Pancreatic Injury after COVID-19 Vaccine—A Case Report
Abstract
1. Introduction
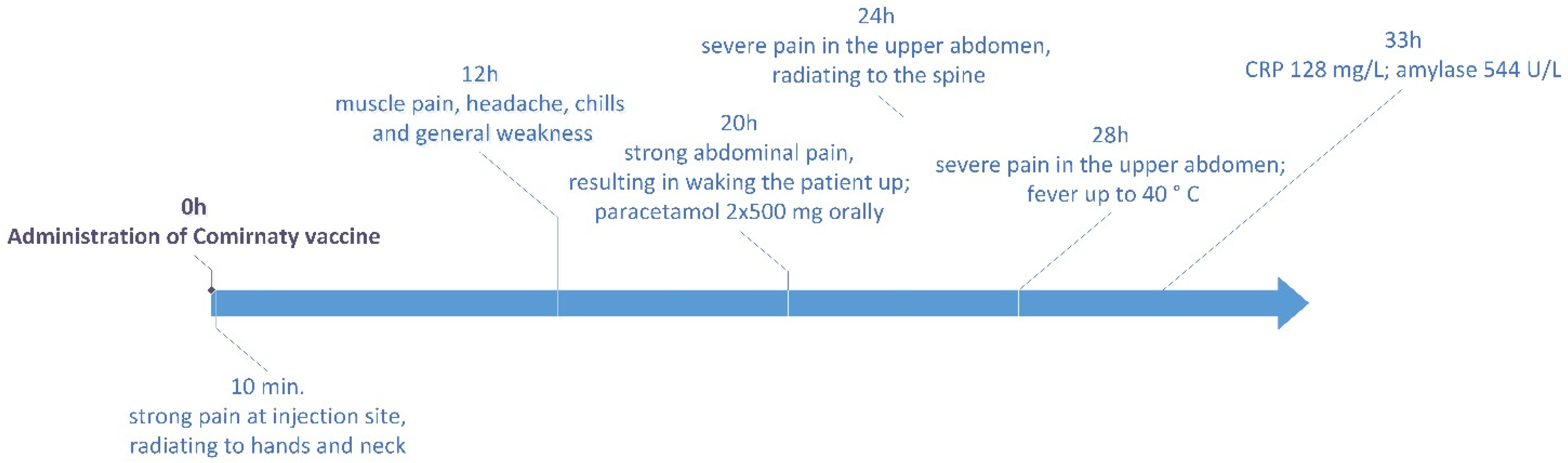
2. Case
3. Discussion
- the chronology of events associated with the onset of adverse reaction;
- positive rechallenge and exacerbation of symptoms after repeated exposure to the vaccine;
- similar case reports describing pancreatitis after using the same vaccine;
- a probable causal relationship of the vaccine to other kinds of autoimmune diseases;
- case reports describing pancreatitis observed after different vaccines;
- probable mechanism between the vaccine and acute pancreatitis.
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Worldometer COVID-19 Coronavirus Pandemic. Available online: https://www.worldometers.info/coronavirus/?ref=tjournal.ru (accessed on 26 April 2021).
- Comirnaty. Summary of Product Characteristics. Available online: https://www.ema.europa.eu/documents/product-information/comirnaty-epar-product-information_en.pdf (accessed on 25 March 2021).
- COVID-19 Vaccine Moderna. Summary of Product Characteristics. Available online: https://www.ema.europa.eu/documents/product-information/covid-19-vaccine-moderna-epar-product-information_en.pdf (accessed on 25 March 2021).
- COVID-19 Vaccine (ChAdOx1-S [Recombinant]). Summary of Product Characteristics. Available online: https://www.ema.europa.eu/documents/product-information/covid-19-vaccine-astrazeneca-epar-product-information_en.pdf (accessed on 25 March 2021).
- COVID-19 Vaccine (Ad26.COV2-S [Recombinant]). Summary of Product Characteristics. Available online: https://www.ema.europa.eu/documents/product-information/covid-19-vaccine-janssen-epar-product-information_en.pdf (accessed on 25 March 2021).
