Microfiltration Membranes Modified with Silver Oxide by Plasma Treatment
Abstract
1. Introduction
2. Materials and Methods
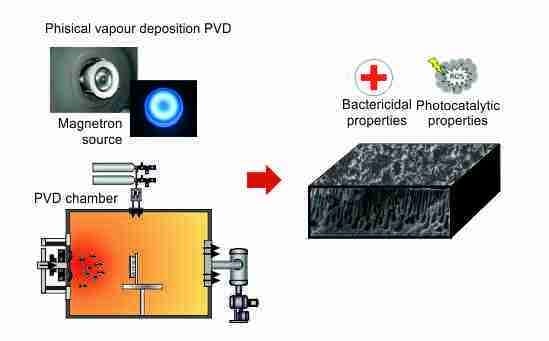
2.1. Coatings Deposition
2.2. Structure Characterization
2.3. Bactericidal Properties
2.4. Photocatalytic Properties
2.5. Filtration Properties
3. Results and Discussion
3.1. Structure Characterization
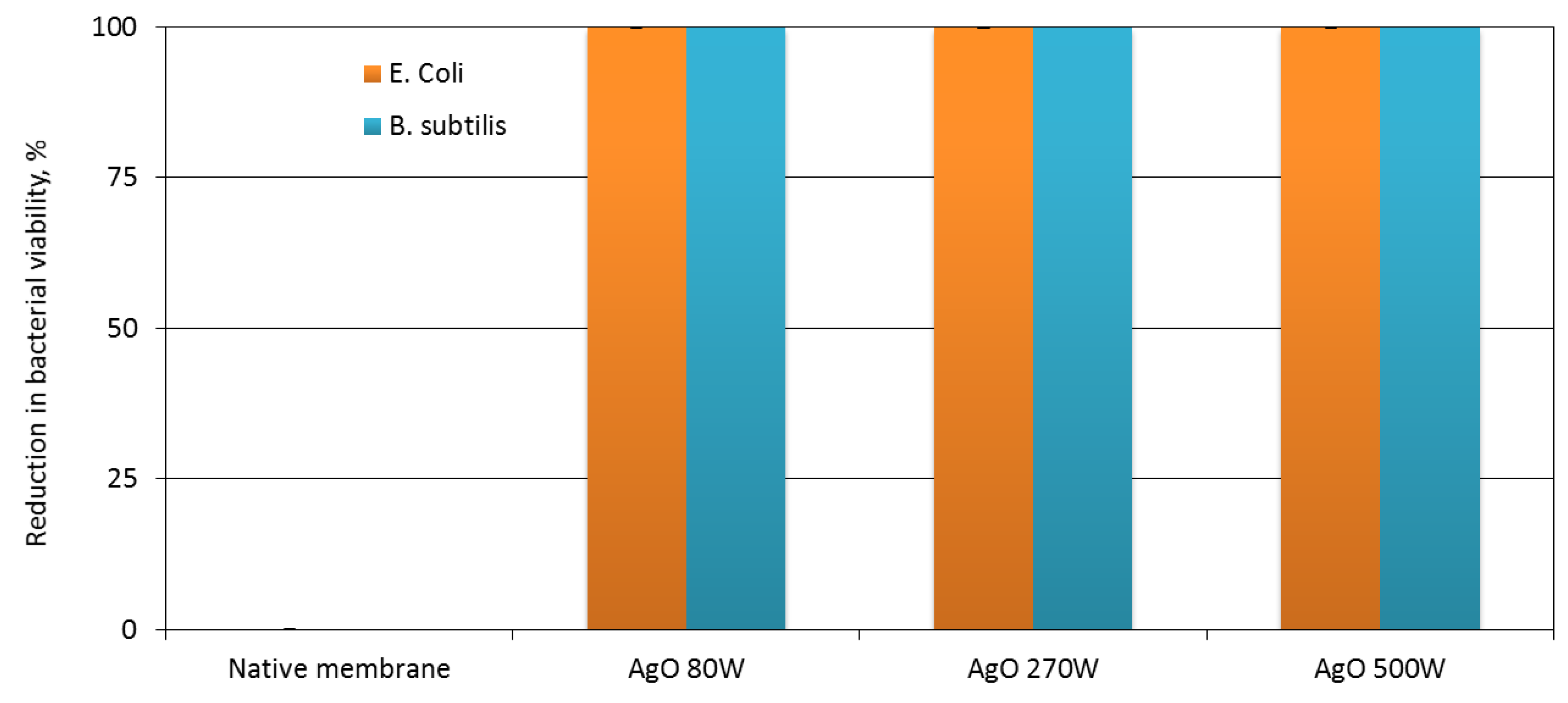
3.2. Antibacterial Properties
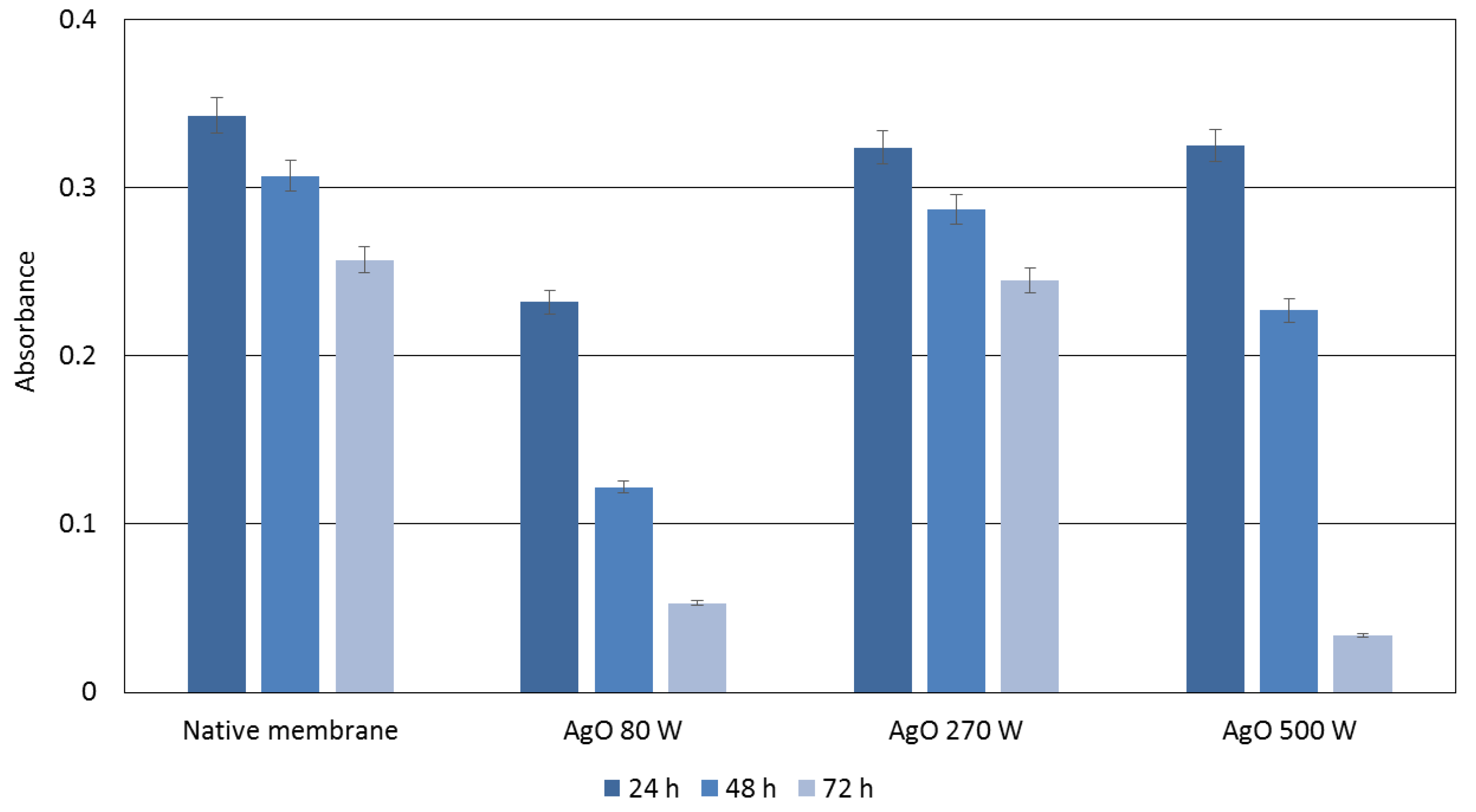
3.3. Photocatalytic Properties
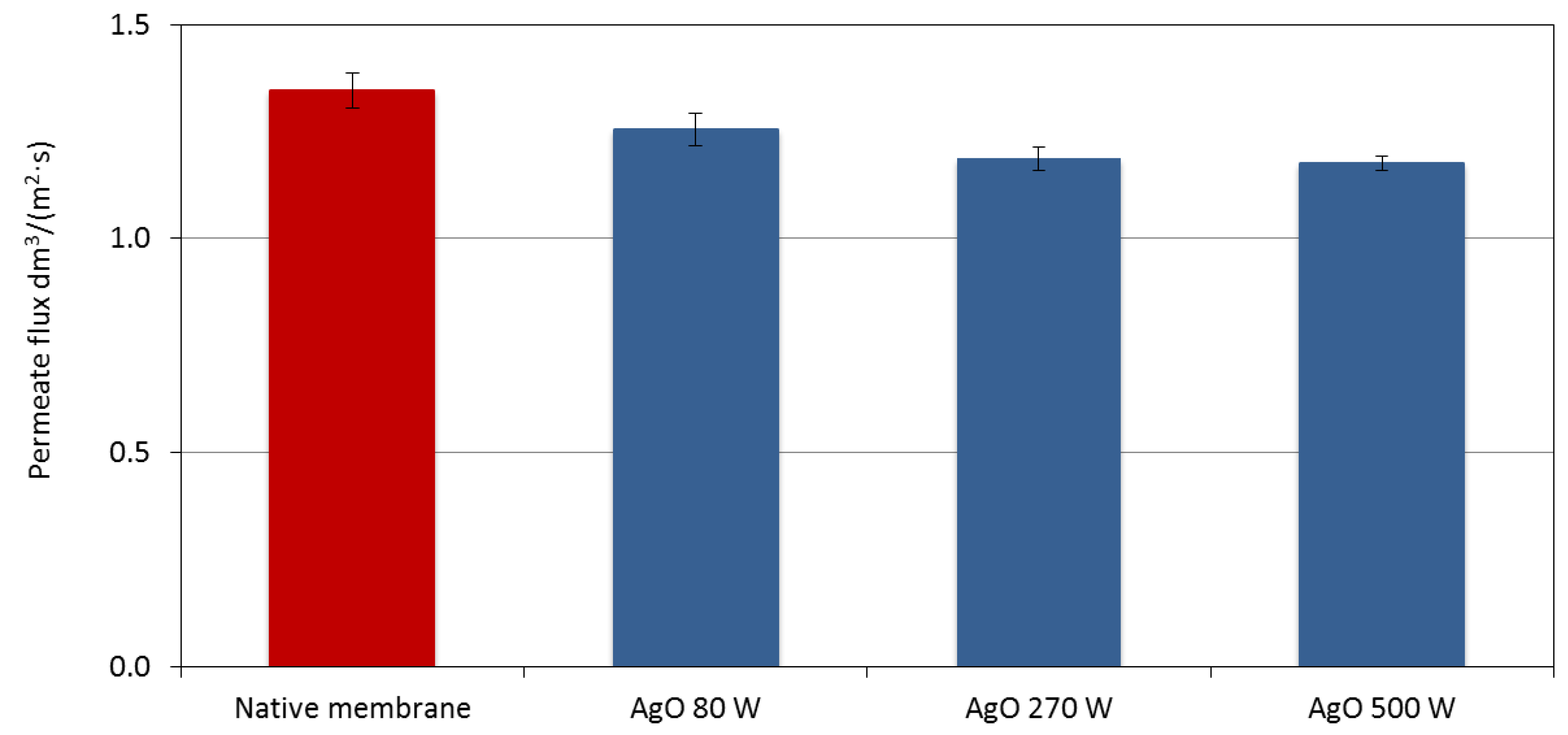
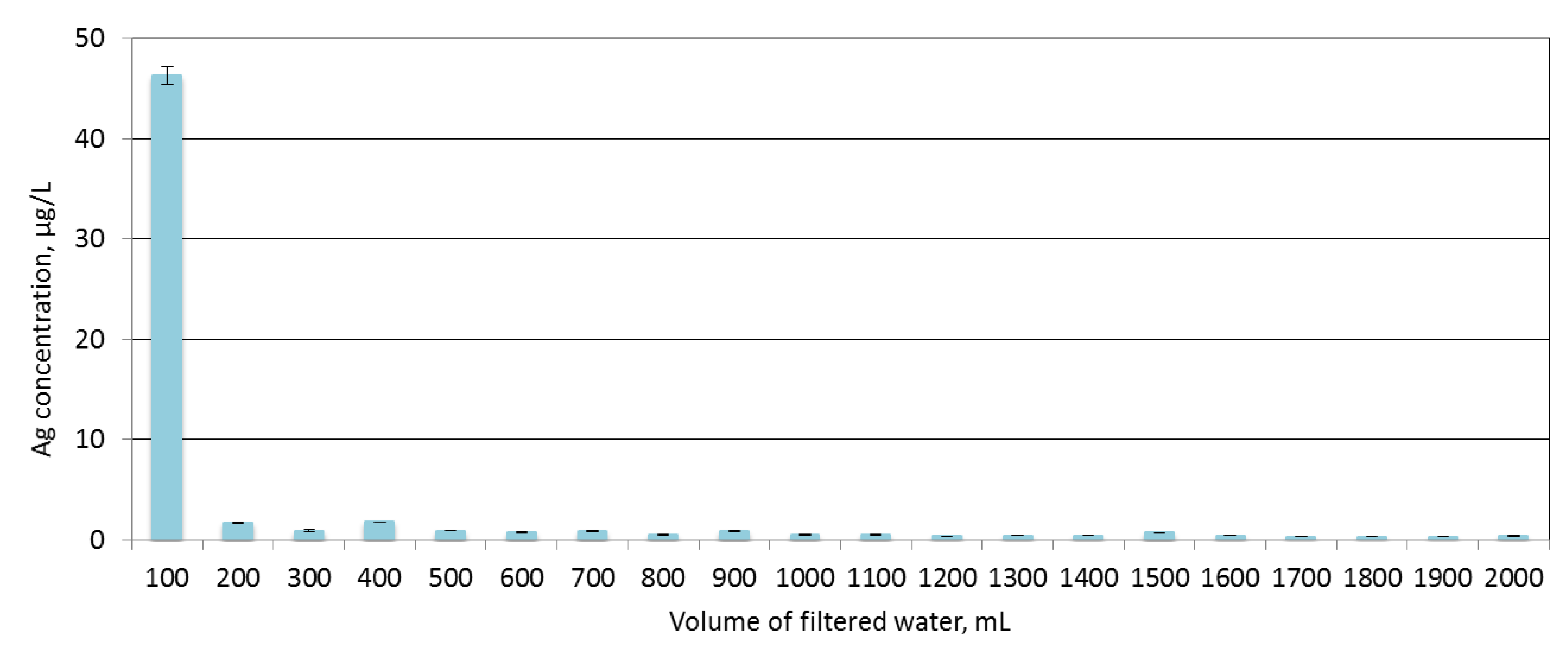
3.4. Filtration Properties and Stability of Coatings
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Kołtuniewicz, A.B.; Drioli, E. Membranes in Clean Technologies. Theory and Practice, Volume 1-2; WILEY-VCH Verlag GmbH & Co: Weinheim, Germany, 2008. [Google Scholar]
- Faria, A.F.; Liu, C.; Xie, M.; Perreault, F.; Nghiem, L.D.; Ma, J.; Elimelech, M. Thin-film composite forward osmosis membranes functionalized with graphene oxide-silver nanocomposite for biofouling control. J. Membr. Sci. 2017, 525, 146–156. [Google Scholar] [CrossRef]
- Nguyen, T.; Roddick, F.A.; Fan, L. Biofouling of Water Treatment Membranes: A Review of the Underlying Causes, Monitoring Techniques and Control Measures. Membranes 2012, 2, 804–840. [Google Scholar] [CrossRef]
