Potential of Anti-MUC1 Antibodies as a Targeted Therapy for Gastrointestinal Cancers
Abstract
1. Global Burden of GI Cancers
2. MUC1 as a Target Antigen in GI Cancers
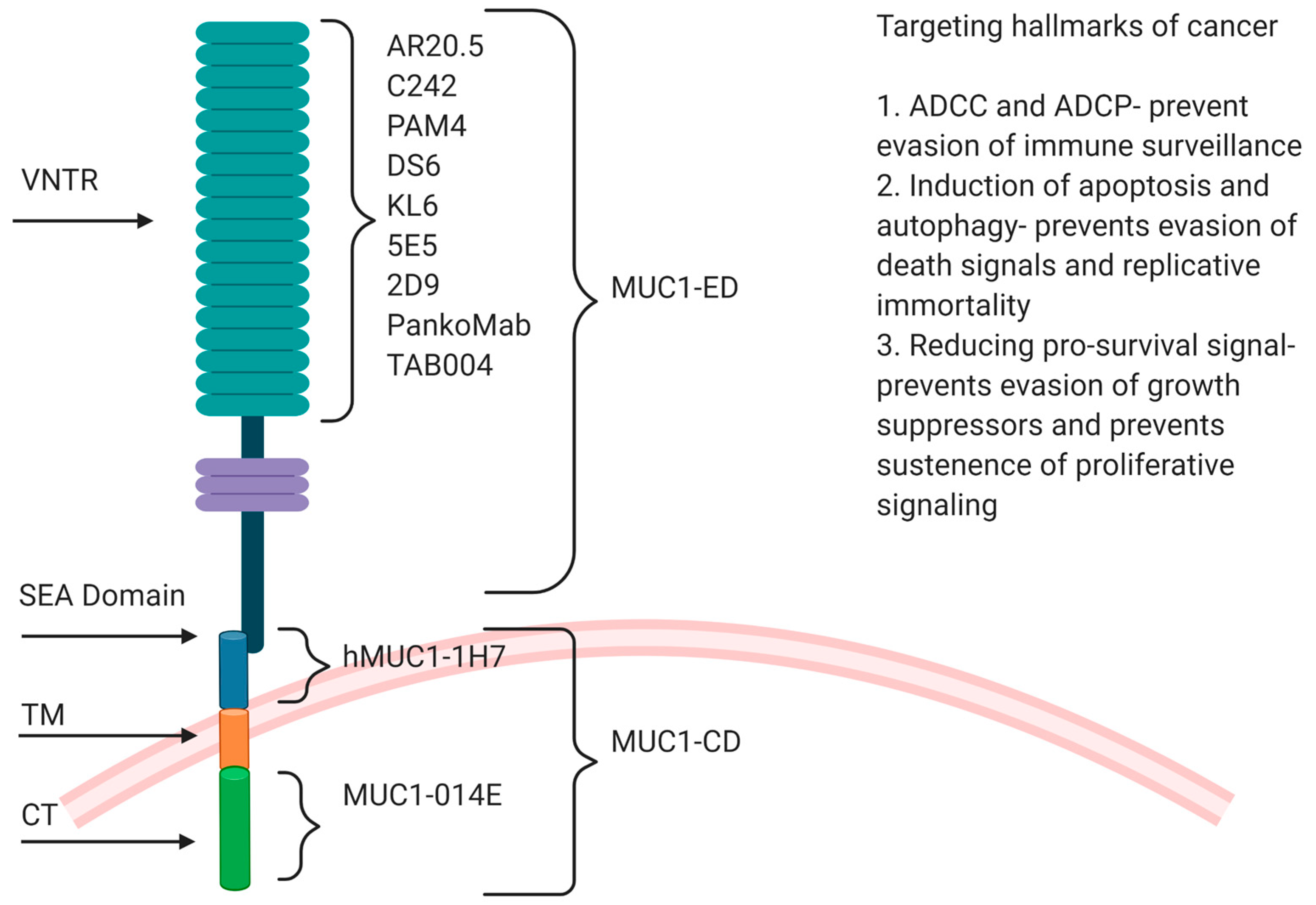
2.1. Structure of MUC1
2.2. Role in GI Tumors
3. Anti-MUC1 Antibodies in Preclinical and Clinical Trials
3.1. Monoclonal Antibodies
3.1.1. Antibodies Recognizing Non-Glycopeptide Epitope
3.1.2. Antibodies Recognizing Glycopeptide Epitopes
3.2. Bispecific Antibodies for MUC1
3.3. CAR-T Cells Targeting MUC1
4. Molecular Interactions between MUC1 and Its Antibodies
5. Concluding Remarks and Future Perspectives
Author Contributions
Funding
Conflicts of Interest
References
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA A Cancer J. Clin. 2018, 68, 394–424. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.; Wu, S.; Huang, Y.; Shao, Y.; Chen, X.Y.; Xian, L.; Zheng, J.; Wen, Y.; Chen, X.; Li, H. Screening for oesophageal cancer. Cochrane Database Syst. Rev. 2012, CD007883. [Google Scholar] [CrossRef] [PubMed]
- Bjelakovic, G.; Nikolova, D.; Simonetti, R.G.; Gluud, C. Antioxidant supplements for preventing gastrointestinal cancers. Cochrane Database Syst. Rev. 2008. [Google Scholar] [CrossRef] [PubMed]
- Linden, S.; Sutton, P.; Karlsson, N.; Korolik, V.; McGuckin, M. Mucins in the mucosal barrier to infection. Mucosal Immunol. 2008, 1, 183–197. [Google Scholar] [CrossRef]
- Ostedgaard, L.S.; Moninger, T.O.; McMenimen, J.D.; Sawin, N.M.; Parker, C.P.; Thornell, I.M.; Powers, L.S.; Gansemer, N.D.; Bouzek, D.C.; Cook, D.P. Gel-forming mucins form distinct morphologic structures in airways. Proc. Natl. Acad. Sci. USA 2017, 114, 6842–6847. [Google Scholar] [CrossRef]
- Rachagani, S.; Torres, M.P.; Moniaux, N.; Batra, S.K. Current status of mucins in the diagnosis and therapy of cancer. Biofactors 2009, 35, 509–527. [Google Scholar] [CrossRef] [PubMed]
- Gendler, S.; Burchell, J.; Duhig, T.; Lamport, D.; White, R.; Parker, M.; Taylor-Papadimitriou, J. Cloning of partial cDNA encoding differentiation and tumor-associated mucin glycoproteins expressed by human mammary epithelium. Proc. Natl. Acad. Sci. USA 1987, 84, 6060–6064. [Google Scholar] [CrossRef]
- Lan, M.S.; Batra, S.K.; Qi, W.-N.; Metzgar, R.S.; Hollingsworth, M.A. Cloning and sequencing of a human pancreatic tumor mucin cDNA. J. Biol. Chem. 1990, 265, 15294–15299. [Google Scholar]
- Ligtenberg, M.; Vos, H.; Gennissen, A.; Hilkens, J. Episialin, a carcinoma-associated mucin, is generated by a polymorphic gene encoding splice variants with alternative amino termini. J. Biol. Chem. 1990, 265, 5573–5578. [Google Scholar]
- Siddiqui, J.; Abe, M.; Hayes, D.; Shani, E.; Yunis, E.; Kufe, D. Isolation and sequencing of a cDNA coding for the human DF3 breast carcinoma-associated antigen. Proc. Natl. Acad. Sci. USA 1988, 85, 2320–2323. [Google Scholar] [CrossRef] [PubMed]
- Hattrup, C.L.; Gendler, S.J. Structure and function of the cell surface (tethered) mucins. Annu. Rev. Physiol. 2008, 70, 431–457. [Google Scholar] [CrossRef]
- Gendler, S.J.; Spicer, A. Epithelial mucin genes. Annu. Rev. Physiol. 1995, 57, 607–634. [Google Scholar] [CrossRef]
- Gendler, S.J. MUC1, the renaissance molecule. J. Mammary Gland Biol. Neoplasia 2001, 6, 339–353. [Google Scholar] [CrossRef]
- Chang, J.-F.; Zhao, H.-L.; Phillips, J.; Greenburg, G. The epithelial mucin, MUC1, is expressed on resting T lymphocytes and can function as a negative regulator of T cell activation. Cell. Immunol. 2000, 201, 83–88. [Google Scholar] [CrossRef]
- Yolken, R.H.; Peterson, J.A.; Vonderfecht, S.L.; Fouts, E.T.; Midthun, K.; Newburg, D.S. Human milk mucin inhibits rotavirus replication and prevents experimental gastroenteritis. J. Clin. Investig. 1992, 90, 1984–1991. [Google Scholar] [CrossRef]
- Schroten, H.; Hanisch, F.; Plogmann, R.; Hacker, J.; Uhlenbruck, G.; Nobis-Bosch, R.; Wahn, V. Inhibition of adhesion of S-fimbriated Escherichia coli to buccal epithelial cells by human milk fat globule membrane components: A novel aspect of the protective function of mucins in the nonimmunoglobulin fraction. Infect. Immun. 1992, 60, 2893–2899. [Google Scholar] [CrossRef] [PubMed]
- Kufe, D.W. Mucins in cancer: Function, prognosis and therapy. Nat. Rev. Cancer 2009, 9, 874–885. [Google Scholar] [CrossRef] [PubMed]
- Baruch, A.; Hartmann, M.-L.; Yoeli, M.; Adereth, Y.; Greenstein, S.; Stadler, Y.; Skornik, Y.; Zaretsky, J.; Smorodinsky, N.I.; Keydar, I. The breast cancer-associated MUC1 gene generates both a receptor and its cognate binding protein. Cancer Res. 1999, 59, 1552–1561. [Google Scholar]
- Stasyk, T.; Huber, L.A. Spatio-temporal parameters of endosomal signaling in cancer: Implications for new treatment options. J. Cell. Biochem. 2016, 117, 836–843. [Google Scholar] [CrossRef]
- Levitin, F.; Stern, O.; Weiss, M.; Gil-Henn, C.; Ziv, R.; Prokocimer, Z.; Smorodinsky, N.I.; Rubinstein, D.B.; Wreschner, D.H. The MUC1 SEA module is a self-cleaving domain. J. Biol. Chem. 2005, 280, 33374–33386. [Google Scholar] [CrossRef]
- Parry, S.; Silverman, H.S.; McDermott, K.; Willis, A.; Hollingsworth, M.A.; Harris, A. Identification of MUC1 proteolytic cleavage sites in vivo. Biochem. Biophys. Res. Commun. 2001, 283, 715–720. [Google Scholar] [CrossRef]
