COVID-19 Breakthrough Infections and Transmission Risk: Real-World Data Analyses from Germany’s Largest Public Health Department (Cologne)
Abstract
:1. Introduction
2. Methods
2.1. Study Design
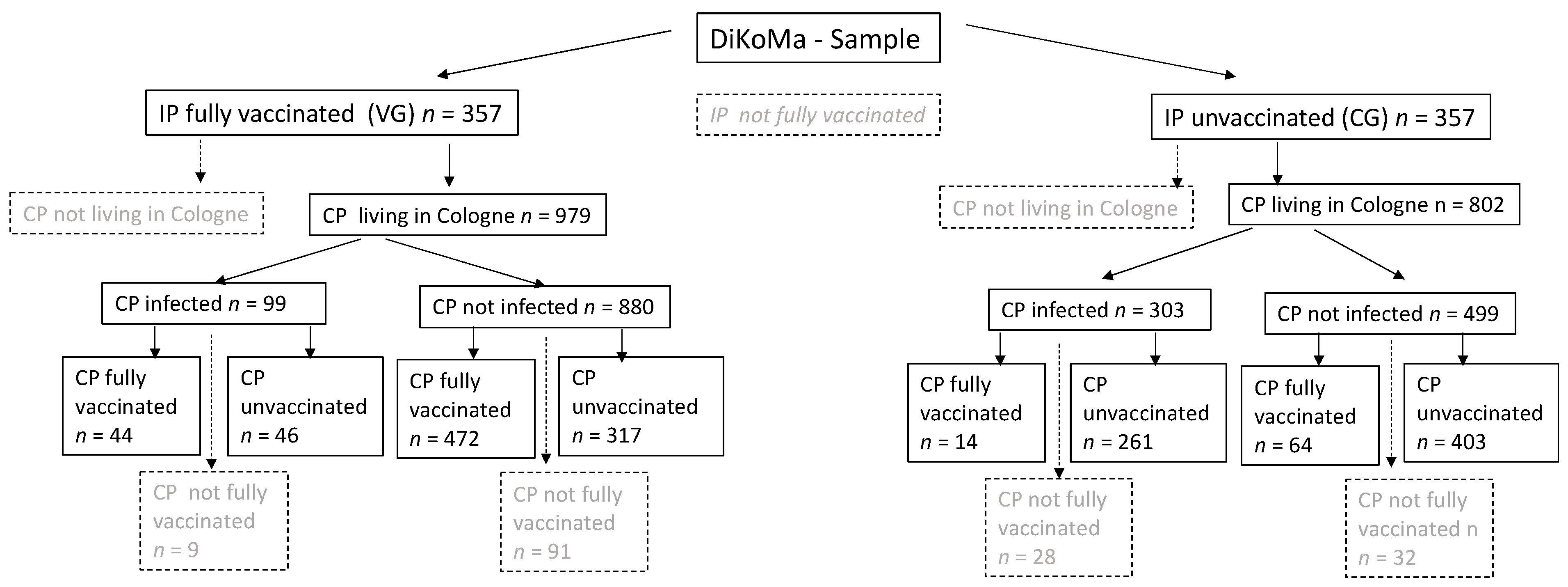
2.2. Study Population
2.3. Matching
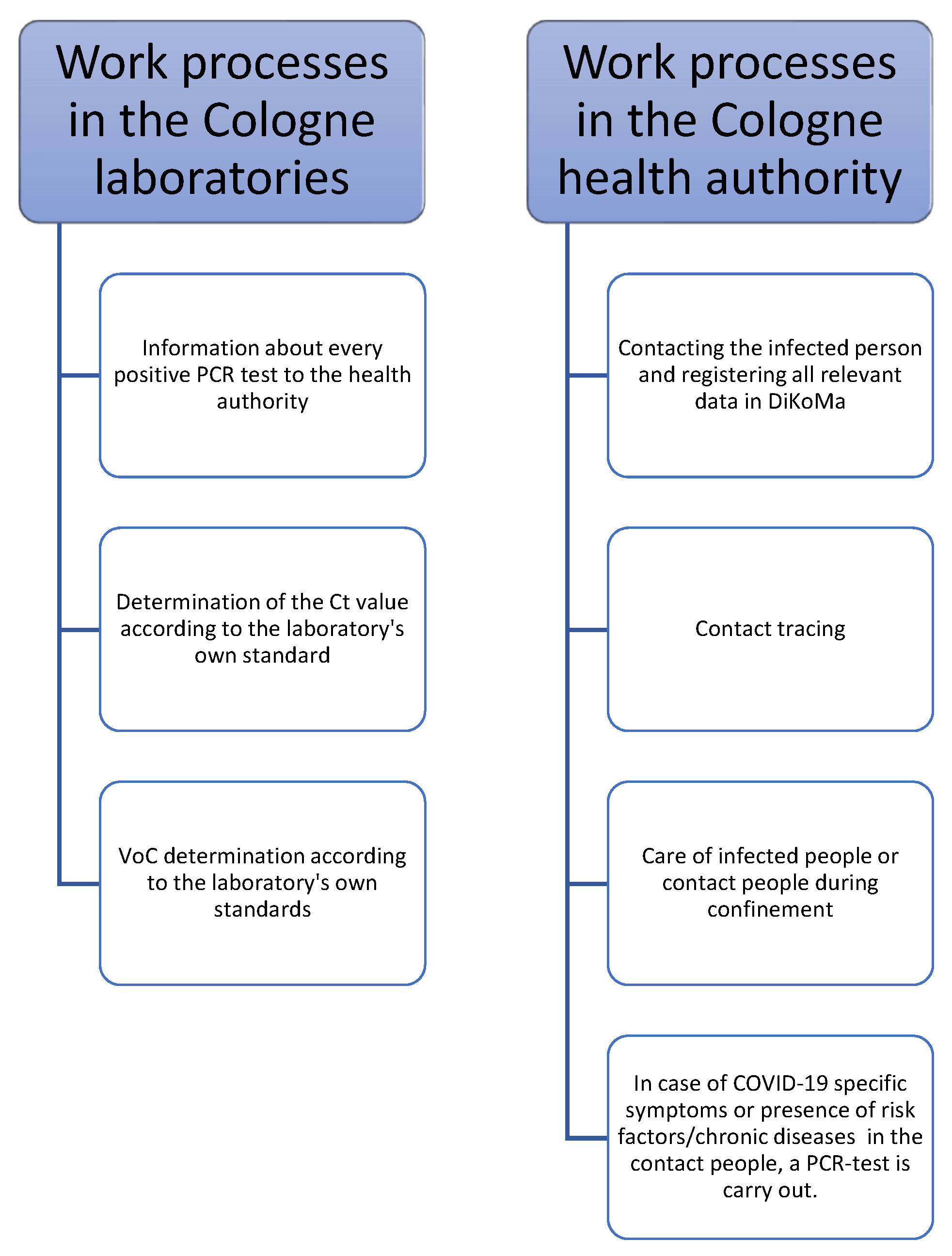
2.4. Laboratory Analyses
2.5. Transmission Risk
2.6. Statistical Analyses
3. Results
VG versus CG
4. Discussion
Strength and Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Robert-Koch-Institut. Wöchentlicher Lagebericht des RKI zur Coronavirus-Krankheit-2019 (COVID-19) 16.09.2021—AKTUALISIERTER STAND FÜR DEUTSCHLAND. Available online: https://www.rki.de/DE/Content/InfAZ/N/Neuartiges_Coronavirus/Situationsberichte/Wochenbericht/Wochenbericht_2021-09-16.pdf?__blob=publicationFile (accessed on 3 October 2021).
