Dynamic Modeling Using Artificial Neural Network of Bacillus Velezensis Broth Cross-Flow Microfiltration Enhanced by Air-Sparging and Turbulence Promoter
Abstract
1. Introduction
2. Materials and Methods
2.1. Preparation of Bacillus Velezensis Cultivation Broth
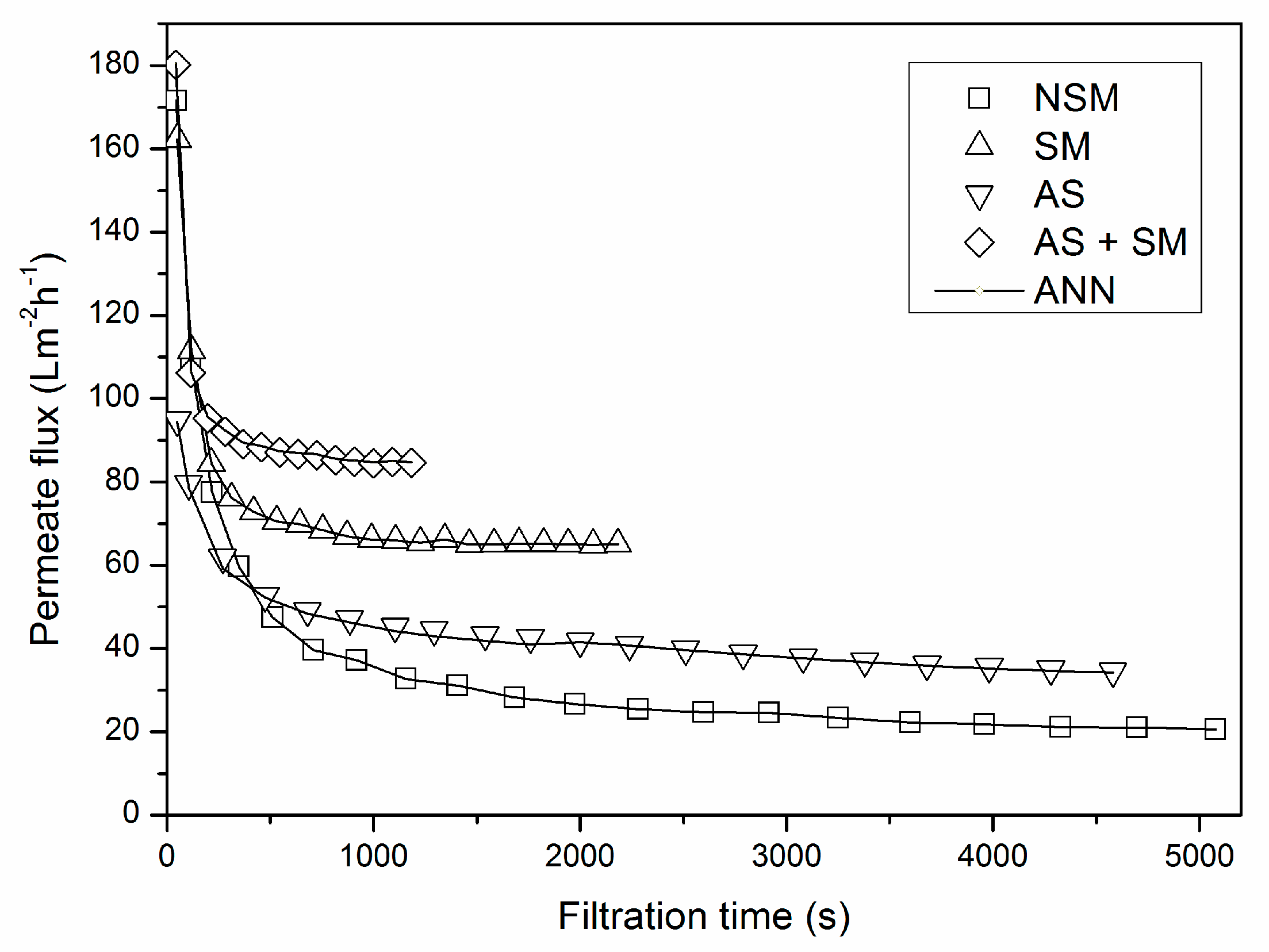
2.2. Microfiltration Experiments
2.3. Data Compilation
2.4. Artificial Neural Network Modelling
3. Results and Discussion
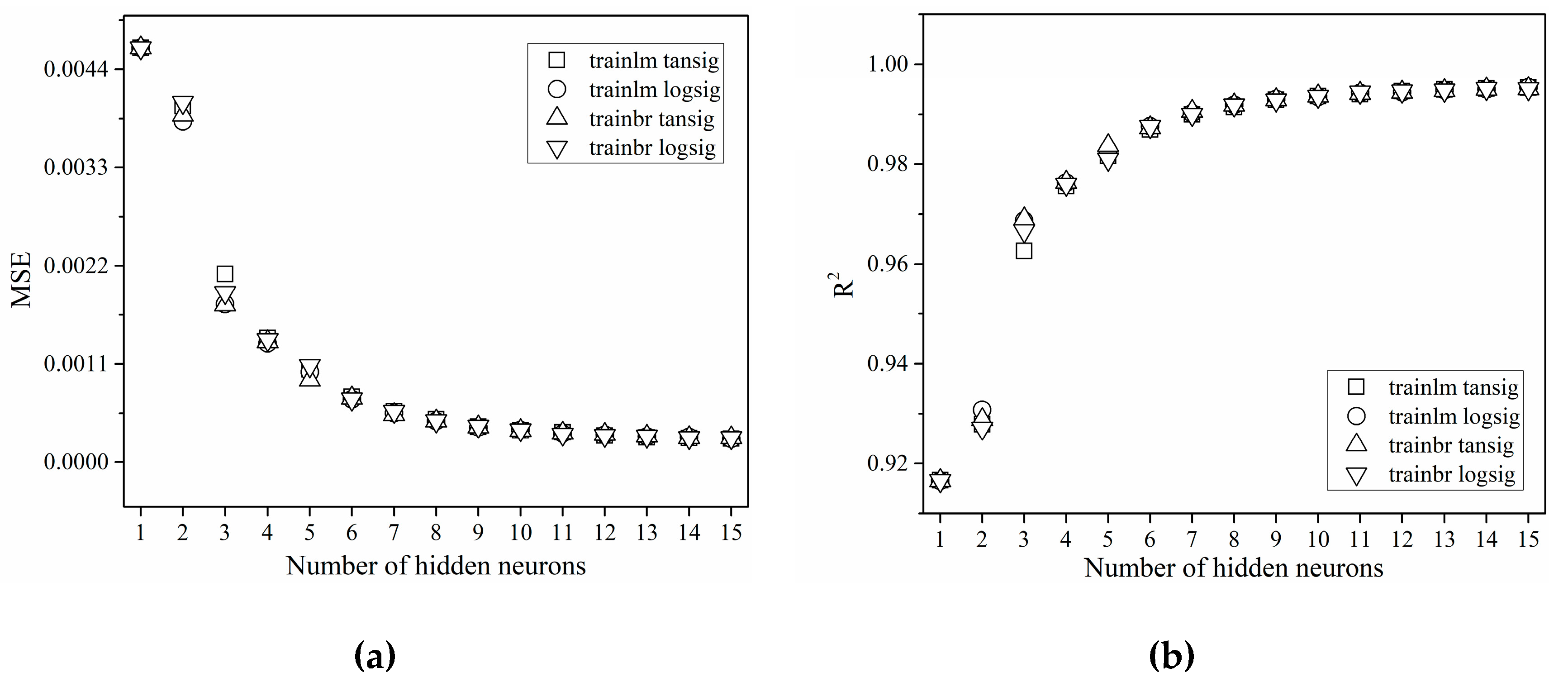
3.1. Effect of Learning Algorithm, Transfer Function and Number of Hidden Layer Neurons
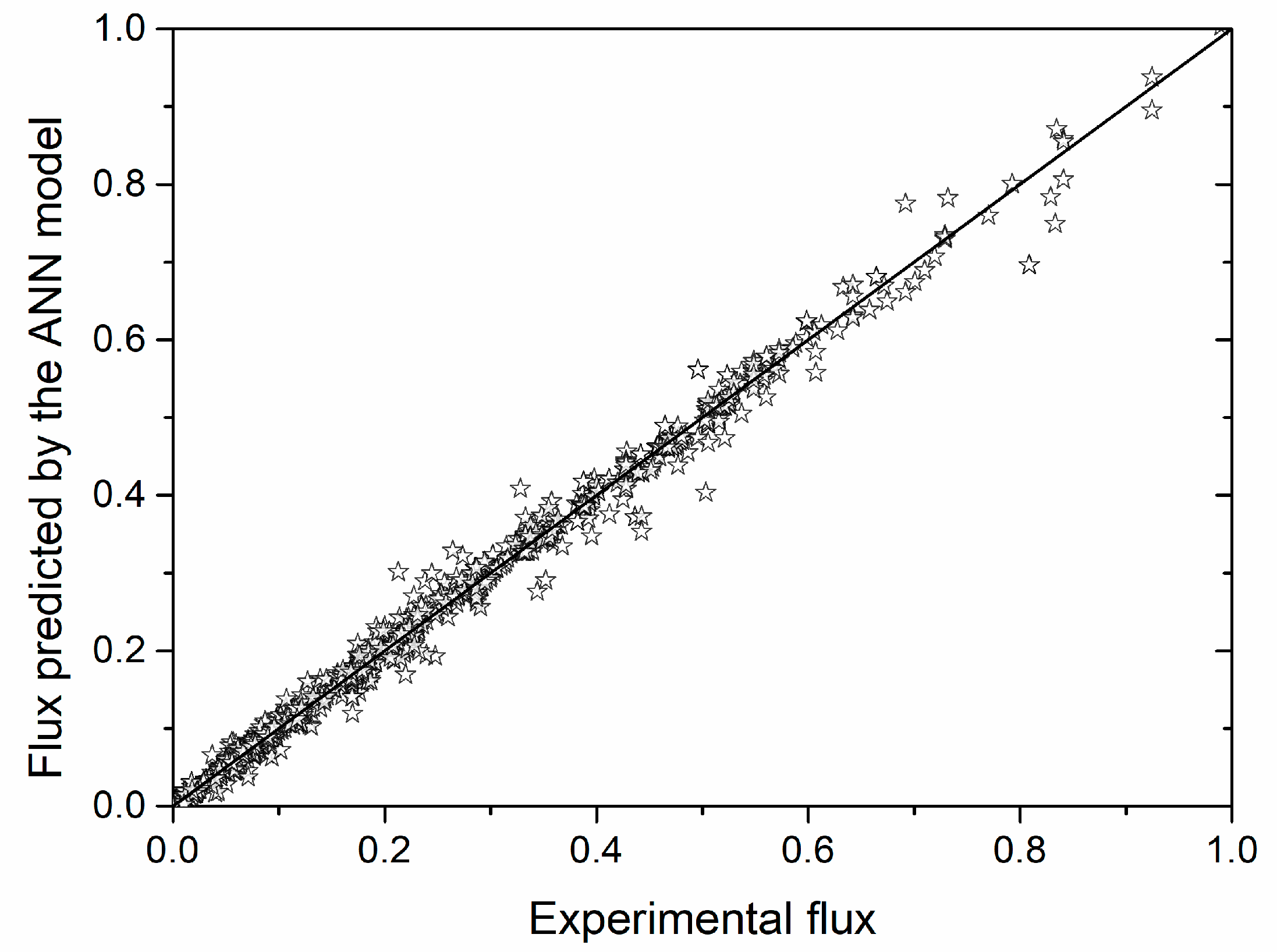
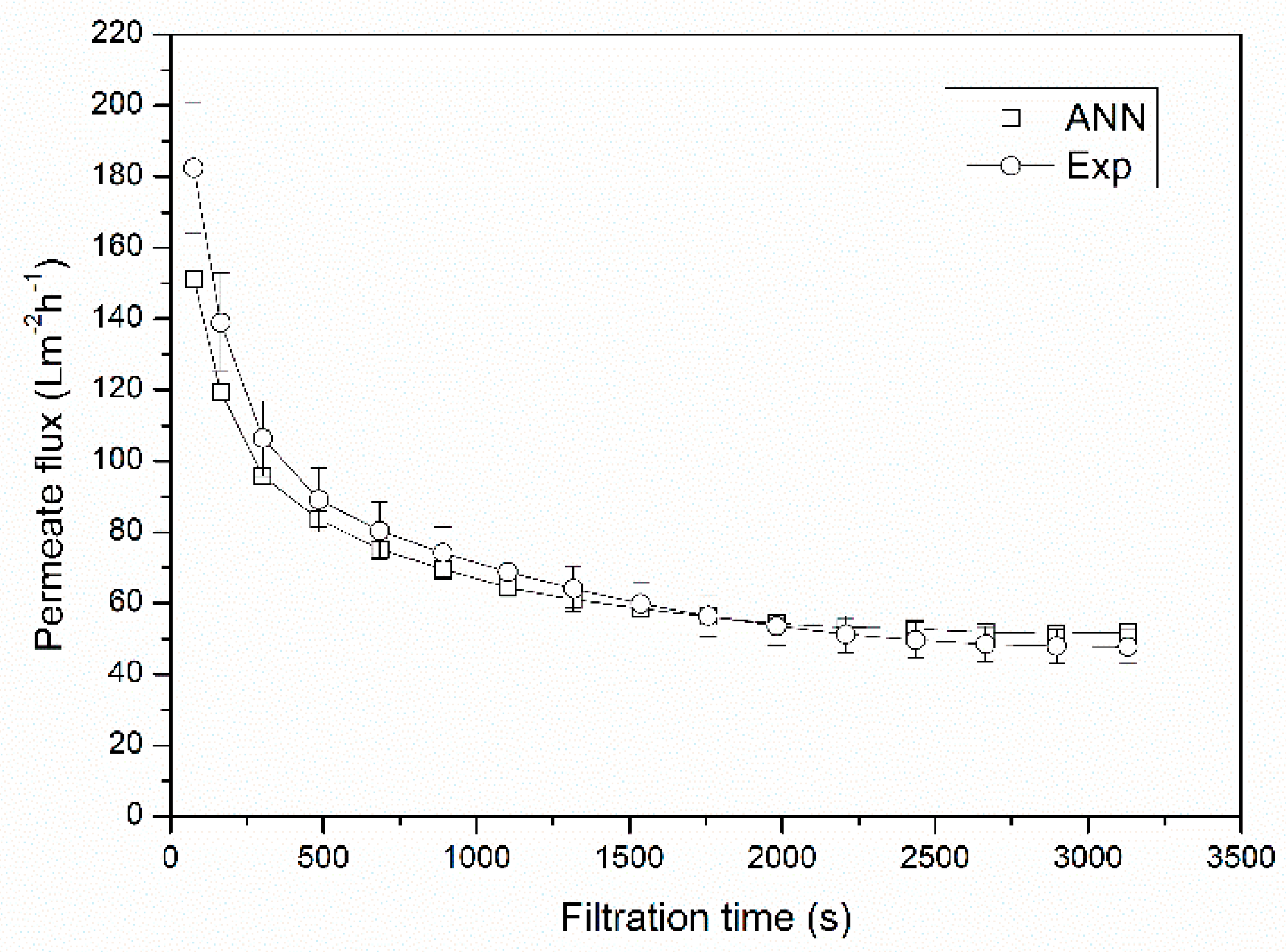
3.2. Verification of the Neural Network Model
3.3. Relative Importance of the Input Variables
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Ruiz-García, C.; Béjar, V.; Martínez-Checa, F.; Llamas, I.; Quesada, E. Bacillus velezensis sp. nov., a surfactant-producing bacterium isolated from the river Vélez in Málaga, southern Spain. Int. J. Syst. Evol. Microbiol. 2005, 55, 191–195. [Google Scholar] [CrossRef]
- Fan, B.; Wang, C.; Song, X.; Ding, X.; Wu, L.; Wu, H.; Gao, X.; Borriss, R. Bacillus velezensis FZB42 in 2018: The Gram-Positive Model Strain for Plant Growth Promotion and Biocontrol. Front. Microbiol. 2018, 9, 2491. [Google Scholar] [CrossRef] [PubMed]
- Meena, K.R.; Tandon, T.; Sharma, A.; Kanwar, S.S. Lipopeptide antibiotic production by Bacillus velezensis KLP2016. J. Appl. Pharm. Sci. 2018, 8, 91–98. [Google Scholar] [CrossRef]
- Wang, C.; Zhao, D.; Qi, G.; Mao, Z.; Hu, X.; Du, B.; Liu, K.; Ding, Y. Effects of Bacillus velezensis FKM10 for Promoting the Growth of Malus hupehensis Rehd. and Inhibiting Fusarium verticillioides. Front. Microbiol. 2020, 10, 2889. [Google Scholar] [CrossRef] [PubMed]