- Kufe, D.W. MUC1-C oncoprotein as a target in breast cancer: Activation of signaling pathways and therapeutic approaches. Oncogene 2013, 32, 1073–1081. [Google Scholar]
- Bennett, E.P.; Mandel, U.; Clausen, H.; Gerken, T.A.; Fritz, T.A.; Tabak, L.A. Control of mucin-type O-glycosylation: A classification of the polypeptide GalNAc-transferase gene family. Glycobiology 2012, 22, 736–756. [Google Scholar] [CrossRef]
- Movahedin, M.; Brooks, T.M.; Supekar, N.T.; Gokanapudi, N.; Boons, G.-J.; Brooks, C.L. Glycosylation of MUC1 influences the binding of a therapeutic antibody by altering the conformational equilibrium of the antigen. Glycobiology 2017, 27, 677–687. [Google Scholar] [CrossRef] [PubMed]
- Piyush, T.; Rhodes, J.M.; Yu, L.-G. MUC1 O-glycosylation contributes to anoikis resistance in epithelial cancer cells. Cell Death Discov. 2017, 3, 1–9. [Google Scholar] [CrossRef]
- Parry, S.; Hanisch, F.G.; Leir, S.-H.; Sutton-Smith, M.; Morris, H.R.; Dell, A.; Harris, A. N-Glycosylation of the MUC1 mucin in epithelial cells and secretions. Glycobiology 2006, 16, 623–634. [Google Scholar] [CrossRef]
- Tian, E.; Ten Hagen, K.G. Recent insights into the biological roles of mucin-type O-glycosylation. Glycoconj. J. 2009, 26, 325–334. [Google Scholar] [CrossRef]
- Ju, T.; Wang, Y.; Aryal, R.P.; Lehoux, S.D.; Ding, X.; Kudelka, M.R.; Cutler, C.; Zeng, J.; Wang, J.; Sun, X. T n and sialyl-Tn antigens, aberrant O-glycomics as human disease markers. Proteom. Clin. Appl. 2013, 7, 618–631. [Google Scholar] [CrossRef]
- Clausen, H.; Bennett, E.P. A family of UDP-GalNAc: Polypeptide N-acetylgalactosaminyl-transferases control the initiation of mucin-type O-linked glycosylation. Glycobiology 1996, 6, 635–646. [Google Scholar] [CrossRef]
- Breloy, I.; Hanisch, F.-G. Functional Roles of O-Glycosylation. Molecules 2018, 3063. [Google Scholar] [CrossRef]
- Brockhausen, I. Mucin-type O-glycans in human colon and breast cancer: Glycodynamics and functions. EMBO Rep. 2006, 7, 599–604. [Google Scholar] [CrossRef] [PubMed]
- Hanson, R.L.; Hollingsworth, M.A. Functional consequences of differential O-glycosylation of MUC1, MUC4, and MUC16 (downstream effects on signaling). Biomolecules 2016, 6, 34. [Google Scholar] [CrossRef]
- Chandrasekaran, E.; Xue, J.; Xia, J.; Locke, R.D.; Patil, S.A.; Neelamegham, S.; Matta, K.L. Characterization of cancer associated mucin type O-glycans using the exchange sialylation properties of mammalian sialyltransferase ST3Gal-II. J. Proteome Res. 2012, 11, 2609–2618. [Google Scholar] [CrossRef]
- Saeland, E.; Belo, A.I.; Mongera, S.; van Die, I.; Meijer, G.A.; van Kooyk, Y. Differential glycosylation of MUC1 and CEACAM5 between normal mucosa and tumour tissue of colon cancer patients. Int. J. Cancer 2012, 131, 117–128. [Google Scholar] [CrossRef]
- Horm, T.M.; Schroeder, J.A. MUC1 and metastatic cancer: Expression, function and therapeutic targeting. Cell Adhes. Migr. 2013, 7, 187–198. [Google Scholar] [CrossRef]
- Ghosh, S.K.; Pantazopoulos, P.; Medarova, Z.; Moore, A. Expression of underglycosylated MUC1 antigen in cancerous and adjacent normal breast tissues. Clin. Breast Cancer 2013, 13, 109–118. [Google Scholar] [CrossRef] [PubMed]
- Taylor-Papadimitriou, J.; Burchell, J.M.; Graham, R.; Beatson, R. Latest developments in MUC1 immunotherapy. Biochem. Soc. Trans. 2018, 46, 659–668. [Google Scholar] [CrossRef]
- Posey, A.D., Jr.; Schwab, R.D.; Boesteanu, A.C.; Steentoft, C.; Mandel, U.; Engels, B.; Stone, J.D.; Madsen, T.D.; Schreiber, K.; Haines, K.M. Engineered CAR T cells targeting the cancer-associated Tn-glycoform of the membrane mucin MUC1 control adenocarcinoma. Immunity 2016, 44, 1444–1454. [Google Scholar] [CrossRef]
- Pourjafar, M.; Samadi, P.; Khoshinani, H.M.; Saidijam, M. Are mimotope vaccines a good alternative to monoclonal antibodies? Immunotherapy 2019, 11, 795–800. [Google Scholar] [CrossRef]
- Lau, S.K.; Weiss, L.M.; Chu, P.G. Differential expression of MUC1, MUC2, and MUC5AC in carcinomas of various sites: An immunohistochemical study. Am. J. Clin. Pathol. 2004, 122, 61–69. [Google Scholar] [CrossRef]
- Nath, S.; Mukherjee, P. MUC1: A multifaceted oncoprotein with a key role in cancer progression. Trends Mol. Med. 2014, 20, 332–342. [Google Scholar] [CrossRef]
- Bose, M.; Mukherjee, P. Microbe–MUC1 Crosstalk in Cancer-Associated Infections. Trends Mol. Med. 2020, 26, 324–336. [Google Scholar] [CrossRef] [PubMed]
- Cheever, M.A.; Allison, J.P.; Ferris, A.S.; Finn, O.J.; Hastings, B.M.; Hecht, T.T.; Mellman, I.; Prindiville, S.A.; Viner, J.L.; Weiner, L.M. The prioritization of cancer antigens: A national cancer institute pilot project for the acceleration of translational research. Clin. Cancer Res. 2009, 15, 5323–5337. [Google Scholar] [CrossRef]
- Zeng, Y.; Zhang, Q.; Zhang, Y.; Lu, M.; Liu, Y.; Zheng, T.; Feng, S.; Hao, M.; Shi, H. MUC1 predicts colorectal cancer metastasis: A systematic review and meta-analysis of case controlled studies. PLoS ONE 2015, 10, e0138049. [Google Scholar] [CrossRef]
- Agata, N.; Ahmad, R.; Kawano, T.; Raina, D.; Kharbanda, S.; Kufe, D. MUC1 oncoprotein blocks death receptor–mediated apoptosis by inhibiting recruitment of caspase-8. Cancer Res. 2008, 68, 6136–6144. [Google Scholar] [CrossRef]
