The Impact of Rubella Vaccine Introduction on Rubella Infection and Congenital Rubella Syndrome: A Systematic Review of Mathematical Modelling Studies
Abstract
1. Introduction
2. Materials and Methods
2.1. Inclusion Criteria
- Participants: Individuals eligible for rubella vaccination of any age in any country
- Intervention: Rubella-containing vaccine introduction scenarios
- Comparison: No rubella vaccine or different vaccine introduction scenarios
- Outcomes: We included studies that reported at least one of the following outcomes of RCVs at a population level: time (in years) to the elimination of CRS, time (in years) to rubella elimination, description of trends in rubella and CRS incidence, number of vaccinated individuals per CRS case averted, and cost-effectiveness of vaccine introduction strategy.
- Time horizon: We included studies in which the time horizon from the year of vaccine introduction to the end of the simulation is at least five years. We assumed it is unlikely that any meaningful impact of rubella vaccine introduction will be measurable within a shorter period.
2.2. Exclusion Criteria
2.3. Search Strategy
2.4. Study Selection and Data Extraction
2.5. Risk of Bias Assessment
2.6. Data Analysis
2.7. Ethical Considerations
3. Results
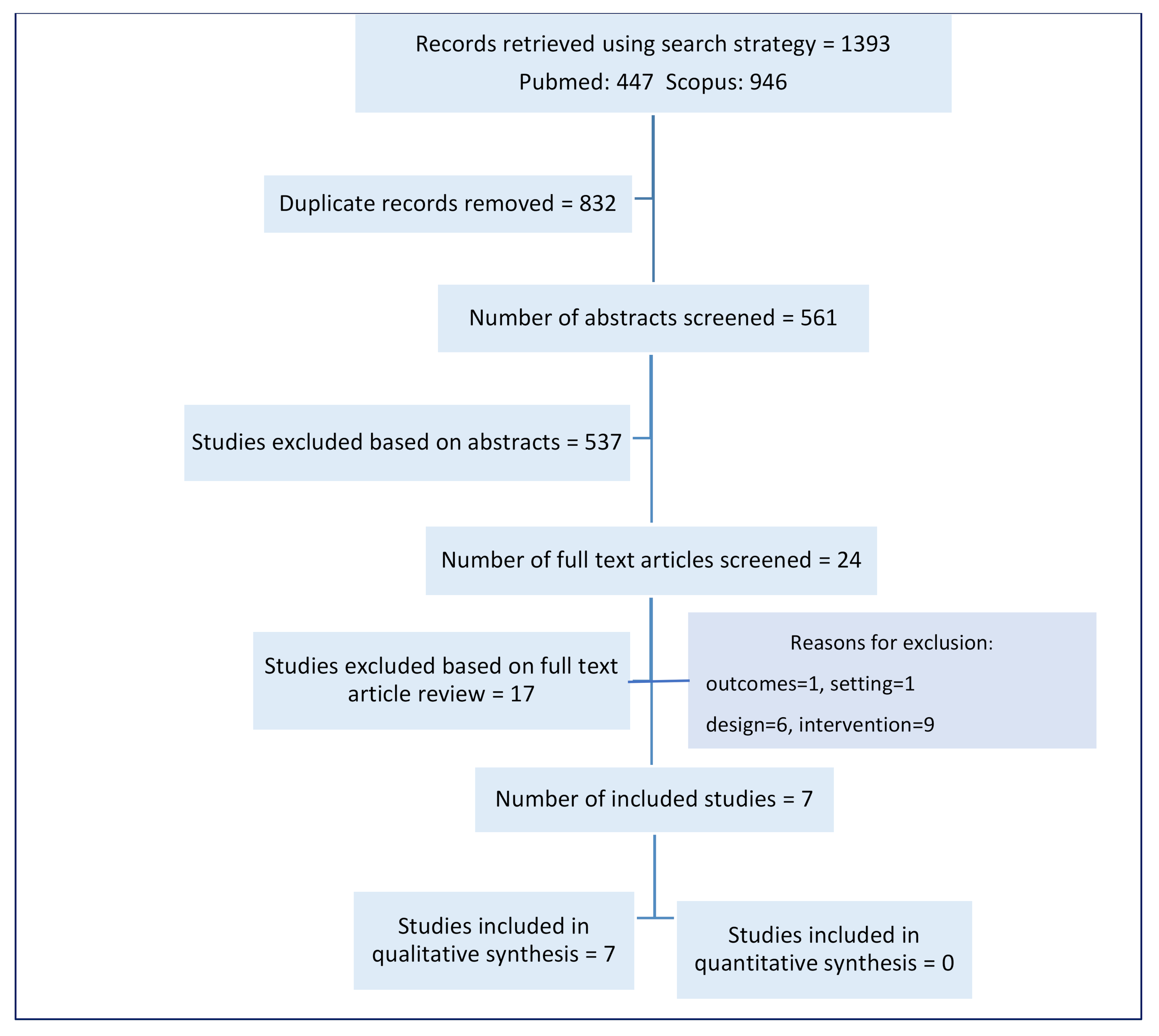
3.1. Included Studies
3.1.1. Characteristics of Included Studies
