Oral Vaccination of Grass Carp (Ctenopharyngodon idella) with Baculovirus-Expressed Grass Carp Reovirus (GCRV) Proteins Induces Protective Immunity against GCRV Infection
Abstract
1. Introduction
2. Materials and Methods
2.1. Ethics Statement
2.2. Fish, Virus, Cells and Plasmid
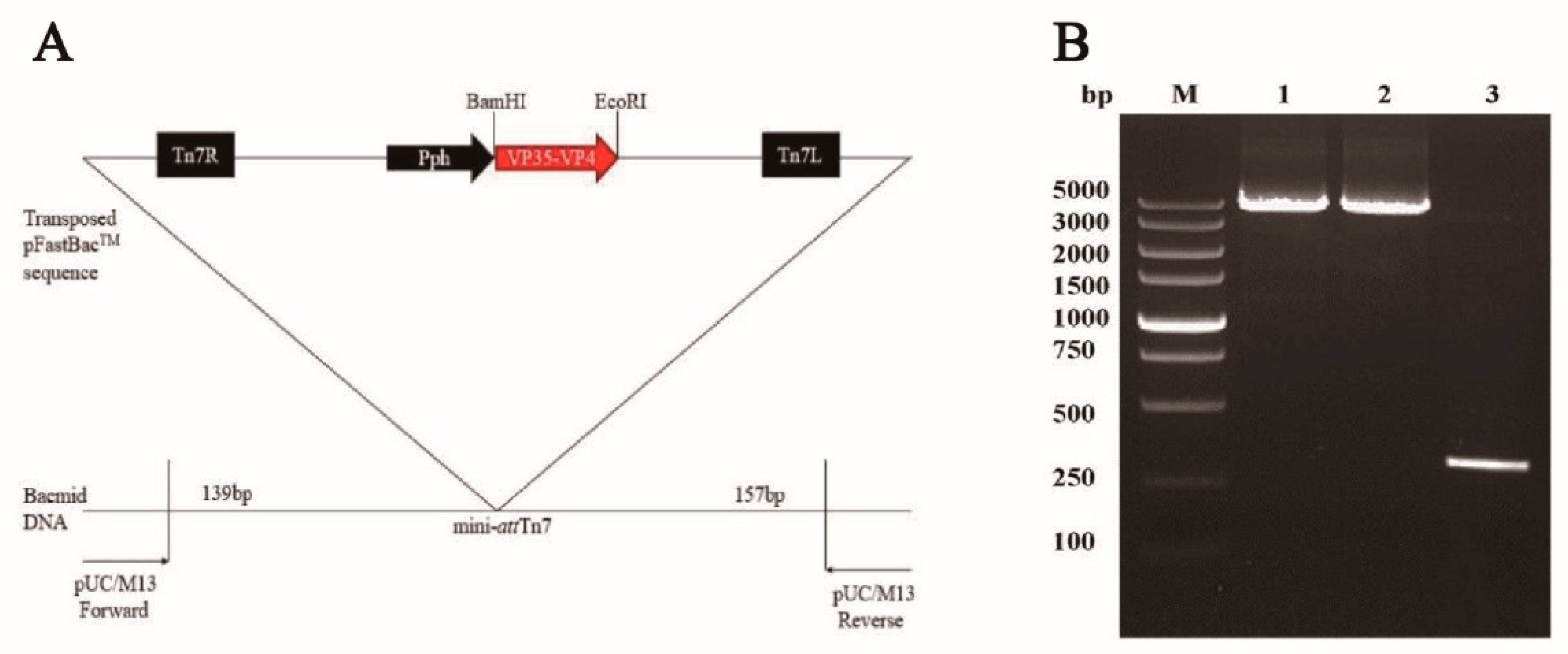
2.3. Generation of Recombinant Baculovirus
2.4. Analysis of VP35-VP4 Expression in BmE Cells by Western Blotting
2.5. Detection of VP35-VP4 Protein Expression in BmE Cells by IFA
2.6. Multiplication and Detection of rVP35-VP4 Protein in Silkworm Pupae
2.7. Oral Immunization and Sample Collection
2.8. Counting of Blood Cells and Differential Leukocytes
2.9. Detection of Serum Antibody Titer by Enzyme-Linked Immunosorbent Assay (ELISA)
2.10. Determination of Immune-Related Genes Expression by qRT-PCR
2.11. Challenge Test
2.12. Statistical Analysis
3. Results
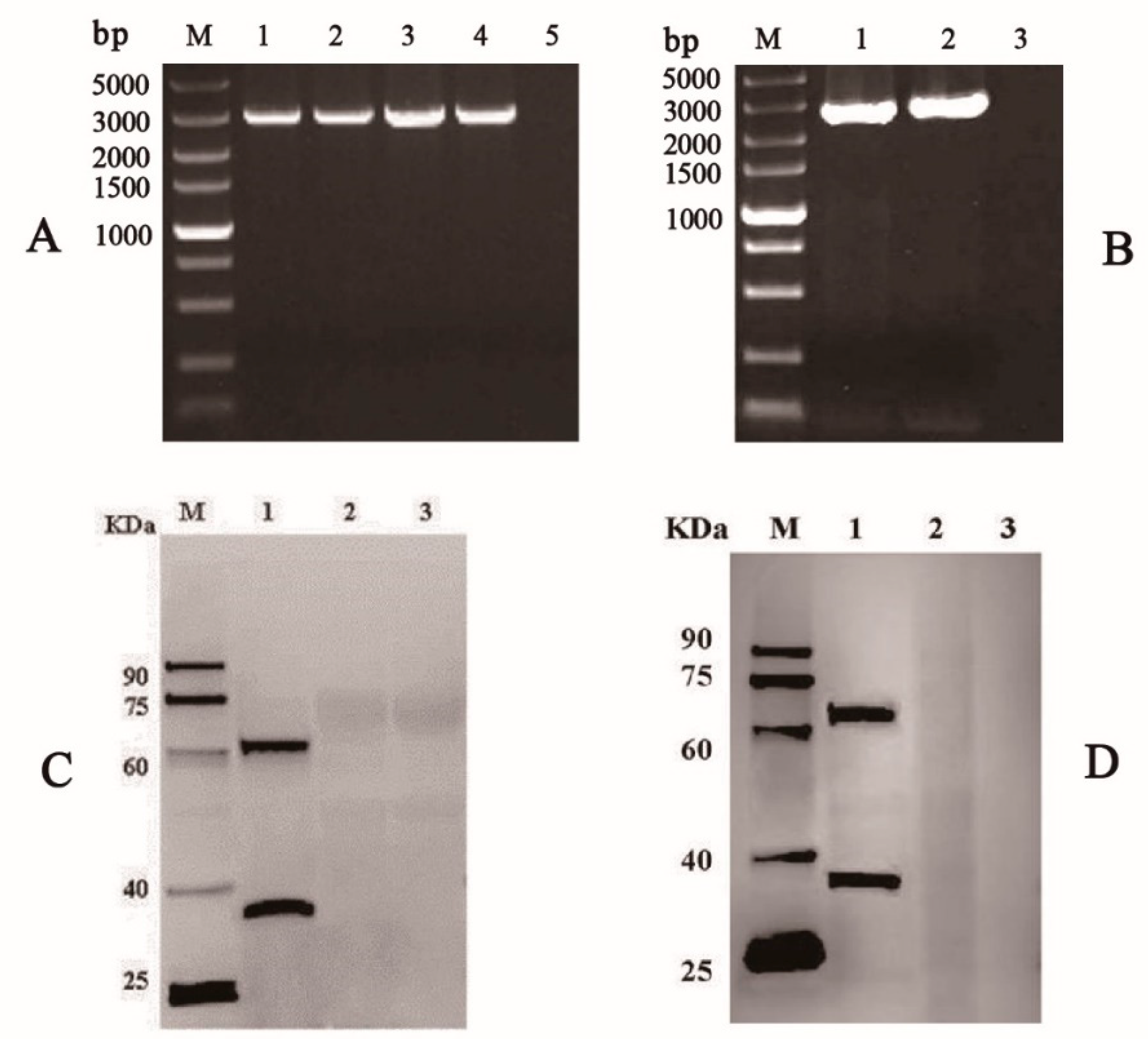
3.1. Generation of Recombinant Baculoviruses in BmE Cells
3.2. Expression of VP35-VP4 Protein in the BmE Cells and Silkworm upae
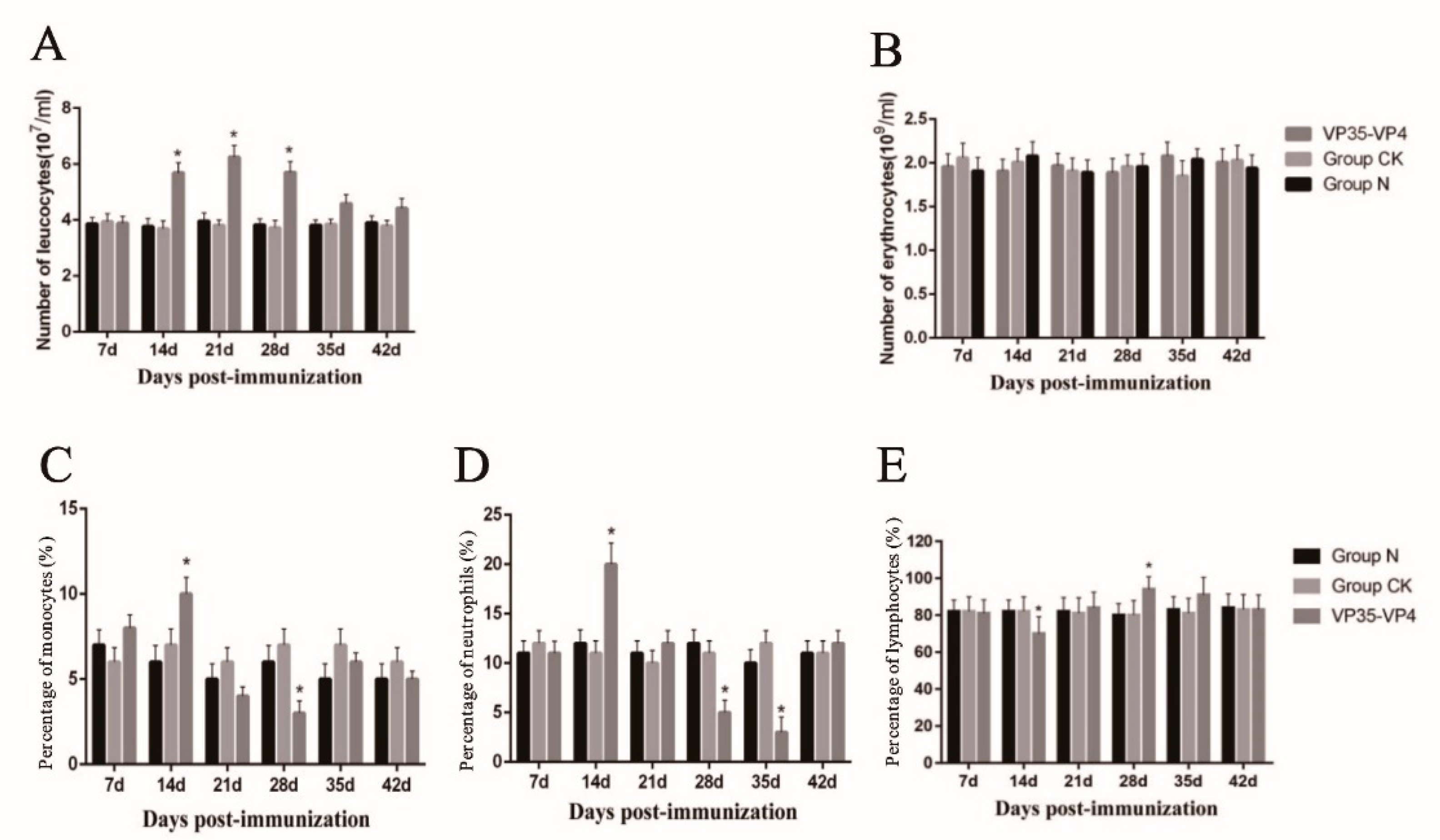
3.3. Blood Cell Counting
3.4. Differential Leukocyte Count
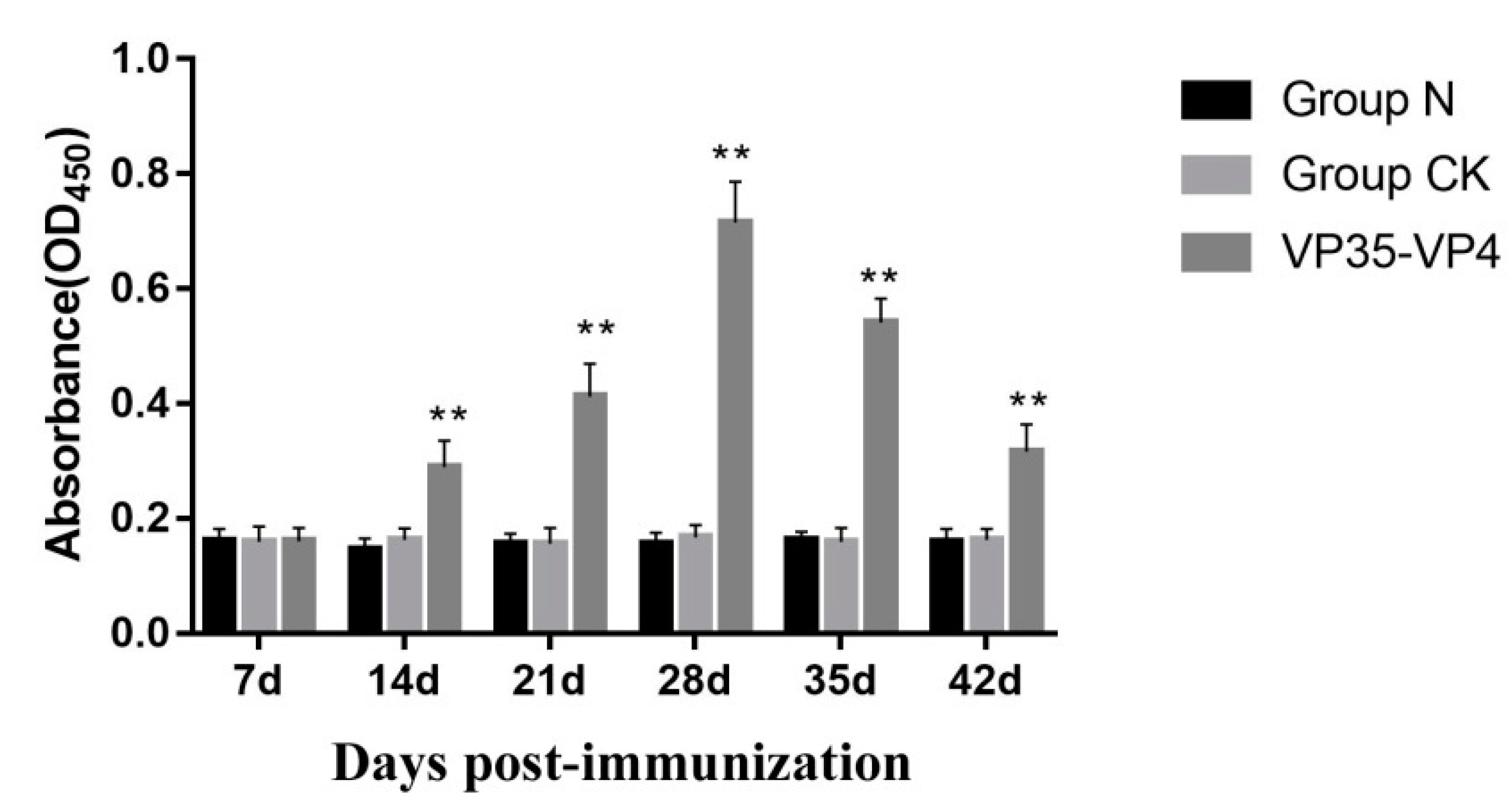
3.5. Detection of Serum Antibody Titer by ELISA
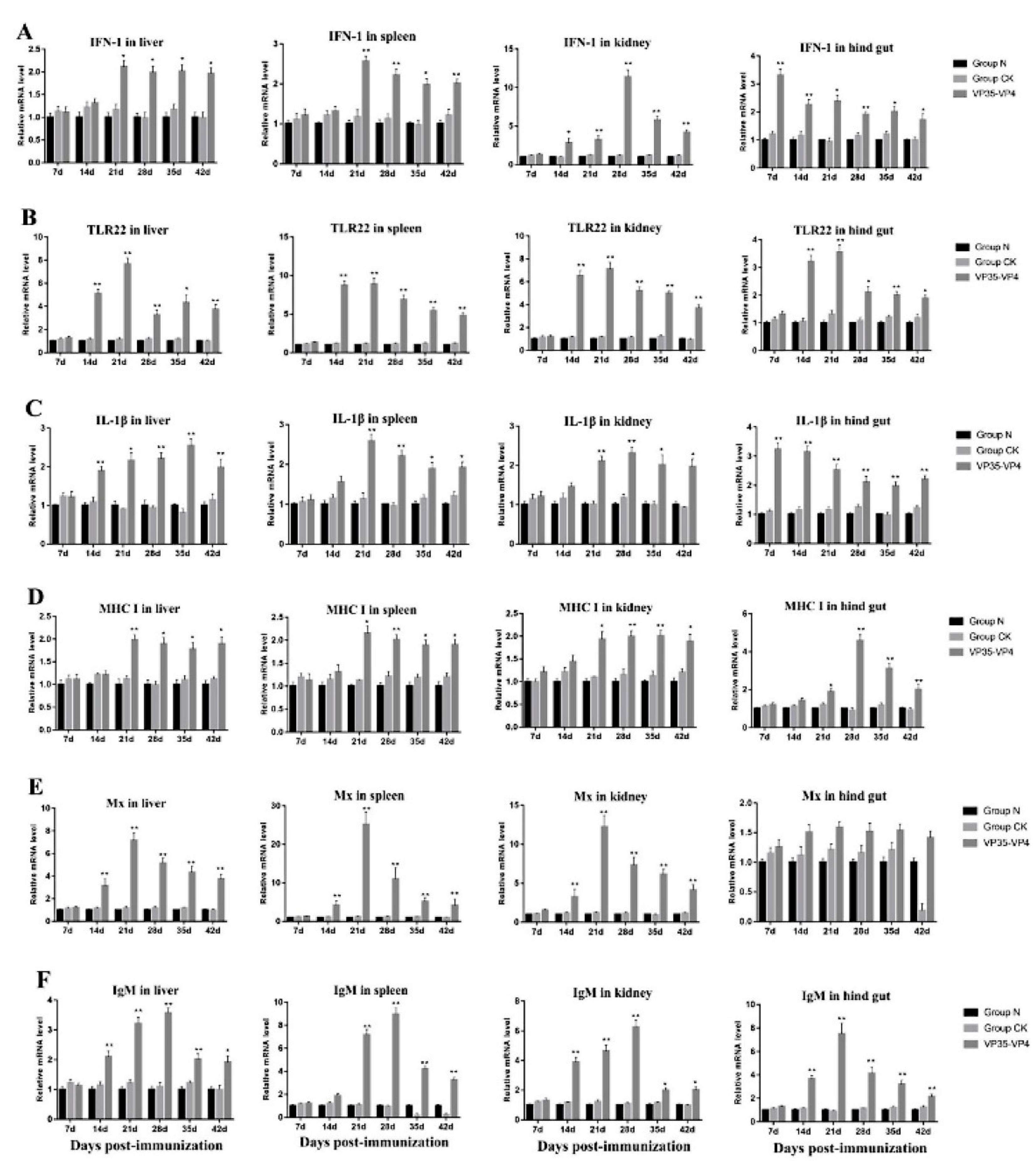
3.6. Expression of Immune-Related Genes
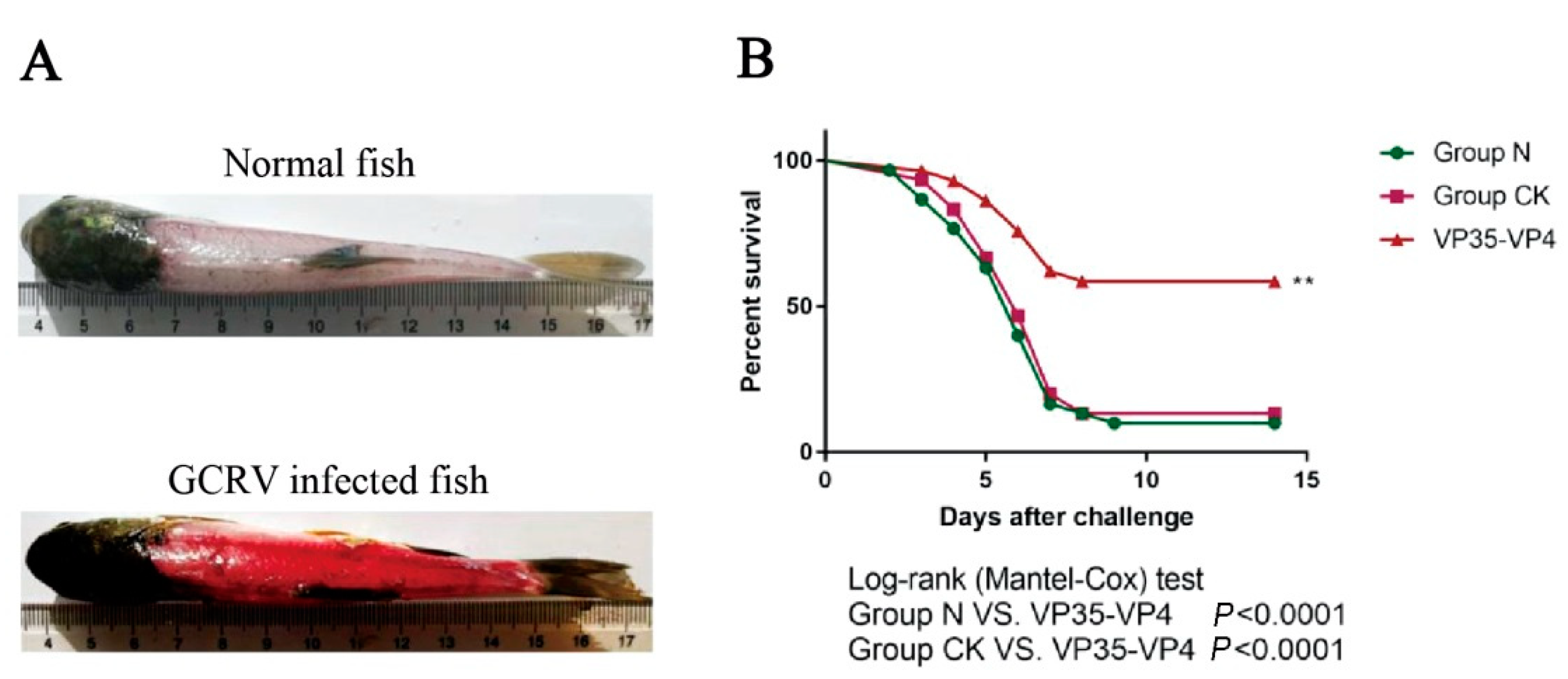
3.7. GCRV Challenge Test
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Rao, Y.; Su, J. Insights into the antiviral immunity against grass carp (Ctenopharyngodon idella) reovirus (GCRV) in grass carp. J. Immunol. Res. 2015, 2015, 670437. [Google Scholar] [CrossRef] [PubMed]