- Rabbee, M.F.; Ali, S.; Choi, J.; Hwang, B.S.; Jeong, S.C.; Baek, K.-H. Bacillus velezensis: A Valuable Member of Bioactive Molecules within Plant Microbiomes. Molecules 2019, 24, 1046. [Google Scholar] [CrossRef] [PubMed]
- Pajčin, I.; Vlajkov, V.; Frohme, M.; Grebinyk, S.; Grahovac, M.S.; Mojićević, M.; Grahovac, J.A. Pepper Bacterial Spot Control by Bacillus velezensis: Bioprocess Solution. Microorganisms 2020, 8, 1463. [Google Scholar] [CrossRef]
- Grahovac, J.; Pajčin, I.; Vlajkov, V.; Rončević, Z.; Dodić, J.; Cvetković, D.; Jokić, A. Xanthomonas campestris biocontrol agent: Selection, medium formulation and bioprocess kinetic analysis. Chem. Ind. Chem. Eng. Q 2020, 32. [Google Scholar] [CrossRef]
- Tomczak, W.; Gryta, M. Cross-Flow Microfiltration of Glycerol Fermentation Broths with Citrobacter freundii. Membranes 2020, 10, 67. [Google Scholar] [CrossRef]
- Chew, J.W.; Kilduff, J.; Belfort, G. The behavior of suspensions and macromolecular solutions in crossflow microfiltration: An update. J. Membr. Sci. 2020, 601, 117865. [Google Scholar] [CrossRef]
- Chang, M.; Zhou, S.; Sun, Q.; Li, T.; Ni, J. Recovery ofBacillus thuringiensisbased biopesticides from fermented sludge by cross-flow microfiltration. Desalination Water Treat. 2012, 43, 17–28. [Google Scholar] [CrossRef]
- Fan, R.; Ebrahimi, M.; Quitmann, H.; Czermak, P. Lactic acid production in a membrane bioreactor system with thermophilicBacillus coagulans: Fouling analysis of the used ceramic membranes. Sep. Sci. Technol. 2015, 50, 2177–2189. [Google Scholar] [CrossRef]
- Zhang, Y.; Fu, Q. Algal fouling of microfiltration and ultrafiltration membranes and control strategies: A review. Sep. Purif. Technol. 2018, 203, 193–208. [Google Scholar] [CrossRef]
- Jokić, A.; Zavargo, Z.; Šereš, Z.; Tekić, M. The effect of turbulence promoter on cross-flow microfiltration of yeast suspensions: A response surface methodology approach. J. Membr. Sci. 2010, 350, 269–278. [Google Scholar] [CrossRef]
