The Perception and Attitudes toward COVID-19 Vaccines: A Cross-Sectional Study in Poland
Abstract
1. Introduction
2. Materials and Methods
2.1. Survey
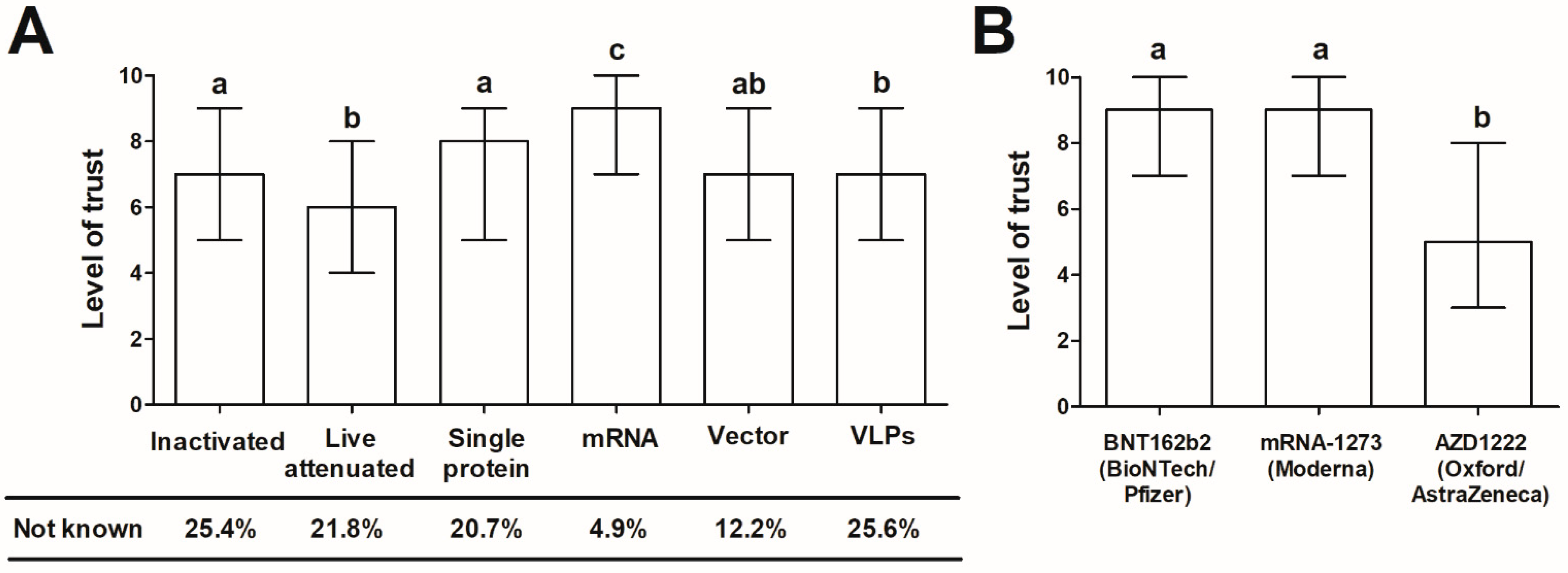
- The general level of trust in vaccines (using a 10-point Likert-type scale, where 1-no trust, 10-very high level of trust) in relation to their type: attenuated, inactivated, mRNA, vector-based, protein-based, and virus-like particles (VLPs). The mechanism of action of each vaccine was explained to the surveyed individuals using non-specialist language;
- The level of acceptance of the COVID-19 vaccines already approved and in use in Poland during the time of the study: BNT162b (BioNTech/Pfizer, Mainz/ Sandton, P O Box, Germany/USA), mRNA-1273 (Moderna, Cambridge, MA, USA) and AZD1222 (Oxford/AstraZeneca, Oxford, UK);