- Robert-Koch-Institut. Wöchentlicher Lagebericht des RKI zur Coronavirus-Krankheit-2019 (COVID-19) 30.09.2021—AKTUALISIERTER STAND FÜR DEUTSCHLAND. Available online: https://www.rki.de/DE/Content/InfAZ/N/Neuartiges_Coronavirus/Situationsberichte/Wochenbericht/Wochenbericht_2021-09-30.pdf?__blob=publicationFile (accessed on 3 October 2021).
- Haas, E.J.; Angulo, F.J.; McLaughlin, J.M.; Anis, E.; Singer, S.R.; Khan, F.; Brooks, N.; Smaja, M.; Mircus, G.; Pan, K.; et al. Impact and effectiveness of mRNA BNT162b2 vaccine against SARS-CoV-2 infections and COVID-19 cases, hospitalisations, and deaths following a nationwide vaccination campaign in Israel: An observational study using national surveillance data. Lancet 2021, 397, 1819–1829. [Google Scholar] [CrossRef]
- Lopez Bernal, J.; Andrews, N.; Gower, C. Effectiveness of the Pfizer-BioNTech and Oxford-AstraZeneca vaccines on covid-19 related symptoms, hospital admissions, and mortality in older adults in England: Test negative case-control study. BMJ 2021, 373, n1088. [Google Scholar] [CrossRef] [PubMed]
- Menni, C.; Klaser, K.; May, A. Vaccine side-effects and SARS-CoV-2 infection after vaccination in users of the COVID Symptom Study app in the UK: A prospective observational study. Lancet Infect. Dis. 2021, 21, 939–949. [Google Scholar] [CrossRef]
- Dagan, N.; Barda, N.; Kepten, E.; Miron, O.; Perchik, S.; Katz, M.A.; Hernán, M.A.; Lipsitch, M.; Reis, B.; Balicer, R.D. BNT162b2 mRNA Covid-19 Vaccine in a Nationwide Mass Vaccination Setting. N. Engl. J. Med. 2021, 384, 1412–1423. [Google Scholar] [CrossRef] [PubMed]
- Levine-Tiefenbrun, M.; Yelin, I.; Katz, R.; Herzel, E.; Golan, Z.; Schreiber, L.; Wolf, T.; Nadler, V.; Ben-Tov, A.; Kuint, J.; et al. Initial report of decreased SARS-CoV-2 viral load after inoculation with the BNT162b2 vaccine. Nat. Med. 2021, 27, 790–792. [Google Scholar] [CrossRef] [PubMed]
- Riemersma, K.K.; Grogan, B.E.; Kita-Yarbro, A.; Halfmann, P.J.; Segaloff, H.E.; Kocharian, A.; Florek, K.R.; Westergaard, R.; Bateman, A.; Jeppson, G.E.; et al. Shedding of Infectious SARS-CoV-2 Despite Vaccination when the Delta Variant is Prevalent—Wisconsin, July 2021 [Internet]. bioRxiv 2021. [Preprint]. [Google Scholar] [CrossRef]
- Chia, P.Y.; Xiang Ong, S.W.; Chiew, C.J.; Ang, L.W.; Chavatte, J.M.; Mak, T.M.; Cui, L.; Kalimuddin, S.; Chia, W.N.; Tan, C.W.; et al. Virological and serological kinetics of SARS-CoV-2 Delta variant vaccine-breakthrough infections: A multi-center cohort study [Internet]. bioRxiv 2021. Preprint. [Google Scholar] [CrossRef]
- Hsu, L.; Hurraß, J.; Kossow, A.; Klobucnik, J.; Nießen, J.; Wiesmüller, G.A.; Grüne, B.; Joisten, C. Breakthrough infections with the delta variant: Vaccinations halved transmission risk. Public Health 2021. under review. [Google Scholar]
- European Centre for Disease Prevention and Control (ECDC). Guidance for Discharge and Ending of Isolation of People with COVID-19. Available online: https://www.ecdc.europa.eu/en/publications-data/covid-19-guidance-discharge-and-ending-isolation (accessed on 3 October 2021).