- Yin, L.; Kosugi, M.; Kufe, D. Inhibition of the MUC1-C oncoprotein induces multiple myeloma cell death by down-regulating TIGAR expression and depleting NADPH. Blood J. Am. Soc. Hematol. 2012, 119, 810–816. [Google Scholar] [CrossRef]
- Pochampalli, M.; El Bejjani, R.; Schroeder, J. MUC1 is a novel regulator of ErbB1 receptor trafficking. Oncogene 2007, 26, 1693–1701. [Google Scholar] [CrossRef]
- Liu, X.; Caffrey, T.; Steele, M.; Mohr, A.; Singh, P.; Radhakrishnan, P.; Kelly, D.L.; Wen, Y.; Hollingsworth, M.A. MUC1 regulates cyclin D1 gene expression through p120 catenin and β-catenin. Oncogenesis 2014, 3, e107. [Google Scholar] [CrossRef]
- Wei, X.; Xu, H.; Kufe, D. Human mucin 1 oncoprotein represses transcription of the p53 tumor suppressor gene. Cancer Res. 2007, 67, 1853–1858. [Google Scholar] [CrossRef]
- Yin, L.; Huang, L.; Kufe, D. MUC1 oncoprotein activates the FOXO3a transcription factor in a survival response to oxidative stress. J. Biol. Chem. 2004, 279, 45721–45727. [Google Scholar] [CrossRef]
- Ren, J.; Bharti, A.; Raina, D.; Chen, W.; Ahmad, R.; Kufe, D. MUC1 oncoprotein is targeted to mitochondria by heregulin-induced activation of c-Src and the molecular chaperone HSP90. Oncogene 2006, 25, 20–31. [Google Scholar] [CrossRef] [PubMed]
- Raina, D.; Kharbanda, S.; Kufe, D. The MUC1 oncoprotein activates the anti-apoptotic phosphoinositide 3-kinase/Akt and Bcl-xL pathways in rat 3Y1 fibroblasts. J. Biol. Chem. 2004, 279, 20607–20612. [Google Scholar] [CrossRef]
- Satoh, S.; Hinoda, Y.; Hayashi, T.; Burdick, M.D.; Imai, K.; Hollingsworth, M.A. Enhancement of metastatic properties of pancreatic cancer cells by MUC1 gene encoding an anti-adhesion molecule. Int. J. Cancer 2000, 88, 507–518. [Google Scholar] [CrossRef]
- Regimbald, L.H.; Pilarski, L.M.; Longenecker, B.M.; Reddish, M.A.; Zimmermann, G.; Hugh, J.C. The breast mucin MUC1 as a novel adhesion ligand for endothelial intercellular adhesion molecule 1 in breast cancer. Cancer Res. 1996, 56, 4244–4249. [Google Scholar]
- Nath, S.; Daneshvar, K.; Roy, L.; Grover, P.; Kidiyoor, A.; Mosley, L.; Sahraei, M.; Mukherjee, P. MUC1 induces drug resistance in pancreatic cancer cells via upregulation of multidrug resistance genes. Oncogenesis 2013, 2, e51. [Google Scholar] [CrossRef]
- Singh, P.K.; Wen, Y.; Swanson, B.J.; Shanmugam, K.; Kazlauskas, A.; Cerny, R.L.; Gendler, S.J.; Hollingsworth, M.A. Platelet-derived growth factor receptor β-mediated phosphorylation of MUC1 enhances invasiveness in pancreatic adenocarcinoma cells. Cancer Res. 2007, 67, 5201–5210. [Google Scholar] [CrossRef]
- Roy, L.D.; Sahraei, M.; Subramani, D.B.; Besmer, D.; Nath, S.; Tinder, T.L.; Bajaj, E.; Shanmugam, K.; Lee, Y.Y.; Hwang, S.I. MUC1 enhances invasiveness of pancreatic cancer cells by inducing epithelial to mesenchymal transition. Oncogene 2011, 30, 1449–1459. [Google Scholar] [CrossRef] [PubMed]
- Sahraei, M.; Roy, L.D.; Curry, J.M.; Teresa, T.L.; Nath, S.; Besmer, D.; Kidiyoor, A.; Dalia, R.; Gendler, S.J.; Mukherjee, P. MUC1 regulates PDGFA expression during pancreatic cancer progression. Oncogene 2012, 31, 4935–4945. [Google Scholar] [CrossRef]
- Grover, P.; Nath, S.; Nye, M.D.; Zhou, R.; Ahmad, M.; Mukherjee, P. SMAD4-independent activation of TGF-β signaling by MUC1 in a human pancreatic cancer cell line. Oncotarget 2018, 9, 6897. [Google Scholar] [CrossRef] [PubMed]
- Grover, P.; Nath, S.; Bose, M.; Sanders, A.J.; Brouwer, C.; Fnu, N.; Zhou, R.; Yazdanifar, M.; Ahmed, M.; Wu, S.-T. Tumor-Associated MUC1 Regulates TGF-β Signaling and Function in Pancreatic Ductal Adenocarcinoma. bioRxiv 2020. [Google Scholar] [CrossRef]
- Wang, X.-T.; Kong, F.-B.; Mai, W.; Li, L.; Pang, L.-M. MUC1 immunohistochemical expression as a prognostic factor in gastric cancer: Meta-analysis. Dis. Markers 2016, 9421571. [Google Scholar] [CrossRef] [PubMed]
- Scott, A.M.; Allison, J.P.; Wolchok, J.D. Monoclonal antibodies in cancer therapy. Cancer Immun. Arch. 2012, 12, 14–22. [Google Scholar]
- Weiner, L.M.; Murray, J.C.; Shuptrine, C.W. Antibody-based immunotherapy of cancer. Cell 2012, 148, 1081–1084. [Google Scholar] [CrossRef]
- Redman, J.; Hill, E.; AlDeghaither, D.; Weiner, L. Mechanisms of action of therapeutic antibodies for cancer. Mol. Immunol. 2015, 67, 28–45. [Google Scholar] [CrossRef]
- Shuptrine, C.W.; Surana, R.; Weiner, L.M. Monoclonal antibodies for the treatment of cancer. In Seminars in Cancer Biology; Elsevier: Amsterdam, The Netherlands, 2012; pp. 3–13. [Google Scholar]
- Samadi, P.; Saki, S.; Dermani, F.K.; Pourjafar, M.; Saidijam, M. Emerging ways to treat breast cancer: Will promises be met? Cell. Oncol. 2018, 41, 605–621. [Google Scholar] [CrossRef]
- Doi, M.; Yokoyama, A.; Kondo, K.; Ohnishi, H.; Ishikawa, N.; Hattori, N.; Kohno, N. Anti-tumor effect of the anti-KL-6/MUC1 monoclonal antibody through exposure of surface molecules by MUC1 capping. Cancer Sci. 2006, 97, 420–429. [Google Scholar] [CrossRef]
- Ohyabu, N.; Hinou, H.; Matsushita, T.; Izumi, R.; Shimizu, H.; Kawamoto, K.; Numata, Y.; Togame, H.; Takemoto, H.; Kondo, H. An essential epitope of anti-MUC1 monoclonal antibody KL-6 revealed by focused glycopeptide library. J. Am. Chem. Soc. 2009, 131, 17102–17109. [Google Scholar] [CrossRef]