- Jokić, A.; Pajčin, I.; Grahovac, J.A.; Lukić, N.L.; Dodić, J.; Rončević, Z.; Šereš, Z. Energy efficient turbulence promoter flux-enhanced microfiltration for the harvesting of rod-shaped bacteria using tubular ceramic membrane. Chem. Eng. Res. Des. 2019, 150, 359–368. [Google Scholar] [CrossRef]
- Jokić, A.; Nikolić, N.; Lukić, N.L.; Grahovac, J.A.; Dodić, J.; Rončević, Z.; Šereš, Z. Dynamic Modeling of Streptomyces hygroscopicus Fermentation Broth Microfiltration by Artificial Neural Networks. Period. Polytech. Chem. Eng. 2019, 63, 541–547. [Google Scholar] [CrossRef]
- Jokić, A.; Pajčin, I.; Grahovac, J.A.; Lukić, N.; Dodić, J.; Rončević, Z.; Šereš, Z. Improving energy efficiency of Bacillus velezensis broth microfiltration in tubular ceramic membrane by air sparging and turbulence promoter. J. Chem. Technol. Biotechnol. 2019, 95, 1110–1115. [Google Scholar] [CrossRef]
- Hartinger, M.; Napiwotzki, J.; Schmid, E.-M.; Hoffmann, D.; Kurz, F.; Kulozik, U. Influence of Spacer Design and Module Geometry on the Filtration Performance during Skim Milk Microfiltration with Flat Sheet and Spiral-Wound Membranes. Membranes 2020, 10, 57. [Google Scholar] [CrossRef]
- Krstić, D.M.; Tekić, M.N.; Carić, M.Đ.; Milanović, S.D. The effect of turbulence promoter on cross-flow microfiltration of skim milk. J. Membr. Sci. 2002, 208, 303–314. [Google Scholar] [CrossRef]
- Ogunbiyi, O.O.; Miles, N.J.; Hilal, N. Comparison of Different Pitch Lengths on Static Promoters for Flux Enhancement in Tubular Ceramic Membrane. Sep. Sci. Technol. 2007, 42, 1945–1963. [Google Scholar] [CrossRef]
- Liu, Y.; He, G.; Tan, M.; Nie, F.; Li, B. Artificial neural network model for turbulence promoter-assisted crossflow microfiltration of particulate suspensions. Desalination 2014, 338, 57–64. [Google Scholar] [CrossRef]
