Correlates of COVID-19 Vaccine Hesitancy among a Community Sample of African Americans Living in the Southern United States
Abstract
:1. Introduction
1.1. Social Susceptibility to COVID-19
1.2. Health Decision-Making Susceptibility and Prevention of COVID-19
2. Materials and Methods
2.1. Study Design and Population
2.2. Primary Outcome of Interest
2.3. Participant Characteristics
2.4. Statistical Analyses
3. Results
3.1. Characteristics of Study Population
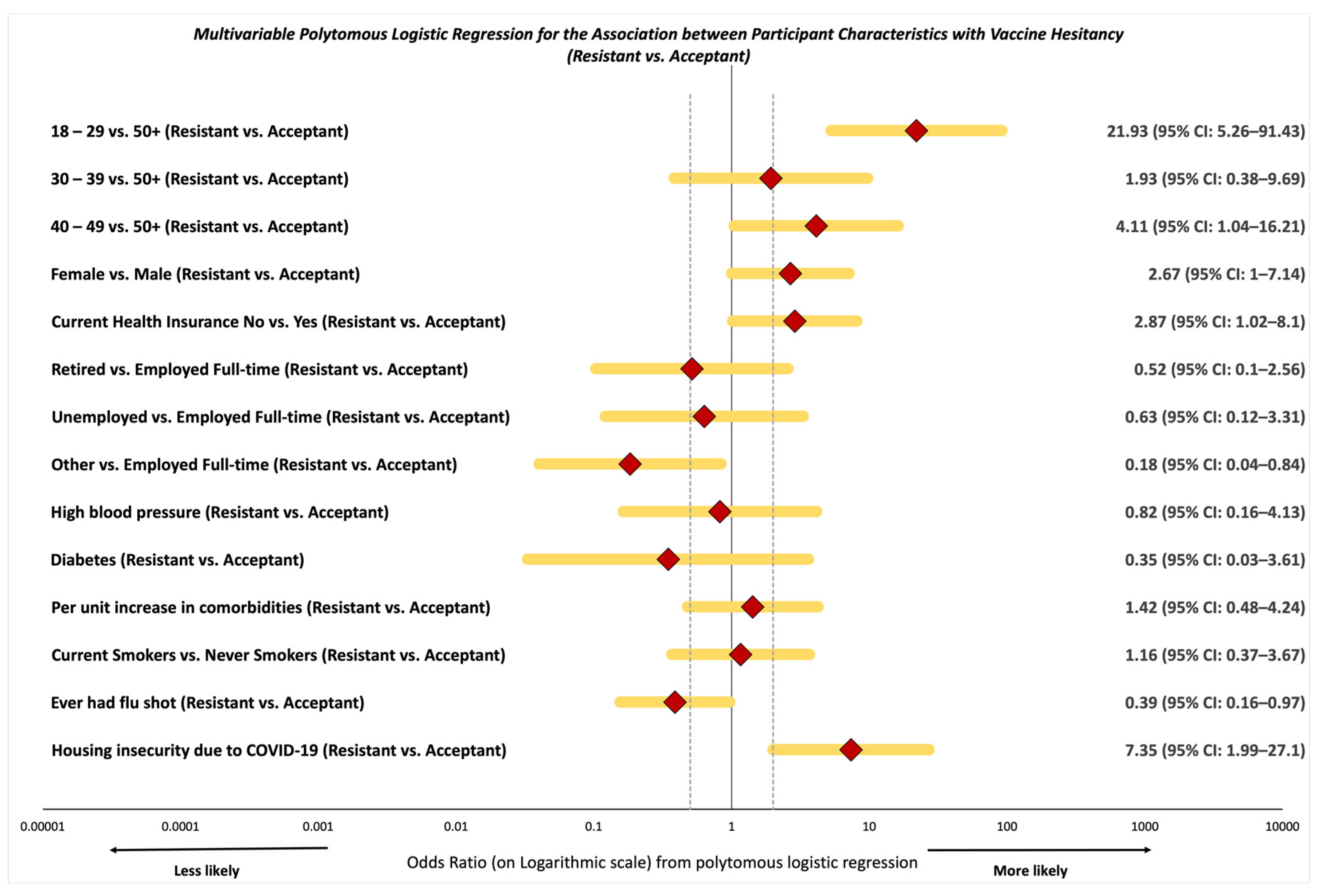
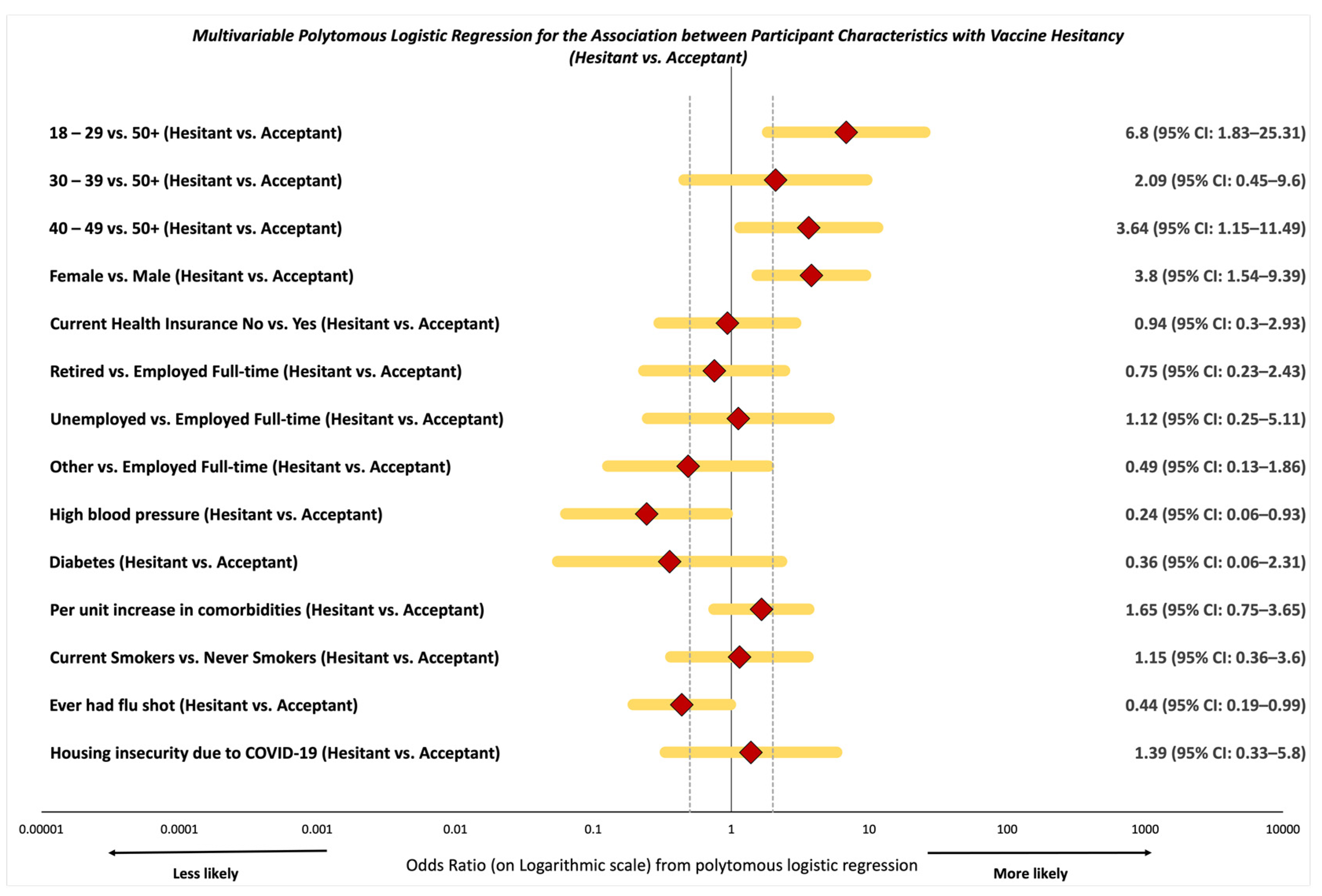
3.2. Likelihood of Vaccine Resistance and Hesitancy
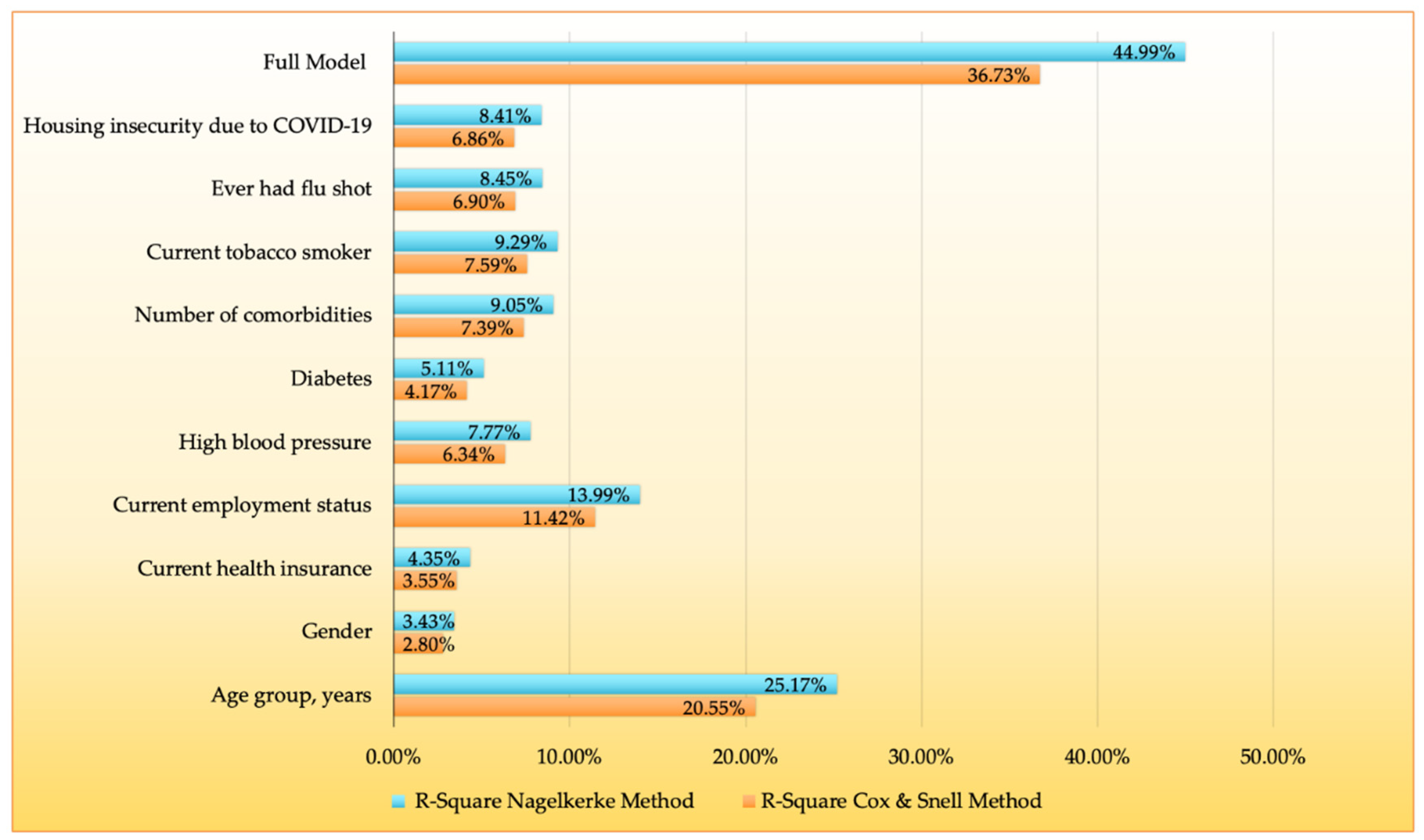
3.3. Variation in Vaccine Resistance and Hesitancy Explained by Factors
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Dong, E.; Du, H.; Gardner, L. An interactive web-based dashboard to track COVID-19 in real time. Lancet Infect. Dis. 2020, 20, 533–534. [Google Scholar] [CrossRef]
- Khanijahani, A. Racial, ethnic, and socioeconomic disparities in confirmed COVID-19 cases and deaths in the United States: A county-level analysis as of November 2020. Ethn. Health 2021, 26, 22–35. [Google Scholar] [CrossRef]
- Khose, S.; Moore, J.X.; Wang, H.E. Epidemiology of the 2020 pandemic of COVID-19 in the State of Texas: The first month of community spread. J. Community Health 2020, 45, 696–701. [Google Scholar] [CrossRef]
- Khose, S.; Chan, H.K.; Wang, H.E.; Moore, J.X. Predictors for county level variations in initial 4-week COVID-19 incidence and case fatality risk in the United States. Res. Sq. 2020. [Google Scholar] [CrossRef]
- Moore, J.X.; Langston, M.E.; George, V.; Coughlin, S.S. Epidemiology of the 2020 pandemic of COVID-19 in the state of Georgia: Inadequate critical care resources and impact after 7 weeks of community spread. J. Am. Coll. Emerg. Physicians Open 2020, 1, 527–532. [Google Scholar] [CrossRef]
