A Possible Role for HSV-1-Specific Humoral Response and PILRA rs1859788 Polymorphism in the Pathogenesis of Parkinson’s Disease
Abstract
:1. Introduction
2. Materials and Methods
2.1. Patients and Controls
2.2. ELISA
2.2.1. α-Synuclein Measurement
2.2.2. Anti-Herpetic IgG Antibody Measurements
2.3. PILRA rs1859788 Genotyping
2.4. Statistical Analysis
3. Results
3.1. Clinical Characterization of the Study Population
3.2. Plasmatic α-Synuclein Concentration
3.3. Virological Results
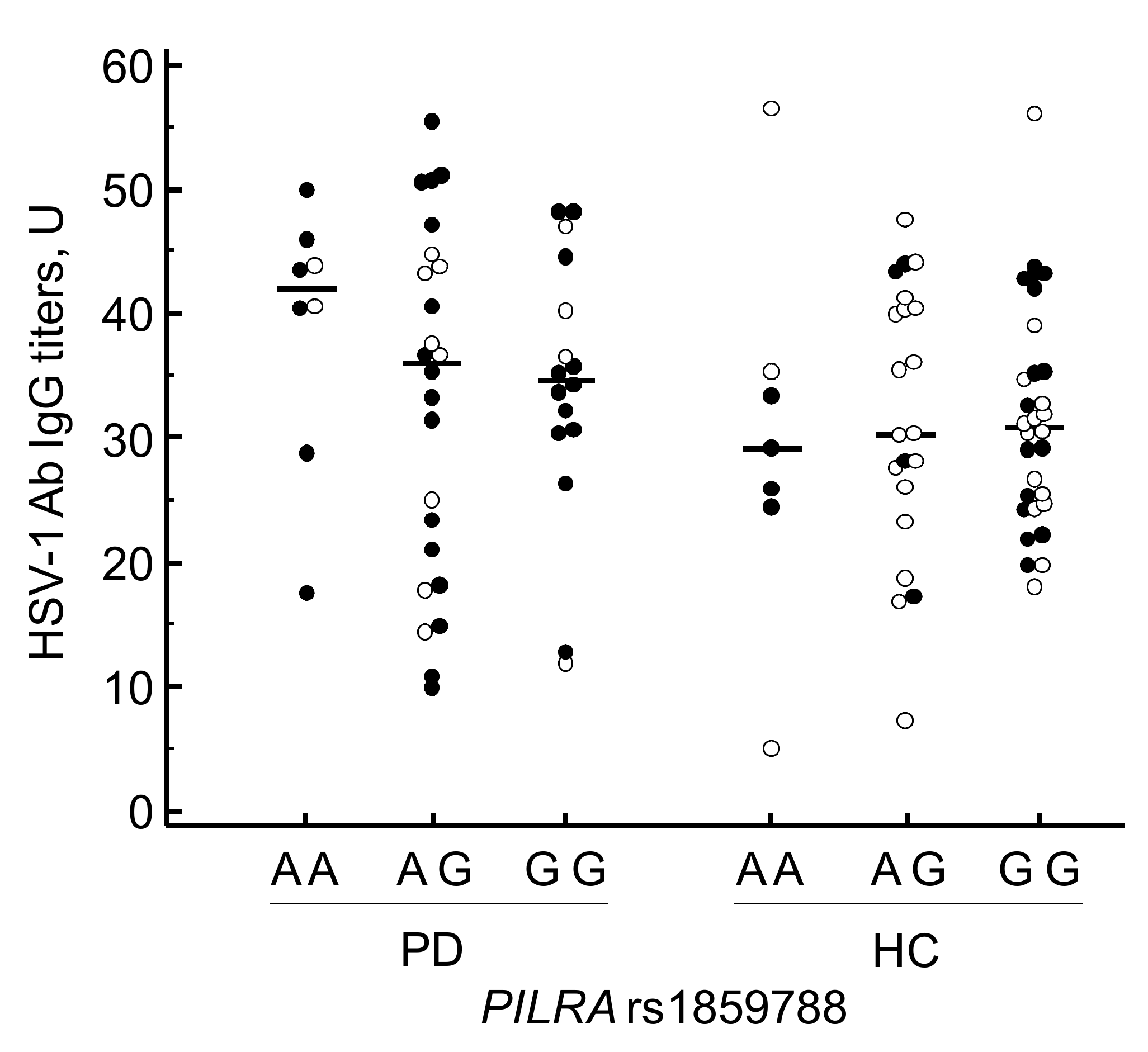
3.4. PILRA rs1859788 Genotyping
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Simon, D.K.; Tanner, C.M.; Brundin, P. Parkinson disease epidemiology, pathology, genetics, and pathophysiology. Clin. Geriatr. Med. 2020, 36, 1–12. [Google Scholar] [CrossRef]
- Pfeiffer, R.F. Non-motor symptoms in Parkinson’s disease. Parkinsonism Relat. Disord. 2016, 22, S119–S122. [Google Scholar] [CrossRef]
- Nussbaum, R.L.; Ellis, C.E. Alzheimer’s disease and Parkinson’s disease. N. Engl. J. Med. 2003, 348, 1356–1364. [Google Scholar] [CrossRef] [Green Version]
- Emamzadeh, F.N.; Surguchov, A. Parkinson’s disease: Biomarkers, treatment, and risk factors. Front. Neurosci. 2018, 12, 612. [Google Scholar] [CrossRef] [PubMed]
- Du, X.Y.; Xie, X.X.; Liu, R.T. The role of α-Synuclein oligomers in Parkinson’s disease. Int. J. Mol. Sci. 2020, 21, 8645. [Google Scholar] [CrossRef]
- Gelders, G.; Baekelandt, V.; Van der Perren, A. Linking neuroinflammation and neurodegeneration in Parkinson’s disease. J. Immunol. Res. 2018, 2018, 4784268. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Piancone, F.; Saresella, M.; La Rosa, F.; Marventano, I.; Meloni, M.; Navarro, J.; Clerici, M. Inflammatory responses to monomeric and aggregated α-synuclein in peripheral blood of Parkinon’s disease patients. Front. Neurosci. 2021, 15, 639646. [Google Scholar] [CrossRef]
- Olsen, L.K.; Dowd, E.; McKernan, D.P. A role for viral infections in Parkinson’s etiology? Neuronal. Signal. 2018, 2. [Google Scholar] [CrossRef] [PubMed]
- Elizan, T.S.; Madden, D.L.; Noble, G.R.; Herrmann, K.L.; Gardner, J.; Schwartz, J.; Smith, H., Jr.; Sever, J.L.; Yahr, M.D. Viral antibodies in serum and CSF of Parkinsonian patients and controls. Arch. Neurol. 1979, 36, 529–534. [Google Scholar] [CrossRef] [PubMed]
- Wu, W.Y.Y.; Kang, K.H.; Chen, S.L.S.; Chiu, S.Y.H.; Yen, A.M.F.; Fann, J.C.Y.; Su, C.W.; Liu, H.C.; Fu, W.M.; Chen, H.H.; et al. Hepatitis C virus infection: A risk factor for Parkinson’s disease. J. Viral. Hepatol. 2015, 22, 784–791. [Google Scholar] [CrossRef] [PubMed]
- Sa, A.C.C.; Madsen, H.; Brown, J.R. Shared molecular signatures across neurodegenerative diseases and herpes virus infections highlights potential mechanisms for maladaptive innate immune responses. Sci. Rep. 2019, 9, 8795. [Google Scholar]
