasma_C8 | 126.5 ± 6.2 | None | 8 | 7 | |||||
| (PDMS)-NaCl | 167.5 ± 5.6 | None | 13 | 11 | |||||
| PDMS_Al2O3_nat | 85.8 ± 11.2 | None | 10 | 10 | |||||
| PDMS_Al2O3_mod._C8 | 129.5 ± 10.9 | None | 21 | 11 | |||||
| PDMS/PVDF | 177.3 ± 13.2 | 24 | 8 | 9 | |||||
| PDMS (PervapTM4060) | 5.5 | 178 | 201 | 9 | |||||
| PDMS | @ | @ | Acetonitrile | 40 | 5913 ppm | 4–15 mbar | 31.56 | 28.2 | [25] |
| PDMS | 250 | None | Phenol | 40 | 0.5 %wt. | 200 Pa | 4 | 15 | [26] |
| 70 | 7 | 3 | |||||||
| PDMS + oleyl alcohol (5%) | 40 | 3 | 3.5 | ||||||
| 70 | 11 | 6.5 | |||||||
| PDMS (PervapTM4060) | 5 | 169 | Acetone | 30 | 3 %wt. | 5.5 kPa | 2.7 | 100 | [30] |
| 60 | 8.4 | 30 | |||||||
| Acetonitrile | 30 | 1.2 | 11 | ||||||
| 60 | 4.2 | 10 | |||||||
| Ethanol | 30 | 0.3 | 5 | ||||||
| 60 | 1.2 | 4 | |||||||
| PDMS + PES | 20 | 200 | Toluene | 30 | 150 ppm | 1 mbar | 3.5 | 2200 | [34] |
| 50 | 300 ppm | 7.5 | 1300 | ||||||
| PDMS + PTFE | 35 | 15 | Acetone | 30 | 0.99 %wt. | 15 mmHg | 8 (kg µm /m2 h) | 55 | [35] |
| Butanone | 10 | 125 | |||||||
| Cyclohexane | 6 | 85 | |||||||
| Ethanol | 6 | 5 | |||||||
| Isopropanol | 7 | 15 | |||||||
| n-butyl alcohol | 9 | 40 | |||||||
| Acetic acid | 6 | 2.5 | |||||||
| Ethyl acetate | 14 | 90 | |||||||
| PDMS + PVDF | 32 | 25 | Ethanol | 35 | 9 %wt. | 10 mmHg | 367 | 6.6 | [36] |
| PDMS | 30 | None | Butanol | 55 | 1.5 %wt. | 240 | 43 | [37] |
| Membrane | ThAL (µm) | ThSL (µm) | T (°C) | Con. | Pressure | C6H6 Flux (g/m2 h) | S.F. | Ref. |
|---|---|---|---|---|---|---|---|---|
| PDMS | 140–200 | None | 60 | 750 ppm | 0.2 kPa | 180 | 9000 | [52] |
| PDMS | 200 | None | 60 | 1400 ppm | 1–10 kPa | 126 | 3302 | [53] |
| CA-f-PDMS | 150 | 116 | 5604 | |||||
| PDMS (composite) | 100 | 100 | 365 | 4600 | ||||
| CA-f-PDMS composite | 50 | 407 | 5913 | |||||
| PDMS + DMMA | 270 | None | 40 | 500 ppm | 0.01 mmHg | 51.4 | 1853 | [54] |
| PDMS + DVB | 314 | 45.5 | 3099 | |||||
| PDMS + DVS | 276 | 70.9 | 2886 | |||||
| PDMS + EGDM | 357 | 49.6 | 2011 | |||||
| PDMS + PES | 11 | None | 25 | 150ppm | 5 mbar | 66 | 972 | [55] |
| PDMS + PES | 0.2 | 140 | 25 | Benzene 2 %wt. + Methanol 50 %wt. in water | @ | 4.2 | 1 | [56] |
| 0.5 | 7 | 1.5 | ||||||
| 2 | 6.4 | 2.5 | ||||||
| 3 | 7 | 4 | ||||||
| 8 | 7.3 | 7.5 | ||||||
| 35 | 8.1 | 15 | ||||||
| 150 | 10 | 20 | ||||||