- Namba, M.; Hattori, N.; Hamada, H.; Yamaguchi, K.; Okamoto, Y.; Nakashima, T.; Masuda, T.; Sakamoto, S.; Horimasu, Y.; Miyamoto, S. Anti-KL-6/MUC1 monoclonal antibody reverses resistance to trastuzumab-mediated antibody-dependent cell-mediated cytotoxicity by capping MUC1. Cancer Lett. 2019, 442, 31–39. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, M.; Bhavanandan, V.; Nakamori, S.; Irimura, T. A novel monoclonal antibody specific for sialylated MUC1 mucin. Jpn. J. Cancer Res. 1996, 87, 488–496. [Google Scholar] [CrossRef]
- Suzuki, H.; Shoda, J.; Kawamoto, T.; Shinozaki, E.; Miyahara, N.; Hotta, S.; Iizuka, Y.; Nakahara, A.; Tanaka, N.; Yanaka, A. Expression of MUC1 recognized by monoclonal antibody MY. 1E12 is a useful biomarker for tumor aggressiveness of advanced colon carcinoma. Clin. Exp. Metastasis 2004, 21, 321–329. [Google Scholar] [CrossRef]
- Yoshimura, Y.; Denda-Nagai, K.; Takahashi, Y.; Nagashima, I.; Shimizu, H.; Kishimoto, T.; Noji, M.; Shichino, S.; Chiba, Y.; Irimura, T. Products of Chemoenzymatic Synthesis Representing MUC1 Tandem Repeat Unit with T-, ST-or STn-antigen Revealed Distinct Specificities of Anti-MUC1 Antibodies. Sci. Rep. 2019, 9, 1–12. [Google Scholar] [CrossRef]
- Muguruma, N.; Ito, S. Labeled anti-mucin antibody detectable by infrared-fluorescence endoscopy. Cancer Biomark. 2008, 4, 321–328. [Google Scholar] [CrossRef]
- Tarp, M.A.; Sørensen, A.L.; Mandel, U.; Paulsen, H.; Burchell, J.; Taylor-Papadimitriou, J.; Clausen, H. Identification of a novel cancer-specific immunodominant glycopeptide epitope in the MUC1 tandem repeat. Glycobiology 2007, 17, 197–209. [Google Scholar] [CrossRef] [PubMed]
- Wu, G.; Maharjan, S.; Kim, D.; Kim, J.N.; Park, B.K.; Koh, H.; Moon, K.; Lee, Y.; Kwon, H.-J. A novel monoclonal antibody targets Mucin1 and attenuates growth in pancreatic cancer model. Int. J. Mol. Sci. 2018, 19, 2004. [Google Scholar] [CrossRef]
- Wu, G.; Kim, D.; Kim, J.N.; Park, S.; Maharjan, S.; Koh, H.; Moon, K.; Lee, Y.; Kwon, H.-J. A Mucin1 C-terminal subunit-directed monoclonal antibody targets overexpressed Mucin1 in breast cancer. Theranostics 2018, 8, 78. [Google Scholar] [CrossRef]
- Tinder, T.L.; Subramani, D.B.; Basu, G.D.; Bradley, J.M.; Schettini, J.; Million, A.; Skaar, T.; Mukherjee, P. MUC1 enhances tumor progression and contributes toward immunosuppression in a mouse model of spontaneous pancreatic adenocarcinoma. J. Immunol. 2008, 181, 3116–3125. [Google Scholar] [CrossRef]
- Mukherjee, P.; Madsen, C.S.; Ginardi, A.R.; Tinder, T.L.; Jacobs, F.; Parker, J.; Agrawal, B.; Longenecker, B.M.; Gendler, S.J. Mucin 1-specific immunotherapy in a mouse model of spontaneous breast cancer. J. Immunother. 2003, 26, 47–62. [Google Scholar] [CrossRef]
- Dréau, D.; Moore, L.J.; Wu, M.; Roy, L.D.; Dillion, L.; Porter, T.; Puri, R.; Momin, N.; Wittrup, K.D.; Mukherjee, P. Combining the specific anti-MUC1 antibody TAB004 and Lip-MSA-IL-2 limits pancreatic cancer progression in immune competent murine models of pancreatic ductal adenocarcinoma. Front. Oncol. 2019, 9, 330. [Google Scholar] [CrossRef]
- Moore, L.J.; Roy, L.D.; Zhou, R.; Grover, P.; Wu, S.-T.; Curry, J.M.; Dillon, L.M.; Puri, P.M.; Yazdanifar, M.; Puri, R. Antibody-guided in vivo imaging for early detection of mammary gland tumors. Transl. Oncol. 2016, 9, 295–305. [Google Scholar] [CrossRef]
- Bose, M.; Mukherjee, P. A Novel Antibody Blocks Anti-Apoptotic Activity of MUC1 in Pancreatic Cancer Cell Lines. Cancer Res. 2019, 79 (Suppl. 13), 2052. [Google Scholar] [CrossRef]
- Yazdanifar, M.; Zhou, R.; Grover, P.; Williams, C.; Bose, M.; Moore, L.J.; Wu, S.-t.; Maher, J.; Dreau, D.; Mukherjee, P. Overcoming Immunological Resistance Enhances the Efficacy of a Novel Anti-tMUC1-CAR T Cell Treatment against Pancreatic Ductal Adenocarcinoma. Cells 2019, 8, 1070. [Google Scholar] [CrossRef]
- Calvete, J.A.; Newell, D.R.; Wright, A.F.; Rose, M.S. In vitro and in vivo antitumor activity of ZENECA ZD0490, a recombinant ricin A-chain immunotoxin for the treatment of colorectal cancer. Cancer Res. 1994, 54, 4684–4690. [Google Scholar]
- Baeckström, D.; Hansson, G.C.; Nilsson, O.; Johansson, C.; Gendler, S.J.; Lindholm, L. Purification and characterization of a membrane-bound and a secreted mucin-type glycoprotein carrying the carcinoma-associated sialyl-Lea epitope on distinct core proteins. J. Biol. Chem. 1991, 266, 21537–21547. [Google Scholar]
- Tolcher, A.W.; Ochoa, L.; Hammond, L.A.; Patnaik, A.; Edwards, T.; Takimoto, C.; Smith, L.; de Bono, J.; Schwartz, G.; Mays, T. Cantuzumab mertansine, a maytansinoid immunoconjugate directed to the CanAg antigen: A phase, I.; pharmacokinetic, and biologic correlative study. J. Clin. Oncol. 2003, 21, 211–222. [Google Scholar] [CrossRef]
- Rodon, J.; Garrison, M.; Hammond, L.A.; De Bono, J.; Smith, L.; Forero, L.; Hao, D.; Takimoto, C.; Lambert, J.M.; Pandite, L. Cantuzumab mertansine in a three-times a week schedule: A phase I and pharmacokinetic study. Cancer Chemother. Pharmacol. 2008, 62, 911–919. [Google Scholar] [CrossRef]
- Mita, M.; Ricart, A.; Mita, A.; Patnaik, A.; Sarantopoulos, J.; Sankhala, K.; Fram, R.; Qin, A.; Watermill, J.; Tolcher, A. A phase I study of a CanAg-targeted immunoconjugate, huC242-DM4, in patients with Can Ag-expressing solid tumors. J. Clin. Oncol. 2007, 25 (Suppl. 18), 3062. [Google Scholar] [CrossRef]