- The level of trust in other COVID-19 vaccines authorized outside the European Union or in advanced phases of clinical trials (using a 10-point Likert-type scale, where 1-no trust, 10-very high level of trust). These included vector vaccines Sputnik V (Gamaleya Research Institute, Moscow, Russia), Ad26.COV2.S (Janssen/Johnson&Johnson, Beerse/ New Brunswick, NJ, Belgium/USA), Ad5-nCoV (CanSino Biologics, Tianjin, China), mRNA vaccine CVnCoV (CureVac, Tübingen, Germany), protein vaccine NVX-CoV2373 (Novavax, Gaithersburg, MD, USA), inactivated vaccines CoronaVac (Sinovac Biotech, Beijing, China), BBIBP-CorV (Sinopharm, Beijing, China) and Covaxin (Bharat Biotech, Telagana, India);
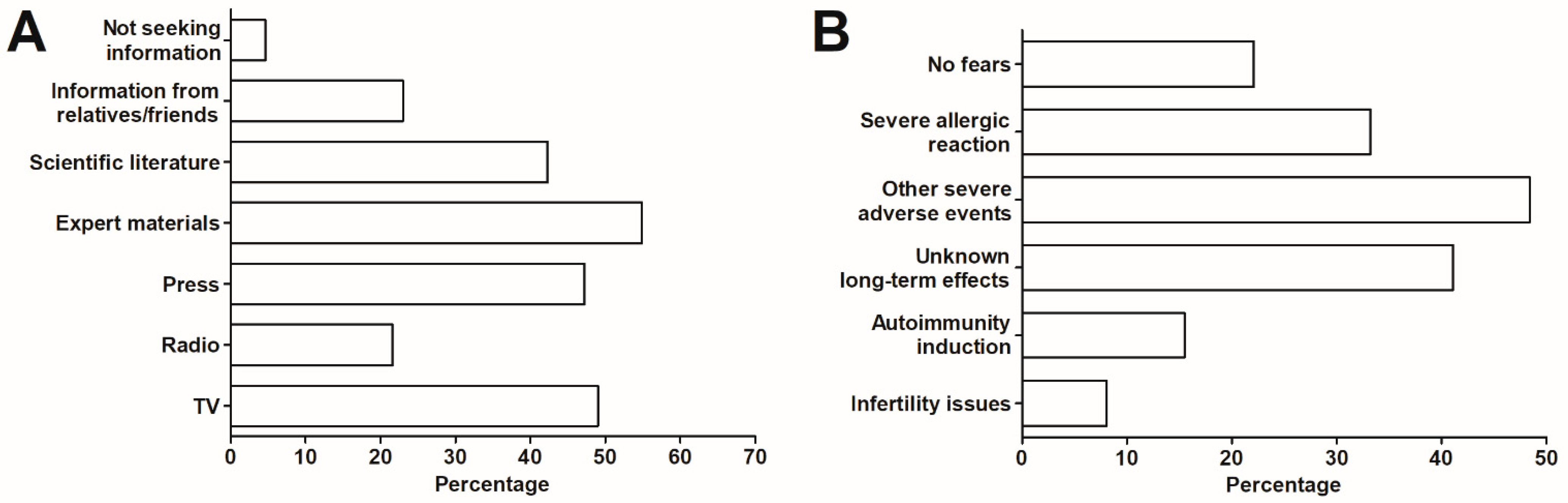
- The level of fear prior to the vaccination against COVID-19 (using a 10-point Likert-type scale, where 1-no fear, 10-very high level of fear) and primary reasons behind this fear;
- The primary reasons behind the willingness to vaccinate against COVID-19;
- The primary sources of information on the COVID-19 vaccines in the surveyed group.