- Coughlin, S.S.; Moore, J.X.; George, V.; Johnson, J.A.; Hobbs, J. COVID-19 among African Americans: From preliminary epidemiological surveillance data to public health action. Am. J. Public Health 2020, 110, 1157–1159. [Google Scholar] [CrossRef]
- Xu, J.J.; Chen, J.T.; Belin, T.R.; Brookmeyer, R.S.; Suchard, M.A.; Ramirez, C.M. Racial and ethnic disparities in years of potential life lost attributable to COVID-19 in the United States: An analysis of 45 states and the district of Columbia. Int. J. Environ. Res. Public Health 2021, 18, 2921. [Google Scholar] [CrossRef] [PubMed]
- CDC. Risk for COVID-19 Infection, Hospitalization, and Death by Race/Ethnicity. 12 March 2021. Available online: https://www.cdc.gov/coronavirus/2019-ncov/covid-data/investigations-discovery/hospitalization-death-by-race-ethnicity.html (accessed on 26 March 2021).
- Shah, P.; Smith, H.; Olarewaju, A.; Jani, Y.; Cobb, A.; Owens, J.; Moore, J.; Chenna, A.; Hess, D. Is cardiopulmonary resuscitation futile in Coronavirus disease 2019 patients experiencing in-hospital cardiac arrest? Crit. Care Med. 2021, 49, 201–208. [Google Scholar] [CrossRef] [PubMed]
- Shah, P.; Owens, J.; Franklin, J.; Mehta, A.; Heymann, W.; Sewell, W.; Hill, J.; Barfield, K.; Doshi, R. Demographics, comorbidities and outcomes in hospitalized Covid-19 patients in rural southwest Georgia. Ann. Med. 2020, 52, 354–360. [Google Scholar] [CrossRef]
- Matthews, K.A.; Ullrich, F.; Gaglioti, A.H.; Dugan, S.; Chen, M.S.; Hall, D.M. Nonmetropolitan COVID-19 incidence and mortality rates surpassed metropolitan rates within the first 24 weeks of the pandemic declaration: United States, March 1-October 18, 2020. J. Rural Health 2021, 37, 272–277. [Google Scholar] [CrossRef] [PubMed]