- Goff, L.; Papadopoulos, K.; Posey, J.; Phan, A.; Patnaik, A.; Miller, J.; Zildjian, S.; O’Leary, J.; Qin, A.; Tolcher, A. A phase II study of IMGN242 (huC242-DM4) in patients with CanAg-positive gastric or gastroesophageal (GE) junction cancer. J. Clin. Oncol. 2009, 27 (Suppl. 15), e15625. [Google Scholar] [CrossRef]
- Gold, D.V.; Cardillo, T.; Vardi, Y.; Blumenthal, R. Radioimmunotherapy of experimental pancreatic cancer with 131I-labeled monoclonal antibody PAM4. Int. J. Cancer 1997, 71, 660–667. [Google Scholar] [CrossRef]
- Cardillo, T.M.; Ying, Z.; Gold, D.V. Therapeutic advantage of 90yttrium-versus 131iodine-labeled PAM4 antibody in experimental pancreatic cancer. Clin. Cancer Res. 2001, 7, 3186–3192. [Google Scholar] [PubMed]
- Gold, D.V.; Karanjawala, Z.; Modrak, D.E.; Goldenberg, D.M.; Hruban, R.H. PAM4-reactive MUC1 is a biomarker for early pancreatic adenocarcinoma. Clin. Cancer Res. 2007, 13, 7380–7387. [Google Scholar] [CrossRef] [PubMed]
- Gulec, S.; Pennington, K.; Bruetman, D.; Garl, S.; Horne, H.; Gold, D.; Wegener, W.; Goldenberg, D. A phase-I study of 90Y-hPAM4 (humanized anti-MUC1 monoclonal antibody) in patients with unresectable and metastatic pancreatic cancer. J. Nucl. Med. 2007, 48 (Suppl. 2), 393p. [Google Scholar]
- Picozzi, V.J.; Ramanathan, R.K.; Lowery, M.A.; Ocean, A.J.; Mitchel, E.P.; O’Neil, B.H.; Guarino, M.J.; Conkling, P.R.; Cohen, S.J.; Bahary, N. 90Y-clivatuzumab tetraxetan with or without low-dose gemcitabine: A phase Ib study in patients with metastatic pancreatic cancer after two or more prior therapies. Eur. J. Cancer 2015, 51, 1857–1864. [Google Scholar] [CrossRef]
- Dian, D.; Janni, W.; Kuhn, C.; Mayr, D.; Karsten, U.; Mylonas, I.; Friese, K.; Jeschke, U. Evaluation of a novel anti-mucin 1 (MUC1) antibody (PankoMab) as a potential diagnostic tool in human ductal breast cancer; comparison with two established antibodies. Oncol. Res. Treat. 2009, 32, 238–244. [Google Scholar] [CrossRef]
- Danielczyk, A.; Stahn, R.; Faulstich, D.; Löffler, A.; Märten, A.; Karsten, U.; Goletz, S. PankoMab: A potent new generation anti-tumour MUC1 antibody. Cancer Immunol. Immunother. 2006, 55, 1337–1347. [Google Scholar] [CrossRef]
- Fiedler, W.; DeDosso, S.; Cresta, S.; Weidmann, J.; Tessari, A.; Salzberg, M.; Dietrich, B.; Baumeister, H.; Goletz, S.; Gianni, L. A phase I study of PankoMab-GEX, a humanised glyco-optimised monoclonal antibody to a novel tumour-specific MUC1 glycopeptide epitope in patients with advanced carcinomas. Eur. J. Cancer 2016, 63, 55–63. [Google Scholar] [CrossRef] [PubMed]
- Ledermann, J.; Sehouli, J.; Zurawski, B.; Raspagliesi, F.; De Giorgi, U.; Banerjee, S.; Arranz Arija, J.; Romeo Marin, M.; Lisyanskaya, A.; Póka, R. LBA41A double-blind, placebo-controlled, randomized, phase 2 study to evaluate the efficacy and safety of switch maintenance therapy with the anti-TA-MUC1 antibody PankoMab-GEX after chemotherapy in patients with recurrent epithelial ovarian carcinoma. Ann. Oncol. 2017, 28 (Suppl. 5), v605–v649. [Google Scholar] [CrossRef]
- Runcie, K.; Budman, D.R.; John, V.; Seetharamu, N. Bi-specific and tri-specific antibodies-the next big thing in solid tumor therapeutics. Mol. Med. 2018, 24, 50. [Google Scholar] [CrossRef]
- Mehla, K.; Tremayne, J.; Grunkemeyer, J.A.; O’Connell, K.A.; Steele, M.M.; Caffrey, T.C.; Zhu, X.; Yu, F.; Singh, P.K.; Schultes, B.C. Combination of mAb-AR20. 5, anti-PD-L1 and PolyICLC inhibits tumor progression and prolongs survival of MUC1. Tg mice challenged with pancreatic tumors. Cancer Immunol. Immunother. 2018, 67, 445–457. [Google Scholar] [CrossRef]
- De Bono, J.; Rha, S.Y.; Stephenson, J.; Schultes, B.; Monroe, P.; Eckhardt, G.; Hammond, L.; Whiteside, T.; Nicodemus, C.; Cermak, J. Phase I trial of a murine antibody to MUC1 in patients with metastatic cancer: Evidence for the activation of humoral and cellular antitumor immunity. Ann. Oncol. 2004, 15, 1825–1833. [Google Scholar] [CrossRef]
- Ibrahim, N.K.; Yariz, K.O.; Bondarenko, I.; Manikhas, A.; Semiglazov, V.; Alyasova, A.; Komisarenko, V.; Shparyk, Y.; Murray, J.L.; Jones, D. Randomized phase II trial of letrozole plus anti-MUC1 antibody AS1402 in hormone receptor–positive locally advanced or metastatic breast cancer. Clin. Cancer Res. 2011, 17, 6822–6830. [Google Scholar] [CrossRef]
- Corraliza-Gorjón, I.; Somovilla-Crespo, B.; Santamaria, S.; Garcia-Sanz, J.A.; Kremer, L. New strategies using antibody combinations to increase cancer treatment effectiveness. Front. Immunol. 2017, 8, 1804. [Google Scholar] [CrossRef]
- Venepalli, N.K.; Gandhi, C.C.; Ozer, H.; Ho, D.; Lu, Y.; Xie, H.; Berg, S.A.; Chowdhery, R.A.; Gargano, M.A.; Braun, A.H. Phase Ib study of PGG beta glucan in combination with anti-MUC1 antibody (BTH1704) and gemcitabine for the treatment of advanced pancreatic cancer. Am. Soc. Clin. Oncol. 2015, 33. [Google Scholar] [CrossRef]
- Gold, D.V.; Newsome, G.; Liu, D.; Goldenberg, D.M. Mapping PAM4 (clivatuzumab), a monoclonal antibody in clinical trials for early detection and therapy of pancreatic ductal adenocarcinoma, to MUC5AC mucin. Mol. Cancer 2013, 12, 143. [Google Scholar] [CrossRef]