- Hao, K.; Chen, X.; Qi, X.; Yu, X.; Du, E.; Ling, F.; Zhu, B.; Wang, G. Protective immunity of grass carp induced by DNA vaccine encoding capsid protein gene (vp7) of grass carp reovirus using bacterial ghost as delivery vehicles. Fish. Shellfish Immunol. 2017, 64, 414–425. [Google Scholar] [CrossRef] [PubMed]
- Tian, Y.; Ye, X.; Zhang, L.; Deng, G.; Bai, Y. Development of a novel candidate subunit vaccine against Grass carp reovirus Guangdong strain (GCRV-GD108). Fish. Shellfish Immunol. 2013, 35, 351–356. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Jiang, Y. Morphological and physico-chemical characterization of the hemorrhagic virus of grass carp. Sci. Bull. 1984, 6, 832–835. [Google Scholar]
- Wang, Q.; Zeng, W.; Liu, C.; Zhang, C.; Wang, Y.; Shi, C.; Wu, S. Complete genome sequence of a reovirus isolated from grass carp, indicating different genotypes of GCRV in China. J. Virol. 2012, 86, 12466. [Google Scholar] [CrossRef] [PubMed]
- He, L.; Zhang, A.; Pei, Y.; Chu, P.; Li, Y.; Huang, R.; Liao, L.; Zhu, Z.; Wang, Y. Differences in responses of grass carp to different types of grass carp reovirus (GCRV) and the mechanism of hemorrhage revealed by transcriptome sequencing. BMC Genomics. 2017, 18, 452. [Google Scholar] [CrossRef]
- Gao, Y.; Pei, C.; Sun, X.; Zhang, C.; Li, L.; Kong, X. Plasmid pcDNA3.1-s11 constructed based on the S11 segment of grass carp reovirus as DNA vaccine provides immune protection. Vaccine. 2018, 36, 3613–3621. [Google Scholar] [CrossRef]