- Baltrus, P.T.; Douglas, M.; Li, C.; Caplan, L.S.; Blount, M.; Mack, D.; Gaglioti, A.H. Percentage of black population and primary care shortage areas associated with higher COVID-19 case and death rates in Georgia counties. South Med. J. 2021, 114, 57–62. [Google Scholar] [CrossRef] [PubMed]
- Webster, G.; Bowman, J. Quantitatively delineating the Black Belt geographic region. Southeast. Geogr. 2008, 48, 3–18. [Google Scholar] [CrossRef]
- Chafe, W.H.; Gavins, R.; Korstad, R.; Ortiz, P.; Parrish, R.; Ritterhouse, J.; Roberts, K.; Waligora-Davis, N. Remembering Jim Crow: African Americans Tell About Life in the Segregated South; The New Press: New York, NY, USA, 2011. [Google Scholar]
- Kennedy, S. Jim Crow Guide to the U.S.A.: The Laws, Customs, and Etiquette Governing the Conduct of Nonwhites and Other Minorities as Second-Class Citizens; The University of Alabama Press: Tuscaloosa, AL, USA, 1990. [Google Scholar]
- Moore, J.X.; Donnelly, J.P.; Griffin, R.; Howard, G.; Safford, M.M.; Wang, H.E. Defining sepsis mortality clusters in the United States. Crit. Care Med. 2016, 44, 1380–1387. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- El Bcheraoui, C.; Mokdad, A.H.; Dwyer-Lindgren, L.; Bertozzi-Villa, A.; Stubbs, R.W.; Morozoff, C.; Shirude, S.; Naghavi, M.; Murray, C.J.L. Trends and patterns of differences in infectious disease mortality among US counties, 1980–2014. JAMA 2018, 319, 1248–1260. [Google Scholar] [CrossRef]
- Roth, G.A.; Dwyer-Lindgren, L.; Bertozzi-Villa, A.; Stubbs, R.W.; Morozoff, C.; Naghavi, M.; Mokdad, A.H.; Murray, C.J.L. Trends and patterns of geographic variation in cardiovascular mortality among US counties, 1980–2014. JAMA 2017, 317, 1976–1992. [Google Scholar] [CrossRef] [PubMed]
- Howard, G.; Howard, V.J. Twenty years of progress toward understanding the stroke belt. Stroke 2020, 51, 742–750. [Google Scholar] [CrossRef]