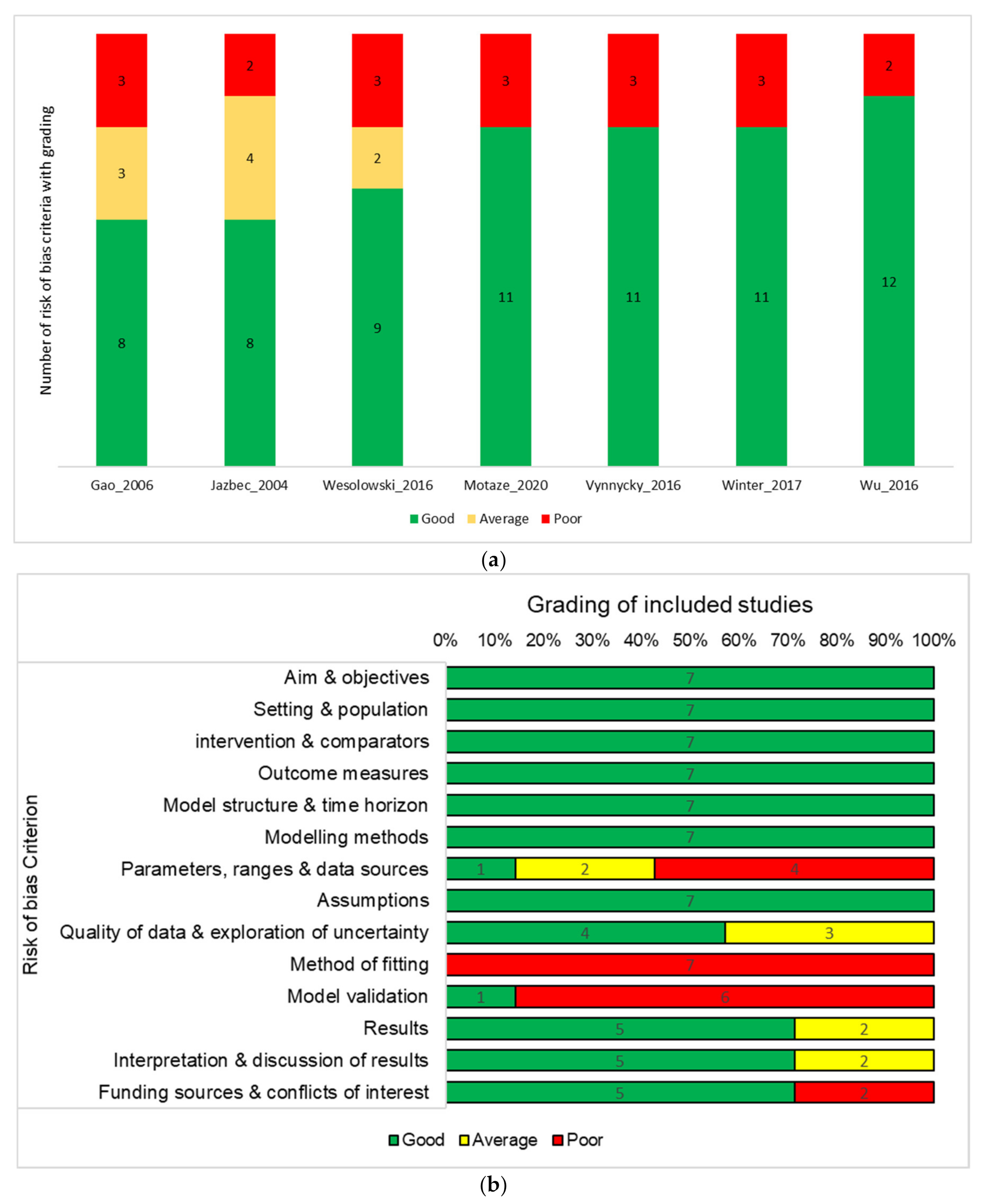
3.1.2. Risk of Bias in Included Studies
3.2. Effect of Rubella Vaccine Introduction
3.2.1. Time to Elimination of CRS
3.2.2. Trends in CRS Incidence
3.2.3. Time to Elimination of Rubella
3.2.4. Trends in Rubella Incidence
3.2.5. Number of Vaccinated Individuals per CRS Case Averted
3.3. Economic Evaluation Measure
3.3.1. Disability-Adjusted Life Years (DALYs) Averted
3.3.2. Vaccine Cost per CRS Cases Averted
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Khan, K.; Kunz, R.; Kleijnen, J.; Antes, G. Five steps to conducting a systematic review. J. R. Soc. Med. 2003, 96, 118–121. [Google Scholar] [CrossRef]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G.; Group TP. Preferred Reporting Items for Systematic Reviews and Meta-Analyses: The PRISMA Statement. PLoS Med. 2009, 6. [Google Scholar] [CrossRef]
- Stroup, D.F.; Berlin, J.A.; Morton, S.C.; Olkin, I.; Williamson, G.D.; Rennie, D.; Moher, D.; Becker, B.J.; Sipe, T.A.; Thacker, S.B.; et al. Meta-analysis of observational studies in epidemiology: A proposal for reporting. J. Am. Med. Assoc. 2000, 283, 2008–2012. [Google Scholar] [CrossRef]
- Fone, D.L.; Hollinghurst, S.; Temple, M.; Round, A.; Lester, N.; Weightman, A.L.; Roberts, K.; Coyle, E.; Bevan, G.; Palmer, S. Systematic review of the use and value of computer simulation modelling in population health and health care delivery. J. Public Health 2003, 25, 325–335. [Google Scholar] [CrossRef]