- Šereš, L.; Dokić, L.; Ikonić, B.; Šoronja-Simović, D.; Djordjević, M.; Žana, Š.; Maravić, N. Data-driven Modelling of Microfiltration Process with Embedded Static Mixer for Steepwater from Corn Starch Industry. Period. Polytech. Chem. Eng. 2017, 62, 114–122. [Google Scholar] [CrossRef]
- Pospísil, P. Shear stress-based modelling of steady state permeate flux in microfiltration enhanced by two-phase flows. Chem. Eng. J. 2004, 97, 257–263. [Google Scholar] [CrossRef]
- Hwang, K.-J.; Hsu, C.-E. Effect of gas–liquid flow pattern on air-sparged cross-flow microfiltration of yeast suspension. Chem. Eng. J. 2009, 151, 160–167. [Google Scholar] [CrossRef]
- Hwang, K.-J.; Chen, L. Effect of air-sparging on the cross-flow microfiltration of microbe/protein bio-suspension. J. Taiwan Inst. Chem. Eng. 2010, 41, 564–569. [Google Scholar] [CrossRef]
- Armbruster, S.; Brochard, A.; Lölsberg, J.; Yüce, S.; Wessling, M. Aerating static mixers prevent fouling. J. Membr. Sci. 2019, 537–546. [Google Scholar] [CrossRef]
- Vatai, G.N.; Krstić, D.M.; Höflinger, W.; Koris, A.K.; Tekic, M.N. Combining air sparging and the use of a static mixer in cross-flow ultrafiltration of oil/water emulsion. Desalination 2007, 204, 255–264. [Google Scholar] [CrossRef]
- Asghari, M.; Dashti, A.; Rezakazemi, M.; Jokar, E.; Halakoei, H. Application of neural networks in membrane separation. Rev. Chem. Eng. 2020, 36, 265–310. [Google Scholar] [CrossRef]
- Hermia, J. Blocking Filtration. Application to Non-Newtonian Fluids. In Mathematical Models and Design Methods in Solid-Liquid Separation; Springer Science and Business Media LLC: Berlin/Heidelberg, Germany, 1985; pp. 83–89. [Google Scholar]
- Bowen, W.; Jenner, F. Theoretical descriptions of membrane filtration of colloids and fine particles: An assessment and review. Adv. Colloid Interface Sci. 1995, 56, 141–200. [Google Scholar] [CrossRef]