- Mokdad, A.H.; Dwyer-Lindgren, L.; Fitzmaurice, C.; Stubbs, R.W.; Bertozzi-Villa, A.; Morozoff, C.; Charara, R.; Allen, C.; Naghavi, M.; Murray, C.J. Trends and patterns of disparities in cancer mortality among US counties, 1980–2014. JAMA 2017, 317, 388–406. [Google Scholar] [CrossRef] [Green Version]
- Moore, J.X.; Royston, K.J.; Langston, M.E.; Griffin, R.; Hidalgo, B.; Wang, H.E.; Colditz, G.; Akinyemiju, T. Mapping hot spots of breast cancer mortality in the United States: Place matters for Blacks and Hispanics. Cancer Causes Control 2018, 29, 737–750. [Google Scholar] [CrossRef]
- Porter, G.; Desai, K.; George, V.; Coughlin, S.S.; Moore, J.X. Racial disparities in the epidemiology of COVID-19 in Georgia: Trends since state-wide reopening. Health Equity 2021, 5, 91–99. [Google Scholar] [CrossRef]
- Cheng, K.J.G.; Sun, Y.; Monnat, S.M. COVID-19 death rates are higher in rural counties with larger shares of Blacks and Hispanics. J. Rural Health 2020, 36, 602–608. [Google Scholar] [CrossRef]
- Tsai, A.C.; Mendenhall, E.; Trostle, J.A.; Kawachi, I. Co-occurring epidemics, syndemics, and population health. Lancet 2017, 389, 978–982. [Google Scholar] [CrossRef] [Green Version]
- Singer, M.; Bulled, N.; Ostrach, B.; Mendenhall, E. Syndemics and the biosocial conception of health. Lancet 2017, 389, 941–950. [Google Scholar] [CrossRef]
- Sharma, A. Syndemics: Health in context. Lancet 2017, 389, 881. [Google Scholar]
- Nunn, A.; Jeffries, W.L.t.; Foster, P.; McCoy, K.; Sutten-Coats, C.; Willie, T.C.; Ransome, Y.; Lanzi, R.G.; Jackson, E.; Berkley-Patton, J.; et al. Reducing the African American HIV disease burden in the Deep South: Addressing the role of faith and spirituality. AIDS Behav. 2019, 23 (Suppl. 3), 319–330. [Google Scholar] [CrossRef]
- CDC Social Vulnerability Index. 2021. Available online: https://www.atsdr.cdc.gov/placeandhealth/svi/data_documentation_download.html (accessed on 26 March 2021).