- Robert-Koch-Institut. Kontaktpersonen-Nachverfolgung bei SARS-CoV-2-Infektionen. Available online: https://www.rki.de/DE/Content/InfAZ/N/Neuartiges_Coronavirus/Kontaktperson/Management.html (accessed on 3 October 2021).
- Neuhann, F.; Buess, M.; Wolff, A.; Pusch, L.; Kossow, A.; Winkler, M.; Demir, J.; Beyé, M.; Wiesmüller, G.; Nießen, J.; et al. Entwicklung einer Software zur Unterstützung der Prozesse im Gesundheitsamt der Stadt Köln in der SARS-CoV-2-Pandemie. Epid. Bull. 2020, 23. [Google Scholar] [CrossRef]
- Curran, J.; Dol, J.; Boulos, L.; Somerville, M.; Reynolds, B.; Gallant, A.; Saxinger, L.; Doroshenko, A.; Shin, D.; Wong, H.; et al. Public Health and Health Systems Impacts of SARS-CoV-2 Variants of Concern: A Rapid Scoping Review. medRxiv 2021. Preprint. [Google Scholar] [CrossRef]
- Müller, L.; Andrée, M.; Moskorz, W.; Drexler, I.; Walotka, L.; Grothmann, R.; Ptok, J.; Hillebrandt, J.; Ritchie, A.; Rabl, D.; et al. Age-dependent immune response to the Biontech/Pfizer BNT162b2 COVID-19 vaccination. Clin. Infect. Dis. 2021, ciab381. [Google Scholar] [CrossRef] [PubMed]
- El Bagoury, M.; Tolba, M.M.; Nasser, H.A.; Jabbar, A.; Elagouz, A.M.; Aktham, Y.; Hutchinson, A. The find of COVID-19 vaccine: Challenges and opportunities. J. Infect. Public Health 2021, 14, 389–416. [Google Scholar] [CrossRef] [PubMed]
- Singanayagam, A.; Patel, M.; Charlett, A.; Bernal, J.L.; Saliba, V.; Ellis, J.; Ladhani, S.; Zambon, M.; Gopal, R. Duration of infectiousness and correlation with RT-PCR cycle threshold values in cases of COVID-19, England, January to May 2020. Eurosurveillance 2020, 25, 2001483. [Google Scholar] [CrossRef] [PubMed]