- Von Economo, C. Die Encephalitis Lethargica, ihre Nachkrankheiten und ihren Behandlung; Urban & Schwarzenberg: Vienna, Austria, 1929; pp. 394–474. [Google Scholar]
- Jang, H.; Bolz, D.; Strum-Ramirez, K.; Shepher, K.R.; Jiao, Y.; Webster, R.; Smeyne, R.J. Highly pathogenic H5N1 influenza virus can enter the central nervous system and induce neuroinflammation and neurodegeneration. Proc. Natl. Acad. Sci. USA 2009, 106, 14063–14068. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Marttila, R.; Arstila, P.; Nikoskelainen, J.; Halonen, P.; Rinne, U. Viral antibodies in the sera from patients with Parkinson disease. Eur. Neurol. 1977, 15, 25–33. [Google Scholar] [CrossRef] [PubMed]
- Marttila, R.J.; Rinne, U.K. Herpes simplex virus antibodies in patients with Parkinson’s disease. J. Neurol. Sci. 1978, 35, 375–379. [Google Scholar] [CrossRef]
- Marttila, R.J.; Rinne, U.K.; Halonen, P.; Madden, D.L.; Sever, J.L. Herpes viruses and Parkinsonism: Herpes simplex virus types 1 and 2, and cytomegalovirus antibodies in serum and CSF. Arch. Neurol. 1981, 38, 19–21. [Google Scholar] [CrossRef]
- Marttila, R.J.; Rinne, U.K.; Tiilikainen, A. Virus antibodies in Parkinson’s disease: Herpes simplex and measles virus antibodies in serum and CSF and their relation to HLA types. J. Neurol. Sci. 1982, 54, 227–238. [Google Scholar] [CrossRef]
- Bu, X.L.; Yao, X.Q.; Jiao, S.S.; Zeng, F.; Liu, Y.H.; Xiang, Y.; Liang, C.R.; Wang, Q.H.; Wang, X.; Cao, H.Y.; et al. A study on the association between infectious burden and Alzheimer’s disease. Eur. J. Neurol. 2015, 22, 1519–1525. [Google Scholar] [CrossRef] [PubMed]
- Harris, M.A.; Tsui, J.K.; Marion, S.A.; Shen, H.; Teschke, K. Association of Parkinson’s disease with infections and occupational exposure to possible vectors. Mov. Disord. 2012, 27, 1111–1117. [Google Scholar] [CrossRef] [PubMed]
- Fatahzadeh, M.; Schwartz, R.A. Human herpes simplex virus infections: Epidemiology, pathogenesis, symptomatology, diagnosis, and management. J. Am. Acad. Dermatol. 2007, 57, 737–763. [Google Scholar] [CrossRef]
- Nicoll, M.P.; Proenca, J.T.; Efstathiou, S. The molecular basis of herpes simplex virus latency. FEMS. Microbiol. Rev. 2012, 26, 684–705. [Google Scholar] [CrossRef] [PubMed]
- World Health Organizaton. Herpes Simplex Virus. 2020. Available online: http://www.who.int/mediacentre/factsheets/fs400/en/ (accessed on 1 June 2021).
- Caggiu, E.; Paulus, K.; Arru, G.; Piredda, R.; Sechi, G.P.; Sechi, A. Humoral cross reactivity between a-synuclein and herpes simplex-1 epitope in Parkinson’s disease, a triggering role in the disease? J. Neuroloimmunol. 2016, 291, 110–114. [Google Scholar] [CrossRef]
- Caggiu, E.; Paulus, K.; Galleri, G.; Arru, G.; Manetti, R.; Sechi, G.P.; Sechi, L.A. Homologous HSV1 and alpha-synuclein peptides stimulate a T cell response in Parkinson’s disease. J. Neuroimmunol. 2017, 310, 26–31. [Google Scholar] [CrossRef] [PubMed]