- Kind, A.J.H.; Buckingham, W.R. Making neighborhood-disadvantage metrics accessible–The neighborhood atlas. N. Engl. J. Med. 2018, 378, 2456–2458. [Google Scholar] [CrossRef]
- Rogers, T.N.; Rogers, C.R.; VanSant-Webb, E.; Gu, L.Y.; Yan, B.; Qeadan, F. Racial disparities in COVID-19 mortality among essential workers in the United States. World Med. Health Policy 2020, 12, 311–327. [Google Scholar] [CrossRef]
- Ford, C.L.; Griffith, D.M.; Bruce, M.A.; Gilbert, K. Racism: Science & Tools for the Public Health Professional; American Public Health Association Press: Washington, DC, USA, 2019. [Google Scholar]
- Williams, D.R. Miles to go before we sleep: Racial inequities in health. J. Health Soc. Behav. 2012, 53, 279–295. [Google Scholar] [CrossRef]
- Williams, D.R.; Lawrence, J.A.; Davis, B.A.; Vu, C. Understanding how discrimination can affect health. Health Serv. Res. 2019, 54 (Suppl. 2), 1374–1388. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gee, G.C.; Hing, A.; Mohammed, S.; Tabor, D.C.; Williams, D.R. Racism and the life course: Taking time seriously. Am. J. Public Health 2019, 109 (Suppl. S1), S43–S47. [Google Scholar] [CrossRef] [PubMed]
- Yearby, R.; Mohapatra, S. Law, structural racism, and the COVID-19 pandemic. J. Law Biosci. 2020, 7, lsaa036. [Google Scholar] [CrossRef] [PubMed]
- Yearby, R. Structural racism and health disparities: Reconfiguring the social determinants of health framework to include the root cause. J. Law Med. Ethics 2020, 48, 518–526. [Google Scholar] [CrossRef]
- Quinn, S.C.; Thomas, S.B. The national Negro health week, 1915 to 1951: A descriptive account. Minority Health Today 2001, 2, 44–49. [Google Scholar]
- Gamble, V.N. Under the shadow of Tuskegee: African Americans and health care. Am. J. Public Health 1997, 87, 1773–1778. [Google Scholar] [CrossRef] [Green Version]
- Scharff, D.P.; Mathews, K.J.; Jackson, P.; Hoffsuemmer, J.; Martin, E.; Edwards, D. More than Tuskegee: Understanding mistrust about research participation. J. Health Care Poor Underserved 2010, 21, 879–897. [Google Scholar] [CrossRef]
- Marrett, C.B. Racial disparities and COVID-19: The social context. J. Racial Ethn. Health Disparities 2021, 8, 794–797. [Google Scholar] [CrossRef]
- Thomas, S.B.; Casper, E. The burdens of race and history on Black people’s health 400 years after Jamestown. Am. J. Public Health 2019, 109, 1346–1347. [Google Scholar] [CrossRef] [PubMed]
- Heart, M.Y.; Chase, J.; Elkins, J.; Altschul, D.B. Historical trauma among Indigenous Peoples of the Americas: Concepts, research, and clinical considerations. J. Psychoact. Drugs 2011, 43, 282–290. [Google Scholar] [CrossRef] [PubMed]
- Evanega, S.; Lynas, M.; Adams, J.; Smolenyak, K. Coronavirus Misinformation: Quantifying Sources and Themes in the COVID-19 ‘Infodemic’. 2020. Available online: https://www.uncommonthought.com/mtblog/wp-content/uploads/2020/12/Evanega-et-al-Coronavirus-misinformation-submitted_07_23_20-1.pdf (accessed on 26 March 2021).
- Brennen, S.J.; Simon, F.M.; Howard, P.N.; Nielsen, R.K. Types, Sources, and Claims of COVID-19 Misinformation; Reuters Institute for the Study of Journalism: Oxford, UK; Oxford Internet Institute: Oxford, UK, 2020. [Google Scholar]
- Burger, A.E.; Reither, E.N.; Mamelund, S.E.; Lim, S. Black-white disparities in 2009 H1N1 vaccination among adults in the United States: A cautionary tale for the COVID-19 pandemic. Vaccine 2021, 39, 943–951. [Google Scholar] [CrossRef]
- Funk, C.; Tyson, A. Growing Share of Americans Say They Plan To Get a COVID-19 Vaccine—Or Already Have; Pew Research Center: Washington, DC, USA, 2021. [Google Scholar]
- Seehusen, D.A.; Ledford, C.J.W. A clarion call to our family medicine colleagues. Fam. Med. 2020, 52, 471–473. [Google Scholar] [CrossRef]
- Ledford, C.J.W.; Anderson, L.N. Communication strategies for family physicians practicing throughout emerging public health crises. Fam. Med. 2020, 52, 48–50. [Google Scholar] [CrossRef]
- Fisher, K.A.; Bloomstone, S.J.; Walder, J.; Crawford, S.; Fouayzi, H.; Mazor, K.M. Attitudes toward a potential SARS-CoV-2 vaccine: A survey of U.S. adults. Ann. Intern. Med. 2020, 173, 964–973. [Google Scholar] [CrossRef] [PubMed]
- Freeman, D.; Loe, B.S.; Chadwick, A.; Vaccari, C.; Waite, F.; Rosebrock, L.; Jenner, L.; Petit, A.; Lewandowsky, S.; Vanderslott, S.; et al. COVID-19 vaccine hesitancy in the UK: The Oxford coronavirus explanations, attitudes, and narratives survey (Oceans) II. Psychol. Med. 2020, 1–15. [Google Scholar] [CrossRef]
- Tang, Q.; Liu, Y.; Fu, Y.; Di, Z.; Xu, K.; Tang, B.; Wu, H.; Di, M. A comprehensive evaluation of early potential risk factors for disease aggravation in patients with COVID-19. Sci. Rep. 2021, 11, 8062. [Google Scholar] [CrossRef] [PubMed]
- Tenforde, M.W.; Fisher, K.A.; Patel, M.M. Identifying COVID-19 risk through observational studies to inform control measures. JAMA 2021, 325, 1464–1465. [Google Scholar] [CrossRef]
- Britto, M.T.; Pandzik, G.M.; Meeks, C.S.; Kotagal, U.R. Combining evidence and diffusion of innovation theory to enhance influenza immunization. Jt. Comm. J. Qual. Patient Saf. 2006, 32, 426–432. [Google Scholar] [CrossRef]
- Jamison, A.M.; Quinn, S.C.; Freimuth, V.S. “You don’t trust a government vaccine“: Narratives of institutional trust and influenza vaccination among African American and white adults. Soc. Sci. Med. 2019, 221, 87–94. [Google Scholar] [CrossRef] [PubMed]
- Livingston, E.H.; Malani, P.N.; Creech, C.B. The Johnson & Johnson vaccine for COVID-19. JAMA 2021, 325, 1575. [Google Scholar]
- Baden, L.R.; El Sahly, H.M.; Essink, B.; Kotloff, K.; Frey, S.; Novak, R.; Diemert, D.; Spector, S.A.; Rouphael, N.; Creech, C.B.; et al. Efficacy and safety of the mRNA-1273 SARS-CoV-2 vaccine. N. Engl. J. Med. 2021, 384, 403–416. [Google Scholar] [CrossRef]
- Polack, F.P.; Thomas, S.J.; Kitchin, N.; Absalon, J.; Gurtman, A.; Lockhart, S.; Perez, J.L.; Perez Marc, G.; Moreira, E.D.; Zerbini, C.; et al. Safety and efficacy of the BNT162b2 mRNA Covid-19 vaccine. N. Engl. J. Med. 2020, 383, 2603–2615. [Google Scholar] [CrossRef]
- CDC. Demographic Characteristics of People Receiving COVID-19 Vaccinations in the United States. 2021. Available online: https://covid.cdc.gov/covid-data-tracker/#vaccination-demographic (accessed on 26 March 2021).