- Robert-Koch-Institut. Anzahl und Anteile von VOC und VOI in Deutschland. Available online: https://www.rki.de/DE/Content/InfAZ/N/Neuartiges_Coronavirus/Daten/VOC_VOI_Tabelle.html (accessed on 3 October 2021).
- Harris, R.J.; Hall, J.A.; Zaidi, A.; Andrews, N.J.; Dunbar, J.K.; Dabrera, G. Effect of Vaccination on Household Transmission of SARS-CoV-2 in England. N. Engl. J. Med. 2021, 385, 759–760. [Google Scholar] [CrossRef]
- Braeye, T.; Cornelissen, L.; Catteau, L.; Haarhuis, F.; Proesmans, K.; De Ridder, K.; Djiena, A.; Mahieu, R.; De Leeuw, F.; Dreuw, A.; et al. Vaccine effectiveness against infection and onwards transmission of COVID-19: Analysis of Belgian contact tracing data, January–June 2021. Vaccine 2021, 39, 5456–5460. [Google Scholar] [CrossRef] [PubMed]
- Brown, C.M.; Vostok, J.; Johnson, H.; Burns, M.; Gharpure, R.; Sami, S.; Sabo, R.T.; Hall, N.; Foreman, A.; Schubert, P.L.; et al. Outbreak of SARS-CoV-2 Infections, Including COVID-19 Vaccine Breakthrough Infections, Associated with Large Public Gatherings—Barnstable County, Massachusetts, July 2021. MMWR Morb. Mortal. Wkly. Rep. 2021, 70, 1059–1062. [Google Scholar] [CrossRef] [PubMed]
- Ioannou, P.; Karakonstantis, S.; Astrinaki, E.; Saplamidou, S.; Vitsaxaki, E.; Hamilos, G.; Sourvinos, G.; Kofteridis, D.P. Transmission of SARS-CoV-2 variant B.1.1.7 among vaccinated health care workers. Infect. Dis. 2021, 53, 876–879. [Google Scholar] [CrossRef]
- Eyre, D.W.; Taylor, D.; Purver, M.; Chapman, D.; Fowler, T.; Pouwels, K.; Walker, A.S.; Peto, T.E. The impact of SARS-CoV-2 vaccination on Alpha & Delta variant transmission. medRxiv 2021. [Preprint]. [Google Scholar] [CrossRef]