- Anis, S.; Hashaikeh, R.; Hilal, N. Microfiltration membrane processes: A review of research trends over the past decade. J. Water Process. Eng. 2019, 32, 100941. [Google Scholar] [CrossRef]
- Jokić, A.I.; Šereš, L.L.; Milović, N.R.; Šereš, Z.I.; Maravić, N.R.; Šaranovic, Ž.; Dokić, L.P. Modelling of starch industry wastewater microfiltration parameters by neural network. Membr. Water Treat 2018, 9, 115–121. [Google Scholar] [CrossRef]
- Al-Abri, M.; Hilal, N. Artificial neural network simulation of combined humic substance coagulation and membrane filtration. Chem. Eng. J. 2008, 141, 27–34. [Google Scholar] [CrossRef]
- Avarzaman, E.M.; Zarafshan, P.; Mirsaeedghazi, H.; Alaeddini, B. Intelligent Modeling of Permeate Flux during Membrane Clarification of Pomegranate Juice. Nutr. Food Sci. Res. 2017, 4, 29–38. [Google Scholar] [CrossRef]
- Da Silva, I.N.; Flauzino, R.A. An approach based on neural networks for estimation and generalization of crossflow filtration processes. Appl. Soft Comput. 2008, 8, 590–598. [Google Scholar] [CrossRef]
- Dornier, M.; Decloux, M.; Trystram, G.; Lebert, A. Dynamic modeling of crossflow microfiltration using neural networks. J. Membr. Sci. 1995, 98, 263–273. [Google Scholar] [CrossRef]
- Hamachi, M.; Cabassud, M.; Davin, A.; Peuchot, M.M. Dynamic modelling of crossflow microfiltration of bentonite suspension using recurrent neural networks. Chem. Eng. Process. Process. Intensif. 1999, 38, 203–210. [Google Scholar] [CrossRef]
- Chellam, S. Artificial neural network model for transient crossflow microfiltration of polydispersed suspensions. J. Membr. Sci. 2005, 258, 35–42. [Google Scholar] [CrossRef]
- Aydiner, C.; Demir, I.; Yildiz, E. Modeling of flux decline in crossflow microfiltration using neural networks: The case of phosphate removal. J. Membr. Sci. 2005, 248, 53–62. [Google Scholar] [CrossRef]