- Gold, D.V.; Cardillo, T.; Goldenberg, D.M.; Sharkey, R.M. Localization of pancreatic cancer with radiolabeled monoclonal antibody PAM4. Crit. Rev. Oncol./Hematol. 2001, 39, 147–154. [Google Scholar] [CrossRef]
- Hisatsune, A.; Nakayama, H.; Kawasaki, M.; Horie, I.; Miyata, T.; Isohama, Y.; Kim, K.C.; Katsuki, H. Anti-MUC1 antibody inhibits EGF receptor signaling in cancer cells. Biochem. Biophys. Res. Commun. 2011, 405, 377–381. [Google Scholar] [CrossRef] [PubMed]
- Hollingsworth, M.A.; Swanson, B.J. Mucins in cancer: Protection and control of the cell surface. Nat. Rev. Cancer 2004, 4, 45–60. [Google Scholar] [CrossRef] [PubMed]
- Vivero-Escoto, J.L.; Jeffords, L.M.; Dréau, D.; Alvarez-Berrios, M.; Mukherjee, P. Mucin1 antibody-conjugated dye-doped mesoporous silica nanoparticles for breast cancer detection in vivo. In Colloidal Nanoparticles for Biomedical Applications XII: 2017; International Society for Optics and Photonics: San Francisco, CA, USA, 2017; p. 100780B. [Google Scholar]
- Curry, J.M.; Thompson, K.J.; Rao, S.G.; Besmer, D.M.; Murphy, A.M.; Grdzelishvili, V.Z.; Ahrens, W.A.; McKillop, I.H.; Sindram, D.; Iannitti, D.A. The use of a novel MUC1 antibody to identify cancer stem cells and circulating MUC1 in mice and patients with pancreatic cancer. J. Surg. Oncol. 2013, 107, 713–722. [Google Scholar] [CrossRef]
- Kelly, V.J.; Wu, S.-T.; Gottumukkala, V.; Coelho, R.; Palmer, K.; Nair, S.; Erick, T.; Puri, R.; Ilovich, O.; Mukherjee, P. Preclinical evaluation of an 111In/225Ac theranostic targeting transformed MUC1 for triple negative breast cancer. Theranostics 2020, 10, 6946. [Google Scholar] [CrossRef]
- Yonezawa, S.; Kitajima, S.; Higashi, M.; Osako, M.; Horinouchi, M.; Yokoyama, S.; Kitamoto, S.; Yamada, N.; Tamura, Y.; Shimizu, T. A novel anti-MUC1 antibody against the MUC1 cytoplasmic tail domain: Use in sensitive identification of poorly differentiated cells in adenocarcinoma of the stomach. Gastric Cancer 2012, 15, 370–381. [Google Scholar] [CrossRef][Green Version]
- Qi, W.; Schultes, B.C.; Liu, D.; Kuzma, M.; Decker, W.; Madiyalakan, R. Characterization of an anti-MUC1 monoclonal antibody with potential as a cancer vaccine. Hybrid. Hybridomics 2001, 20, 313–324. [Google Scholar] [CrossRef]
- Kearse, K.P.; Smith, N.L.; Semer, D.A.; Eagles, L.; Finley, J.L.; Kazmierczak, S.; Kovacs, C.J.; Rodriguez, A.A.; Kellogg-Wennerberg, A.E. Monoclonal antibody DS6 detects a tumor-associated sialoglycotope expressed on human serous ovarian carcinomas. Int. J. Cancer 2000, 88, 866–872. [Google Scholar] [CrossRef]
- Trombe, M.; Caron, A.; Tellier, A.; Carrez, C.; Guérif, S.; Clavier, S.; Karst, N.; Saarinen, J.; Satomaa, T.; Pitkänen, V. Preclinical Activity of an Antibody Drug Conjugate Targeting Tumor Specificmuc1 Structural Peptide-Glycotope. Cancer Res. 2019, 235. [Google Scholar] [CrossRef]
- Gomez-Roca, C.A.; Boni, V.; Moreno, V.; Morris, J.C.; Delord, J.-P.; Calvo, E.; Papadopoulos, K.P.; Rixe, O.; Cohen, P.; Tellier, A. A phase I study of SAR566658, an anti CA6-antibody drug conjugate (ADC), in patients (Pts) with CA6-positive advanced solid tumors (STs) (NCT01156870). Am. Soc. Clin. Oncol. 2016, 34, 2511. [Google Scholar] [CrossRef]
- St. Croix, B.; Sheehan, C.; Rak, J.W.; Flørenes, V.A.; Slingerland, J.M.; Kerbel, R.S. E-cadherin–dependent growth suppression is mediated by the cyclin-dependent kinase inhibitor p27KIP1. J. Cell Biol. 1998, 142, 557–571. [Google Scholar] [CrossRef] [PubMed]
- Matsumura, K.; Niki, I.; Tian, H.; Takuma, M.; Hongo, N.; Matsumoto, S.; Mori, H. Radioimmunoscintigraphy of pancreatic cancer in tumor-bearing athymic nude mice using 99 m technetium-labeled anti-KL-6/MUC1 antibody. Radiat. Med. 2008, 26, 133–139. [Google Scholar] [CrossRef]
- Li, Y.; Zhou, C.; Li, J.; Liu, J.; Lin, L.; Li, L.; Cao, D.; Li, Q.; Wang, Z. Single domain based bispecific antibody, Muc1-Bi-1, and its humanized form, Muc1-Bi-2, induce potent cancer cell killing in muc1 positive tumor cells. PLoS ONE 2018, 13, e0191024. [Google Scholar] [CrossRef]
- Kodama, H.; Suzuki, M.; Katayose, Y.; Shinoda, M.; Sakurai, N.; Takemura, S.-I.; Yoshida, H.; Saeki, H.; Asano, R.; Ichiyama, M. Specific and effective targeting cancer immunotherapy with a combination of three bispecific antibodies. Immunol. Lett. 2002, 81, 99–106. [Google Scholar] [CrossRef]
- Hinoda, Y.; Nakagawa, N.; Ohe, Y.; Kakiuchi, H.; Tsujisaki, M.; Imai, K.; Yachi, A. Recognition of the polypeptide core of mucin by monoclonal antibody MUSE11 against an adenocarcinoma-associated antigen. Jpn. J. Cancer Res. 1990, 81, 1206–1209. [Google Scholar] [CrossRef]
- Katayose, Y.; Kudo, T.; Suzuki, M.; Shinoda, M.; Saijyo, S.; Sakurai, N.; Saeki, H.; Fukuhara, K.; Imai, K.; Matsuno, S. MUC1-specific targeting immunotherapy with bispecific antibodies: Inhibition of xenografted human bile duct carcinoma growth. Cancer Res. 1996, 56, 4205–4212. [Google Scholar]
- Takemura, S.-I.; Asano, R.; Tsumoto, K.; Ebara, S.; Sakurai, N.; Katayose, Y.; Kodama, H.; Yoshida, H.; Suzuki, M.; Imai, K. Construction of a diabody (small recombinant bispecific antibody) using a refolding system. Protein Eng. 2000, 13, 583–588. [Google Scholar] [CrossRef][Green Version]