- Michalakis, Y.; Sofonea, M.T.; Alizon, S.; Bravo, I.G. SARS-CoV-2 viral RNA levels are not ‘viral load’. Trends Microbiol. 2021, 29, 970–972. [Google Scholar] [CrossRef] [PubMed]
- Rzymski, P.; Pazgan-Simon, M.; Simon, K.; Łapiński, T.; Zarębska-Michaluk, D.; Szczepańska, B.; Chojnicki, M.; Mozer-Lisewska, I.; Flisiak, R. Clinical Characteristics of Hospitalized COVID-19 Patients Who Received at Least One Dose of COVID-19 Vaccine. Vaccines 2021, 9, 781. [Google Scholar] [CrossRef]
- Favresse, J.; Bayart, J.L.; Mullier, F.; Elsen, M.; Eucher, C.; Van Eeckhoudt, S.; Roy, T.; Wieers, G.; Laurent, C.; Dogné, J.-M.; et al. Antibody titres decline 3-month post-vaccination with BNT162b2. Emerg. Microbes Infect. 2021, 10, 1495–1498. [Google Scholar] [CrossRef] [PubMed]
- Thomas, S.J.; Moreira, E.D., Jr.; Kitchin, N.; Absalon, J.; Gurtman, A.; Lockhart, S.; Perez, J.L.; Perez Marc, G.; Polack, F.P.; Zerbini, C.; et al. C4591001 Clinical Trial Group. Six Month Safety and Efficacy of the BNT162b2 mRNA COVID-19 Vaccine. medRxiv 2021. Preprint. [Google Scholar] [CrossRef]
| VG n (%) | CG n (%) | p-Value | |
|---|---|---|---|
| Female | 231 (64.7%) | 231 (64.7%) | n.s. * |
| Male | 126 (35.3%) | 126 (35.3%) | |
| Age (years) Mean ± SD | 48.6 ± 22.1 | 46.7 ± 21.0 | n.s. ** |
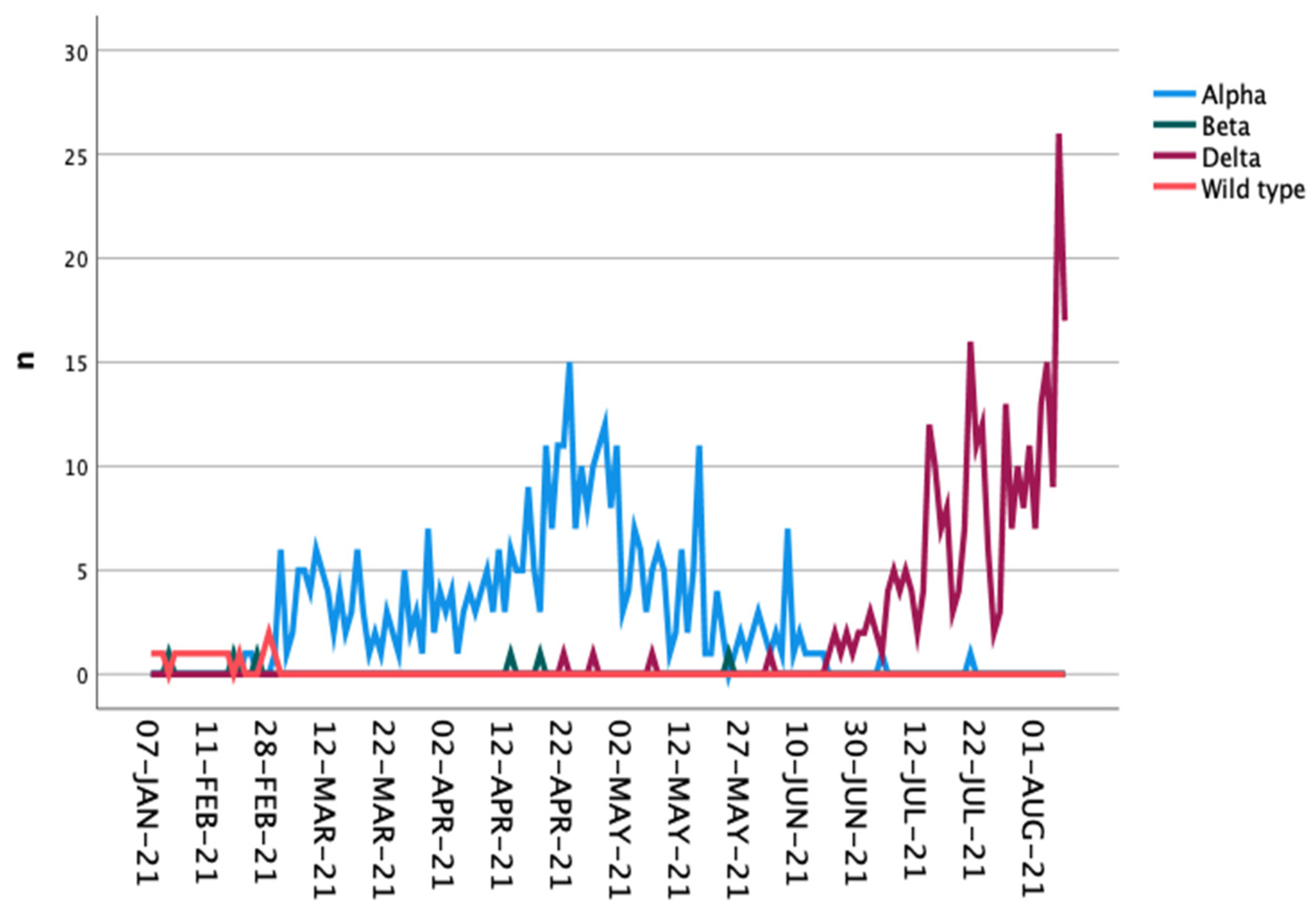
| VoC/virus type | |||
| Wild type | 9 (2.5%) | 9 (2.5%) | n.s. * |
| Alpha | 202 (56.6%) | 202 (56.6%) | |
| Beta | 3 (0.8%) | 3 (0.8%) | |
| Delta | 143 (40.1%) | 143 (40.1%) | |
| Symptoms | |||
| Yes | 147 (41.2%) | 287 (77.9%) | <0.001 * |
| No | 210 (58.8%) | 79 (22.1%) |
| a | |||||
|---|---|---|---|---|---|
| Group (n) | Mean | SD | p-Value * | ||