- Nguyen, L.H.; Joshi, A.D.; Drew, D.A.; Merino, J.; Ma, W.; Lo, C.H.; Kwon, S.; Wang, K.; Graham, M.S.; Polidori, L.; et al. Racial and ethnic differences in COVID-19 vaccine hesitancy and uptake. medRxiv 2021. [Google Scholar] [CrossRef]
- Harris, P.A.; Taylor, R.; Thielke, R.; Payne, J.; Gonzalez, N.; Conde, J.G. Research electronic data capture (REDCap)—A metadata-driven methodology and workflow process for providing translational research informatics support. J. Biomed. Inform. 2009, 42, 377–381. [Google Scholar] [CrossRef] [Green Version]
- Harris, P.A.; Taylor, R.; Minor, B.L.; Elliott, V.; Fernandez, M.; O’Neal, L.; McLeod, L.; Delacqua, G.; Delacqua, F.; Kirby, J.; et al. The REDCap consortium: Building an international community of software platform partners. J. Biomed. Inform. 2019, 95, 103208. [Google Scholar] [CrossRef]
- MacDonald, N.E. Vaccine hesitancy: Definition, scope and determinants. Vaccine 2015, 33, 4161–4164. [Google Scholar] [CrossRef] [PubMed]
- Glynn, R.J.; Rosner, B. Methods to evaluate risks for composite end points and their individual components. J. Clin. Epidemiol. 2004, 57, 113–122. [Google Scholar] [CrossRef]
- Cox, D.R.; Snell, E.J. Analysis of Binary Data, 2nd ed.; Monographs on Statistics and Applied Probability; Chapman and Hall: London, UK; New York, NY, USA, 1989; Volume VII, p. 236. [Google Scholar]
- Nagelkerke, A.J.D. A note on a general definition of the coefficient of determination. Biometrika 1991, 78, 691–692. [Google Scholar] [CrossRef]
- Ledford, C.J.W.; Willett, K.L.; Kreps, G.L. Communicating immunization science: The genesis and evolution of the national network for immunization information. J. Health Commun. 2012, 17, 105–122. [Google Scholar] [CrossRef] [PubMed]
- Larson, H.J.; Schulz, W.S.; Tucker, J.D.; Smith, D.M. Measuring vaccine confidence: Introducing a global vaccine confidence index. PLoS Curr. 2015, 7. [Google Scholar] [CrossRef]
- Quinn, S.C.; Jamison, A.M.; An, J.; Hancock, G.R.; Freimuth, V.S. Measuring vaccine hesitancy, confidence, trust and flu vaccine uptake: Results of a national survey of White and African American adults. Vaccine 2019, 37, 1168–1173. [Google Scholar] [CrossRef]
- Gershon, A.A. Is chickenpox so bad, what do we know about immunity to varicella zoster virus, and what does it tell us about the future? J. Infect. 2017, 74 (Suppl. 1), S27–S33. [Google Scholar] [CrossRef] [Green Version]
- Lee, C.; Whetten, K.; Omer, S.; Pan, W.; Salmon, D. Hurdles to herd immunity: Distrust of government and vaccine refusal in the US, 2002–2003. Vaccine 2016, 34, 3972–3978. [Google Scholar] [CrossRef]
- Razai, M.S.; Oakeshott, P.; Esmail, A.; Wiysonge, C.S.; Viswanath, K.; Mills, M.C. COVID-19 vaccine hesitancy: The five Cs to tackle behavioural and sociodemographic factors. J. R. Soc. Med. 2021, 114, 295–298. [Google Scholar] [CrossRef] [PubMed]
- Betsch, C.; Schmid, P.; Heinemeier, D.; Korn, L.; Holtmann, C.; Böhm, R. Beyond confidence: Development of a measure assessing the 5C psychological antecedents of vaccination. PLoS ONE 2018, 13, e0208601. [Google Scholar] [CrossRef] [Green Version]
- Freimuth, V.S.; Jamison, A.M.; An, J.; Hancock, G.R.; Quinn, S.C. Determinants of trust in the flu vaccine for African Americans and Whites. Soc. Sci. Med. 2017, 193, 70–79. [Google Scholar] [CrossRef]
- Karafillakis, E.; Simas, C.; Jarrett, C.; Verger, P.; Peretti-Watel, P.; Dib, F.; De Angelis, S.; Takacs, J.; Ali, K.A.; Pastore Celentano, L.; et al. HPV vaccination in a context of public mistrust and uncertainty: A systematic literature review of determinants of HPV vaccine hesitancy in Europe. Hum. Vaccin. Immunother. 2019, 15, 1615–1627. [Google Scholar] [CrossRef] [PubMed]