- Harris, R.C.; Sumner, T.; Knight, G.M.; White, R.G. Systematic review of mathematical models exploring the epidemiological impact of future TB vaccines. Hum. Vaccines Immunother. 2016, 12, 2813–2832. [Google Scholar] [CrossRef]
- Brisson, M.; Kim, J.J.; Canfell, K.; Drolet, M.; Gingras, G.; Burger, E.A.; Martin, D.; Simms, K.T.; Bénard, E.; Boily, M.-C. Impact of HPV vaccination and cervical screening on cervical cancer elimination: A comparative modelling analysis in 78 low-income and lower-middle-income countries. Lancet 2020, 395, 575–590. [Google Scholar] [CrossRef]
- Reef, S.E.; Plotkin, S.A. Rubella Vaccines. In Vaccines, 7th ed.; Elsevier Inc.: Amsterdam, The Netherlands, 2018; Volume 295, pp. 970–1000. [Google Scholar] [CrossRef]
- Reef, S.E.; Plotkin, S.A.; Cooper, D.L.; Alford, C. 29–Rubella. In Remington and Klein’s Infectious Diseases of the Fetus and Newborn Infant; Saunders: Philadelphia, PA, USA, 2016; pp. 894–932. [Google Scholar] [CrossRef]
- World Health Organization. Global Vaccine Action Plan 2011–2020; World Health Organization: Geneva, Switzerland, 2013. [Google Scholar] [CrossRef]
- World Health Organization. Global Measles and Rubella Strategic Plan. 2012. Available online: https://apps.who.int/iris/bitstream/handle/10665/44855/9789241503396_eng.pdf?sequence=1 (accessed on 3 December 2020).