| Ct value | VG (300) | 29.5 | 7.5 | <0.001 | |
| CG (287) | 25.7 | 6.6 | |||
| Number of CPs per IP | VG (357) | 2.74 | 3.11 | 0.008 | |
| CG (357) | 2.24 | 1.63 | |||
| Number of infected CPs per IP | VG (357) | 0.27 | 0.69 | <0.001 | |
| CG (357) | 0.79 | 1.06 | |||
| Number of infected CPs to total number of CPs per IP | VG (357) | 0.13 | 0.31 | <0.001 | |
| CG (357) | 0.37 | 0.43 | |||
| b | |||||
| Ct-value | VoC | n | Mean | SD | p-Value * |
| VG | Alpha | 156 | 33.1 | 6.0 | <0.001 |
| Delta | 133 | 25.0 | 6.7 | ||
| CG | Alpha | 154 | 26.9 | 6.4 | <0.001 |
| Delta | 124 | 24.1 | 6.4 | ||
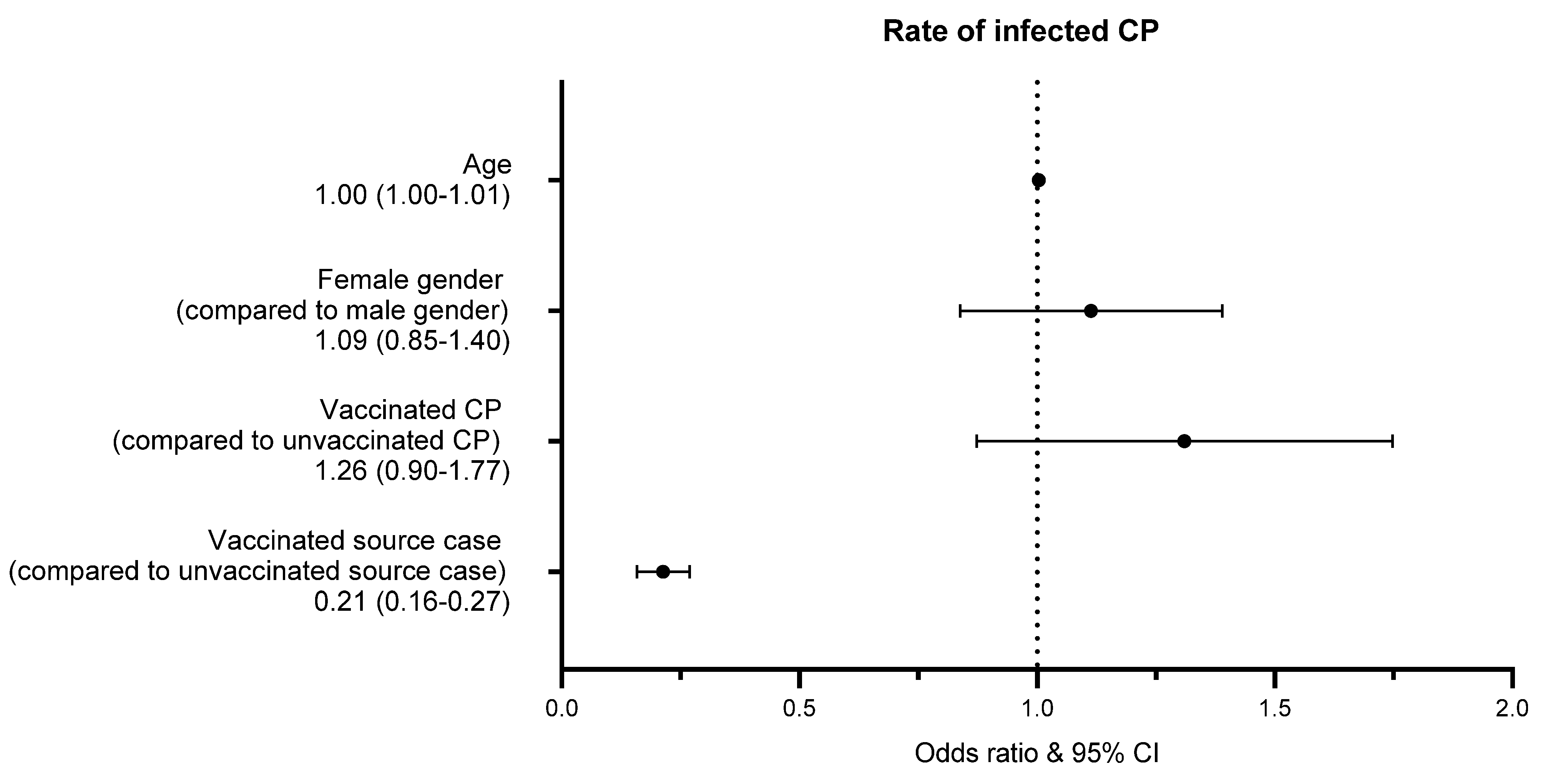
| Model | Non-Standardized Coefficients | Standardized Coefficients | Sig. | 95% Confidence Interval | |||
|---|---|---|---|---|---|---|---|
| Regression Coefficient (B) | Std. Error | β | Lower Limit | Upper Limit | |||
| Baseline model | Age (years) | 0.002 | 0.001 | 0.087 | 0.034 | 0.00 | 0.00 |
| Gender (female = 1; male = 2) | 0.065 | 0.033 | 0.080 | 0.046 | 0.00 | 0.13 | |
| Ct value | −0.006 | 0.002 | −0.115 | 0.011 | −0.01 | 0.00 | |
| B.1.617.2 versus others | 0.030 | 0.036 | 0.038 | 0.396 | −0.04 | 0.10 | |
| VG (1) versus CG (2) | 0.191 | 0.033 | 0.243 | <0.001 | 0.13 | 0.26 | |
| Symptomatic (yes = 1; no = 2) | −0.093 | 0.037 | −0.116 | 0.013 | −0.17 | −0.02 | |
| Final model | Age | 0.001 | 0.001 | 0.079 | 0.049 | 0.00 | 0.00 |
| Gender (female = 1; male = 2) | 0.070 | 0.032 | 0.085 | 0.032 | 0.01 | 0.13 | |
| Ct value | −0.007 | 0.002 | −0.125 | 0.005 | −0.01 | 0.00 | |
| VG (1) versus CG (2) | 0.186 | 0.033 | 0.237 | <0.001 | 0.12 | 0.25 | |
| Symptomatic (yes = 1; no = 2) | −0.100 | 0.036 | −0.125 | 0.006 | −0.17 | −0.03 | |
