Detection of Urinary Antibodies and Its Application in Epidemiological Studies for Parasitic Diseases
Abstract
1. Introduction
2. Characteristics of Urine Examination for Parasite-Specific Antibodies
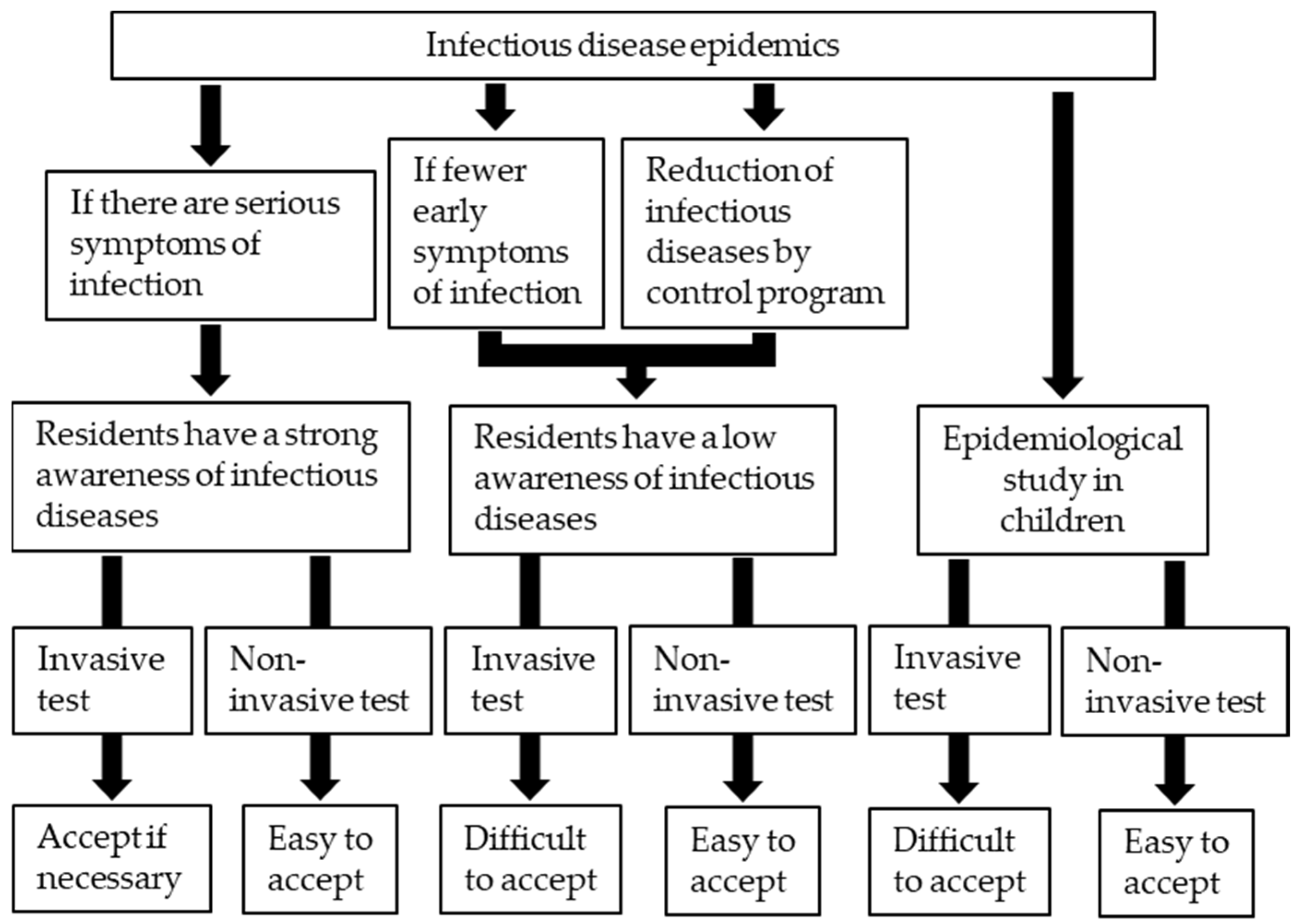
2.1. Efficacy of Antibody Examination in Epidemiological Studies
2.2. Advantages of Antibody Examination Using Urine Samples
3. Practical Examples of Application of Urine for Antibody Detection in Parasitic Diseases
3.1. Visceral Leishmaniasis
3.2. Lymphatic Filariasis
| Parasite Species | Antigen | Gold Standard | Detected Antibody | SEN (%) | SPE (%) | Sample Storage | Author, Year [Reference No] |
|---|---|---|---|---|---|---|---|
| L. donovani | acetone-treated L. donovani promastigote antigen | visceral leishmaniasis patients | IgG | 95.0 | 95.3 | 4 °C | Islam et al., 2002 [10] |
| L. donovani promastigote membrane antigen | visceral leishmaniasis patients | IgG | 97.94 | 100 | 4 °C | Ejazi et al., 2016 [28] | |
| rKRP42 (recombinant antigen) | visceral leishmaniasis patients | IgG | 94.0 | 99.6 | 4 °C | Islam et al., 2008 [29] | |
| rK28 (recombinant antigen) | visceral leishmaniasis patients | IgG | 95.4 | 96.3 | 4 °C | Ghosh et al., 2016 [30] | |
| rK39 (recombinant antigen) | 94.3 | 97.5 | |||||
| rKRP42 (recombinant antigen) | 90.8 | 96.3 | |||||
| W. bancrofti | adult female B. pahangi crude antigen | mf +/Og4C3 + | IgG4 | 95.6 | 99.0 | 4 °C | Itoh et al., 2001 [4] |
| recWb-SXP1 (recombinant antigen) | mf +, ICT + | IgG4 | 96.8 | 100 | 4 °C | Samad et al., 2013 [35] | |
| mf +/-, ICT + | 84.8 | ||||||
| S. japonicum | S. japonicum soluble egg antigen | serum antibody + | IgG | 86.8 * | ― | 4 °C | Itoh et al., 2003 [11] |
| P. westermani | P. westermani adult worm crude antigen | paragonimiasis patients | IgG | 100 | ― | 4 °C | Qiu et al., 2016 [12] |
| IgG4 | 90 | ― | |||||
| E. granulosus & E. multilocularis | hydatid antigen from cysts | cystic echinococcosis patients | IgG | 84 | 76 | −20 °C | Sunita et al., 2007 [13] |
| hydatid antigen from cyst | cystic echinococcosis patients | IgG | 80.48 | 93.75 | −70 °C | Chirag et al., 2015 [21] | |
| IgM | 48.78 | 100 | |||||
| IgG1 | 56.09 | 100 | |||||
| IgG2 | 53.65 | 100 | |||||
| IgG3 | 43.9 | 100 | |||||