2.2. Statistical Analysis
3. Results and Discussion
3.1. Demographic Characteristics
3.2. The General Trust in Vaccines in Relation to Their Type
3.3. The Trust in COVID-19 Vaccines Authorized for Use in Poland
3.4. The Trust in COVID-19 Vaccines Unauthorized in the European Union
3.5. Sources of Information on the COVID-19 Vaccines
3.6. The Fears Related to COVID-19 Vaccination and Primary Reasons behind the Willingness to Receive a Vaccine
3.7. Limitations
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Lenzen, M.; Li, M.; Malik, A.; Pomponi, F.; Sun, Y.-Y.; Wiedmann, T.; Faturay, F.; Fry, J.; Gallego, B.; Geschke, A.; et al. Global socio-economic losses and environmental gains from the Coronavirus pandemic. PLoS ONE 2020, 15, e0235654. [Google Scholar] [CrossRef] [PubMed]
- Betancourt, J.A.; Rosenberg, M.A.; Zevallos, A.; Brown, J.R.; Mileski, M. The impact of COVID-19 on telemedicine utilization across multiple service lines in the United States. Healthcare 2020, 8, 380. [Google Scholar] [CrossRef] [PubMed]
- Rapanta, C.; Botturi, L.; Goodyear, P.; Guàrdia, L.; Koole, M. Online University Teaching During and After the Covid-19 Crisis: Refocusing Teacher Presence and Learning Activity. Postdigit. Sci. Educ. 2020, 2, 923–945. [Google Scholar] [CrossRef]
- Sidor, A.; Rzymski, P. Dietary Choices and Habits during COVID-19 Lockdown: Experience from Poland. Nutrients 2020, 12, 1657. [Google Scholar] [CrossRef] [PubMed]
- Prati, G.; Mancini, A.D. The psychological impact of COVID-19 pandemic lockdowns: A review and meta-analysis of longitudinal studies and natural experiments. Psychol. Med. 2021, 51, 201–211. [Google Scholar] [CrossRef]
- Nowakowska, J.; Sobocińska, J.; Lewicki, M.; Lemańska, Ż.; Rzymski, P. When science goes viral: The research response during three months of the COVID-19 outbreak. Biomed. Pharmacother. 2020, 129, 110451. [Google Scholar] [CrossRef] [PubMed]
- Rzymski, P.; Nowicki, M.; Mullin, G.E.; Abraham, A.; Rodríguez-Román, E.; Petzold, M.B.; Bendau, A.; Sahu, K.K.; Ather, A.; Naviaux, A.-F.; et al. Quantity does not equal quality: Scientific principles cannot be sacrificed. Int. Immunopharmacol. 2020, 86, 106711. [Google Scholar] [CrossRef] [PubMed]
- Gianola, S.; Jesus, T.S.; Bargeri, S.; Castellini, G. Characteristics of academic publications, preprints, and registered clinical trials on the COVID-19 pandemic. PLoS ONE 2020, 15, e0240123. [Google Scholar] [CrossRef] [PubMed]
- Defendi, H.G.T.; da Silva Madeira, L.; Borschiver, S. Analysis of the COVID-19 Vaccine Development Process: An Exploratory Study of Accelerating Factors and Innovative Environments. J. Pharm. Innov. 2021. [Google Scholar] [CrossRef]
- Burgos, R.M.; Badowski, M.E.; Drwiega, E.; Ghassemi, S.; Griffith, N.; Herald, F.; Johnson, M.; Smith, R.O.; Michienzi, S.M. The race to a COVID-19 vaccine: Opportunities and challenges in development and distribution. Drugs Context 2021, 10. [Google Scholar] [CrossRef]
- Polack, F.P.; Thomas, S.J.; Kitchin, N.; Absalon, J.; Gurtman, A.; Lockhart, S.; Perez, J.L.; Pérez Marc, G.; Moreira, E.D.; Zerbini, C.; et al. Safety and Efficacy of the BNT162b2 mRNA Covid-19 Vaccine. N. Engl. J. Med. 2020, 383, 2603–2615. [Google Scholar] [CrossRef]
- Baden, L.R.; El Sahly, H.M.; Essink, B.; Kotloff, K.; Frey, S.; Novak, R.; Diemert, D.; Spector, S.A.; Rouphael, N.; Creech, C.B.; et al. Efficacy and Safety of the mRNA-1273 SARS-CoV-2 Vaccine. N. Engl. J. Med. 2020, 384, 403–416. [Google Scholar] [CrossRef]
- Voysey, M.; Clemens, S.A.C.; Madhi, S.A.; Weckx, L.Y.; Folegatti, P.M.; Aley, P.K.; Angus, B.; Baillie, V.L.; Barnabas, S.L.; Bhorat, Q.E.; et al. Safety and efficacy of the ChAdOx1 nCoV-19 vaccine (AZD1222) against SARS-CoV-2: An interim analysis of four randomised controlled trials in Brazil, South Africa, and the UK. Lancet 2021, 397, 99–111. [Google Scholar] [CrossRef]