- Zingg, A.; Siegrist, M. Measuring people’s knowledge about vaccination: Developing a one-dimensional scale. Vaccine 2012, 30, 3771–3777. [Google Scholar] [CrossRef]
- Lu, H.; Winneg, K.; Jamieson, K.H.; Albarracín, D. Intentions to seek information about the influenza vaccine: The role of informational subjective norms, anticipated and experienced affect, and information insufficiency among vaccinated and unvaccinated people. Risk Anal. 2020, 40, 2040–2056. [Google Scholar] [CrossRef] [PubMed]
- Viswanath, K.; Lee, E.W.J.; Pinnamaneni, R. We need the lens of equity in COVID-19 communication. Health Commun. 2020, 35, 1743–1746. [Google Scholar] [CrossRef] [PubMed]
- Grimes, D.R. Medical disinformation and the unviable nature of COVID-19 conspiracy theories. PLoS ONE 2021, 16, e0245900. [Google Scholar] [CrossRef] [PubMed]
- Wilson, S.L.; Wiysonge, C. Social media and vaccine hesitancy. BMJ Glob. Health 2020, 5, e004206. [Google Scholar] [CrossRef]
- Avtar, R.; Chakrabarti, R.; Pinkovskiy, M. Understanding the Racial and Income Gap in COVID-19: Essential Workers. 2021. Available online: https://libertystreeteconomics.newyorkfed.org/2021/01/understanding-the-racial-and-income-gap-in-covid-19-essential-workers.html (accessed on 12 January 2021).
- Chaganti, S.; Higgins, A.; Mattingly, M.J. Health Insurance and Essential Service Workers in New England: Who Lacks Access to Care for COVID-19? Federal Reserve Bank of Boston: Boston, MA, USA, 2020. [Google Scholar]
- Abdel-Latif, M.M.M. The enigma of health literacy and COVID-19 pandemic. Public Health 2020, 185, 95–96. [Google Scholar] [CrossRef] [PubMed]
- Paakkari, L.; Okan, O. COVID-19: Health literacy is an underestimated problem. Lancet Public Health 2020, 5, e249–e250. [Google Scholar] [CrossRef]
- Ratzan, S.C.; Sommariva, S.; Rauh, L. Enhancing global health communication during a crisis: Lessons from the COVID-19 pandemic. Public Health Res. Pract. 2020, 30, e3022010. [Google Scholar] [CrossRef] [PubMed]
- Bajaj, S.S.; Stanford, F.C. Beyond Tuskegee—Vaccine distrust and everyday racism. N. Engl. J. Med. 2021, 384, e12. [Google Scholar] [CrossRef] [PubMed]
- Gerussi, V.; Peghin, M.; Palese, A.; Bressan, V.; Visintini, E.; Bontempo, G.; Graziano, E.; De Martino, M.; Isola, M.; Tascini, C. Vaccine hesitancy among Italian patients recovered from COVID-19 infection towards Influenza and Sars-Cov-2 vaccination. Vaccines 2021, 9, 172. [Google Scholar] [CrossRef] [PubMed]
| Characteristic | Acceptant (n = 175) | Hesitant (n = 40) | Resistant (n = 42) | p-Value 1 |
|---|---|---|---|---|
| Median age (years)—IQR 2 | 61.0 (49.0, 70.0) | 46.0 (30.0, 60.5) | 31.0 (27.0, 47.0) | <0.01 |
| Age group, years—no. (%) 3 | ||||
| 18–29 | 10 (5.7) | 10 (25.0) | 18 (42.9) | <0.01 |
| 30–39 | 15 (8.6) | 4 (10.0) | 6 (14.3) | |
| 40–49 | 19 (10.9) | 10 (25.0) | 9 (21.4) | |
| 50+ | 131 (74.9) | 16 (40.0) | 9 (21.4) | |
| Male—no. (%) 3 | 100 (57.1) | 14 (35.0) | 19 (45.2) | 0.03 |
| Black or African American—no. (%) 3 | 168 (96.0) | 38 (95.0) | 42 (100.0) | 0.38 |
| Education—no. (%) 3 | ||||
| Less than high school | 15 (8.6) | 1 (2.5) | 2 (4.8) | 0.80 |
| High school/GED | 40 (22.9) | 11 (27.5) | 11 (26.2) | |
| Some college or Associate’s degree | 59 (33.7) | 16 (40.0) | 15 (35.7) | |
| College graduate | 61 (34.9) | 12 (30.0) | 14 (33.3) | |
| Current employment status—no. (%) 3 | ||||
| Employed full-time | 73 (41.7) | 25 (62.5) | 32 (76.2) | <0.01 |