| IgG4 | 65.85 | 100 | |||||
| E. multilocularis crude antigen from whole cysts | alveolar echinococcosis patients | IgG | 83 | 99 | 4 °C | Itoh et al., 2013 [14] | |
| IgG4 | 91 | 98 | |||||
| recEm18 (recombinant antigen) | IgG | 78 | 85 | ||||
| IgG4 | 78 | 100 | |||||
| S. stercoralis | filariform S. ratti larva crude antigen | S. stercoralis + (agar plate culture) | IgG | 92.7 | 40.7 | 4 °C | Eamudomkarn et al., 2018 [15] |
| O. viverrini | adult O. viverrini crude antigen | O. viverrini egg + | IgG | 43.0 | 64.5 | −70 °C | Tesana et al., 2007 [16] |
| IgG4 | 45.9 | 67.2 |
3.3. Schistosomiasis Japonica
3.4. Paragonimiasis
3.5. Echinococcosis
3.6. Strongyloidiasis
3.7. Opisthorchiasis
4. Application of Urine ELISA to the Sentinel Surveillance System
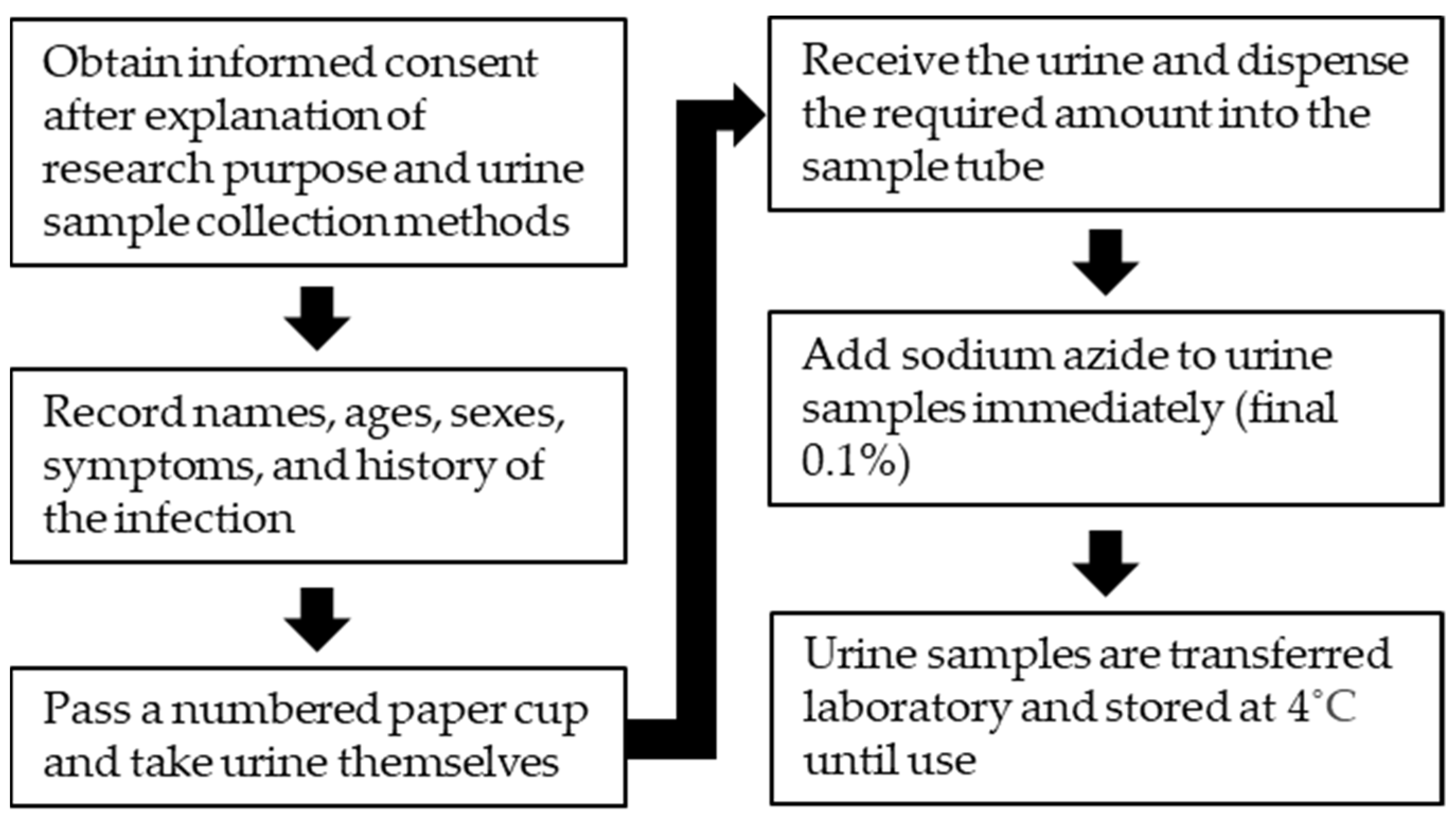
5. Collection, Handling, and Storage of Urine Samples for Antibody Detection
6. Discussion and Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Conflicts of Interest
References
- Zhao, M.; Li, M.; Yang, Y.; Guo, Z.; Sun, Y.; Shao, C.; Sun, W.; Gao, Y. A comprehensive analysis and annotation of human normal urinary proteome. Sci. Rep. 2017, 7, 3024. [Google Scholar] [CrossRef]
- Katsuragi, K.; Noda, A.; Tachikawa, T.; Azuma, A.; Mukai, F.; Murakami, K.; Fujioka, T.; Kato, M.; Asaka, M. Highly sensitive urine-based enzyme-linked immunosorbent assay for detection of antibody to Helicobacter pylori. Helicobacter 1998, 3, 289–295. [Google Scholar] [CrossRef] [PubMed]
- Takagi, A. Detection of H. pylori by serum and urine-based ELISA. Nihon Rinsho 2003, 61, 88–91. (In Japanese) [Google Scholar] [PubMed]
- Itoh, M.; Weerasooriya, M.V.; Qiu, G.; Gunawardena, N.K.; Anantaphruti, M.T.; Tesana, S.; Rattanaxay, P.; Fujimaki, Y.; Kimura, E. Sensitive and specific enzyme-linked immunosorbent assay for the diagnosis of Wuchereria bancrofti infection in urine samples. Am. J. Trop. Med. Hyg. 2001, 65, 362–365. [Google Scholar] [CrossRef] [PubMed]
- Alemohammad, M.M.; Foley, T.J.; Cohen, H. Detection of immunoglobulin G antibodies to Helicobacter pylori in urine by an enzyme immunoassay method. J. Clin. Microbiol. 1993, 31, 2174–2177. [Google Scholar] [CrossRef]