- Liu, H.; Chiou, P.P.; Gong, H.Y.; Chou, H.Y. Cloning of the major capsid protein (MCP) of grouper iridovirus of Taiwan (TGIV) and preliminary evaluation of a recombinant MCP vaccine against TGIV. Int. J. Mol. Sci. 2015, 16, 28647–28656. [Google Scholar] [CrossRef]
- Pei, C.; Gao, Y.; Sun, X.; Li, L.; Kong, X. A developed subunit vaccine based on fiber protein VP56 of grass carp reovirus providing immune protection against grass carp hemorrhagic disease. Fish. Shellfish Immunol. 2019, 90, 12–19. [Google Scholar] [CrossRef]
- Xue, R.; Liu, L.; Cao, G.; Xu, S.; Li, J.; Zou, Y.; Chen, H.; Gong, C. Oral vaccination of BacFish-vp6 against grass carp reovirus evoking antibody response in grass carp. Fish. Shellfish Immunol. 2013, 34, 348–355. [Google Scholar] [CrossRef]
- Yan, X.; Wang, Y.; Xiong, L.; Jian, J.; Wu, Z. Phylogenetic analysis of newly isolated grass carp reovirus. SpringerPlus 2014, 3, 190. [Google Scholar] [CrossRef] [PubMed]
- Su, H.; Liao, Z.; Yuan, G.; Su, J. A plasmid containing CpG ODN as vaccine adjuvant against grass carp reovirus in grass carp Ctenopharyngodon idella. Oncotarget 2017, 8, 86576–86591. [Google Scholar] [CrossRef] [PubMed]
- Seng, E.K.; Fang, Q.; Sin, Y.M.; Lam, T.J. Molecular characterization of a major outer capsid protein encoded by the Threadfin aquareovirus (TFV) gene segment 10 (S10). Arch. Virol. 2005, 150, 2021–2036. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Pei, C.; Gao, X.; Chen, Z.; Zhang, Q. Fish reovirus GCReV-109 VP33 protein elicits protective immunity in rare minnows. Arch. Virol. 2016, 161, 573–582. [Google Scholar] [CrossRef]