- Najjar, A.; Sabri, S.; Al-Gaashani, R.; Atieh, M.A.; Kochkodan, V. Antibiofouling Performance by Polyethersulfone Membranes Cast with Oxidized Multiwalled Carbon Nanotubes and Arabic Gum. Membranes 2019, 9, 32. [Google Scholar] [CrossRef]
- Sabri, S.; Najjar, A.; Manawi, Y.; Eltai, N.O.; Al-Thani, A.; Atieh, M.A.; Kochkodan, V. Antibacterial Properties of Polysulfone Membranes Blended with Arabic Gum. Membranes 2019, 9, 29. [Google Scholar] [CrossRef]
- Kowalik-Klimczak, A.; Stanisławek, E.; Kacprzyńska-Gołacka, J. The surface modification of polyamide membranes using graphene oxide. Colloids Surf. A Physicochem. Eng. Asp. 2020, 587, 124281. [Google Scholar] [CrossRef]
- Kowalik-Klimczak, A.; Stanisławek, E.; Kacprzyńska-Gołacka, J.; Osuch-Słomka, E.; Bednarska, A.; Skowroński, J. The polyamide membranes functionalized by nanoparticles for biofouling control. Desalin. Water Treat. 2018, 128, 243–252. [Google Scholar] [CrossRef]
- Padil, V.V.T.; Černík, M. Poly (vinyl alcohol)/gum karaya electrospun plasma treated membrane for the removal of nanoparticles (Au, Ag Pt, CuO and Fe3O4) from aqueous solutions. J. Hazard. Mater. 2015, 287, 102–110. [Google Scholar] [CrossRef] [PubMed]
- Cruz, M.C.; Ruano, G.; Wolf, M.; Hecker, D.; Castro Vidaurre, E.; Schmittgens, R.; Rajal, V.B. Plasma deposition of silver nanoparticles on ultrafiltration membranes: Antibacterial and anti-biofouling properties. Chem. Eng. Res. Des. 2015, 94, 524–537. [Google Scholar] [CrossRef] [PubMed]