- World Health Organization. Rubella vaccines: WHO position paper-July 2020. Wkly. Epidemiol. Rec. 2020, 27, 306–324. Available online: https://apps.who.int/iris/bitstream/handle/10665/332950/WER9527-eng-fre.pdf?ua=1 (accessed on 30 November 2020).
- World Health Organization. Rubella Vaccines: WHO Position Paper; World Health Organization: Geneva, Switzerland, 2011. [Google Scholar]
- Centre for Reviews and Dissemination. CRD’s Guidance for Undertaking Reviews in Health Care. 2009. Available online: https://www.york.ac.uk/media/crd/Systematic_Reviews.pdf (accessed on 30 November 2020).
- Higgins, J.P.T.; Green, S. Cochrane Handbook for Systematic Reviews of Interventions; Higgins, J.P.T., Thomas, J., Chandler, J., Cumpston, M., Li, T., Page, M.J., Welch, V.A., Eds.; John Wiley & Sons: Chichester, UK, 2019. [Google Scholar] [CrossRef]
- Murad, M.H.; Asi, N.; Alsawas, M.; Alahdab, F. New evidence pyramid. Evid. Based Med. 2016, 21, 125–127. [Google Scholar] [CrossRef]
- Akobeng, A.K. Principles of evidence based medicine. Arch. Dis. Child. 2005, 90, 837–840. [Google Scholar] [CrossRef]
- Takahashi, S.; Metcalf, C.J.E.; Ferrari, M.J.; Tatem, A.J.; Lessler, J. The geography of measles vaccination in the African Great Lakes region. Nat. Commun. 2017, 8, 1–9. [Google Scholar] [CrossRef]
- Wesolowski, A.; Mensah, K.; Brook, C.E.; Andrianjafimasy, M.; Winter, A.; Buckee, C.O.; Razafindratsimandresy, R.; Tatem, A.J.; Heraud, J.-M.; Metcalf, C.J.E. Introduction of rubella-containing-vaccine to Madagascar: Implications for roll-out and local elimination. J. R. Soc. Interface 2016, 13. [Google Scholar] [CrossRef]
- Winter, A.K.; Pramanik, S.; Lessler, J.; Ferrari, M.; Grenfell, B.T.; Metcalf, C.J.E. Rubella vaccination in India: Identifying broad consequences of vaccine introduction and key knowledge gaps. Epidemiol. Infect. 2018, 146, 65–77. [Google Scholar] [CrossRef]
- Metcalf, C.J.E.; Cohen, C.; Lessler, J.; McAnerney, J.M.; Ntshoe, G.M.; Puren, A.; Klepac, P.; Tatem, A.; Grenfell, B.T.; Bjørnstad, O.N. Implications of spatially heterogeneous vaccination coverage for the risk of congenital rubella syndrome in South Africa. J. R. Soc. Interface 2013, 10. [Google Scholar] [CrossRef]
- Bellan, S.E.; Pulliam, J.R.; Scott, J.C.; Dushoff, J. How to make epidemiological training infectious. PLoS Biol. 2012, 10. [Google Scholar] [CrossRef]
- Elveback, L.; Varma, A. Simulation of mathematical models for public health problems. Public Health Rep. 1965, 80, 1067–1076. [Google Scholar] [CrossRef]
- Metcalf, C.J.E.; Lessler, J.; Klepac, P.; Morice, A.; Grenfell, B.T.; Bjørnstad, O.N. Structured models of infectious disease: Inference with discrete data. Theor. Popul. Biol. 2012, 82, 275–282. [Google Scholar] [CrossRef]
- Caro, J.; Eddy, D.M.; Kan, H.; Kaltz, C.; Patel, B.; Eldessouki, R.; Briggs, A.H. Questionnaire to assess relevance and credibility of modeling studies for informing health care decision making: An ISPOR-AMCP-NPC good practice task force report. Value Health 2014, 17, 174–182. [Google Scholar] [CrossRef]
- Team RC. R: A Language and Environment for Statistical Computing. 2020. Available online: https://www.r-project.org (accessed on 30 November 2020).