- Vasileiou, E.; Simpson, C.R.; Robertson, C.; Shi, T.; Kerr, S.; Agrawal, U.; Akbari, A.; Bedston, S.; Beggs, J.; Bradley, D. Effectiveness of First Dose of COVID-19 Vaccines against Hospital Admissions in Scotland: National Prospective Cohort Study of 5.4 Million People. SSRN 2021. [Google Scholar] [CrossRef]
- Coustasse, A.; Kimble, C.; Maxik, K. COVID-19 and Vaccine Hesitancy: A Challenge the United States Must Overcome. J. Ambul. Care Manag. 2021, 44, 71–75. [Google Scholar] [CrossRef]
- Dubé, E.; Laberge, C.; Guay, M.; Bramadat, P.; Roy, R.; Bettinger, J. Vaccine hesitancy: An overview. Hum. Vaccines Immunother. 2013, 9, 1763–1773. [Google Scholar] [CrossRef] [PubMed]
- Baldo, V.; Reno, C.; Cocchio, S.; Fantini, M.P. SARS-CoV-2/COVID-19 Vaccines: The Promises and the Challenges Ahead. Vaccines 2021, 9, 21. [Google Scholar] [CrossRef] [PubMed]
- Loomba, S.; de Figueiredo, A.; Piatek, S.J.; de Graaf, K.; Larson, H.J. Measuring the impact of COVID-19 vaccine misinformation on vaccination intent in the UK and USA. Nat. Hum. Behav. 2021. [Google Scholar] [CrossRef]
- Schiavo, R. Vaccine communication in the age of COVID-19: Getting ready for an information war. J. Commun. Healthc. 2020, 13, 73–75. [Google Scholar] [CrossRef]
- Johnson, N.F.; Velásquez, N.; Restrepo, N.J.; Leahy, R.; Gabriel, N.; El Oud, S.; Zheng, M.; Manrique, P.; Wuchty, S.; Lupu, Y. The online competition between pro- and anti-vaccination views. Nature 2020, 582, 230–233. [Google Scholar] [CrossRef]
- Saied, S.M.; Saied, E.M.; Kabbash, I.A.; Abdo, S.A.E.-F. Vaccine hesitancy: Beliefs and barriers associated with COVID-19 vaccination among Egyptian medical students. J. Med. Virol 2021. [Google Scholar] [CrossRef] [PubMed]
- Gagneux-Brunon, A.; Detoc, M.; Bruel, S.; Tardy, B.; Rozaire, O.; Frappe, P.; Botelho-Nevers, E. Intention to get vaccinations against COVID-19 in French healthcare workers during the first pandemic wave: A cross sectional survey. J. Hosp. Infect. 2020. [Google Scholar] [CrossRef]
- Gadoth, A.; Halbrook, M.; Martin-Blais, R.; Gray, A.; Tobin, N.H.; Ferbas, K.G.; Aldrovandi, G.M.; Rimoin, A.W. Assessment of COVID-19 vaccine acceptance among healthcare workers in Los Angeles. medRxiv 2020. [Google Scholar] [CrossRef]
- Nzaji, M.K.; Ngombe, L.K.; Mwamba, G.N.; Ndala, D.B.B.; Miema, J.M.; Lungoyo, C.L.; Mwimba, B.L.; Bene, A.C.M.; Musenga, E.M. Acceptability of Vaccination Against COVID-19 among Healthcare Workers in the Democratic Republic of the Congo. Pragmat Obs. Res. 2020, 11, 103–109. [Google Scholar] [CrossRef]
- Boytchev, H. Why did a German newspaper insist the Oxford AstraZeneca vaccine was inefficacious for older people—Without evidence? BMJ 2021, 372, n414. [Google Scholar] [CrossRef]
- Rzymski, P.; Borkowski, L.; Drąg, M.; Flisiak, R.; Jemielity, J.; Krajewski, J.; Mastalerz-Migas, A.; Matyja, A.; Pyrć, K.; Simon, K.; et al. The Strategies to Support the COVID-19 Vaccination with Evidence-Based Communication and Tackling Misinformation. Vaccines 2021, 9, 109. [Google Scholar] [CrossRef]
- Quinn, S.C.; Jamison, A.M.; Freimuth, V. Communicating Effectively About Emergency Use Authorization and Vaccines in the COVID-19 Pandemic. Am. J. Public Health 2021, 111, 355–358. [Google Scholar] [CrossRef] [PubMed]
- Bazan, D.; Nowicki, M.; Rzymski, P. Medical students as the volunteer workforce during the COVID-19 pandemic: Polish experience. Int. J. Disaster Risk Reduct. 2021, 55, 102109. [Google Scholar] [CrossRef] [PubMed]
- Geldsetzer, P. Use of Rapid Online Surveys to Assess People’s Perceptions During Infectious Disease Outbreaks: A Cross-sectional Survey on COVID-19. J. Med. Internet Res. 2020, 22, e18790. [Google Scholar] [CrossRef] [PubMed]
