Changing Attitudes toward the COVID-19 Vaccine among North Carolina Participants in the COVID-19 Community Research Partnership
Abstract
:1. Introduction
2. Materials and Methods
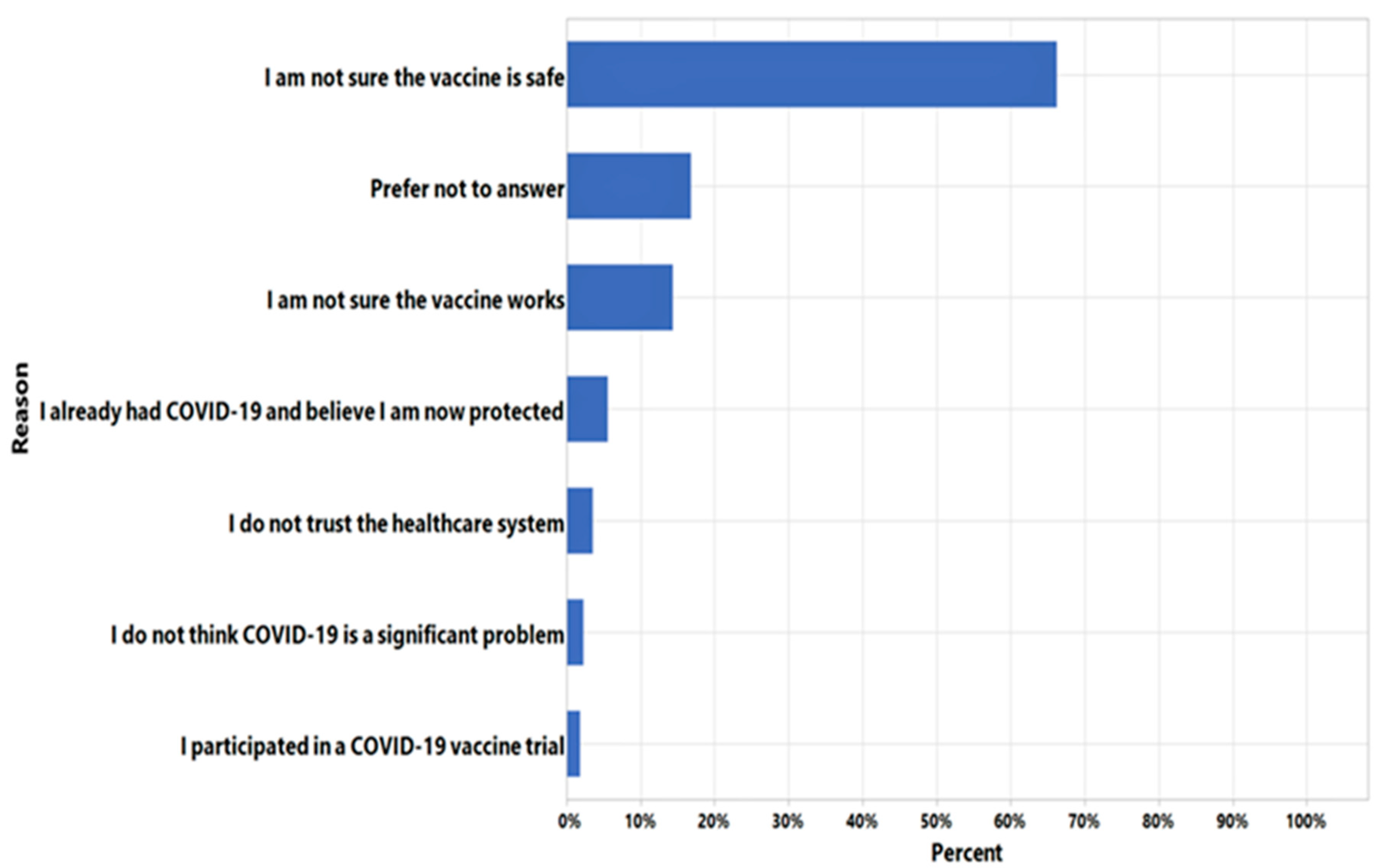
3. Results
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| BIPOC | Blacks, Indigenous and People of color |
| CCRP | COVID-19 Community Research Partnership |
| CDC | Centers for Disease Control and Prevention |
| COVID-19 | Coronavirus disease 2019 |
| EUA | Emergency Use Authorization |
| FDA | Food and Drug Administration |
| NC | North Carolina |
| US | United States |
References
- COVID-19 Vaccines. Available online: https://www.fda.gov/emergency-preparedness-and-response/coronavirus-disease-2019-covid-19/covid-19-vaccines (accessed on 27 April 2021).