- Gao, H.; Wang, Y.; Li, N.; Peng, W.; Sun, Y.; Tong, G.; Qiu, H. Efficient gene delivery into mammalian cells mediated by a recombinant baculovirus containing a whispovirus ie1 promoter, a novel shuttle promoter between insect cells and mammalian cells. J. Biotechnol. 2007, 131, 138–143. [Google Scholar] [CrossRef]
- Chen, C.; Lin, C.; Chen, G.; Hu, Y. Baculovirus as a gene delivery vector: Recent understandings of molecular alterations in transduced cells and latest applications. Biotechnol. Adv. 2011, 29, 618–631. [Google Scholar] [CrossRef]
- Kato, T.; Sugioka, S.; Itagaki, K.; Park, E.Y. Gene transduction in mammalian cells using Bombyx mori nucleopolyhedrovirus assisted by glycoprotein 64 of Autographa californica multiple nucleopolyhedrovirus. Sci. Rep. UK 2016, 6, 32283. [Google Scholar] [CrossRef]
- Galindo-Villegas, J.; Mulero, I.; García-Alcazar, A.; Muñoz, I.; Peñalver-Mellado, M.; Streitenberger, S.; Meseguer, J.; Mulero, V. Recombinant TNFα as oral vaccine adjuvant protects European sea bass against vibriosis: Insights into the role of the CCL25/CCR9 axis. Fish. Shellfish Immunol. 2013, 35, 1260–1271. [Google Scholar] [CrossRef]
- Mu, C.; Vakharia, V.N.; Zhou, Y.; Jiang, N.; Liu, W.; Meng, Y.; Li, Y.; Xue, M.; Zhang, J.; Zeng, L.; et al. Novel Subunit Vaccine Based on Outer Capsid Proteins of Grass Carp Reovirus (GCRV) Provides Protective Immunity against GCRV Infection in Rare Minnow (Gobiocypris rarus). Pathogens 2020, 9, E945. [Google Scholar] [CrossRef]
- Blaxhall, P.C.; Daisley, K.W. Routine haematological methods for use with fish blood. J. Fish. Biol. 1973, 5, 771–781. [Google Scholar] [CrossRef]