- Ren, P.-F.; Fang, Y.; Wan, L.-S.; Ye, X.-Y.; Xu, Z.-K. Surface modification of polypropylene microfiltration membrane by grafting poly(sulfobetaine methacrylate) and poly(ethylene glycol): Oxidative stability and antifouling capability. J. Membr. Sci. 2015, 492, 249–256. [Google Scholar] [CrossRef]
- Shahkaramipour, N.; Tran, T.N.; Ramanan, S.; Lin, H. Membranes with Surface-Enhanced Antifouling Properties for Water Purification. Membranes 2017, 7, 13. [Google Scholar] [CrossRef]
- Ahmad, A.L.; Sugumaran, J.; Shoparwe, N.F. Antifouling Properties of PES Membranes by Blending with ZnO Nanoparticles and NMP–Acetone Mixture as Solvent. Membranes 2018, 8, 131. [Google Scholar] [CrossRef] [PubMed]
- Akbari, A.; Derikvandi, Z.; Mojallali Rostami, S.M. Influence of chitosan coating on the separation performance, morphology and anti-fouling properties of the polyamide nanofiltration membranes. J. Ind. Eng. Chem. 2015, 28, 268–276. [Google Scholar] [CrossRef]
- Muppalla, R.; Rana, H.H.; Devi, S.; Jewrajka, S.K. Adsorption of pH-responsive amphiphilic copolymer micelles and gel on membrane surface as an approach for antifouling coating. Appl. Surf. Sci. 2013, 268, 355–367. [Google Scholar] [CrossRef]
- Liu, L.-F.; Yu, S.-C.; Wu, L.-G.; Gao, C.-J. Study on a novel polyamide-urea reverse osmosis composite membrane (ICIC-MPD): II. Analysis of membrane antifouling performance. J. Membr. Sci. 2006, 283, 133–146. [Google Scholar] [CrossRef]