- Hamel, L. KFF COVID-19 Vaccine Monitor; Kaiser Family Foundation (KFF). Available online: https://www.kff.org/coronavirus-covid-19/poll-finding/kff-covid-19-vaccine-monitor-april-2021/ (accessed on 15 May 2021).
- Salvanto, A.; De Pinto, J.; Backus, F.; Khanna, K. Many Would Get the COVID-19 Vaccine, But Hesitancy Remains—CBS News Poll. 26 February 2021. Available online: https://www.cbsnews.com/news/covid-vaccine-hesitancy-opinion-poll/ (accessed on 27 April 2021).
- Debruyn, J. Polls Show North Carolinians Are Increasingly Willing to Get COVID-19 Vaccine. 16 February 2021. Available online: https://www.wunc.org/health/2021-02-16/polls-show-north-carolinians-are-increasingly-willing-to-get-covid-19-vaccine (accessed on 27 April 2021).
- COVID-19 Community Research Partnership. Available online: https://www.wakehealth.edu/Coronavirus/COVID-19-Community-Research-Partnership (accessed on 19 July 2021).
- NC Rural Center. North Carolina Counties. Available online: https://www.ncruralcenter.org/about-us/ (accessed on 27 April 2021).
- Lazarus, J.V.; Ratzan, S.C.; Palayew, A.; Gostin, L.O.; Larson, H.J.; Rabin, K.; Kimball, S.; El-Mohandes, A. A Global Survey of Potential acceptance of a COVID-19 Vaccine. Nat. Med. 2021, 27, 225–228. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, K.H.; Srivastav, A.; Razzaghi, H.; Williams, W.; Lindley, M.C.; Jorgensen, C.; Abad, N.; Singleton, J.A. COVID-19 Vaccination Intent, Perceptions, and Reasons for Not Vaccinating Among Groups Prioritized for Early Vaccination—United States, September and December 2020. MMWR 2021, 70, 217–222. [Google Scholar] [PubMed]
- Funk, G.; Tyson, A. Growing Share of Americans Say They Plan to Get a COVID-19 Vaccine—Or Already Have—77% Think Vaccinations Will Benefit U.S. Economy. Pew Center Research Science and Society. 5 March 2021. Available online: https://www.pewresearch.org/science/2021/03/05/growing-share-of-americans-say-they-plan-to-get-a-covid-19-vaccine-or-already-have/ (accessed on 30 April 2021).
- Khubchandani, J.; Sharma, S.; Price, J.H.; Wiblishauser, M.J.; Sharma, M.; Webb, F.J. COVID-19 Vaccination Hesitancy in the United States: A Rapid National Assessment. J. Community Health 2020, 46, 270–277. [Google Scholar] [CrossRef] [PubMed]
- Gold, J.A.; Rossen, L.M.; Ahmad, F.B.; Sutton, P.; Li, Z.; Salvatore, P.P.; Coyle, J.; DeCuir, J.; Baack, B.; Durant, T.; et al. Race, Ethnicity, and Age Trends in Persons Who Died from COVID-19—United States, May–August 2020. MMWR 2020, 69, 1517–1521. [Google Scholar] [PubMed]
- APM Research Lab Staff. The Color of Coronavirus: COVID-19 Deaths by Race and Ethnicity in the U.S. 5 March 2021. Available online: https://www.apmresearchlab.org/covid/deaths-by-race (accessed on 28 April 2021).
- Fletcher, M.A. New poll shows Black Americans see a racist health care system setting the stage for pandemic’s impact even as they lose family members and jobs, most are reluctant to try a vaccine. Undefeated 2020. Available online: https://theundefeated.com/features/new-poll-shows-black-americans-see-a-racist-health-care-system-setting-the-stage-for-pandemics-impact/ (accessed on 28 April 2021).