- Chandak, S.; Mandal, A.; Singh, A. Acute pancreatitis and pancreatic pseudocyst in a toddler following mumps, measles and rubella vaccine. Clin. Pediatr. 2017, 2, 1–4. Available online: https://www.longdom.org/open-access/acute-pancreatitis-and-pancreatic-pseudocyst-in-a-toddler-followingmumps-measles-and-rubella-vaccine.pdf (accessed on 25 March 2021). [CrossRef]
- Shlomovitz, E.; Davies, W.; Cairns, E.; Brintnell, W.C.; Goldszmidt, M.; Dresser, G.K. Severe necrotising pancreatitis following combined hepatitis A and B vaccination. CMAJ 2007, 176, 339–342. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Bizjak, M.; Bruck, O.; Praprotnik, S.; Dahan, S.; Shoenfeld, Y. Pancreatitis after human papillomavirus vaccination: A matter of molecular mimicry. Immunol. Res. 2017, 65, 164–167. [Google Scholar] [CrossRef] [PubMed]
- VRBPAC Briefing Document. Pfizer-BioNTech COVID-19 Vaccine (BNT162, PF-07302048) Vaccines and Related Biological Products Advisory Committee. Briefing Document. Meeting Date: 10 December 2020. US Food and Drug Administration 2020:14 December. [Ref. ID 103835]. Available online: https://www.fda.gov/media/144246/download (accessed on 8 April 2021).
- COVID-19 mRNA Pfizer-BioNTech Vaccine Analysis Print. All UK Spontaneous Reports Received between 9/12/20 and 21/03/21 for mRNA Pfizer/BioNTech Vaccine Analysis Print. Available online: https://assets.publishing.service.gov.uk/government/uploads/system/uploads/attachment_data/file/975808/COVID-19_mRNA_Pfizer-BioNTech_Vaccine_Analysis_Print.pdf (accessed on 8 April 2021).
- Bogdanos, D.P.; Smith, H.; Ma, Y.; Baum, H.; Mieli-Vergani, G.; Vergani, D. A study of molecular mimicry and immunological cross-reactivity between hepatitis B surface antigen and myelin mimics. Clin. Dev. Immunol. 2005, 12, 217–224. [Google Scholar] [CrossRef] [PubMed]
- Vojdani, A.; Kharrazian, D. Potential Antigenic Cross-Reactivity between SARS-CoV-2 and Human Tissue with a Possible Link to an Increase in Autoimmune Diseases. Clin. Immunol. 2020, 217, 108480. Available online: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7246018/ (accessed on 19 May 2021). [CrossRef] [PubMed]
- Vojdani, A.; Vojdani, E.; Kharrazian, D. Reaction of Human Monoclonal Antibodies to SARS-CoV-2 Proteins with Tissue Antigens: Implications for Autoimmune Diseases. Front. Immunol. 2021, 11, 617089. Available online: https://www.frontiersin.org/articles/10.3389/fimmu.2020.617089/full#B23 (accessed on 19 May 2021). [CrossRef] [PubMed]
- Kottanattu, L.; Lava, S.A.G.; Helbling, R.; Simonetti, G.D.; Bianchetti, M.G.; Milani, G.P. Pancreatitis and cholecystitis in primary acute symptomatic Epstein-Barr virus infection - Systematic review of the literature. J. Clin. Virol. 2016, 82, 51–55. [Google Scholar] [CrossRef] [PubMed]
- Rawla, P.; Bandaru, S.S.; Vellipuram, A.R. Review of Infectious Etiology of Acute Pancreatitis. Gastroenterol. Res. 2017, 10, 153–158. [Google Scholar] [CrossRef] [PubMed]
- Guan, W.J.; Ni, Z.Y.; Hu, Y.; Liang, W.H.; Ou, C.Q.; He, J.X.; Liu, L.; Shan, H.; Lei, C.L.; Hui, D.S.C.; et al. China Medical Treatment Expert Group for COVID-19. Clinical Characteristics of Coronavirus Disease 2019 in China. N. Engl. J. Med. 2020, 382, 1708–1720. [Google Scholar] [CrossRef] [PubMed]
- Perisetti, A.; Gajendran, M.; Boregowda, U.; Bansal, P.; Goyal, H. COVID-19 and gastrointestinal endoscopies: Current insights and emergent strategies. Dig. Endosc. 2020, 32, 715–722. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.; Wang, H.; Fan, J.; Zhang, Y.; Wang, H.; Zhao, Q. Pancreatic Injury Patterns in Patients with Coronavirus Disease 19 Pneumonia. Gastroenterology 2020, 159, 367–370. [Google Scholar] [CrossRef] [PubMed]