- Albano, L.; Matuozzo, A.; Marinelli, P.; Di Giuseppe, G. Knowledge, attitudes and behaviour of hospital health-care workers regarding influenza A/H1N1: A cross sectional survey. BMC Infect. Dis. 2014, 14, 208. [Google Scholar] [CrossRef]
- Costantino, C.; Amodio, E.; Vitale, F.; Trucchi, C.; Maida, C.M.; Bono, S.E.; Caracci, F.; Sannasardo, C.E.; Scarpitta, F.; Vella, C.; et al. Human Papilloma Virus Infection and Vaccination: Pre-Post Intervention Analysis on Knowledge, Attitudes and Willingness to Vaccinate Among Preadolescents Attending Secondary Schools of Palermo, Sicily. Int. J. Environ. Res. Public Health 2020, 17, 5362. [Google Scholar] [CrossRef]
- Dubé, E.; Gagnon, D.; Ouakki, M.; Bettinger, J.A.; Guay, M.; Halperin, S.; Wilson, K.; Graham, J.; Witteman, H.O.; MacDonald, S.; et al. Understanding Vaccine Hesitancy in Canada: Results of a Consultation Study by the Canadian Immunization Research Network. PLoS ONE 2016, 11, e0156118. [Google Scholar] [CrossRef] [PubMed]
- Di Giuseppe, G.; Abbate, R.; Liguori, G.; Albano, L.; Angelillo, I.F. Human papillomavirus and vaccination: Knowledge, attitudes, and behavioural intention in adolescents and young women in Italy. Br. J. Cancer 2008, 99, 225–229. [Google Scholar] [CrossRef]
- Napolitano, F.; Napolitano, P.; Angelillo, I.F. Seasonal influenza vaccination in pregnant women: Knowledge, attitudes, and behaviors in Italy. BMC Infect. Dis. 2017, 17, 48. [Google Scholar] [CrossRef]
- Alholm, Z.; Ault, K.; Zwick, R.; Fitzgerald, S.; Satterwhite, C. Pregnant Women’s Acceptance of Hypothetical Zika Vaccine. Open Forum Infect. Dis. 2017, 4, S458. [Google Scholar] [CrossRef]
- CBOS. Research Reports. Available online: https://www.cbos.pl/EN/publications/reports.php (accessed on 13 March 2021).
- Cochran, W.G. Sampling Techniques, 3rd ed.; Wiley: Hoboken, NJ, USA, 1977. [Google Scholar]
- Le, T.T.; Cramer, J.P.; Chen, R.; Mayhew, S. Evolution of the COVID-19 vaccine development landscape. Nat. Rev. Drug Discov. 2020, 19, 667–668. [Google Scholar] [CrossRef] [PubMed]
- Forni, G.; Mantovani, A.; Forni, G.; Mantovani, A.; Moretta, L.; Rappuoli, R.; Rezza, G.; Bagnasco, A.; Barsacchi, G.; Bussolati, G.; et al. COVID-19 vaccines: Where we stand and challenges ahead. Cell Death Differ. 2021, 28, 626–639. [Google Scholar] [CrossRef] [PubMed]
- Plotkin, S. History of vaccination. Proc. Natl. Acad. Sci. USA 2014, 111, 12283–12287. [Google Scholar] [CrossRef]
- Minor, P.D. Live attenuated vaccines: Historical successes and current challenges. Virology 2015, 479–480, 379–392. [Google Scholar] [CrossRef]
- Zarobkiewicz, M.K.; Zimecka, A.; Zuzak, T.; Cieślak, D.; Roliński, J.; Grywalska, E. Vaccination among Polish university students. Knowledge, beliefs and anti-vaccination attitudes. Hum. Vaccines Immunother. 2017, 13, 2654–2658. [Google Scholar] [CrossRef]
- Kraśnicka, J.; Krajewska-Kułak, E.; Klimaszewska, K.; Cybulski, M.; Guzowski, A.; Kowalewska, B.; Jankowiak, B.; Rolka, H.; Doroszkiewicz, H.; Kułak, W. Mandatory and recommended vaccinations in Poland in the views of parents. Hum. Vaccines Immunother. 2018, 14, 2884–2893. [Google Scholar] [CrossRef]
- Nitsch-Osuch, A.; Gołębiak, I.; Wyszkowska, D.; Rosińska, R.; Kargul, L.; Szuba, B.; Tyszko, P.; Brydak, L.B. Influenza Vaccination Coverage Among Polish Patients with Chronic Diseases. Adv. Exp. Med. Biol. 2017, 968, 19–34. [Google Scholar] [CrossRef]
- Kar-Gupta, S. EU Must Be United over Russian, Chinese COVID-19 Vaccines: French Minister. Available online: https://www.reuters.com/article/us-health-coronavirus-eu-france-idUSKBN2AX0NF (accessed on 14 March 2021).