- O’Neal, L. Half of Black adults say they won’t take a coronavirus vaccine. Undefeated 2020. Available online: https://theundefeated.com/features/half-of-black-adults-say-they-wont-take-a-coronavirus-vaccine/ (accessed on 28 April 2021).
- Summers, J. Little Difference in Vaccine Hesitancy among White and Black Americans, Poll Finds. 12 March 2021. Available online: https://www.npr.org/ (accessed on 28 April 2021).
- Huetteman, E. Covid vaccine hesitancy drops among all Americans, new survey shows. KHN 2021. Available online: https://www.healthleadersmedia.com/covid-19/covid-vaccine-hesitancy-drops-among-all-americans-new-survey-shows (accessed on 29 April 2021).
- Violette, R.; Pullagura, G.R. Vaccine hesitancy: Moving practice beyond binary vaccination outcomes in community pharmacy. Can. Pharm. J. 2019, 152, 391–394. [Google Scholar] [CrossRef] [PubMed]
- Mascola, J.R.; Graham, B.S.; Fauci, A.S. SARS-CoV-2 Viral Variants—Tackling a Moving Target. JAMA 2021, 325, 1261–1262. [Google Scholar] [CrossRef] [PubMed]
- Bunn, C. ‘Getting a clearer picture’: Black Americans on the Factors That Overcame Their Vaccine Hesitancy. NBC News, 12 April 2021. Available online: https://www.nbcnews.com/news/nbcblk/getting-clearer-picture-black-americans-factors-overcame-their-vaccine-hesitancy-n1263787(accessed on 29 April 2021).
- Working Group on Equity in COVID-19 Vaccination; John Hopkins School of Public Health. Equity in Vaccination: A Plan to Work with Communities of Color toward COVID-19 Recovery and Beyond. February 2021. Available online: https://www.centerforhealthsecurity.org/ (accessed on 30 April 2021).
- CDC COVID-19 Task Force. Building Confidence in COVID-19 Vaccines among Your Patients. Tips for Healthcare Teams. January 2021. Available online: https://www.cdc.gov/vaccines/covid-19/downloads/VaccinateWConfidence-TipsForHCTeams_508.pdf (accessed on 30 April 2021).
- Craig Palosky. Vaccine Monitor: Some Who Were Hesitant to Get a Vaccine in January Say They Changed Their Mind Because of Family, Friends and Their Personal Doctors. 13 July 2020. Available online: https://www.kff.org/coronavirus-covid-19/press-release/vaccine-monitor-some-who-were-hesitant-to-get-a-vaccine-in-january-say-they-changed-their-mind-because-of-family-friends-and-their-personal-doctors/ (accessed on 30 April 2021).
| Total | Vaccine Status | |||||||
|---|---|---|---|---|---|---|---|---|
| Vaccinated | Non-Vaccinated | |||||||
| n | n | Percent | n | Percent | Relative Risk | 95% CI | p-Value | |
| TOTAL | 18,874 | 17,461 | 92.5 | 1413 | 7.5 | |||
| Vaccine Intent | ||||||||
| Yes | 14,810 | 14,582 | 98.5 | 228 | 1.5 | ref. | ref. | ref. |
| No | 1358 | 715 | 52.7 | 643 | 47.3 | 0.65 | 0.62–0.67 | <0.0001 |
| Undecided | 2403 | 1929 | 80.3 | 474 | 19.7 | 0.86 | 0.84–0.87 | <0.0001 |
| Prefer no to answer | 303 | 235 | 77.6 | 68 | 1.6 | 0.83 | 0.79–0.88 | <0.0001 |