- La Rosa, F.; Agostini, S.; Bianchi, A.; Nemni, R.; Piancone, F.; Marventano, I.; Mancuso, R.; Saresella, M.; Clerici, M. Herpes simplex virus-1 (HSV-1) infection induces a potent but ineffective IFN-l production in immune cells of AD and PD patients. J. Trans. Med. 2019, 17, 286. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hawkes, C.H.; Del Tredici, K.; Braak, H. Parkinson’s disease: The dual hit theory reviseted. Ann. N. Y. Acad. Sci. 2009, 1170, 615–622. [Google Scholar] [CrossRef]
- Mori, I. Herpes simplex virus type 1 in the aged brain through hypothetical expression of accessory genes. J. Neurovirol. 2010, 16, 203–207. [Google Scholar] [CrossRef] [PubMed]
- Mori, I. Viremic attack explains the dual-hit theory of Parkinon’s disease. Med. Hypothesis 2017, 101, 33–36. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, N.; Nagata, T.; Shinagawa, S.; Oka, N.; Shimada, K.; Shimizu, A.; Tatebayashi, Y.; Yamada, H.; Nakayama, K.; Kondo, K. Increase in the IgG avidity index due to herpes simplex virus type 1 reactivation and its relationship with cognitive function in amnestic mild cognitive impairment and Alzheimer’s disease. Biochem. Biophys. Res. Commun. 2013, 840, 907–911. [Google Scholar] [CrossRef] [PubMed]
- Mancuso, R.; Baglio, F.; Cabinio, M.; Calabrese, E.; Hernis, A.; Nemni, R.; Clerici, M. Titers of herpes simplex virus type 1 antibodies positively correlate with grey matter volumes in Alzheimer’s disease. J. Alzheimers Dis. 2014, 33, 741–745. [Google Scholar] [CrossRef]
- Agostini, S.; Mancuso, R.; Baglio, F.; Cabinio, M.; Hernis, A.; Costa, A.S.; Calabrese, E.; Nemni, R.; Clerici, M. High avidity HSV-1 antibodies correlate with absence of amnestic Mild Cognitive Impairment conversion to Alzheimer’s disease. Brain Behav. Immun. 2016, 58, 254–260. [Google Scholar] [CrossRef]
- Mancuso, R.; Cabinio, M.; Agostini, S.; Baglio, F.; Clerici, M. HSV-1 specific IgG3 titers correlate with brain cortical thinning in individuals with Mild Cognitive Impairment and Alzheimer’s disease. Vaccines 2020, 8, 255. [Google Scholar] [CrossRef]
- Wozniak, M.A.; Itzhaki, R.F.; Shipley, S.J.; Dobson, C.B. Herpes simplex virus infection causes cellular β-amyloid accumulation and secretase upregulation. Neurosci. Lett. 2007, 429, 95–100. [Google Scholar] [CrossRef]
- Alvarez, G.; Aldudo, J.; Alonso, M.; Santana, S.; Valdivieso, F. Herpes simplex virus type 1 induces nuclear accumulation of hyperphosphorylated tau in neuronal cells. J. Neurosci. Res. 2012, 90, 1020–1029. [Google Scholar] [CrossRef]
- De Chiara, G.; Piacentini, R.; Fabiani, M.; Mastrodonato, A.; Marcocci, M.E.; Limongi, D.; Napoletani, G.; Protto, V.; Coluccio, P.; Celestino, I.; et al. Recurrent herpes simplex virus-1 infection induces hallmarks of neurodegeneration and cognitive deficits in mice. PLoS Pathog. 2019, 15, e1007617. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Santana, S.; Bullido, M.J.; Recuero, M.; Valdivieso, F.; Aldudo, J. Herpes simplex virus type I induces an incomplete autophagic response in human neuroblatoma cells. J. Alzheimer’s Dis. 2012, 30, 815–831. [Google Scholar] [CrossRef] [Green Version]