- Wise, J. Covid-19: European countries suspend use of Oxford-AstraZeneca vaccine after reports of blood clots. BMJ 2021, 372, n699. [Google Scholar] [CrossRef]
- Ring, S.; Fourcade, M. Europe Restarts AstraZeneca Vaccines after Safety Endorsement. Available online: https://www.bloomberg.com/news/articles/2021-03-18/italy-france-to-restart-astra-vaccines-after-safety-endorsement (accessed on 5 April 2021).
- CBOS. Attitudes to Vaccination against Covid-19. Available online: https://www.cbos.pl/EN/publications/reports/2020/154_20.pdf (accessed on 13 March 2021).
- Persad, G.; Peek, M.E.; Emanuel, E.J. Fairly Prioritizing Groups for Access to COVID-19 Vaccines. JAMA 2020, 324, 1601–1602. [Google Scholar] [CrossRef]
- Ribas, A.; Sengupta, R.; Locke, T.; Zaidi, S.K.; Campbell, K.M.; Carethers, J.M.; Jaffee, E.M.; Wherry, E.J.; Soria, J.-C.; D’Souza, G. Priority COVID-19 Vaccination for Patients with Cancer while Vaccine Supply Is Limited. Cancer Discov. 2021, 11, 233–236. [Google Scholar] [CrossRef]
- Logunov, D.Y.; Dolzhikova, I.V.; Zubkova, O.V.; Tukhvatulin, A.I.; Shcheblyakov, D.V.; Dzharullaeva, A.S.; Grousova, D.M.; Erokhova, A.S.; Kovyrshina, A.V.; Botikov, A.G.; et al. Safety and immunogenicity of an rAd26 and rAd5 vector-based heterologous prime-boost COVID-19 vaccine in two formulations: Two open, non-randomised phase 1/2 studies from Russia. Lancet 2020, 396, 887–897. [Google Scholar] [CrossRef]
- Logunov, D.Y.; Dolzhikova, I.V.; Shcheblyakov, D.V.; Tukhvatulin, A.I.; Zubkova, O.V.; Dzharullaeva, A.S.; Kovyrshina, A.V.; Lubenets, N.L.; Grousova, D.M.; Erokhova, A.S.; et al. Safety and efficacy of an rAd26 and rAd5 vector-based heterologous prime-boost COVID-19 vaccine: An interim analysis of a randomised controlled phase 3 trial in Russia. Lancet 2021, 397, 671–681. [Google Scholar] [CrossRef]
- Bucci, E.; Andreev, K.; Björkman, A.; Calogero, R.A.; Carafoli, E.; Carninci, P.; Castagnoli, P.; Cossarizza, A.; Mussini, C.; Guerin, P.; et al. Safety and efficacy of the Russian COVID-19 vaccine: More information needed. Lancet 2020, 396, e53. [Google Scholar] [CrossRef]
- Burki, T.K. The Russian vaccine for COVID-19. Lancet Respir. Med. 2020, 8, e85–e86. [Google Scholar] [CrossRef]
- Balakrishnan, V.S. The arrival of Sputnik V. Lancet Infect. Dis. 2020, 20, 1128. [Google Scholar] [CrossRef]
- Thorp, H.H. A dangerous rush for vaccines. Science 2020, 369, 885. [Google Scholar] [CrossRef]
- Pew Research Center. Russia’s Global Image Negative amid Crisis in Ukraine. Available online: https://www.pewresearch.org/global/2014/07/09/russias-global-image-negative-amid-crisis-in-ukraine/ (accessed on 14 March 2021).