- Metcalf, C.J.E.; Lessler, J.; Klepac, P.; Cutts, F.; Grenfell, B.T. Impact of birth rate, seasonality and transmission rate on minimum levels of coverage needed for rubella vaccination. Epidemiol. Infect. 2012, 140, 2290–2301. [Google Scholar] [CrossRef]
- Thompson, K.M.; Badizadegan, N.D. Modeling the Transmission of Measles and Rubella to Support Global Management Policy Analyses and Eradication Investment Cases. Risk Anal. 2017, 37, 1109–1131. [Google Scholar] [CrossRef]
- Thompson, K.M.; Odahowski, C.L. The Costs and Valuation of Health Impacts of Measles and Rubella Risk Management Policies. Risk Anal. 2016, 36, 1357–1382. [Google Scholar] [CrossRef]
- Thompson, K.M.; Odahowski, C.L.; Goodson, J.L.; Reef, S.E.; Perry, R.T. Synthesis of Evidence to Characterize National Measles and Rubella Exposure and Immunization Histories. Risk Anal. 2016, 36, 1427–1458. [Google Scholar] [CrossRef]
- Metcalf, C.J.E.; Munayco, C.V.; Chowell, G.; Grenfell, B.T.; Bjørnstad, O.N. Rubella metapopulation dynamics and importance of spatial coupling to the risk of congenital rubella syndrome in Peru. J. R. Soc. Interface 2011, 8, 369–376. [Google Scholar] [CrossRef]
- Reinert, P.; Soubeyrand, B.; Gauchoux, R. Évaluation de 35 années de vaccination rougeole-oreillons-rubéole en France. Arch. Pediatr. 2003, 10, 948–954. [Google Scholar] [CrossRef]
- Lessler, J.; Metcalf, C.J.E. Balancing Evidence and Uncertainty when Considering Rubella Vaccine Introduction. PLoS ONE 2013, 8. [Google Scholar] [CrossRef]
- Hincapié-Palacio, D.; Ospina-Giraldo, J.; Gómez-Arias, R.D.; Uyi-Afuwape, A.; Chowell-Puente, G. Simulating measles and rubella elimination levels according to social stratification and interaction. Rev. Salud Pública 2010, 12, 103–115. [Google Scholar]
- Hachiya, M.; Miyano, S.; Mori, Y.; Vynnycky, E.; Keungsaneth, P.; Vongphrachanh, P.; Xeuatvongsa, A.; Sisouk, T.; Som-Oulay, V.; Khamphaphongphane, B.; et al. Evaluation of nationwide supplementary immunization in Lao people’s democratic republic: Population-based seroprevalence survey of anti-measles and anti-rubella igg in children and adults, mathematical modelling and a stability testing of the vaccine. PLoS ONE 2018, 13, e0194931. [Google Scholar] [CrossRef]
- Béraud, G.; Abrams, S.; Levy-Bruhl, D.; Antona, D.; Beutels, P.; Dervaux, B.; Hens, N. Resurgence risk for measles, mumps and rubella in France in 2016. Med. Mal. Infect. 2016, 46, 4. [Google Scholar] [CrossRef]
