A Critical Study of the Effect of Polymeric Fibers on the Performance of Supported Liquid Membranes in Sample Microextraction for Metals Analysis
Abstract
:1. Introduction
2. Materials and Methods
2.1. Reagents and Solutions
2.2. Apparatus
2.3. Set up of the Hollow Fiber-Supported Liquid Membranes Experiments
- A piece of fiber of the desired length was cut using a blade.
- One of the fiber’s ends was thermally sealed using a hot tip and tweezers.
- The fiber was filled with the acceptor solution and the other end was thermally sealed to make-up the 3SBME.
- The 3SBME was placed into an organic solution containing the carrier for impregnation of the pores.
- The fiber was left free in the sample for extraction.
- The desired fiber length was cut.
- The lumen of the fiber was filled with the acceptor solution using a microsyringe.
- In order to impregnate the pores, the fiber was immersed in the organic solution, still connected to the syringe.
- The lumen was again flushed with acceptor solution and the fiber ends were put together and sealed using paraffin film.
- The fiber was placed into the sample for extraction.
2.4. Quantification of Transport Efficiency
3. Results and Discussion
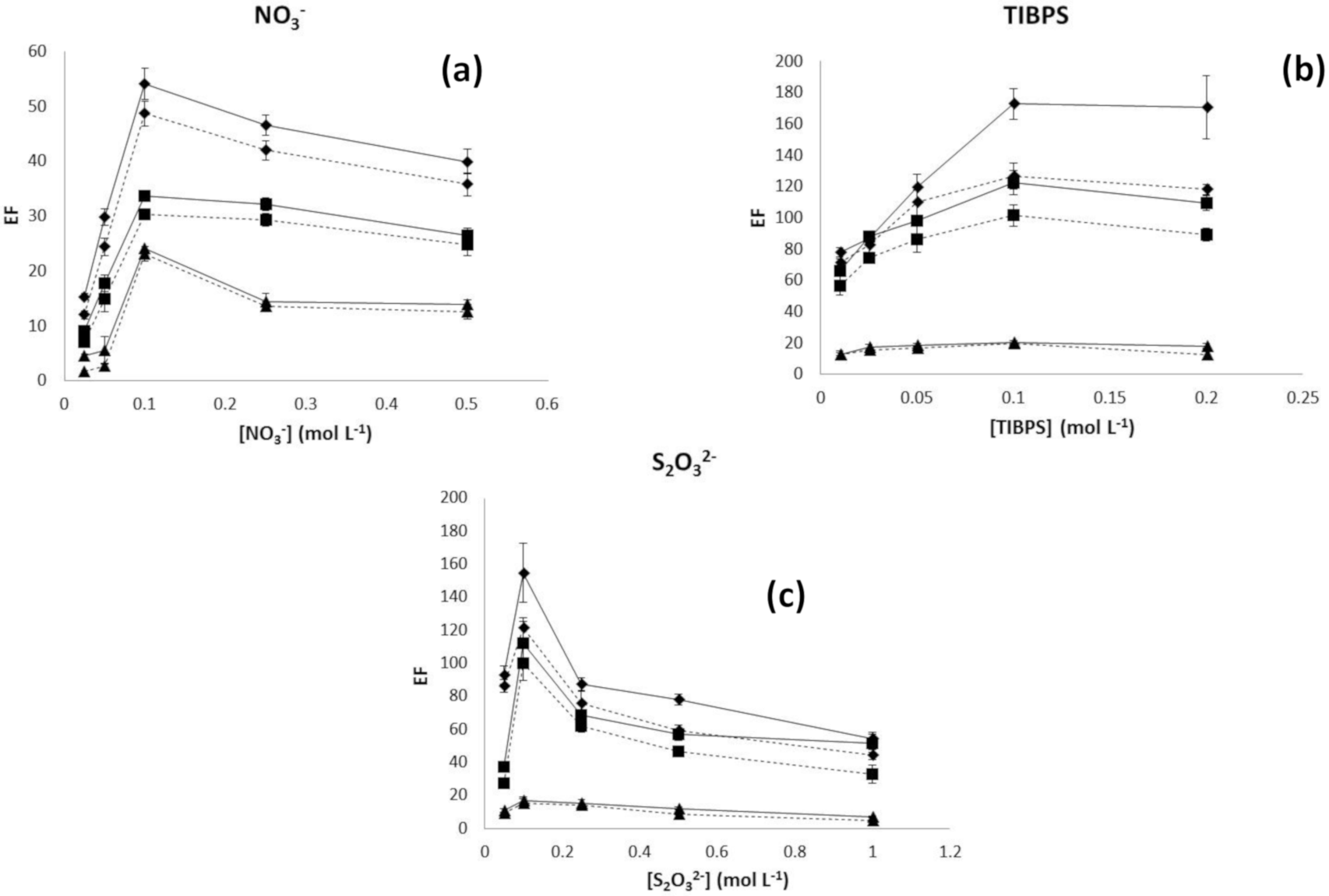
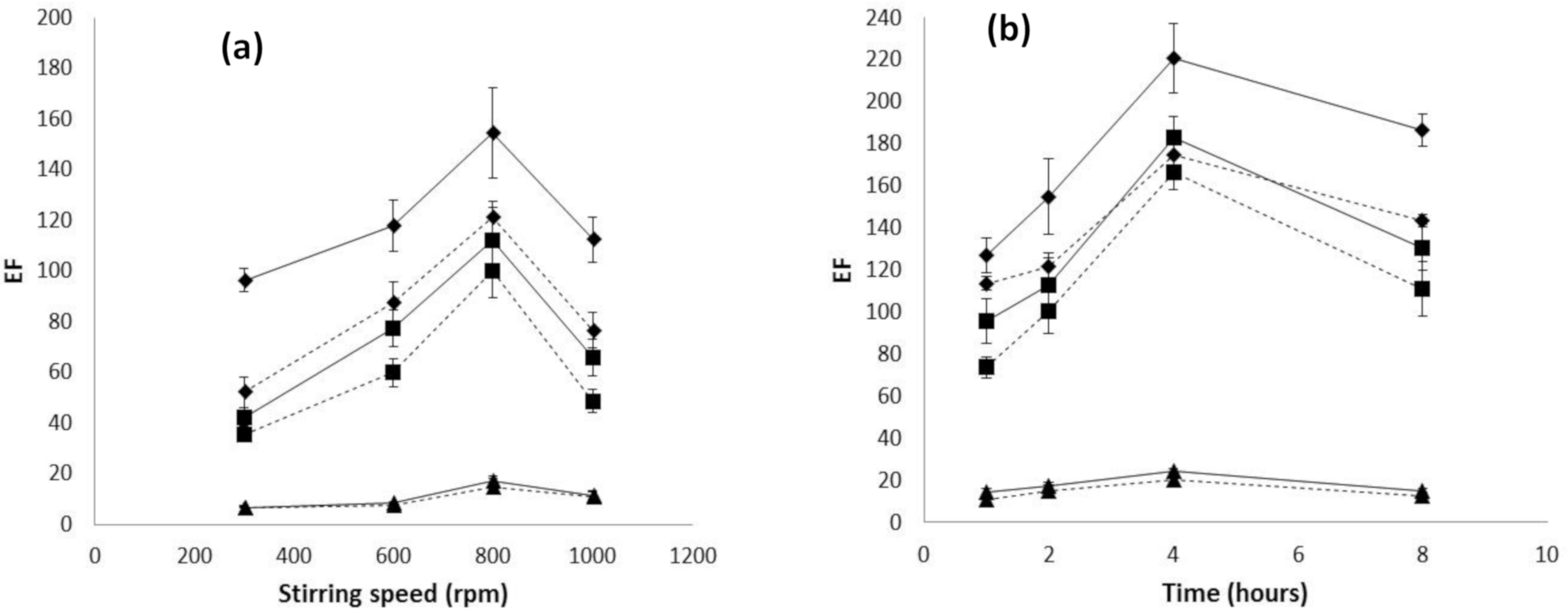
3.1. Influence of Hollow Fiber Characteristics on Transport Efficiency
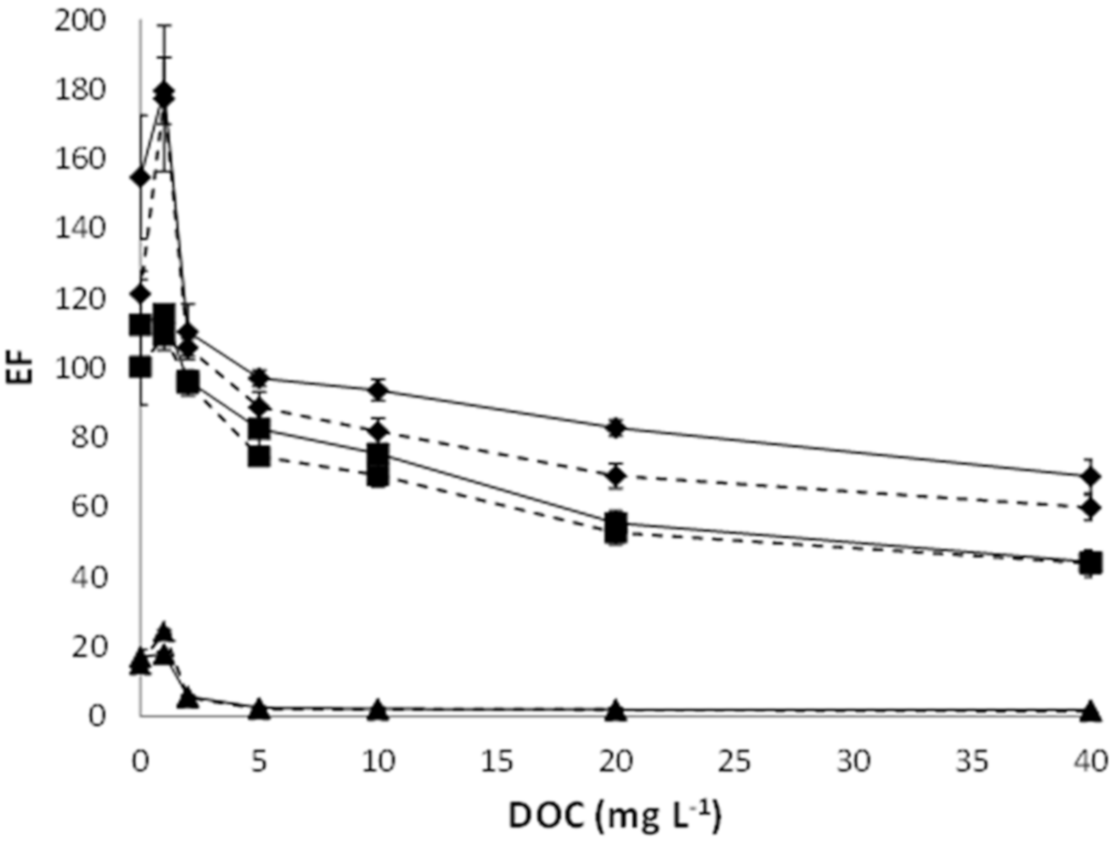
3.2. Relationship between the Effect of Organic Matter Concentration and Polymeric Support
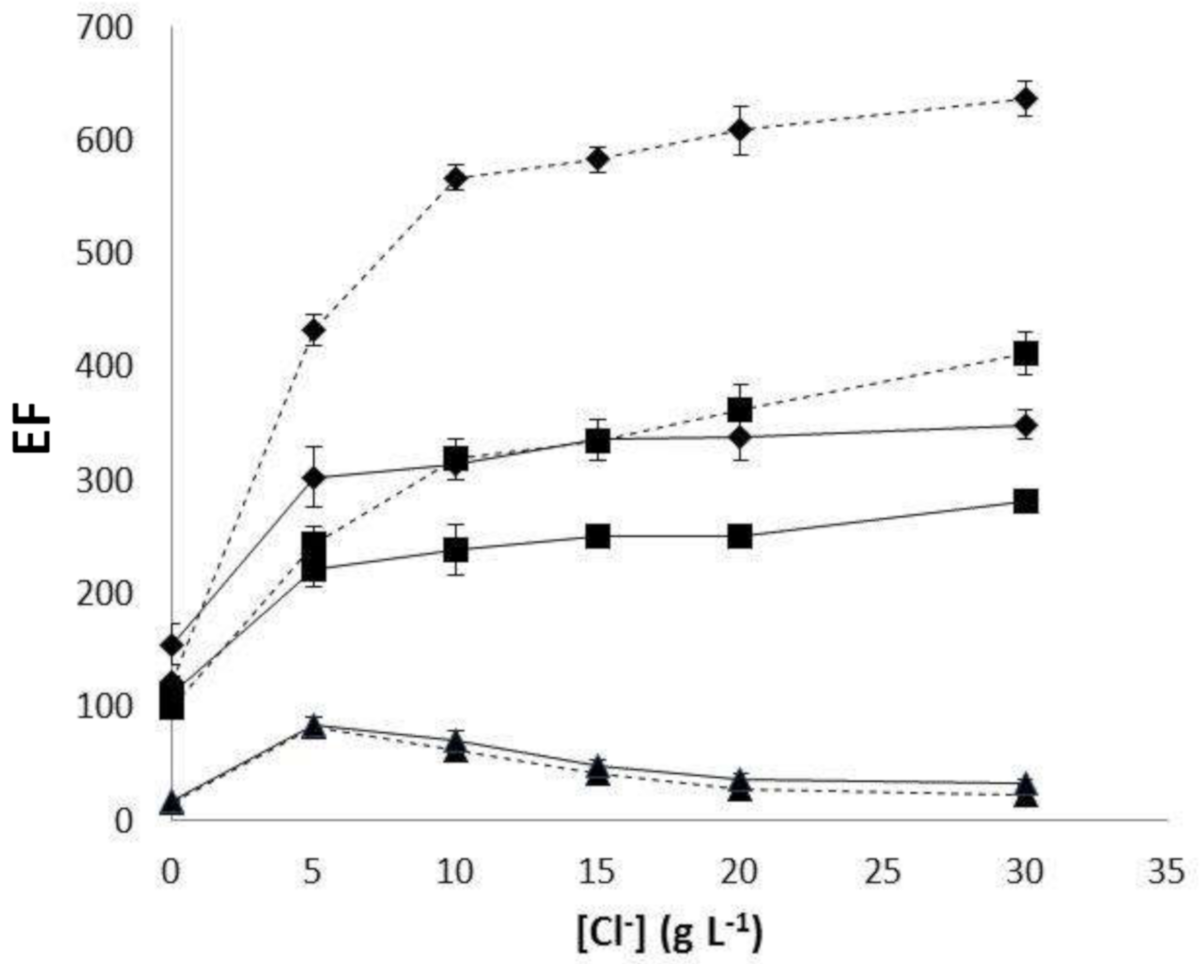
3.3. Relationships between the Effect of Sample Salinity and Polymeric Support