- European Medicines Agency. EMA Starts Rolling Review of the Sputnik V COVID-19 Vaccine. Available online: https://www.ema.europa.eu/en/news/ema-starts-rolling-review-sputnik-v-covid-19-vaccine (accessed on 14 March 2021).
- McLean, C.P.; Anderson, E.R. Brave men and timid women? A review of the gender differences in fear and anxiety. Clin. Psychol. Rev. 2009, 29, 496–505. [Google Scholar] [CrossRef]
- Napolitano, C.L.; Ogunseitan, O.A. Gender Differences in the Perception of Genetic Engineering Applied to Human Reproduction. Soc. Indic. Res. 1999, 46, 191–204. [Google Scholar] [CrossRef]
- Simon, R.M. Gender differences in knowledge and attitude towards biotechnology. Public Underst. Sci. 2010, 19, 642–653. [Google Scholar] [CrossRef]
- Dolgin, E. How COVID unlocked the power of RNA vaccines. Nature 2021, 589, 189–191. [Google Scholar] [CrossRef]
- Male, V. Are COVID-19 vaccines safe in pregnancy? Nat. Rev. Immunol. 2021. [Google Scholar] [CrossRef]
- Ferdinand, K.C.; Nedunchezhian, S.; Reddy, T.K. The COVID-19 and Influenza “Twindemic”: Barriers to Influenza Vaccination and Potential Acceptance of SARS-CoV2 Vaccination in African Americans. J. Natl. Med. Assoc. 2020, 112, 681–687. [Google Scholar] [CrossRef]
- Brownlie, J.; Howson, A. ‘Between the demands of truth and government’: Health practitioners, trust and immunisation work. Soc. Sci. Med. 2006, 62, 433–443. [Google Scholar] [CrossRef]
- Larson, H.J.; Cooper, L.Z.; Eskola, J.; Katz, S.L.; Ratzan, S. Addressing the vaccine confidence gap. Lancet 2011, 378, 526–535. [Google Scholar] [CrossRef]
| Age | Mean ± SD (Min–Max) | 45 ± 15 (17–85) |
|---|---|---|
| <50 | %/n | 64.2 (655) |
| ≥50 | 35.8 (365) | |
| Gender | %/n | |
| Female | 61.6 (628) | |
| Male | 38.4 (392) | |
| Place of living | %/n | |
| Rural | 14.9 (152) | |
| Urban < 50,000 | 16.3 (166) | |
| Urban 50,000–150,000 | 7.5 (77) | |
| Urban 150,000–500,000 | 16.3 (166) | |
| Urban > 500,000 | 45.0 (459) | |
| Education | %/n | |
| Primary | 0.2 (2) | |
| Secondary | 22.0 (222) | |
| Vocational | 3.8 (38) | |
| Tertiary | 74.0 (748) | |
| History of SARS-CoV-2 infection | %/n | 14.9 (152) |
| Fatal case of COVID-19 in the family | %/n | 12.4 (126) |
| Vaccine Type | Age | χ2 p-Value | Gender | χ2 p-Value | Education | χ2 p-Value | Place of Living | χ2 p-Value | ||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| <50 | ≥50 | Male | Female | Tertiary | Other | Urban | Rural | |||||
| Inactivated | 81.4 | 62.5 | <0.001 | 73.2 | 76.8 | ns | 77.7 | 66.2 | 0.002 | 75.2 | 71.1 | ns |
| Attenuated | 84.3 | 67.4 | <0.001 | 78.5 | 77.8 | ns | 81.8 | 68.4 | <0.001 | 78.0 | 79.6 | ns |
| Single protein | 84.6 | 69.9 | <0.001 | 78.5 | 80.6 | ns | 81.7 | 72.8 | <0.001 | 79.1 | 80.3 | ns |