| Retired | 72 (41.1) | 7 (17.5) | 3 (7.1) | |
| Unemployed | 8 (4.6) | 4 (10.0) | 4 (9.5) | |
| Other 4 | 22 (12.6) | 4 (10.0) | 3 (7.1) | |
| Current health insurance—no. (%) 3 | 147 (84.0) | 33 (82.5) | 26 (61.9) | <0.01 |
| Annual household income—no. (%) 3 | ||||
| Less than USD $20,000 | 28 (16.0) | 12 (30.0) | 7 (16.7) | 0.24 |
| USD $20,001–35,000 | 19 (10.9) | 6 (15.0) | 5 (11.9) | |
| USD $35,001–50,000 | 45 (25.7) | 4 (10.0) | 11 (26.2) | |
| USD $50,001–75,000 | 28 (16.0) | 5 (12.5) | 4 (9.5) | |
| USD $75,001+ | 29 (16.6) | 5 (12.5) | 4 (9.5) | |
| Not reported | 26 (14.9) | 8 (20.0) | 11 (26.2) |
| Characteristic | Acceptant (n = 175) | Hesitant (n = 40) | Resistant (n = 42) | p-Value 1 |
|---|---|---|---|---|
| Baseline comorbidities—no. (%) 2 | ||||
| High blood pressure | 89 (50.9) | 9 (22.5) | 11 (26.2) | <0.01 |
| Chronic liver disease | 2 (1.1) | 0 (0.0) | 0 (0.0) | 0.62 |
| Coronary artery disease | 2 (1.1) | 1 (2.5) | 0 (0.0) | 0.57 |
| Congestive heart failure | 6 (3.4) | 0 (0.0) | 0 (0.0) | 0.24 |
| Diabetes | 37 (21.1) | 3 (7.5) | 2 (4.8) | <0.01 |
| Chronic kidney disease | 5 (2.9) | 0 (0.0) | 0 (0.0) | 0.30 |
| High Cholesterol | 37 (21.1) | 7 (17.5) | 5 (11.9) | 0.38 |
| Cancer | 9 (5.1) | 1 (2.5) | 0 (0.0) | 0.27 |
| Median number of comorbidities 3 (IQR) | 1 (0, 2) | 0 (0, 1) | 0 (0, 1) | <0.01 |
| Current Alcohol Use—no. (%) 2 | 70 (40.0) | 18 (45.0) | 25 (59.5) | 0.16 |
| Current Tobacco Smoker—no. (%) 2 | 21 (12.0) | 6 (15.0) | 12 (28.6) | <0.01 |
| Ever experienced racial discrimination during medical care—no. (%) 2 | 12 (6.9) | 1 (2.5) | 4 (9.5) | 0.43 |
| Had prior COVID-19 test—no. (%) 2 | 92 (52.6) | 19 (47.5) | 20 (47.6) | 0.76 |
| Knowledge of COVID-19 spread—no. (%) 2 | 146 (83.4) | 28 (70.0) | 34 (81.0) | 0.15 |
| COVID-19 safety practices—no. (%) 3 | 171 (97.7) | 39 (97.5) | 42 (100.0) | 0.61 |
| Ever had flu shot—no. (%) 2 | 102 (58.3) | 14 (35.0) | 11 (26.2) | <0.01 |
| Job loss due to COVID-19—no. (%) 2 | 23 (13.1) | 3 (7.5) | 9 (21.4) | 0.18 |
| Housing insecurity due to COVID-19—no. (%) 2 | 12 (6.9) | 4 (10.0) | 14 (33.3) | <0.01 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Moore, J.X.; Gilbert, K.L.; Lively, K.L.; Laurent, C.; Chawla, R.; Li, C.; Johnson, R.; Petcu, R.; Mehra, M.; Spooner, A.; et al. Correlates of COVID-19 Vaccine Hesitancy among a Community Sample of African Americans Living in the Southern United States. Vaccines 2021, 9, 879. https://doi.org/10.3390/vaccines9080879
Moore JX, Gilbert KL, Lively KL, Laurent C, Chawla R, Li C, Johnson R, Petcu R, Mehra M, Spooner A, et al. Correlates of COVID-19 Vaccine Hesitancy among a Community Sample of African Americans Living in the Southern United States. Vaccines. 2021; 9(8):879. https://doi.org/10.3390/vaccines9080879
Chicago/Turabian StyleMoore, Justin Xavier, Keon L. Gilbert, Katie L. Lively, Christian Laurent, Rishab Chawla, Cynthia Li, Ryan Johnson, Robert Petcu, Mehul Mehra, Antron Spooner, and et al. 2021. "Correlates of COVID-19 Vaccine Hesitancy among a Community Sample of African Americans Living in the Southern United States" Vaccines 9, no. 8: 879. https://doi.org/10.3390/vaccines9080879
APA StyleMoore, J. X., Gilbert, K. L., Lively, K. L., Laurent, C., Chawla, R., Li, C., Johnson, R., Petcu, R., Mehra, M., Spooner, A., Kolhe, R., & Ledford, C. J. W. (2021). Correlates of COVID-19 Vaccine Hesitancy among a Community Sample of African Americans Living in the Southern United States. Vaccines, 9(8), 879. https://doi.org/10.3390/vaccines9080879