3.4. Selection of Fiber Type
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Khan, W.A.; Arain, M.B.; Yamini, Y.; Shah, N.; Kazi, T.G.; Pedersen-Bjergaard, S.; Tajik, M. Hollow fiber-based liquid phase microextraction followed by analytical instrumental techniques for quantitative analysis of heavy metal ions and pharmaceuticals. J. Pharm. Anal. 2020, 10, 109–122. [Google Scholar] [CrossRef] [PubMed]
- Aguirre, M.A.; Baile, P.; Vidal, L.; Canals, A. Metal applications of liquid phase micro-extraction. TRAC Trends Anal. Chem. 2019, 112, 241–247. [Google Scholar] [CrossRef]
- López-López, J.A.; Mendiguchía, C.; Pinto, J.J.; Moreno, C. Application of solvent-bar micro-extraction for the determination of organic and inorganic compounds. TRAC Trends Anal. Chem. 2019, 110, 57–65. [Google Scholar] [CrossRef]
- Herce-Sesa, B.; Herce-Sesa, B.; López-López, J.A.; Moreno, C. Selective ionic liquid solvent bar micro-extraction for estimation of ultra-trace silver fractions in marine waters. Sci. Total Environ. 2019, 650, 27–33. [Google Scholar] [CrossRef] [PubMed]
- Guo, X.; He, M.; Chen, B.; Hu, B. Phase transfer hollow fiber liquid phase microextraction combined with electrothermal vaporization inductively coupled plasma mass spectrometry for the determination of trace heavy metals in environmental and biological samples. Talanta 2012, 101, 516–523. [Google Scholar] [CrossRef] [PubMed]
- Nomngongo, P.N.; Ngila, J.C.; Msagati, T.A.M.; Moodley, B. Chemometric optimization of hollow fiber-liquid phase microextraction for preconcentration of trace elements in diesel and gasoline prior to their ICP-OES determination. Microchem. J. 2014, 114, 141–147. [Google Scholar] [CrossRef]