- Takahashi, S.; Machikawa, F.; Noda, A.; Oda, T.; Tachikawa, T. Detection of immunoglobulin G and A antibodies to rubella virus in urine and antibody responses to vaccine-induced infection. Clin. Diagn. Lab. Immunol. 1998, 5, 24–27. [Google Scholar] [CrossRef] [PubMed]
- Vazquez, S.; Cabezas, S.; Perez, A.B.; Pupo, M.; Ruiz, D.; Calzada, N.; Bernardo, L.; Castro, O.; Gonzalez, D.; Serrano, T.; et al. Kinetics of antibodies in sera, saliva, and urine samples from adult patients with primary or secondary dengue 3 virus infections. Int. J. Infect. Dis. 2007, 11, 256–262. [Google Scholar] [CrossRef]
- Elsana, S.; Sikuler, E.; Yaari, A.; Shemer-Avni, Y.; Abu-Shakra, M.; Buskila, D.; Katzman, P.; Naggan, L.; Margalith, M. HCV antibodies in saliva and urine. J. Med. Virol. 1998, 55, 24–27. [Google Scholar] [CrossRef]
- Cao, Y.; Friedman-Kien, A.E.; Chuba, J.V.; Mirabile, M.; Hosein, B. IgG antibodies to HIV-1 in urine of HIV-1 seropositive individuals. Lancet 1988, 1, 831–832. [Google Scholar] [CrossRef]
- Islam, M.Z.; Itoh, M.; Shamsuzzaman, S.M.; Mirza, R.; Matin, F.; Ahmed, I.; Shamsuzzaman Choudhury, A.K.; Hossain, M.A.; Qiu, X.G.; Begam, N.; et al. Diagnosis of visceral leishmaniasis by enzyme-linked immunosorbent assay using urine samples. Clin. Diagn. Lab. Immunol. 2002, 9, 789–794. [Google Scholar] [CrossRef][Green Version]
- Itoh, M.; Ohta, N.; Kanazawa, T.; Nakajima, Y.; Sho, M.; Minai, M.; Daren, Z.; Chen, Y.; He, H.; He, Y.K.; et al. Sensitive enzyme-linked immunosorbent assay with urine samples: A tool for surveillance of schistosomiasis japonica. Southeast Asian J. Trop. Med. Public Health 2003, 34, 469–472. [Google Scholar]
- Qiu, X.G.; Nakamura-Uchiyama, F.; Nawa, Y.; Itoh, M. A tool for mass-screening of paragonimiasis: An enzyme-linked immunosorbent assay with urine samples. Trop. Med. Health 2016, 44, 19. [Google Scholar] [CrossRef]
- Sunita, T.; Dubey, M.L.; Khurana, S.; Malla, N. Specific antibody detection in serum, urine and saliva samples for the diagnosis of cystic echinococcosis. Acta Trop. 2007, 101, 187–191. [Google Scholar] [CrossRef]
- Itoh, M.; Sako, Y.; Itoh, S.; Ishikawa, Y.; Akabane, H.; Nakaya, K.; Nagaoka, F.; Ito, A. Immunodiagnosis of alveolar echinococcosis using urine samples. Parasitol. Int. 2013, 62, 514–516. [Google Scholar] [CrossRef]