- Buonomo, B. A simple analysis of vaccination strategies for rubella. Math. Biosci. Eng. 2011, 8, 677–687. [Google Scholar] [CrossRef]
- Edmunds, W.J.; Van De Heijden, O.G.; Eerola, M.; Gay, N.J. Modelling rubella in Europe. Epidemiol. Infect. 2000, 125, 617–634. [Google Scholar] [CrossRef]
- Feng, Z.; Feng, Y.; Glasser, J.W. Influence of demographically-realistic mortality schedules on vaccination strategies in age-structured models. Theor. Popul. Biol. 2020, 132, 24–32. [Google Scholar] [CrossRef]
- Gao, Z.; Wood, J.G.; Burgess, M.A.; Menzies, R.I.; McIntyre, P.B.; MacIntyre, C.R. Models of strategies for control of rubella and congenital rubella syndrome-A 40 year experience from Australia. Vaccine 2013, 31, 691–697. [Google Scholar] [CrossRef]
- Edmunds, W.J.; Gay, N.J.; Kretzschmar, M.; Pebody, R.G.; Wachmann, H. The pre-vaccination epidemiology of measles, mumps and rubella in Europe: Implications for modelling studies. Epidemiol. Infect. 2000, 125, 635–650. [Google Scholar] [CrossRef] [PubMed]
- Motaze, N.V.; Edoka, I.; Wiysonge, C.S.; Metcalf, C.J.E.; Winter, A.K. Rubella vaccine introduction in the South African public vaccination schedule: Mathematical modelling for decision making. Vaccines 2020, 8, 383. [Google Scholar] [CrossRef]
- Jazbec, A.; Delimar, M.; Vrzić, V.S. Simulation model of rubella—The effects of vaccination strategies. Appl. Math. Comput. 2004, 153, 75–96. [Google Scholar] [CrossRef]
- Gao, L.; Hethcote, H. Simulations of rubella vaccination strategies in China. Math. Biosci. 2006, 202, 371–385. [Google Scholar] [CrossRef]
- Vynnycky, E.; Yoshida, L.M.; Huyen, D.T.; Trung, N.D.; Toda, K.; Van Cuong, N.; Hong, D.T.; Ariyoshi, K.; Miyakawa, M.; Moriuchi, H.; et al. Modeling the impact of rubella vaccination in Vietnam. Hum. Vaccines Immunother. 2015, 12, 150–158. [Google Scholar] [CrossRef][Green Version]
- Wu, Y.; Wood, J.; Khandaker, G.; Waddington, C.; Snelling, T. Informing rubella vaccination strategies in East Java, Indonesia through transmission modelling. Vaccine 2016, 34, 5636–5642. [Google Scholar] [CrossRef]
- World Bank. World Bank Country and Lending Groups. 2020. Available online: https://datahelpdesk.worldbank.org/knowledgebase/articles/906519-world-bank-country-and-lending-groups (accessed on 7 December 2020).