- Lei, J.; Ulbricht, M. Macroinitiator-mediated photoreactive coating of membrane surfaces with antifouling hydrogel layers. J. Membr. Sci. 2014, 455, 207–218. [Google Scholar] [CrossRef]
- Zinadini, S.; Zinatizadeh, A.A.; Rahimi, M.; Vatanpour, V.; Zangeneh, H.; Beygzadeh, M. Novel high flux antifouling nanofiltration membranes for dye removal containing carboxymethyl chitosan coated Fe3O4 nanoparticles. Desalination 2014, 349, 145–154. [Google Scholar] [CrossRef]
- Bai, L.; Liang, H.; Crittenden, J.; Qu, F.; Ding, A.; Ma, J.; Du, X.; Guo, S.; Li, G. Surface modification of UF membranes with functionalized MWCNTs to control membrane fouling by NOM fractions. J. Membr. Sci. 2015, 492, 400–411. [Google Scholar] [CrossRef]
- Alammar, A.; Park, S.-H.; Williams, C.J.; Derby, B.; Szekely, G. Oil-in-water separation with graphene-based nanocomposite membranes for produced water treatment. J. Membr. Sci. 2020, 603, 118007. [Google Scholar] [CrossRef]
- Abdulazeez, I.; Matin, A.; Khan, M.; Khaled, M.M.; Ansari, M.A.; Akhtar, S.; Rehman, S. Facile preparation of antiadhesive and biocidal reverse osmosis membranes using a single coating for efficient water purification. J. Membr. Sci. 2019, 591, 117299. [Google Scholar] [CrossRef]
- Kacprzyńska-Gołacka, J.; Kowalik-Klimczak, A.; Skowroński, J.; Rajewska, P.; Wieciński, P.; Smolik, J. Możliwości wykorzystania plazmowych technik inżynierii powierzchni do modyfikacji membran polimerowych. Polimery 2018, 5, 353–361. [Google Scholar]