| PDMS (PervapTM4060) | 5.5 | 178 | 30 | 1000 ppm | 2 kPa | 7.5 | 47 | The present work |
| Std Order | Temp. (°C) | Conc. (ppm) | Flow Rate (L/min) | Flux (g/m2·h) | S.F. |
|---|---|---|---|---|---|
| 6 | 50 | 100 | 3.5 | 0.24442 | 6.5231 |
| 12 | 40 | 1000 | 2.5 | 6.53818 | 23.7613 |
| 17 | 40 | 550 | 2.5 | 3.20917 | 23.7200 |
| 1 | 30 | 100 | 1.5 | 0.05733 | 2.2940 |
| 10 | 50 | 550 | 2.5 | 3.40000 | 18.8843 |
| 9 | 30 | 550 | 2.5 | 3.00000 | 32.3700 |
| 14 | 40 | 550 | 3.5 | 3.50000 | 20.0000 |
| 4 | 50 | 1000 | 1.5 | 7.05867 | 19.7256 |
| 13 | 40 | 550 | 1.5 | 3.12402 | 21.2793 |
| 3 | 30 | 1000 | 1.5 | 6.00000 | 36.9546 |
| 16 | 40 | 550 | 2.5 | 3.20917 | 23.7200 |
| 20 | 40 | 550 | 2.5 | 3.20917 | 23.7200 |
| 15 | 40 | 550 | 2.5 | 3.20917 | 23.7200 |
| 19 | 40 | 550 | 2.5 | 3.20917 | 23.7200 |
| 5 | 30 | 100 | 3.5 | 0.18333 | 8.8097 |
| 11 | 40 | 100 | 2.5 | 0.08583 | 4.1221 |
| 2 | 50 | 100 | 1.5 | 0.20035 | 6.2494 |
| 18 | 40 | 550 | 2.5 | 3.20917 | 23.7200 |
| 7 | 30 | 1000 | 3.5 | 6.77333 | 40.0000 |
| A0 | A1 | A2 | A3 | A4 | A5 | A6 | A7 | A8 | A9 | |
|---|---|---|---|---|---|---|---|---|---|---|
| −0.662 | 0.0134 | 4.66 × 10−3 | 0.236 | 0.00125 | 1 × 10−6 | 0.1245 | 3.6 × 10−5 | −0.00886 | 0.000208 | |
| 26 | −2.467 | 0.11601 | 13.34 | 0.03609 | −4 × 10−5 | −1.378 | −0.0011 | −0.1247 | −0.00123 |
| Source | DF | Adj SS | Adj MS | F-Value | p-Value |
|---|---|---|---|---|---|
| Model | 9 | 108.871 | 12.097 | 2393.22 | 0.000 |
| Linear | 3 | 108.263 | 36.088 | 7139.59 | 0.000 |
| Temp. | 1 | 0.439 | 0.439 | 86.79 | 0.000 |
| Conc. | 1 | 107.610 | 107.610 | 21289.47 | 0.000 |
| Flow | 1 | 0.215 | 0.215 | 42.50 | 0.000 |
| Square | 3 | 0.268 | 0.089 | 17.65 | 0.000 |
| Temp. × Temp. | 1 | 0.000 | 0.000 | 0.09 | 0.776 |
| Conc. × Conc. | 1 | 0.043 | 0.043 | 8.44 | 0.016 |
| Flow × Flow | 1 | 0.043 | 0.043 | 8.44 | 0.016 |
| 2-Way Interaction | 3 | 0.340 | 0.113 | 22.41 | 0.000 |
| Temp. × Conc. | 1 | 0.207 | 0.207 | 40.91 | 0.000 |
| Temp. × Flow | 1 | 0.063 | 0.063 | 12.43 | 0.005 |
| Conc. × Flow | 1 | 0.070 | 0.070 | 13.90 | 0.004 |
| Error | 10 | 0.051 | 0.005 | - | - |
| Lack-of-Fit | 5 | 0.051 | 0.010 | - | - |
| Pure Error | 5 | 0.000 | 0.000 | - | - |
| Total | 19 | 108.921 | - | - | - |
| Source | DF | Adj SS | Adj MS | F-Value | p-Value |
|---|---|---|---|---|---|