| mRNA | 95.6 | 94.3 | ns | 94.0 | 96.9 | ns | 96.0 | 92.7 | 0.011 | 94.9 | 96.1 | ns |
| Vector | 88.6 | 86.6 | ns | 85.8 | 91.0 | ns | 90.1 | 81.6 | <0.001 | 87.9 | 87.5 | ns |
| VLP | 77.1 | 69.6 | 0.034 | 74.8 | 73.2 | ns | 78.7 | 62.5 | <0.001 | 75.6 | 67.8 | ns |
| Vaccine Type | Age | χ2 p-Value | Gender | χ2 p-Value | Education | χ2 p-Value | Place of Living | χ2 p-Value | ||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| <50 | ≥50 | Male | Female | Tertiary | Other | Urban | Rural | |||||
| Inactivated | 76.9 | 41.2 | <0.001 | 69.7 | 63.9 | ns | 66.9 | 63.8 | ns | 66.5 | 64.8 | ns |
| Attenuated | 64.1 | 30.5 | <0.001 | 56.4 | 52.1 | ns | 55.1 | 49.5 | ns | 53.5 | 55.4 | ns |
| Single protein | 78.7 | 45.9 | <0.001 | 71.8 | 66.1 | ns | 70.2 | 62.6 | ns | 69.0 | 64.7 | ns |
| mRNA | 85.1 | 81.4 | ns | 85.5 | 82.7 | ns | 84.5 | 81.7 | ns | 82.9 | 89.0 | ns |
| Vector | 65.6 | 52.5 | <0.001 | 60.7 | 61.6 | ns | 64.2 | 51.3 | 0.002 | 61.5 | 58.6 | ns |
| VLP | 71.5 | 51.6 | <0.001 | 65.4 | 64.5 | ns | 67.9 | 54.1 | 0.003 | 64.6 | 66.0 | ns |
| Vaccine | Type | Manufacturer | Not Known [%] | Level of Trust |
|---|---|---|---|---|
| Sputnik V | Vector | Gamaleya Research Institute, Russia | 17.0 | 2.0 (1.0–5.0) a |
| Ad26.COV2.S | Vector | Janssen/ Johnson&Johnson, The Netherlands/USA | 13.9 | 5.0 (3.0–7.0) b |
| CVnCoV | mRNA | CureVac, Germany | 42.7 | 7.0 (4.0–8.0) c |
| NVX-CoV2373 | Single protein | Novavax, USA | 49.1 | 5.0 (3.0–7.0) b |
| CoronaVac | Inactivated | Sinovac Biotech, China | 55.4 | 4.0 (2.0–5.0) d |
| BBIBP-CorV | Inactivated | Sinopharm, China | 57.0 | 5.0 (2.0–5.0) d |
| Covaxin | Inactivated | Bharat Biotech, India | 63.2 | 5.0 (2.0–6.0) d |
| Ad5-nCoV | Vector | Cansino Biologics, China | 63.3 | 4.0 (2.0–5.0) d |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rzymski, P.; Zeyland, J.; Poniedziałek, B.; Małecka, I.; Wysocki, J. The Perception and Attitudes toward COVID-19 Vaccines: A Cross-Sectional Study in Poland. Vaccines 2021, 9, 382. https://doi.org/10.3390/vaccines9040382
Rzymski P, Zeyland J, Poniedziałek B, Małecka I, Wysocki J. The Perception and Attitudes toward COVID-19 Vaccines: A Cross-Sectional Study in Poland. Vaccines. 2021; 9(4):382. https://doi.org/10.3390/vaccines9040382
Chicago/Turabian StyleRzymski, Piotr, Joanna Zeyland, Barbara Poniedziałek, Ilona Małecka, and Jacek Wysocki. 2021. "The Perception and Attitudes toward COVID-19 Vaccines: A Cross-Sectional Study in Poland" Vaccines 9, no. 4: 382. https://doi.org/10.3390/vaccines9040382
APA StyleRzymski, P., Zeyland, J., Poniedziałek, B., Małecka, I., & Wysocki, J. (2021). The Perception and Attitudes toward COVID-19 Vaccines: A Cross-Sectional Study in Poland. Vaccines, 9(4), 382. https://doi.org/10.3390/vaccines9040382