- Woskowicz, E.; Łożyńska, M.; Kowalik-Klimczak, A.; Kacprzyńska-Gołacka, J.; Osuch-Słomka, E.; Piasek, A.; Gradoń, L. Plasma deposition of antimicrobial coatings based on silver and copper on polypropylene. Polimery 2020, 1, 33–43. [Google Scholar] [CrossRef]
- Maurer, C.; Schulz, U. Solid particle erosion of thick PVD coatings on CFRP. Wear 2014, 317, 246–253. [Google Scholar] [CrossRef]
- Prabhu, S.; Poulose, E.K. Silver nanoparticles: Mechanism of antimicrobial action, synthesis, medical application, and toxicity effects. Int. Nano Lett. 2009, 84, 741–749. [Google Scholar] [CrossRef]
- Shen, W.; Li, P.; Feng, H.; Ge, Y.; Liu, Z.; Feng, L. The bactericidal mechanism of action against Staphylococcus aureus for AgO nanoparticles. Mater. Sci. Eng. C 2017, 75, 610–619. [Google Scholar] [CrossRef]
- Danilczuk, M.; Lund, A.; Sadlo, J.; Yamadac, H.; Michalik, J. Conduction electron spin resonance of small silver particles. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2009, 63, 189–191. [Google Scholar] [CrossRef]
- Qing, Y.; Cheng, L.; Li, R.; Liu, G.; Zhang, Y.; Tang, X.; Wang, J.; Liu, H.; Qin, Y. Potential antibacterial mechanism of silver nanoparticles and the optimization of orthopedic implants by advanced modification technologies. Int. J. Nanomed. 2018, 13, 3311–3327. [Google Scholar] [CrossRef]
- Kim, J.S.; Kuk, E.; Yu, K.; Kim, J.H.; Park, S.J.; Lee, H.J.; Kim, S.H.; Park, Y.K.; Park, Y.H.; Hwang, C.Y.; et al. Antimicrobial effects of silver nanoparticles. Nanomedicine 2007, 3, 95–101. [Google Scholar] [CrossRef]