- Eamudomkarn, C.; Sithithaworn, P.; Kamamia, C.; Yakovleva, A.; Sithithaworn, J.; Kaewkes, S.; Techasen, A.; Loilome, W.; Yongvanit, P.; Wangboon, C.; et al. Diagnostic performance of urinary IgG antibody detection: A novel approach for population screening of strongyloidiasis. PLoS ONE 2018, 13, e0192598. [Google Scholar] [CrossRef] [PubMed]
- Tesana, S.; Srisawangwong, T.; Sithithaworn, P.; Itoh, M.; Phumchaiyothin, R. The ELISA-based detection of anti-Opisthorchis viverrini IgG and IgG4 in samples of human urine and serum from an endemic area of north-eastern Thailand. Ann. Trop. Med. Parasitol. 2007, 101, 585–591. [Google Scholar] [CrossRef] [PubMed]
- Chanteau, S.; Glaziou, P.; Moulia-Pelat, J.P.; Plichart, C.; Luquiaud, P.; Cartel, J.L. Low positive predictive value of anti-Brugia malayi IgG and IgG4 serology for the diagnosis of Wuchereria bancrofti. Trans. R. Soc. Trop. Med. Hyg. 1994, 88, 661–662. [Google Scholar] [CrossRef]
- Bergquist, R.; Johansen, M.V.; Utzinger, J. Diagnostic dilemmas in helminthology: What tools to use and when? Trends Parasitol. 2009, 25, 151–156. [Google Scholar] [CrossRef]
- Weerasooriya, M.V.; Itoh, M.; Mudalige, M.P.; Qiu, X.G.; Kimura, E.; Gunawardena, N.K.; Fujimaki, Y. Human infection with Wuchereria bancrofti in Matara, Sri Lanka: The use, in parallel, of an ELISA to detect filaria-specific IgG4 in urine and of ICT card tests to detect filarial antigen in whole blood. Ann. Trop. Med. Parasitol. 2003, 97, 179–185. [Google Scholar] [CrossRef]
- Kuriyama, M. The detection of urinary immunoglobulins with E.L.I.S.A. (author’s transl). Nihon Hinyokika Gakkai Zasshi 1979, 70, 228–234. (In Japanese) [Google Scholar] [CrossRef]
- Chirag, S.; Fomda, B.A.; Khan, A.; Malik, A.A.; Lone, G.N.; Khan, B.A.; Zahoor, D. Detection of hydatid-specific antibodies in the serum and urine for the diagnosis of cystic echinococcosis in patients from the Kashmir Valley, India. J. Helminthol. 2015, 89, 232–237. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.; Sharma, U.; Mishra, J. Post-kala-azar dermal leishmaniasis: Recent developments. Int. J. Derm. 2011, 50, 1099–1108. [Google Scholar] [CrossRef]