- Wang, W.Z.; Fang, X.H.; Cheng, X.M.; Jiang, D.H.; Lin, Z.J. A case-control study on the environmental risk factors of Parkinsons’s Disease in Tianjin, China. Neuroepidemiology 1993, 12, 209–218. [Google Scholar] [CrossRef]
- Bu, X.L.; Wang, X.; Xiang, Y.; Shen, L.L.; Wang, Q.H.; Liu, Y.H.; Jiao, S.S.; Wang, Y.R.; Cao, H.Y.; Yi, X.; et al. The association between infectious burden and Parkinson’s disease: A case-control study. Parkinsonsism Relat. Disord. 2015, 12, 877–881. [Google Scholar] [CrossRef]
- Cury, R.G.; Lopez, W.O. Bilateral striatal lesion due to herpesvirus-6 infection. J. Neurol. Sci. 2015, 358, 538–539. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Shiratori, I.; Uehori, J.; Ikawa, M.; Arase, H. Neutrophil infiltration during inflammation is regulated by PILRA via modulation of integrin activation. Nat. Immunol. 2014, 2, 860–870. [Google Scholar]
- Kohyama, M.; Matsuoka, S.; Shida, K.; Sugihara, F.; Aoshi, T.; Kishida, K.; Ishii, K.J.; Arase, H. Monocyte infiltration into obese and fibrilized tissues is regulated by PILRα. Eur. J. Immunol. 2014, 46, 1214–1223. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lambert, J.C.; Ibrahim-Verbaas, C.A.; Harold, D.; Naj, A.C.; Sims, R.; Bellenguez, C.; DeStafano, A.L.; Bis, J.C.; Beecham, G.W.; Grenier-Boley, B.; et al. Meta-analysis of 74,046 individuals identifies 11 new susceptibility loci for Alzheimer’s disease. Nat. Gen. 2013, 45, 1452–1458. [Google Scholar] [CrossRef] [Green Version]
- Rathore, N.; Ramani, S.R.; Pantua, H.; Payandeh, J.; Bhangale, T.; Wuster, A.; Kapoor, M.; Sun, Y.; Kapadia, S.B.; Gonzales, L.; et al. Paired immunoglobulin-like type 2 receptos alpha G78R variant alters ligand binding and confers protection to Alzheimer’s disease. PLoS Genet. 2018, 14, e1007427. [Google Scholar] [CrossRef] [Green Version]
- Satoh, T.; Arii, J.; Suenaga, T.; Wang, J.; Kogure, A.; Uehori, J.; Arase, N.; Shiratori, I.; Tanaka, S.; Kawaguchi, Y.; et al. PILRα is a herpes simplex virus-1 entry co-receptors that associated with glycoprotein B. Cell 2008, 132, 935–944. [Google Scholar] [CrossRef] [Green Version]
- Agostini, S.; Costa, A.S.; Mancuso, R.; Guerini, F.R.; Nemni, R.; Clerici, M. The PILRA G78R variant correlated with higher HSV-1 specific IgG titers in Alzheimer’s disease. Cell. Mol. Neurobiol. 2019, 39, 1217–1221. [Google Scholar] [CrossRef]
- Postuma, R.B.; Berg, D.; Stern, M.; Powe, W.; Olanow, C.W.; Oertel, W.; Obeso, J.; Marek, K.; Litvan, I.; Lang, A.E.; et al. MDS clinical diagostic criteria for Parkinson’s disease. Mov. Disord. 2015, 30, 1591–1601. [Google Scholar] [CrossRef] [PubMed]