- Fu, R.; Xu, T.; Pan, Z. Modelling of the adsorption of bovine serum albumin on porous polyethylene membrane by back-propagation artificial neural network. J. Membr. Sci. 2005, 251, 137–144. [Google Scholar] [CrossRef]
- Cheng, L.-H.; Cheng, Y.; Chen, J. Predicting effect of interparticle interactions on permeate flux decline in CMF of colloidal suspensions: An overlapped type of local neural network. J. Membr. Sci. 2008, 308, 54–65. [Google Scholar] [CrossRef]
- Hilal, N.; Ogunbiyi, O.O.; Al-Abri, M. Neural network modeling for separation of bentonite in tubular ceramic membranes. Desalination 2008, 228, 175–182. [Google Scholar] [CrossRef]
- Liu, Q.-F.; Kim, S.H.; Lee, S. Prediction of microfiltration membrane fouling using artificial neural network models. Sep. Purif. Technol. 2009, 70, 96–102. [Google Scholar] [CrossRef]
- Guadix, A.; Zapata, J.E.; Almécija, M.C.; Guadix, E.M. Predicting the flux decline in milk cross-flow ceramic ultrafiltration by artificial neural networks. Desalination 2010, 250, 1118–1120. [Google Scholar] [CrossRef]
- Hwang, T.-M.; Choi, Y.; Nam, S.-H.; Lee, S.; Oh, H.; Hyun, K.; Choung, Y.-K. Prediction of membrane fouling rate by neural network modeling. Desalination Water Treat. 2010, 15, 134–140. [Google Scholar] [CrossRef]
- Nandi, B.; Moparthi, A.; Uppaluri, R.; Purkait, M. Treatment of oily wastewater using low cost ceramic membrane: Comparative assessment of pore blocking and artificial neural network models. Chem. Eng. Res. Des. 2010, 88, 881–892. [Google Scholar] [CrossRef]
- Mhurchú, J.N.; Foley, G.; Havel, J. Modeling process dynamics using a novel neural network architecture: Application to stirred cell microfiltration. Chem. Eng. Commun. 2010, 197, 1152–1162. [Google Scholar] [CrossRef]
- Soleimani, R.; Shoushtari, N.A.; Mirza, B.; Salahi, A. Experimental investigation, modeling and optimization of membrane separation using artificial neural network and multi-objective optimization using genetic algorithm. Chem. Eng. Res. Des. 2013, 91, 883–903. [Google Scholar] [CrossRef]