| Model | 9 | 1975.38 | 219.49 | 80.03 | 0.000 |
| Linear | 3 | 1499.05 | 499.68 | 182.19 | 0.000 |
| Temp. | 1 | 250.10 | 250.10 | 91.19 | 0.000 |
| Conc. | 1 | 1242.76 | 1242.76 | 453.12 | 0.000 |
| Flow | 1 | 6.19 | 6.19 | 2.26 | 0.164 |
| Square | 3 | 262.81 | 87.60 | 31.94 | 0.000 |
| Temp. × Temp. | 1 | 35.82 | 35.82 | 13.06 | 0.005 |
| Conc. × Conc. | 1 | 179.37 | 179.37 | 65.40 | 0.000 |
| Flow × Flow | 1 | 5.22 | 5.22 | 1.90 | 0.198 |
| 2-Way Interaction | 3 | 213.52 | 71.17 | 25.95 | 0.000 |
| Temp. × Conc. | 1 | 198.62 | 198.62 | 72.42 | 0.000 |
| Temp. × Flow | 1 | 12.44 | 12.44 | 4.54 | 0.059 |
| Conc. × Flow | 1 | 2.46 | 2.46 | 0.90 | 0.366 |
| Error | 10 | 27.43 | 2.74 | - | - |
| Lack-of-Fit | 5 | 27.43 | 5.49 | - | - |
| Pure Error | 5 | 0.00 | 0.00 | - | - |
| Total | 19 | 2002.81 | - | - | - |
| Parameters | Stand. Dev. | R2 | R2(adj) | R2(pred) |
|---|---|---|---|---|
| Permeate flux | 0.0710957 | 99.95% | 99.91% | 99.11% |
| Separation factor | 1.65611 | 98.63% | 97.40% | 90.47% |
| Temp. (°C) | Conc. (ppm) | Flow (L/min.) | S.F.Fit | Flux Fit (g/m2·h) | Composite Desirability |
|---|---|---|---|---|---|
| 30 | 1000 | 3.5 | 39.8049 | 6.70111 | 0.961608 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rasheed, S.H.; Ibrahim, S.S.; Alsalhy, Q.F.; Salih, I.K. Separation of Soluble Benzene from an Aqueous Solution by Pervaporation Using a Commercial Polydimethylsiloxane Membrane. Membranes 2022, 12, 1040. https://doi.org/10.3390/membranes12111040
Rasheed SH, Ibrahim SS, Alsalhy QF, Salih IK. Separation of Soluble Benzene from an Aqueous Solution by Pervaporation Using a Commercial Polydimethylsiloxane Membrane. Membranes. 2022; 12(11):1040. https://doi.org/10.3390/membranes12111040
Chicago/Turabian StyleRasheed, Salam H., Salah S. Ibrahim, Qusay F. Alsalhy, and Issam K. Salih. 2022. "Separation of Soluble Benzene from an Aqueous Solution by Pervaporation Using a Commercial Polydimethylsiloxane Membrane" Membranes 12, no. 11: 1040. https://doi.org/10.3390/membranes12111040
APA StyleRasheed, S. H., Ibrahim, S. S., Alsalhy, Q. F., & Salih, I. K. (2022). Separation of Soluble Benzene from an Aqueous Solution by Pervaporation Using a Commercial Polydimethylsiloxane Membrane. Membranes, 12(11), 1040. https://doi.org/10.3390/membranes12111040