- Gedda, M.R.; Singh, B.; Kumar, D.; Singh, A.K.; Madhukar, P.; Upadhyay, S.; Singh, O.P.; Sundar, S. Post kala-azar dermal leishmaniasis: A threat to elimination program. PLoS Negl. Trop. Dis. 2020, 14, e0008221. [Google Scholar] [CrossRef] [PubMed]
- Van Griensven, J.; Diro, E. Visceral Leishmaniasis: Recent Advances in Diagnostics and Treatment Regimens. Infect. Dis. Clin. N. Am. 2019, 33, 79–99. [Google Scholar] [CrossRef] [PubMed]
- Mohammed, E.A.; Wright, E.P.; Kager, P.A.; Laarman, J.J.; Pondman, K.W. ELISA using intact promastigotes for immunodiagnosis of kala-azar. Trans. R. Soc. Trop. Med. Hyg. 1985, 79, 344–350. [Google Scholar] [CrossRef]
- Zijlstra, E.E.; Daifalla, N.S.; Kager, P.A.; Khalil, E.A.; El-Hassan, A.M.; Reed, S.G.; Ghalib, H.W. rK39 enzyme-linked immunosorbent assay for diagnosis of Leishmania donovani infection. Clin. Diagn. Lab. Immunol. 1998, 5, 717–720. [Google Scholar] [CrossRef]
- Harith, A.E.; Kolk, A.H.; Kager, P.A.; Leeuwenburg, J.; Muigai, R.; Kiugu, S.; Laarman, J.J. A simple and economical direct agglutination test for serodiagnosis and sero-epidemiological studies of visceral leishmaniasis. Trans. R. Soc. Trop. Med. Hyg. 1986, 80, 583–586. [Google Scholar] [CrossRef]
- Ejazi, S.A.; Bhattacharya, P.; Bakhteyar, M.A.; Mumtaz, A.A.; Pandey, K.; Das, V.N.; Das, P.; Rahaman, M.; Goswami, R.P.; Ali, N. Noninvasive Diagnosis of Visceral Leishmaniasis: Development and Evaluation of Two Urine-Based Immunoassays for Detection of Leishmania donovani Infection in India. PLoS Negl. Trop. Dis. 2016, 10, e0005035. [Google Scholar] [CrossRef] [PubMed]
- Islam, M.Z.; Itoh, M.; Takagi, H.; Islam, A.U.; Ekram, A.R.; Rahman, A.; Takesue, A.; Hashiguchi, Y.; Kimura, E. Enzyme-linked immunosorbent assay to detect urinary antibody against recombinant rKRP42 antigen made from Leishmania donovani for the diagnosis of visceral leishmaniasis. Am. J. Trop. Med. Hyg. 2008, 79, 599–604. [Google Scholar] [CrossRef]
- Ghosh, P.; Bhaskar, K.R.; Hossain, F.; Khan, M.A.; Vallur, A.C.; Duthie, M.S.; Hamano, S.; Salam, M.A.; Huda, M.M.; Khan, M.G.; et al. Evaluation of diagnostic performance of rK28 ELISA using urine for diagnosis of visceral leishmaniasis. Parasit. Vectors 2016, 9, 383. [Google Scholar] [CrossRef]
- Nutman, T.B. Insights into the pathogenesis of disease in human lymphatic filariasis. Lymphat. Res. Biol. 2013, 11, 144–148. [Google Scholar] [CrossRef]