- Nourbakhsh, H.; Emam-Djomeh, Z.; Omid, M.; Mirsaeedghazi, H.; Moini, S. Prediction of red plum juice permeate flux during membrane processing with ANN optimized using RSM. Comput. Electron. Agric. 2014, 102, 1–9. [Google Scholar] [CrossRef]
- Shahriari, S.; Hakimzadeh, V.; Shahidi, M. Modeling the efficiency of microfiltration process in reducing the hardness, improvement the non-sugar component rejection and purity of raw sugar beet juice. Ukr. Food J. 2017, 6, 648–660. [Google Scholar] [CrossRef]
- Corbatón-Báguena, M.-J.; Vincent-Vela, M.; Gozálvez-Zafrilla, J.-M.; Álvarez-Blanco, S.; Lora, J.; Catalán-Martínez, D. Comparison between artificial neural networks and Hermia’s models to assess ultrafiltration performance. Sep. Purif. Technol. 2016, 170, 434–444. [Google Scholar] [CrossRef]
- Demuth, H.; Beale, M. Neural Network Toolbox User’s Guide, 4th ed.; The MathWorks, Inc.: Natick, MA, USA, 2004. [Google Scholar]
- Tanaka, T.; Abe, K.-I.; Asakawa, H.; Yoshida, H.; Nakanishi, K. Filtration characteristics and structure of cake in crossflow filtration of bacterial suspension. J. Ferment. Bioeng. 1994, 78, 455–461. [Google Scholar] [CrossRef]
| Input Variable | Value | |
|---|---|---|
| Without Static Mixer | With Static Mixer | |
| Static mixer (-) | 0 | 1 |
| Transmembrane pressure (bar) | 0.2; 0.6; 1.0 | 0.2; 0.6; 1.0 |
| Superficial feed velocity (m∙s−1) | 0.43; 0.87; 1.30 | 0.53; 1.06; 1.59 |
| Superficial air velocity (m∙s−1) | 0.0; 0.2; 0.4 | 0.0; 0.23; 0.46 |
| Filtration time (s) | 0—time to reach stationary flux | |
| ANN Type | Training Algorithm | Transfer Function | |
|---|---|---|---|
| Input-Hidden Layer | Hidden-Output Layer | ||
| A | trainlm | logsig | puerlin |
| B | trainlm | tansig | |
| C | trainbr | logsig | |