- Goetz, C.G.; Tilley, B.C.; Shaftman, S.R.; Stebbins, G.T.; Fahn, S.; Martinez-Martin, P.; Poewe, W.; Sampaio, C.; Stern, M.B.; Dodel, R.; et al. Movement disorder society-sponsored revision of the unified Parkinson’s disease rating scale (MDS-UPDRS): Scale presentation and clinimetric testing results. Mov. Disord. 2008, 23, 2129–2170. [Google Scholar] [CrossRef] [PubMed]
- Tomilson, C.L.; Stowe, R.; Patel, S.; Rick, C.; Gay, R.; Clarke, C.E. Systematic review of Levodopa dose equivalency reporting in Parkinson’s disease. Mov. Disord. 2010, 25, 2649–2653. [Google Scholar]
- Yuan, H.; You, J.; You, H.; Zheng, C. Herpes Simplex Virus 1 UL36USP antagonizes type I interferon-mediated antiviral innate immunity. J. Virol. 2018, 92. [Google Scholar] [CrossRef] [Green Version]
- De Virgilio, A.; Greco, A.; Fabbrini, G.; Inghilleri, M.; Rizzo, M.I.; Gallo, A.; Conte, M.; Rosato, C.; Ciniglio Appiani, M.; de Vincentiis, M. Parkinson’s disease: Autoimmunity and neuroinflammation. Autoimmun. Rev. 2016, 15, 1005–1011. [Google Scholar] [CrossRef] [Green Version]
- Garretti, F.; Agalliu, D.; Lindestam Arlehamn, C.S.; Sette, A.; Sulzer, D. Autoimmunity in Parkinson’s disease: The role of α-synuclein-specific T cells. Front. Immunol. 2019, 10, 303. [Google Scholar] [CrossRef] [Green Version]
- Mahdavi, T. Inflammation in Parkinson’s disease. In Scientific and Clinical Aspects; Thomas, M., Ed.; Springer International Publishing: Cham, Switzerland, 2014; pp. 105–126. [Google Scholar]
- Sulzer, D.; Alcalay, R.N.; Garretti, F.; Cote, L.; Kanter, E.; Agin-Liebes, J.; Liong, C.; McMurtrey, C.; Hildebrand, W.H.; Mao, X.; et al. T cells from patients with Parkinson’s disease recognize α-synuclein peptides. Nature 2017, 546, 656–661. [Google Scholar] [CrossRef] [Green Version]
- Bougea, A.; Stefai, L.; Paraskeva, G.P.; Emmanouilidou, E.; Vekrelis, K.; Kapaki, E. Plasma alpha-synucelin levels in patients with Parkinson’s disease: A systematic review and meta-analysis. Neurol. Sci. 2019, 40, 929–938. [Google Scholar] [CrossRef] [PubMed]
- Chang, C.W.; Yang, S.Y.; Yang, C.C.; Chang, C.W.; Wu, Y.R. Plasma and serum alpha-synuclein as a biomarker of diagnosis in patients with Parkinson’s disease. Front. Neurol. 2019, 10, 1388. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Agliardi, C.; Meloni, M.; Guerini, F.R.; Zanzottera, M.; Bolognesi, E.; Baglio, F.; Clerici, M. Oligomeric α-Syn and SNARE complex proteins in peripheral extracellular vesicles of neural origin are biomarkers for Parkinson’s disease. Neurobiol. Dis. 2021, 148, 105185. [Google Scholar] [CrossRef] [PubMed]
- Mousseau, D.D.; Banville, D.; L’Abbe, D.; Bouchard, P.; Shen, S.H. PILRα, a novel immunoreceptor tyrosine-based inhibitory motif-bearing protein, recruits SHP-1 upon tyrosine phosphorylation and is paired with the truncated counterpart PILRb. J. Biol. Chem. 2000, 275, 4467–4474. [Google Scholar] [CrossRef] [PubMed] [Green Version]