- Ghasemi, E.; Najafi, N.M.; Raofie, F.; Chassempour, A. Simultaneous speciation and preconcentration of ultratraces of inorganic tellurium and selenium in environmental samples by hollow fiber liquid phase microextraction prior to electrothermal atomic absorption determination. J. Hazard. Mat. 2010, 181, 491–496. [Google Scholar] [CrossRef]
- Parthasarathty, N.; Pelletier, M.; Buffle, J. Transport of lipophilic metal complexes through permeation liquid membrane in relation to natural water analysis: Cu(II)-8-hydroxyquinoline complex as a model compound. J. Mem. Sci. 2010, 355, 78–84. [Google Scholar] [CrossRef]
- Vergel, C.; Montoya, R.; Mendiguchía, C.; García-Vargas, M.; Moreno, C. HFLPME as a green alternative for the preconcentration of nickel in natural waters. Anal. Bioanal. Chem. 2012, 404, 665–670. [Google Scholar] [CrossRef]
- Bautista-Flores, A.N.; Rodríguez de San Miguel, E.; Gyves, J.; Jönsson, J.A. Optimization, evaluation, and characterization of a hollow fiber supported liquid membrane or sampling and speciation of lead (II) from aqueous solutions. J. Membr. Sci. 2010, 363, 180–187. [Google Scholar] [CrossRef]
- López-López, J.A.; Jönsson, J.A.; García-Vargas, M.; Moreno, C. Simple hollow fiber liquid membrane based pre-concentration of silver for atomic absorption spectrometry. Anal. Methods 2014, 6, 1462–1647. [Google Scholar] [CrossRef]