- Zhang, L.; Ma, J.; Fan, Y.; Zhou, Y.; Xu, J.; Liu, W.; Gu, Z.; Zeng, L. Immune response and protection in gibel carp, Carassius gibelio, after vaccination with β-propiolactone inactivated cyprinid herpesvirus 2. Fish. Shellfish Immunol. 2016, 49, 344–350. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Xu, J.; Ma, J.; LaPatra, S.E.; Meng, Y.; Fan, Y.; Zhou, Y.; Yang, X.; Zeng, L. Immunological responses and protection in Chinese giant salamander Andrias davidianus immunized with inactivated iridovirus. Vet. Microbiol. 2014, 174, 382–390. [Google Scholar] [CrossRef] [PubMed]
- Schmittgen, T.D.; Livak, K.J. Analyzing real-time PCR data by the comparative C(T) method. Nat. Protoc. 2008, 3, 1101. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.; Pei, C.; Sun, X.; Zhang, C.; Li, L.; Kong, X. Novel subunit vaccine based on grass carp reovirus VP35 protein provides protective immunity against grass carp hemorrhagic disease. Fish. Shellfish Immunol. 2018, 75, 91–98. [Google Scholar] [CrossRef]
- Oers, M.M.; Pijlman, G.P.; Vlak, J.M. Thirty years of baculovirus–insect cell protein expression: From dark horse to mainstream technology. J. Gen. Virol. 2015, 96, 6–23. [Google Scholar] [CrossRef]
- Kost, T.A.; Kemp, C.W. Fundamentals of Baculovirus Expression and Applications. Springerplus 2016, 896, 187–197. [Google Scholar]
- Liu, B.; Gong, Y.; Li, Z.; Hu, X.; Cao, G.; Xue, R.; Gong, C. Baculovirus-mediated GCRV vp7 and vp6 genes expression in silkworm and grass carp. Mol. Biol. Rep. 2016, 43, 509–515. [Google Scholar] [CrossRef]