- Panagiotopoulos, T.; Antoniadou, L.; Valassi-Adam, E. Increase in congenital rubella occurrence after immunisation in Greece: Retrospective survey and systematic review. Br. Med. J. 1999, 319, 1462–1466. [Google Scholar] [CrossRef]
- Jiménez, G.; Avila-Aguero, M.L.; Morice, A.; Gutiérrez, H.; Soriano, A.; Badilla, X.; Reef, S.; Castillo-Solórzano, C. Estimating the burden of congenital rubella syndrome in Costa Rica, 1996–2001. Pediatr. Infect. Dis. J. 2007, 26, 382–386. [Google Scholar] [CrossRef]
- Dudgeon, J.A. Selective immunization: Protection of the individual. Clin. Infect. Dis. 1985, 7, S185–S190. [Google Scholar] [CrossRef]
- Hanaoka, M.; Yamaguchi, K.; Yamaguchi, S. Impact of vaccination policy on changes in rubella virus antibody in Japanese pregnant women. J. Public Health 2020. [Google Scholar] [CrossRef]
- Kopec, J.A.; Finès, P.; Manuel, D.G.; Buckeridge, D.L.; Flanagan, W.M.; Oderkirk, J.; Abrahamowicz, M.; Harper, S.; Sharif, B.; Okhmatovskaia, A.; et al. Validation of population-based disease simulation models: A review of concepts and methods. BMC Public Health 2010, 10. [Google Scholar] [CrossRef] [PubMed]
| Study | Description of Target Age Groups and Sex for Each Vaccine Introduction Scenario | Setting | WHO Region | World Bank Grading | Previous Private Sector RCV | Time Frame | Classes | Reported Outcomes |
|---|---|---|---|---|---|---|---|---|
| Gao_2016 [43] |
| China | Western Pacific | Upper-middle Income | Yes | 46 years | MSEIRV |
|
| Jazbec_2004 [42] |
| Croatia, Tresnjevka municipality | Europe | High-income | No | 55 years | MSEIRV |
|
| Motaze_2020 [41] |
| South Africa | Africa | Upper-middle income | Yes | 30 years | MSIRV |
|
| Vynnycky_2016 [44] |
| Vietnam | Western Pacific | Lower-middle Income | No | 37 years | MSEIRV |
|
| Wesolowski_2016 [18] |
| Madagascar | Africa | Lowe-middle income | No | 30 years | MSIRV |
|
| Winter_2017 [19] |
| India | South-East Asia | Lower-middle income | Yes | 30 years | MSIRV |
|
| Wu_2016 [45] |
| Indonesia, East Java province | South-East Asia | Upper-middle income | No | 50 years | SEIRV |
|
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Motaze, N.V.; Mthombothi, Z.E.; Adetokunboh, O.; Hazelbag, C.M.; Saldarriaga, E.M.; Mbuagbaw, L.; Wiysonge, C.S. The Impact of Rubella Vaccine Introduction on Rubella Infection and Congenital Rubella Syndrome: A Systematic Review of Mathematical Modelling Studies. Vaccines 2021, 9, 84. https://doi.org/10.3390/vaccines9020084
Motaze NV, Mthombothi ZE, Adetokunboh O, Hazelbag CM, Saldarriaga EM, Mbuagbaw L, Wiysonge CS. The Impact of Rubella Vaccine Introduction on Rubella Infection and Congenital Rubella Syndrome: A Systematic Review of Mathematical Modelling Studies. Vaccines. 2021; 9(2):84. https://doi.org/10.3390/vaccines9020084
Chicago/Turabian StyleMotaze, Nkengafac Villyen, Zinhle E. Mthombothi, Olatunji Adetokunboh, C. Marijn Hazelbag, Enrique M. Saldarriaga, Lawrence Mbuagbaw, and Charles Shey Wiysonge. 2021. "The Impact of Rubella Vaccine Introduction on Rubella Infection and Congenital Rubella Syndrome: A Systematic Review of Mathematical Modelling Studies" Vaccines 9, no. 2: 84. https://doi.org/10.3390/vaccines9020084
APA StyleMotaze, N. V., Mthombothi, Z. E., Adetokunboh, O., Hazelbag, C. M., Saldarriaga, E. M., Mbuagbaw, L., & Wiysonge, C. S. (2021). The Impact of Rubella Vaccine Introduction on Rubella Infection and Congenital Rubella Syndrome: A Systematic Review of Mathematical Modelling Studies. Vaccines, 9(2), 84. https://doi.org/10.3390/vaccines9020084