- Liu, Y.; Wang, Y.; Hu, Y.; Ni, L.; Han, J.; Che, T.; Chen, H.; Liu, Y. Ionic liquid-based hollow fiber liquid-phase microextraction for the determination of trace lead (II) in environmental water and tea drinks samples by graphite furnace atomic absorption spectrometry. J. Iran. Chem. Soc. 2015, 12, 371–377. [Google Scholar] [CrossRef]
- Slaveykova, V.I.; Karadjova, I.B.; Karadjov, M.; Tsalev, D.L. Trace metal speciation and bioavailability in surface waters of the Black Sea coastal area evaluated by HF-PLM and DGT. Environ. Sci. Technol. 2009, 43, 1798–1803. [Google Scholar] [CrossRef]
- Zeng, C.; Lin, Y.; Zhou, N.; Zheng, J.; Zhang, W. Room temperature ionic liquids enhanced the speciation of Cr(VI) and Cr(III) by hollow fiber liquid phase micro-extraction combined with flame atomic absorption spectrometry. J. Hazard. Mater. 2012, 237, 365–370. [Google Scholar] [CrossRef]
- Stanisz, E.; Werner, J.; Zgola-Crzeskowiak, A. Liquid-phase microextraction techniques based on ionic liquids for preconcentration and determination of metals. TRAC Trends Anal. Chem. 2014, 61, 54–66. [Google Scholar] [CrossRef]
- López-García, I.; Rivas, R.E.; Hernández-Córdoba, M. Hollow fiber based liquid-phase microextraction for the determination of mercury traces in water samples by electrothermal atomic absorption spectrometry. Anal. Chim. Acta 2012, 743, 69–74. [Google Scholar] [CrossRef]
- Herce-Sesa, H.; López-López, J.A.; Moreno, C. Ionic liquid solvent bar micro-extraction of CdCln(n−2)− species for ultra-trace Cd determination in seawater. Chemosphere 2018, 193, 306–312. [Google Scholar] [CrossRef]
- Herce-Sesa, B.; López-López, J.A.; Moreno, C. Multi-element ionic liquid-based solvent bar micro-extraction of priority and emerging trace metallic pollutants (Cd, Ag, Pd) in natural waters. J. Hazard. Mater. 2019, 370, 63–69. [Google Scholar] [CrossRef]