| Total | Vaccine Status | ||||
|---|---|---|---|---|---|
| Vaccinated | Non-Vaccinated | ||||
| n | n | Percent | n | Percent | |
| TOTAL | 18,874 | 17,462 | 92.5 | 1413 | 7.5 |
| SEX | |||||
| Women | 12,835 | 11,780 | 91.8 | 1055 | 8.2 |
| Men | 6039 | 5681 | 94.1 | 358 | 5.9 |
| AGE GROUP | |||||
| <30 | 895 | 804 | 89.8 | 91 | 10.2 |
| 30–39 | 2766 | 2483 | 89.8 | 283 | 10.2 |
| 40–49 | 3577 | 3197 | 89.4 | 380 | 10.6 |
| 50–59 | 4001 | 3636 | 90.9 | 365 | 9.1 |
| 60–69 | 4715 | 4503 | 95.5 | 212 | 4.5 |
| 70+ | 2920 | 2838 | 97.2 | 82 | 2.8 |
| ETHNICITY | |||||
| Black or African American | 811 | 721 | 88.9 | 90 | 11.1 |
| Hispanic or Latino | 379 | 350 | 92.3 | 29 | 7.7 |
| Other | 692 | 643 | 92.9 | 49 | 7.1 |
| White (not Hispanic/Latino) | 16,992 | 15,747 | 92.7 | 1245 | 7.3 |
| HEALTHCARE WORKER | |||||
| N | 13,997 | 12,956 | 92.6 | 1041 | 7.4 |
| Y | 4877 | 4505 | 92.4 | 372 | 7.6 |
| PREVIOUS COVID-19 DIAGNOSIS | |||||
| N | 18,482 | 17,140 | 92.7 | 1342 | 7.3 |
| Y | 392 | 321 | 81.9 | 71 | 18.1 |
| SITE | |||||
| Campbell University | 134 | 123 | 91.8 | 11 | 8.2 |
| New Hanover Regional Medical Center | 612 | 576 | 94.1 | 36 | 5.9 |
| Vidant Health | 873 | 804 | 92.1 | 69 | 7.9 |
| Wake Forest Baptist Health | 15,022 | 13,835 | 92.1 | 1187 | 7.9 |
| WakeMed Health and Hospitals | 2233 | 2123 | 95.1 | 110 | 4.9 |
| COUNTY CLASS | |||||
| Rural | 6118 | 5521 | 90.2 | 597 | 9.8 |
| Suburban | 1533 | 1355 | 88.4 | 178 | 11.6 |
| Urban | 11,223 | 10,585 | 94.3 | 638 | 5.7 |
| Total | Vaccine Intent | ||||
|---|---|---|---|---|---|
| No/Undecided/Prefer Not to Answer | Yes | ||||
| n | N | Percent | n | Percent | |
| TOTAL | 20,232 | 4810 | 23.8 | 15,422 | 76.2 |
| SEX | |||||
| Women | 13,784 | 3688 | 26.8 | 10,096 | 73.2 |
| Men | 6448 | 1122 | 17.4 | 5326 | 82.6 |
| AGE GROUP | |||||
| <30 | 1096 | 322 | 29.4 | 774 | 70.6 |
| 30–39 | 3086 | 889 | 28.8 | 2197 | 71.2 |
| 40–49 | 3897 | 1175 | 30.2 | 2722 | 69.8 |
| 50–59 | 4256 | 1180 | 27.7 | 3076 | 72.3 |
| 60–69 | 4898 | 865 | 17.7 | 4033 | 82.3 |
| 70+ | 2999 | 379 | 12.6 | 2620 | 87.4 |
| ETHNICITY/RACE | |||||
| Black or African American | 932 | 426 | 45.7 | 506 | 54.3 |
| Hispanic or Latino | 413 | 122 | 29.5 | 291 | 70.5 |
| Other | 758 | 179 | 23.6 | 579 | 76.4 |
| White (not Hispanic/Latino) | 18,129 | 4083 | 22.5 | 14,046 | 77.5 |
| HEALTHCARE WORKER | |||||
| N | 15,062 | 3466 | 23 | 11,596 | 77 |
| Y | 5170 | 1344 | 26 | 3826 | 74 |
| PREVIOUS COVID-19 DIAGNOSIS | |||||
| N | 19,756 | 4624 | 23.4 | 15,132 | 76.6 |
| Y | 476 | 186 | 39.1 | 290 | 60.9 |
| SITE | |||||
| Campbell University | 147 | 48 | 32.7 | 99 | 67.3 |
| New Hanover Regional Medical Center | 641 | 100 | 15.6 | 541 | 84.4 |
| Vidant Health | 967 | 285 | 29.5 | 682 | 70.5 |
| Wake Forest Baptist Health | 16,058 | 3917 | 24.4 | 12,141 | 75.6 |
| WakeMed Health and Hospitals | 2419 | 460 | 19 | 1959 | 81 |