- Simonsen, P.E.; Niemann, L.; Meyrowitsch, D.W. Wuchereria bancrofti in Tanzania: Microfilarial periodicity and effect of blood sampling time on microfilarial intensities. Trop. Med. Int. Health 1997, 2, 153–158. [Google Scholar] [CrossRef] [PubMed]
- Weil, G.J.; Lammie, P.J.; Weiss, N. The ICT Filariasis Test: A rapid-format antigen test for diagnosis of bancroftian filariasis. Parasitol. Today 1997, 13, 401–404. [Google Scholar] [CrossRef]
- Itoh, M.; Weerasooriya, M.V.; Yahathugoda, T.C.; Takagi, H.; Samarawickrema, W.A.; Nagaoka, F.; Kimura, E. Effects of 5 rounds of mass drug administration with diethylcarbamazine and albendazole on filaria-specific IgG4 titers in urine: 6-year follow-up study in Sri Lanka. Parasitol. Int. 2011, 60, 393–397. [Google Scholar] [CrossRef] [PubMed]
- Samad, M.S.; Itoh, M.; Moji, K.; Hossain, M.; Mondal, D.; Alam, M.S.; Kimura, E. Enzyme-linked immunosorbent assay for the diagnosis of Wuchereria bancrofti infection using urine samples and its application in Bangladesh. Parasitol. Int. 2013, 62, 564–567. [Google Scholar] [CrossRef]
- Gryseels, B. Schistosomiasis. Infect. Dis. Clin. N. Am. 2012, 26, 383–397. [Google Scholar] [CrossRef]
- Ishii, A.; Owhashi, M. Enzyme-linked immunosorbent assay with egg antigens of Schistosoma japonicum. Z. Parasitenkd. 1982, 67, 279–287. [Google Scholar] [CrossRef]
- Lin, D.D.; Xu, J.M.; Zhang, Y.Y.; Liu, Y.M.; Hu, F.; Xu, X.L.; Li, J.Y.; Gao, Z.L.; Wu, H.W.; Kurtis, J.; et al. Evaluation of IgG-ELISA for the diagnosis of Schistosoma japonicum in a high prevalence, low intensity endemic area of China. Acta Trop. 2008, 107, 128–133. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, A.; Doanh, P.N.; Maruyama, H. Paragonimus and paragonimiasis in Asia: An update. Acta Trop. 2019, 199, 105074. [Google Scholar] [CrossRef] [PubMed]
- Imai, J. Evaluation of ELISA for the diagnosis of paragonimiasis westermani. Trans. R. Soc. Trop. Med. Hyg. 1987, 81, 3–6. [Google Scholar] [CrossRef]
- Ikeda, T.; Oikawa, Y.; Nishiyama, T. Enzyme-linked immunosorbent assay using cysteine proteinase antigens for immunodiagnosis of human paragonimiasis. Am. J. Trop. Med. Hyg. 1996, 55, 435–437. [Google Scholar] [CrossRef]