- Wongsawa, T.; Sunsandee, N.; Pancharoen, U.; Lothongkum, A.W. High-efficiency HFSLM for silver–ion pertraction from pharmaceutical wastewater and mass-transport models. Chem. Eng. Res. Des. 2014, 92, 2691–2693. [Google Scholar] [CrossRef]
- Abdullah, S.Z.; Berube, P.; Horne, D.J. SEM imaging of membranes: Importance of sample preparation and imaging parameters. J. Membr. Sci. 2014, 463, 113–125. [Google Scholar] [CrossRef]
- López-López, J.A.; Herce-Sesa, B.; Moreno, C. Three-phase solvent bar micro-extraction as an approach to silver ultra-traces speciation in estuarine water samples. Talanta 2015, 382–386. [Google Scholar] [CrossRef] [PubMed]
- López-López, J.A.; García-Vargas, M.; Moreno, C. A new analytical method for selective pre-concentration of free silver in estuarine waters using liquid membranes. Talanta 2013, 108, 7–10. [Google Scholar] [CrossRef] [PubMed]
- González-Álvarez, R.; Pinto, J.J.; López-López, J.A.; Mendiguchía, C.; Moreno, C. Selective solvent bar micro-extraction as a single-step approach for the measurement of Cu fractions in seawater. Anal. Bioanal. Chem. 2020, 412, 1863–1870. [Google Scholar] [CrossRef]
- Mendiguchía, C.; Moreno, C.; García-Vargas, M. Separation of heavy metal in seawater by liquid membranes: Preconcentration of copper. Sep. Sci. Technol. 2002, 37, 2337–2351. [Google Scholar] [CrossRef]
- López-López, J.A.; Mendiguchía, C.; Pinto, J.J.; Moreno, C. Liquid membranes for quantification and speciation of trace metals in natural waters. TRAC Trends Anal. Chem. 2010, 29, 645–653. [Google Scholar] [CrossRef]