| Demographic and Clinical Characteristics | PD Patients | Healthy Controls |
|---|---|---|
| n. | 51 | 73 |
| Gender (M:F) | 15:36 | 32:41 |
| Age, years (mean ± SD) | 69.5 ± 8.5 | 69.5 ± 8.0 |
| Disease duration (mean ± SD) | 7.4 ± 5.1 | --- |
| MDS-UPDRS III (mean ± SD) | 27.6 ± 13.0 | --- |
| Modified H&Y (median; IQR) | 2.0; 1.5–3.0 | --- |
| LEDD, mg/die (mean ± SD) | 512.8 ± 280.8 | --- |
| α-synuclein, ng/mL (median, IQR) | 19.43; 15.22–23.12 1 | 12.25; 8.00–20.20 |
| Virological Characteristics | PD Patients | Healthy Controls |
|---|---|---|
| HSV-1 seropositivity, % | 98 | 92 |
| HSV-1 IgG, U (median; IQR) | 36.2; 32.8–40.6 1 | 30.3; 27.7–34.7 |
| HSV-1 avidity index, % (median; IQR) | 92.1; 83.4–97.5 | 88.6; 83.8–91.8 |
| CMV seropositivity, % | 88 | 98 |
| CMV IgG, AU/mL (median; IQR) | 161.2; 141.7–196.9 | 193.5; 159.0–221.2 |
| HHV-6 seropositivity, % | 56 | 67 |
| HHV-6, P.I. (median; IQR) | 5.4; 1.7–2.5 | 2.6; 2.0–3.3 |
| Groups | PILRA rs1859788 | PILRA rs1859788 | PILRA rs1859788 | ||||
|---|---|---|---|---|---|---|---|
| Genotype/Allele | A G | A G | G G | A (A + AG) | G G | A | G |
| PD, % | 16.3 | 51.0 | 32.7 | 67.3 | 32.7 | 41.8 | 58.2 |
| HC, % | 12.5 | 37.5 | 50.0 | 50.0 | 50.0 | 31.3 | 68.7 |
| Men | |||||||
| PD, % 1 | 17.7 | 47.0 | 35.3 | 64.7 2 | 35.3 | 41.2 | 58.8 |
| HC, % | 18.5 | 18.5 | 63.0 | 37.1 | 62.9 | 27.8 | 72.2 |
| Women | |||||||
| PD, % | 13.3 | 60.0 | 26.7 | 73.3 | 26.7 | 43.3 | 56.7 |
| HC, % | 8.1 | 51.4 | 40.5 | 59.5 | 40.5 | 33.8 | 66.2 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Agostini, S.; Mancuso, R.; Costa, A.S.; Citterio, L.A.; Guerini, F.R.; Meloni, M.; Navarro, J.; Clerici, M. A Possible Role for HSV-1-Specific Humoral Response and PILRA rs1859788 Polymorphism in the Pathogenesis of Parkinson’s Disease. Vaccines 2021, 9, 686. https://doi.org/10.3390/vaccines9070686
Agostini S, Mancuso R, Costa AS, Citterio LA, Guerini FR, Meloni M, Navarro J, Clerici M. A Possible Role for HSV-1-Specific Humoral Response and PILRA rs1859788 Polymorphism in the Pathogenesis of Parkinson’s Disease. Vaccines. 2021; 9(7):686. https://doi.org/10.3390/vaccines9070686
Chicago/Turabian StyleAgostini, Simone, Roberta Mancuso, Andrea S. Costa, Lorenzo A. Citterio, Franca R. Guerini, Mario Meloni, Jorge Navarro, and Mario Clerici. 2021. "A Possible Role for HSV-1-Specific Humoral Response and PILRA rs1859788 Polymorphism in the Pathogenesis of Parkinson’s Disease" Vaccines 9, no. 7: 686. https://doi.org/10.3390/vaccines9070686
APA StyleAgostini, S., Mancuso, R., Costa, A. S., Citterio, L. A., Guerini, F. R., Meloni, M., Navarro, J., & Clerici, M. (2021). A Possible Role for HSV-1-Specific Humoral Response and PILRA rs1859788 Polymorphism in the Pathogenesis of Parkinson’s Disease. Vaccines, 9(7), 686. https://doi.org/10.3390/vaccines9070686