| COUNTY CLASS | |||||
| Rural | 6645 | 1916 | 28.8 | 4729 | 71.2 |
| Suburban | 1669 | 530 | 31.8 | 1139 | 68.2 |
| Urban | 11,918 | 2364 | 19.8 | 9554 | 80.2 |
| Vaccine Acceptance Multivariate Model Estimates | |||||
|---|---|---|---|---|---|
| Parameter | Estimate | Adj. Relative Risk | Relative Risk 95% CI | Pr > ChiSq | |
| SEX | Women | −0.07 | 0.93 | 0.92–0.94 | <0.0001 |
| Men | ref. | ref. | ref. | ||
| AGE GROUP | <30 | −0.17 | 0.85 | 0.81–0.88 | <0.0001 |
| 30–39 | −0.16 | 0.85 | 0.83–0.87 | <0.0001 | |
| 40–49 | −0.17 | 0.84 | 0.82–0.86 | <0.0001 | |
| 50–59 | −0.15 | 0.86 | 0.84–0.88 | <0.0001 | |
| 60–69 | −0.03 | 0.97 | 0.95–0.98 | 0.0001 | |
| ≥70 | ref. | ref. | ref. | ||
| ETHNICITY/RACE | Hispanic or Latino | 0.27 | 1.31 | 1.20–1.42 | <0.0001 |
| Other | 0.34 | 1.41 | 1.32–1.51 | <0.0001 | |
| White (not Hispanic/Latino) | 0.33 | 1.39 | 1.31–1.48 | <0.0001 | |
| Black or African American | ref. | ref. | ref. | ||
| HEALTHCARE WORKER | No | −0.03 | 0.97 | 0.96–0.99 | 0.0014 |
| Yes | ref. | ref. | ref. | ||
| PREVIOUS COVID-19 DIAGNOSIS | No | 0.18 | 1.20 | 1.11–1.28 | <0.0001 |
| Yes | ref. | ref. | ref. | ||
| COUNTY CLASS | Rural | −0.10 | 0.90 | 0.89–0.92 | <0.0001 |
| Suburban | −0.16 | 0.85 | 0.83–0.88 | <0.0001 | |
| Urban | ref. | ref. | ref. | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Enwezor, C.H.; Peacock, J.E., Jr.; Seals, A.L.; Edelstein, S.L.; Hinkelman, A.N.; Wierzba, T.F.; Munawar, I.; Maguire, P.D.; Lagarde, W.H.; Runyon, M.S.; et al. Changing Attitudes toward the COVID-19 Vaccine among North Carolina Participants in the COVID-19 Community Research Partnership. Vaccines 2021, 9, 916. https://doi.org/10.3390/vaccines9080916
Enwezor CH, Peacock JE Jr., Seals AL, Edelstein SL, Hinkelman AN, Wierzba TF, Munawar I, Maguire PD, Lagarde WH, Runyon MS, et al. Changing Attitudes toward the COVID-19 Vaccine among North Carolina Participants in the COVID-19 Community Research Partnership. Vaccines. 2021; 9(8):916. https://doi.org/10.3390/vaccines9080916
Chicago/Turabian StyleEnwezor, Chukwunyelu H., James E. Peacock, Jr., Austin L. Seals, Sharon L. Edelstein, Amy N. Hinkelman, Thomas F. Wierzba, Iqra Munawar, Patrick D. Maguire, William H. Lagarde, Michael S. Runyon, and et al. 2021. "Changing Attitudes toward the COVID-19 Vaccine among North Carolina Participants in the COVID-19 Community Research Partnership" Vaccines 9, no. 8: 916. https://doi.org/10.3390/vaccines9080916
APA StyleEnwezor, C. H., Peacock, J. E., Jr., Seals, A. L., Edelstein, S. L., Hinkelman, A. N., Wierzba, T. F., Munawar, I., Maguire, P. D., Lagarde, W. H., Runyon, M. S., Gibbs, M. A., Gallaher, T. R., Sanders, J. W., III, & Herrington, D. M. (2021). Changing Attitudes toward the COVID-19 Vaccine among North Carolina Participants in the COVID-19 Community Research Partnership. Vaccines, 9(8), 916. https://doi.org/10.3390/vaccines9080916