- Patel, K.P.; Patel, P.A.; Vunnam, R.R.; Hewlett, A.T.; Jain, R.; Jing, R.; Vunnam, S.R. Gastrointestinal, hepatobiliary, and pancreatic manifestations of COVID-19. J. Clin. Virol. 2020, 128, 104386. [Google Scholar] [CrossRef] [PubMed]
- de-Madaria, E.; Capurso, G. COVID-19 and acute pancreatitis: Examining the causality. Nat. Rev. Gastroenterol. Hepatol. 2021, 18, 3–4. [Google Scholar] [CrossRef] [PubMed]
| Parameter [Unit] (Reference Values) | Result |
|---|---|
| WBC [/L] (4.00–10.00) | 6.72 |
| %NEUT [%] (45.0–70.0) | 75.6 * |
| %LYMPH [%] (20.0–45.0) | 16.1 * |
| %MONO [%] (0.0–9.0) | 4.9 |
| %EOS [%] (0.0–5.0) | 1.3 |
| %BASO [%] (0.0–1.5) | 0.4 |
| %LUC [%] (0.0–4.0) | 1.7 |
| #NEUT [/L] (1.80–7.00) | 5.08 |
| #LYMPH [/L] (0.80–4.50) | 1.09 |
| #MONO [/L] (0.00–0.90) | 0.33 |
| #EOS [/L] (0.00–0.50) | 0.09 |
| #BASO [/L] (0.00–0.15) | 0.03 |
| #LUC [/L] (0.00–0.40) | 0.12 |
| RBC [/L] (4.00–5.00) | 4.29 |
| HGB [mmol/L] (7.45–10.00) | 8.50 |
| HCT [L/L] (0.36–0.47) | 0.39 |
| MCV [fL] (82–97) | 90 |
| MCH [fmol] (1.64–2.08) | 1.98 |
| MCHC [mmol/L] (20.00–22.00) | 22.01 * |
| RDW [%] (11.00–15.00) | 12.60 |
| PLT [/L] (130.0–390.0] | 161 |
| MPV [fL] (7.00–11.00] | 9.40 |
| PCT [L/L] (0.002–0.004) | 0.002 |
| PDW [%] (40.0–60.0) | 67.5 * |
| Parameter [Unit] (Reference Values) | 9 January 2021 | 11 January 2021 | 19 January 2021 |
|---|---|---|---|
| CRP [mg/L] (<5.0) | 128.0 * | 38.0 * | 2.2 |
| Urine amylase [U/L] (30–200) | 544 * | 208 | 240 |
| Serum amylase [U/L] (25–115) | 51 | 58 | 64 |
| ALAT [U/L] (<45) | <7 | 8 | 8 |
| ASPAT [U/L] (<35) | 15 | 14 | 13 |
| Serum creatinine [mg/dL] (0.50–0.90) | 0.68 | - | 0.83 |
| Potassium [mmol/L] (3.5–5.1) | 3.8 | - | 4.85 |
| Sodium [mmol/L] (135.0–145.0) | 139.0 | - | 141.0 |
| Fasting glucose [mg/dL] (70–99) | - | 84 | 89 |
| Total bilirubin [mg/dL] (<1.20) | - | 2.21 * | 1.24 * |
| Direct bilirubin [mg/dL] (<0.3) | - | - | 0.40 * |
| Alkaline phosphatase [U/L] (35–105) | - | 61 | 67 |
| GGT [U/L] (5–36) | - | 12 | 14 |
| Total cholesterol [mg/dL] (<190) | - | 174 | - |
| HDL [mg/dL] (>45.0) | - | 78 | - |
| %HDL [%] (>20.0) | - | 45 | - |
| Triglycerides [mg/dL] (65–150) | - | 45 | - |
| Iron [µg/dL] (37–145) | - | 80 | - |
| UIBC [µg/dL] (112–346) | - | 224 | - |
| TIBC (calculated) [µg/dL] (250–400) | - | 304 | - |
| TSAT (calculated) [%] (20–55) | - | 26 | - |
| CEA [ng/mL] (<5.0/<6.5 smoking/non-smoking) | - | 0.88 | - |
| CA [U/mL] (<39) | - | 4.4 | - |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cieślewicz, A.; Dudek, M.; Krela-Kaźmierczak, I.; Jabłecka, A.; Lesiak, M.; Korzeniowska, K. Pancreatic Injury after COVID-19 Vaccine—A Case Report. Vaccines 2021, 9, 576. https://doi.org/10.3390/vaccines9060576
Cieślewicz A, Dudek M, Krela-Kaźmierczak I, Jabłecka A, Lesiak M, Korzeniowska K. Pancreatic Injury after COVID-19 Vaccine—A Case Report. Vaccines. 2021; 9(6):576. https://doi.org/10.3390/vaccines9060576
Chicago/Turabian StyleCieślewicz, Artur, Magdalena Dudek, Iwona Krela-Kaźmierczak, Anna Jabłecka, Maciej Lesiak, and Katarzyna Korzeniowska. 2021. "Pancreatic Injury after COVID-19 Vaccine—A Case Report" Vaccines 9, no. 6: 576. https://doi.org/10.3390/vaccines9060576
APA StyleCieślewicz, A., Dudek, M., Krela-Kaźmierczak, I., Jabłecka, A., Lesiak, M., & Korzeniowska, K. (2021). Pancreatic Injury after COVID-19 Vaccine—A Case Report. Vaccines, 9(6), 576. https://doi.org/10.3390/vaccines9060576