- Chen, D.; Yao, Y.; Cui, Z.; Zhang, X.; Peng, K.; Guo, X.; Wang, B.; Zhou, Y.; Li, S.; Wu, N.; et al. Comparative study of the immunoprotective effect of two DNA vaccines against grass carp reovirus. Fish. Shellfish Immunol. 2018, 75, 66–73. [Google Scholar] [CrossRef]
- Mir, F.A.; Kaufmann, S.H.; Eddine, A.N. A multicistronic DNA vaccine induces significant protection against tuberculosis in mice and offers flexibility in the expressed antigen repertoire. Clin. Vaccine Immunol. 2009, 16, 1467–1475. [Google Scholar] [CrossRef]
- Szymczak-Workman, A.L.; Vignali, K.M.; Vignali, D.A. Generation of 2A-linked multicistronic cassettes by recombinant PCR. Cold Spring Harbor. Protocols. 2012, 2012, 251–254. [Google Scholar] [CrossRef]
- Zhang, Q.; Xu, B.; Pan, J.; Liu, D.; Lv, R.; Yan, D. Expression and active testing of VP7 from GCRV (Grass carp reovirus) fused with cholera toxin B subunit in rice calli. Protein Expres Purif. 2019, 158, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Uribe, C.; Folch, H.; Enríquez, R.; Moran, G. Innate and adaptive immunity in teleost fish: A review. Vet. Med. Czech. 2011, 56, 486–503. [Google Scholar] [CrossRef]
- Gomez, D.; Sunyer, J.O.; Salinas, I. The mucosal immune system of fish: The evolution of tolerating commensals while fighting pathogens. Fish. Shellfish Immunol. 2013, 35, 1729–1739. [Google Scholar] [CrossRef] [PubMed]
- Siriyappagouder, P.; Galindo-Villegas, J.; Lokesh, J.; Mulero, V.; Fernandes, J.M.O.; Kiron, V. Exposure to Yeast Shapes the Intestinal Bacterial Community Assembly in Zebrafish Larvae. Front. Microbiol. 2018, 9, 1868. [Google Scholar] [CrossRef]