- Kumari, R.M.; Thapa, N.; Gupta, N.; Kumar, A.; Nimesh, S. Antibacterial and photocatalytic degradation efficacy of silver nanoparticles biosynthesized using Cordia dichtoma leaf extract. Adv. Nat. Sci. Nanosci. Nanotechnol. 2016, 7, 045009. [Google Scholar] [CrossRef]
- López-Miranda, J.L.; Cervantes-Chávez, J.A.; Hernández-Martínez, A.R.; Pérez, R.; Esparza, R.; Estévez-González, M. Study on the photocatalytic and antibacterial properties of silver nanoparticles synthesized by a green approach. Mater. Res. Express 2019, 6, 065066. [Google Scholar] [CrossRef]
- Ravichandran, V.; Vasanthi, S.; Shalini, S.; Shah SA, A.; Tripathy, M.; Paliwal, N. Green synthesis, characterization, antibacterial, antioxidant and photocatalytic activity of Parkia speciosa leaves extract mediated silver nanoparticles. Results Phys. 2019, 15, 102565. [Google Scholar] [CrossRef]
- Jiang, W.; Wang, X.; Wu, Z.; Yue, X.; Yuan, S.; Lu, H.; Liang, B. Silver Oxide as Superb and Stable Photocatalyst under Visible and Near-Infrared Light Irradation and Its Photocatalytic Mechanism. Ind. Eng. Chem. Res. 2015, 54, 832–841. [Google Scholar] [CrossRef]
- Vanaja, M.; Paulkumar, K.; Baburaja, M.; Rajeshkumar, S.; Gnanajobitha, G.; Malarkodi, C.; Sivakavinesan, M.; Annadurai, G. Degradation of Methylene Blue Using Biologically Synthesized Silver Nanoparticles. Bioinorg. Chem. Appl. 2014, 742346. [Google Scholar] [CrossRef] [PubMed]