- Takemura, S.-I.; Kudo, T.; Asano, R.; Suzuki, M.; Tsumoto, K.; Sakurai, N.; Katayose, Y.; Kodama, H.; Yoshida, H.; Ebara, S. A mutated superantigen SEA D227A fusion diabody specific to MUC1 and CD3 in targeted cancer immunotherapy for bile duct carcinoma. Cancer Immunol. Immunother. 2002, 51, 33–44. [Google Scholar] [CrossRef]
- Schuhmacher, J.; Klivényi, G.; Kaul, S.; Henze, M.; Matys, R.; Hauser, H.; Clorius, J. Pretargeting of human mammary carcinoma xenografts with bispecific anti-MUC1/anti-Ga chelate antibodies and immunoscintigraphy with PET. Nucl. Med. Biol. 2001, 28, 821–828. [Google Scholar] [CrossRef]
- King, T.; Posey, A. Co-expression of an engineered cell-surface sialidase by CART cells improves anti-cancer activity of NK cells in solid tumors. Cytotherapy 2019, 21, S27. [Google Scholar] [CrossRef]
- Brooks, C.L.; Schietinger, A.; Borisova, S.N.; Kufer, P.; Okon, M.; Hirama, T.; MacKenzie, C.R.; Wang, L.-X.; Schreiber, H.; Evans, S.V. Antibody recognition of a unique tumor-specific glycopeptide antigen. Proc. Natl. Acad. Sci. USA 2010, 107, 10056–10061. [Google Scholar] [CrossRef]
- Borgert, A.; Heimburg-Molinaro, J.; Song, X.; Lasanajak, Y.; Ju, T.; Liu, M.; Thompson, P.; Ragupathi, G.; Barany, G.; Smith, D.F. Deciphering structural elements of mucin glycoprotein recognition. ACS Chem. Biol. 2012, 7, 1031–1039. [Google Scholar] [CrossRef]
- Springer, G.F. Immunoreactive T and Tn epitopes in cancer diagnosis, prognosis, and immunotherapy. J. Mol. Med. 1997, 75, 594–602. [Google Scholar] [CrossRef] [PubMed]
- Kudelka, M.R.; Ju, T.; Heimburg-Molinaro, J.; Cummings, R.D. Simple sugars to complex disease—Mucin-type O-glycans in cancer. In Advances in Cancer Research; Elsevier: Amsterdam, The Netherlands, 2015; Volume 126, pp. 53–135. [Google Scholar]
- Karsten, U.; Serttas, N.; Paulsen, H.; Danielczyk, A.; Goletz, S. Binding patterns of DTR-specific antibodies reveal a glycosylation-conditioned tumor-specific epitope of the epithelial mucin (MUC1). Glycobiology 2004, 14, 681–692. [Google Scholar] [CrossRef]
- Matsushita, T.; Takada, W.; Igarashi, K.; Naruchi, K.; Miyoshi, R.; Garcia-Martin, F.; Amano, M.; Hinou, H.; Nishimura, S.-I. A straightforward protocol for the preparation of high performance microarray displaying synthetic MUC1 glycopeptides. Biochim. Biophys. Acta (BBA)-Gen. Subj. 2014, 1840, 1105–1116. [Google Scholar] [CrossRef]
- Rangappa, S.; Artigas, G.; Miyoshi, R.; Yokoi, Y.; Hayakawa, S.; Garcia-Martin, F.; Hinou, H.; Nishimura, S.-I. Effects of the multiple O-glycosylation states on antibody recognition of the immunodominant motif in MUC1 extracellular tandem repeats. MedChemComm 2016, 7, 1102–1122. [Google Scholar] [CrossRef]
- Martínez-Sáez, N.; Castro-López, J.; Valero-González, J.; Madariaga, D.; Compañón, I.; Somovilla, V.J.; Salvadó, M.; Asensio, J.L.; Jiménez-Barbero, J.; Avenoza, A. Deciphering the Non-equivalence of serine and threonine O-glycosylation points: Implications for molecular recognition of the Tn antigen by an anti-MUC1 antibody. Angew. Chem. Int. Ed. 2015, 54, 9830–9834. [Google Scholar] [CrossRef]
- Wakui, H.; Tanaka, Y.; Ose, T.; Matsumoto, I.; Kato, K.; Min, Y.; Tachibana, T.; Sato, M.; Naruchi, K.; Martin, F.G. A straightforward approach to antibodies recognising cancer specific glycopeptidic neoepitopes. Chem. Sci. 2020, 11, 4999–5006. [Google Scholar] [CrossRef]
- Thie, H.; Toleikis, L.; Li, J.; von Wasielewski, R.; Bastert, G.; Schirrmann, T.; Esteves, I.T.; Behrens, C.K.; Fournes, B.; Fournier, N. Rise and fall of an anti-MUC1 specific antibody. PLoS ONE 2011, 6, e15921. [Google Scholar] [CrossRef]
- Moreno, M.; Bontkes, H.J.; Scheper, R.J.; Kenemans, P.; Verheijen, R.H.; von Mensdorff-Pouilly, S. High level of MUC1 in serum of ovarian and breast cancer patients inhibits huHMFG-1 dependent cell-mediated cytotoxicity (ADCC). Cancer Lett. 2007, 257, 47–55. [Google Scholar] [CrossRef] [PubMed]
- Tang, Y.; Cui, X.; Xiao, H.; Qi, S.; Hu, X.; Yu, Q.; Shi, G.; Zhang, X.; Gu, J.; Yu, Y. Binding of circulating anti-MUC1 antibody and serum MUC1 antigen in stage IV breast cancer. Mol. Med. Rep. 2017, 15, 2659–2664. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Treon, S.P.; Maimonis, P.; Bua, D.; Young, G.; Raje, N.; Mollick, J.; Chauhan, D.; Tai, Y.-T.; Hideshima, T.; Shima, Y. Elevated soluble MUC1 levels and decreased anti-MUC1 antibody levels in patients with multiple myeloma. Blood J. Am. Soc. Hematol. 2000, 96, 3147–3153. [Google Scholar]
- Storr, S.J.; Royle, L.; Chapman, C.J.; Hamid, U.M.A.; Robertson, J.F.; Murray, A.; Dwek, R.A.; Rudd, P.M. The O-linked glycosylation of secretory/shed MUC1 from an advanced breast cancer patient’s serum. Glycobiology 2008, 18, 456–462. [Google Scholar] [CrossRef]
- Pegram, M.; Borges, V.; Fuloria, J.; Ibrahim, N.; Shapiro, C.; Perez, E.; Wang, K.; Schaedeli Stark, F.; Yeon, C.; Courtenay-Luck, N. Phase I pharmacokinetics (PK) of humanized anti-MUC-1 antibody R1550. J. Clin. Oncol. 2006, 24 (Suppl. 18), 2533. [Google Scholar] [CrossRef]
- Zhou, D.; Xu, L.; Huang, W.; Tonn, T. Epitopes of MUC1 tandem repeats in cancer as revealed by antibody crystallography: Toward glycopeptide signature-guided therapy. Molecules 2018, 23, 1326. [Google Scholar] [CrossRef]