| Vaccination Status | p-Value | ||
|---|---|---|---|
| Unvaccinated | Fully Vaccinated | ||
| Non-infected CP | 875 (74.0%) | 381 (86.8%) | <0.001 * |
| Infected CP | 307 (26.0%) | 58 (13.2%) | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hsu, L.; Grüne, B.; Buess, M.; Joisten, C.; Klobucnik, J.; Nießen, J.; Patten, D.; Wolff, A.; Wiesmüller, G.A.; Kossow, A.; et al. COVID-19 Breakthrough Infections and Transmission Risk: Real-World Data Analyses from Germany’s Largest Public Health Department (Cologne). Vaccines 2021, 9, 1267. https://doi.org/10.3390/vaccines9111267
Hsu L, Grüne B, Buess M, Joisten C, Klobucnik J, Nießen J, Patten D, Wolff A, Wiesmüller GA, Kossow A, et al. COVID-19 Breakthrough Infections and Transmission Risk: Real-World Data Analyses from Germany’s Largest Public Health Department (Cologne). Vaccines. 2021; 9(11):1267. https://doi.org/10.3390/vaccines9111267
Chicago/Turabian StyleHsu, Lea, Barbara Grüne, Michael Buess, Christine Joisten, Jan Klobucnik, Johannes Nießen, David Patten, Anna Wolff, Gerhard A. Wiesmüller, Annelene Kossow, and et al. 2021. "COVID-19 Breakthrough Infections and Transmission Risk: Real-World Data Analyses from Germany’s Largest Public Health Department (Cologne)" Vaccines 9, no. 11: 1267. https://doi.org/10.3390/vaccines9111267
APA StyleHsu, L., Grüne, B., Buess, M., Joisten, C., Klobucnik, J., Nießen, J., Patten, D., Wolff, A., Wiesmüller, G. A., Kossow, A., & Hurraß, J. (2021). COVID-19 Breakthrough Infections and Transmission Risk: Real-World Data Analyses from Germany’s Largest Public Health Department (Cologne). Vaccines, 9(11), 1267. https://doi.org/10.3390/vaccines9111267