- Romig, T.; Deplazes, P.; Jenkins, D.; Giraudoux, P.; Massolo, A.; Craig, P.S.; Wassermann, M.; Takahashi, K.; de la Rue, M. Ecology and Life Cycle Patterns of Echinococcus Species. Adv. Parasitol. 2017, 95, 213–314. [Google Scholar] [CrossRef] [PubMed]
- Brunetti, E.; Kern, P.; Vuitton, D.A. Expert consensus for the diagnosis and treatment of cystic and alveolar echinococcosis in humans. Acta Trop. 2010, 114, 1–16. [Google Scholar] [CrossRef]
- Olsen, A.; van Lieshout, L.; Marti, H.; Polderman, T.; Polman, K.; Steinmann, P.; Stothard, R.; Thybo, S.; Verweij, J.J.; Magnussen, P. Strongyloidiasis--the most neglected of the neglected tropical diseases? Trans. R. Soc. Trop. Med. Hyg. 2009, 103, 967–972. [Google Scholar] [CrossRef] [PubMed]
- Requena-Mendez, A.; Chiodini, P.; Bisoffi, Z.; Buonfrate, D.; Gotuzzo, E.; Munoz, J. The laboratory diagnosis and follow up of strongyloidiasis: A systematic review. PLoS Negl. Trop. Dis. 2013, 7, e2002. [Google Scholar] [CrossRef]
- Kaewkes, S. Taxonomy and biology of liver flukes. Acta Trop. 2003, 88, 177–186. [Google Scholar] [CrossRef] [PubMed]
- Thamavit, W.; Pairojkul, C.; Tiwawech, D.; Itoh, M.; Shirai, T.; Ito, N. Promotion of cholangiocarcinogenesis in the hamster liver by bile duct ligation after dimethylnitrosamine initiation. Carcinogenesis 1993, 14, 2415–2417. [Google Scholar] [CrossRef] [PubMed]
- Sripa, B.; Brindley, P.J.; Mulvenna, J.; Laha, T.; Smout, M.J.; Mairiang, E.; Bethony, J.M.; Loukas, A. The tumorigenic liver fluke Opisthorchis viverrini—Multiple pathways to cancer. Trends Parasitol. 2012, 28, 395–407. [Google Scholar] [CrossRef]
- Pinlaor, P.; Pongsamart, P.; Hongsrichan, N.; Sangka, A.; Srilunchang, T.; Mairiang, E.; Sithithaworn, P.; Pinlaor, S. Specific serum IgG, but not IgA, antibody against purified Opisthorchis viverrini antigen associated with hepatobiliary disease and cholangiocarcinoma. Parasitol. Int. 2012, 61, 212–216. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Ending the Neglect to Attain the Sustainable Development Goals: A Road Map for Neglected Tropical Diseases 2021–2030. 2020. Available online: https://www.who.int/teams/control-of-neglected-tropical-diseases/ending-ntds-together-towards-2030 (accessed on 22 April 2021).