| D | trainbr | tansig | |
| Absolute Relative Error (%) | <1 | <5 | <10 | <20 | >20 | Sum |
|---|---|---|---|---|---|---|
| Number of data | 274 | 470 | 199 | 108 | 64 | 1115 |
| Percentage of data (%) | 25 | 42 | 18 | 10 | 6 | 100 |
| Input | Importance (%) | Rank |
|---|---|---|
| Static mixer (-) | 13.13 | 3 |
| Transmembrane pressure (bar) | 9.44 | 5 |
| Superficial air velocity (m∙s−1) | 15.77 | 2 |
| Superficial feed velocity (m∙s−1) | 11.36 | 4 |
| Filtration time (s) | 50.30 | 1 |
| TOTAL: | 100 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jokić, A.; Pajčin, I.; Grahovac, J.; Lukić, N.; Ikonić, B.; Nikolić, N.; Vlajkov, V. Dynamic Modeling Using Artificial Neural Network of Bacillus Velezensis Broth Cross-Flow Microfiltration Enhanced by Air-Sparging and Turbulence Promoter. Membranes 2020, 10, 372. https://doi.org/10.3390/membranes10120372
Jokić A, Pajčin I, Grahovac J, Lukić N, Ikonić B, Nikolić N, Vlajkov V. Dynamic Modeling Using Artificial Neural Network of Bacillus Velezensis Broth Cross-Flow Microfiltration Enhanced by Air-Sparging and Turbulence Promoter. Membranes. 2020; 10(12):372. https://doi.org/10.3390/membranes10120372
Chicago/Turabian StyleJokić, Aleksandar, Ivana Pajčin, Jovana Grahovac, Nataša Lukić, Bojana Ikonić, Nevenka Nikolić, and Vanja Vlajkov. 2020. "Dynamic Modeling Using Artificial Neural Network of Bacillus Velezensis Broth Cross-Flow Microfiltration Enhanced by Air-Sparging and Turbulence Promoter" Membranes 10, no. 12: 372. https://doi.org/10.3390/membranes10120372
APA StyleJokić, A., Pajčin, I., Grahovac, J., Lukić, N., Ikonić, B., Nikolić, N., & Vlajkov, V. (2020). Dynamic Modeling Using Artificial Neural Network of Bacillus Velezensis Broth Cross-Flow Microfiltration Enhanced by Air-Sparging and Turbulence Promoter. Membranes, 10(12), 372. https://doi.org/10.3390/membranes10120372