| Fiber | Internal Diameter (µm) | Pore Size (µm) | Porosity (%) | Wall Thickness (µm) | Effective Surface (mm2) | Length (mm) |
|---|---|---|---|---|---|---|
| 50/280 | 280 | 0.1 | 60 | 50 | 211 | 400 |
| Q3/2 | 600 | 0.2 | 75 | 200 | 127 | 90 |
| S6/2 | 1800 | 0.2 | 72 | 450 | 41 | 10 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
González-Álvarez, R.J.; López-López, J.A.; Pinto, J.J.; Moreno, C. A Critical Study of the Effect of Polymeric Fibers on the Performance of Supported Liquid Membranes in Sample Microextraction for Metals Analysis. Membranes 2020, 10, 275. https://doi.org/10.3390/membranes10100275
González-Álvarez RJ, López-López JA, Pinto JJ, Moreno C. A Critical Study of the Effect of Polymeric Fibers on the Performance of Supported Liquid Membranes in Sample Microextraction for Metals Analysis. Membranes. 2020; 10(10):275. https://doi.org/10.3390/membranes10100275
Chicago/Turabian StyleGonzález-Álvarez, Rafael J., José A. López-López, Juan J. Pinto, and Carlos Moreno. 2020. "A Critical Study of the Effect of Polymeric Fibers on the Performance of Supported Liquid Membranes in Sample Microextraction for Metals Analysis" Membranes 10, no. 10: 275. https://doi.org/10.3390/membranes10100275
APA StyleGonzález-Álvarez, R. J., López-López, J. A., Pinto, J. J., & Moreno, C. (2020). A Critical Study of the Effect of Polymeric Fibers on the Performance of Supported Liquid Membranes in Sample Microextraction for Metals Analysis. Membranes, 10(10), 275. https://doi.org/10.3390/membranes10100275