- Hao, K.; Chen, X.; Qi, X.; Zhu, B.; Wang, G.; Ling, F. Display of GCRV vp7 protein on the surface of Escherichia coli and its immunoprotective effects in grass carp (Ctenopharyngodon idella). Fish. Shellfish Immunol. 2018, 72, 199–209. [Google Scholar] [CrossRef]
- Lorenzen, N.; Lapatra, S.E. Immunity to rhabdoviruses in rainbow trout: The antibody response. Fish. Shellfish Immunol. 1999, 9, 345–360. [Google Scholar] [CrossRef]
- Yan, L.; Guo, H.; Sun, X.; Shao, L.; Fang, Q. Characterization of grass carp reovirus minor core protein VP4. Virol. J. 2012, 9, 89. [Google Scholar] [CrossRef]
- Jiang, H.; Bian, Q.; Zeng, W.; Ren, P.; Sun, H.; Lin, H.; Tang, Z.; Zhou, X.; Wang, Q.; Wang, Y. Oral delivery of Bacillus subtilis spores expressing grass carp reovirus VP4 protein produces protection against grass carp reovirus infection. Fish. Shellfish Immunol. 2019, 84, 768–780. [Google Scholar] [CrossRef]


| Gene | Primers (5′–3′) | Annealing Temperature (°C) | Product Size (bp) | Accession No. |
|---|---|---|---|---|
| TLR22 | F:CCATCCATTTAACAGGTGCATAC R:CAGCAGATGTGGAAAGAGACC | 58 | 174 | JQ670915.1 |
| IL-1 β | F:TGTGACGCTGAGAGACGGAAA R:GAGTTTCAGTGACCTCCTTCAA | 60 | 190 | JX014320 |
| IgM | F:GAGGCATCGGAGGCACATTTC R:TTGGGTCTCGCACCATTTTCTC | 55 | 166 | DQ417927 |
| Mx1 | F:CTGGGGAGGAAGTAAAGTGTTCT R:CAGCATGGATTCTGCCTGG | 57 | 391 | HQ245104 |
| IFN-I | F:AAGCAACGAGTCTTTGAGCCT R:GCGTCCTGGAAATGACACCT | 58 | 78 | DQ357216 |
| MHC I | F:CCTGGCAGAAAAATGGACAAG R:CCAACAACACCAATGACAATC | 56 | 271 | AY391782 |