- World Health Organization. Global programme to eliminate lymphatic filariasis. Wkly. Epidemiol. Rec. 2020, 95, 509–524. [Google Scholar]
- Adachi, K.; Kawamura, A.; Ono, M.; Masuzaki, K.; Takashima, T.; Yuki, M.; Fujishiro, H.; Ishihara, S.; Kinoshita, Y. Comparative evaluation of urine-based and other minimally invasive methods for the diagnosis of Helicobacter pylori infection. J. Gastroenterol. 2002, 37, 703–708. [Google Scholar] [CrossRef]
- Terada, K.; Niizuma, T.; Kataoka, N.; Niitani, Y. Testing for rubella-specific IgG antibody in urine. Pediatr. Infect. Dis. J. 2000, 19, 104–108. [Google Scholar] [CrossRef] [PubMed]
- Lichstein, H.C.; Soule, M.H. Studies of the Effect of Sodium Azide on Microbic Growth and Respiration: I. The Action of Sodium Azide on Microbic Growth. J. Bacteriol. 1944, 47, 221–230. [Google Scholar] [CrossRef] [PubMed]
- Ottesen, E.A.; Skvaril, F.; Tripathy, S.P.; Poindexter, R.W.; Hussain, R. Prominence of IgG4 in the IgG antibody response to human filariasis. J. Immunol. 1985, 134, 2707–2712. [Google Scholar] [PubMed]
- Weil, G.J.; Ogunrinade, A.F.; Chandrashekar, R.; Kale, O.O. IgG4 subclass antibody serology for onchocerciasis. J. Infect. Dis. 1990, 161, 549–554. [Google Scholar] [CrossRef] [PubMed]
- Santra, A.; Bhattacharya, T.; Chowdhury, A.; Ghosh, A.; Ghosh, N.; Chatterjee, B.P.; Mazumder, D.N. Serodiagnosis of ascariasis with specific IgG4 antibody and its use in an epidemiological study. Trans. R. Soc. Trop. Med. Hyg. 2001, 95, 289–292. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nagaoka, F.; Yamazaki, T.; Akashi-Takamura, S.; Itoh, M. Detection of Urinary Antibodies and Its Application in Epidemiological Studies for Parasitic Diseases. Vaccines 2021, 9, 778. https://doi.org/10.3390/vaccines9070778
Nagaoka F, Yamazaki T, Akashi-Takamura S, Itoh M. Detection of Urinary Antibodies and Its Application in Epidemiological Studies for Parasitic Diseases. Vaccines. 2021; 9(7):778. https://doi.org/10.3390/vaccines9070778
Chicago/Turabian StyleNagaoka, Fumiaki, Tatsuya Yamazaki, Sachiko Akashi-Takamura, and Makoto Itoh. 2021. "Detection of Urinary Antibodies and Its Application in Epidemiological Studies for Parasitic Diseases" Vaccines 9, no. 7: 778. https://doi.org/10.3390/vaccines9070778
APA StyleNagaoka, F., Yamazaki, T., Akashi-Takamura, S., & Itoh, M. (2021). Detection of Urinary Antibodies and Its Application in Epidemiological Studies for Parasitic Diseases. Vaccines, 9(7), 778. https://doi.org/10.3390/vaccines9070778