| Parameters | Values |
|---|---|
| Forward power, W | 1548.6 |
| Cool gas flow, dm3/min | 13.956 |
| Auxiliary gas flow, dm3/min | 0.8021 |
| Nebulizer gas flow, dm3/min | 1.02464 |
| Dwell time, s | 0.005 |
| Number of replicates | 3 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kacprzyńska-Gołacka, J.; Kowalik-Klimczak, A.; Woskowicz, E.; Wieciński, P.; Łożyńska, M.; Sowa, S.; Barszcz, W.; Kaźmierczak, B. Microfiltration Membranes Modified with Silver Oxide by Plasma Treatment. Membranes 2020, 10, 133. https://doi.org/10.3390/membranes10060133
Kacprzyńska-Gołacka J, Kowalik-Klimczak A, Woskowicz E, Wieciński P, Łożyńska M, Sowa S, Barszcz W, Kaźmierczak B. Microfiltration Membranes Modified with Silver Oxide by Plasma Treatment. Membranes. 2020; 10(6):133. https://doi.org/10.3390/membranes10060133
Chicago/Turabian StyleKacprzyńska-Gołacka, Joanna, Anna Kowalik-Klimczak, Ewa Woskowicz, Piotr Wieciński, Monika Łożyńska, Sylwia Sowa, Wioletta Barszcz, and Bernadetta Kaźmierczak. 2020. "Microfiltration Membranes Modified with Silver Oxide by Plasma Treatment" Membranes 10, no. 6: 133. https://doi.org/10.3390/membranes10060133
APA StyleKacprzyńska-Gołacka, J., Kowalik-Klimczak, A., Woskowicz, E., Wieciński, P., Łożyńska, M., Sowa, S., Barszcz, W., & Kaźmierczak, B. (2020). Microfiltration Membranes Modified with Silver Oxide by Plasma Treatment. Membranes, 10(6), 133. https://doi.org/10.3390/membranes10060133