| β-actin | F:GATGATGAAATTGCCGCACTG R:TGGTCAGCCCGAAACTATC | 58 | 151 | M25013 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mu, C.; Zhong, Q.; Meng, Y.; Zhou, Y.; Jiang, N.; Liu, W.; Li, Y.; Xue, M.; Zeng, L.; Vakharia, V.N.; et al. Oral Vaccination of Grass Carp (Ctenopharyngodon idella) with Baculovirus-Expressed Grass Carp Reovirus (GCRV) Proteins Induces Protective Immunity against GCRV Infection. Vaccines 2021, 9, 41. https://doi.org/10.3390/vaccines9010041
Mu C, Zhong Q, Meng Y, Zhou Y, Jiang N, Liu W, Li Y, Xue M, Zeng L, Vakharia VN, et al. Oral Vaccination of Grass Carp (Ctenopharyngodon idella) with Baculovirus-Expressed Grass Carp Reovirus (GCRV) Proteins Induces Protective Immunity against GCRV Infection. Vaccines. 2021; 9(1):41. https://doi.org/10.3390/vaccines9010041
Chicago/Turabian StyleMu, Changyong, Qiwang Zhong, Yan Meng, Yong Zhou, Nan Jiang, Wenzhi Liu, Yiqun Li, Mingyang Xue, Lingbing Zeng, Vikram N. Vakharia, and et al. 2021. "Oral Vaccination of Grass Carp (Ctenopharyngodon idella) with Baculovirus-Expressed Grass Carp Reovirus (GCRV) Proteins Induces Protective Immunity against GCRV Infection" Vaccines 9, no. 1: 41. https://doi.org/10.3390/vaccines9010041
APA StyleMu, C., Zhong, Q., Meng, Y., Zhou, Y., Jiang, N., Liu, W., Li, Y., Xue, M., Zeng, L., Vakharia, V. N., & Fan, Y. (2021). Oral Vaccination of Grass Carp (Ctenopharyngodon idella) with Baculovirus-Expressed Grass Carp Reovirus (GCRV) Proteins Induces Protective Immunity against GCRV Infection. Vaccines, 9(1), 41. https://doi.org/10.3390/vaccines9010041