- Sørensen, A.L.; Reis, C.A.; Tarp, M.A.; Mandel, U.; Ramachandran, K.; Sankaranarayanan, V.; Schwientek, T.; Graham, R.; Taylor-Papadimitriou, J.; Hollingsworth, M.A. Chemoenzymatically synthesized multimeric Tn/STn MUC1 glycopeptides elicit cancer-specific anti-MUC1 antibody responses and override tolerance. Glycobiology 2006, 16, 96–107. [Google Scholar] [CrossRef]
- Rivalland, G.; Loveland, B.; Mitchell, P. Update on Mucin-1 immunotherapy in cancer: A clinical perspective. Expert Opin. Biol. Ther. 2015, 15, 1773–1787. [Google Scholar] [CrossRef]
- Singh, R.; Bandyopadhyay, D. MUC1: A target molecule for cancer therapy. Cancer Biol. Ther. 2007, 6, 481–486. [Google Scholar] [CrossRef]
- McGuckin, M.A.; Hurst, T.G.; Ward, B.G. Heterogeneity in production, secretion and glycosylation of MUC1 epithelial mucin by primary cultures of ovarian carcinoma. Int. J. Cancer 1995, 63, 412–418. [Google Scholar] [CrossRef]
- Walsh, M.D.; Luckie, S.M.; Cummings, M.C.; Antalis, T.M.; McGuckin, M.A. Heterogeneity of MUC1 expression by human breast carcinoma cell lines in vivo and in vitro. Breast Cancer Res. Treat. 1999, 58, 253–264. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Q.; Piyush, T.; Chen, C.; Hollingsworth, M.A.; Hilkens, J.; Rhodes, J.M.; Yu, L.-G. MUC1 extracellular domain confers resistance of epithelial cancer cells to anoikis. Cell Death Dis. 2014, 5, e1438. [Google Scholar] [CrossRef] [PubMed]
- Pichinuk, E.; Benhar, I.; Jacobi, O.; Chalik, M.; Weiss, L.; Ziv, R.; Sympson, C.; Karwa, A.; Smorodinsky, N.I.; Rubinstein, D.B. Antibody targeting of cell-bound MUC1 SEA domain kills tumor cells. Cancer Res. 2012, 72, 3324–3336. [Google Scholar] [CrossRef]
- Madsen, C.B.; Wandall, H.H.; Pedersen, A.E. Potential for novel MUC1 glycopeptide-specific antibody in passive cancer immunotherapy. Immunopharmacol. Immunotoxicol. 2013, 35, 649–652. [Google Scholar] [CrossRef]
- Reilly, R.M.; Sandhu, J.; Alvarez-Diez, T.M.; Gallinger, S.; Kirsh, J.; Stern, H. Problems of delivery of monoclonal antibodies. Clin. Pharmacokinet. 1995, 28, 126–142. [Google Scholar] [CrossRef] [PubMed]
- Sonbol, M.B.; Ahn, D.H.; Goldstein, D.; Okusaka, T.; Tabernero, J.; Macarulla, T.; Reni, M.; Li, C.-P.; O’Neil, B.; Van Cutsem, E. CanStem111P trial: A Phase III study of napabucasin plus nab-paclitaxel with gemcitabine. Future Oncol. 2019, 15, 1295–1302. [Google Scholar] [CrossRef]
- Bose, M.; Vora, A.; Colleton, T.; Mukherjee, P. MUC1 Confers Sensitivity to STAT-3 Inhibitor Napabucasin in Pancreatic Ductal Adenocarcinoma Cells. Cancer Res. 2020, 1837. [Google Scholar] [CrossRef]
| Antibody | Epitope | Original Antigen | Treatment under Trial | GI Cancer Type | Year | Reference |
|---|---|---|---|---|---|---|
| KL-6 | a sialylated sugar of Krebs von den Lugen-6 (KL-6) PDTRPAP sequence | a sialylated sugar of Krebs von den Lugen-6 (KL-6) PDTRPAP sequence | 99mTc labeled anti-KL-6/MUC1 | Pancreatic Cancer | 2008 | [67,68,69] |
| MY.1E12 | sialyla2–3galactosylh1-3Nacetylgalactosaminide linked to a distinct threonine residue in the MUC1 tandem repeat | HMFG | 3-ICG-acyl-1,3-thiazolidine-2-thione labeled MY.1E12 | Gastric Cancer | 2008 | [70,71,72,73] |
| 5E5, 2D9 | Tn or STn in the tandem repeat domain | GalNAc-glycosylated MUC1 glycopeptide (VTSAPDTRPAPGSTAPPAHG) conjugated to KLH | 5E5 MUC1-CAR-T cells | Pancreatic Cancer | 2016 2019 | [74] |
| hMUC1-1H7 | extracellular domain of MUC1 C-terminal subunit (MUC1-C) | recombinant human (rh) protein including extracellular region of MUC1- C (rhMUC1-EC192) obtained from MCF7 cells | hMUC1-1H7 | Pancreatic Cancer | 2004 | [75,76] |
| TAB004 | STAPPVHNV within the TR sequence | Protein lysate from MUC1-expressing tumors that developed in a MUC1 transgenic mice (PDA mice) that expressed human MUC1 |
| Pancreatic Cancer | 2008–2019 | [77,78,79,80,81,82] |
| Antibody | Epitope | Original Antigen | Treatment under Trial | GI Cancer Type | Clinical Trial Status | Year | Reference |
|---|---|---|---|---|---|---|---|
| huC242 | Sialyl-Lewis a epitope CanAg glycoprotein which is similar to MUC1 | Human colorectal adenocarcinoma cell line COLO205 | huC242-DM4 |
|
| 2006 2008 | [83,84,85,86,87,88] |
| huPAM4 | Domain located between the amino terminus and start of the repeat domain of a MUC1 antigen (non- VNTR) and also react with MUC5AC | Mucin purified from the xenografted RIP I human pancreatic carcinoma | 111In-huPAM4 | Pancreatic Cancer | Phase I terminated | 2006 | [89,90,91] |
| hPAM4 (Clivatuzumab) | Domain located between the amino terminus and start of the repeat domain of a MUC1 antigen (non-VNTR) and also react with MUC5AC | Mucin purified from the xenografted RIP I human pancreatic carcinoma |
|
|
| 2008 2013 | [92,93] |
| SAR56665 8huDS6-DM4 | O-linked glycans with α2,3-sialylated and β1,4-galactosylated termini in VNTR | Human serous ovarian carcinoma | SAR56665 8huDS6-DM4 | Pancreas | Phase II completed | 2010 | |
| PankoMab-GEX™ (Gatipotuzumab) | Epitope...PDT*RP..., where T* is O-glycosylated with GalNAca1- or a similar short, non-sialylated glycan such as Galb1-3GalNAca1-(core-1) | Tumor MUC1 from a desialylated human breast cancer source |
|
|
| 2010 2017 | [94,95,96,97] |
| PD-1 inhibitor armed with an anti-MUC 1 and anti- CD3 bispecific antibody | Information unavailable | Information unavailable | PD-1 inhibitor armed with an anti-MUC 1 and anti- CD3 bispecific antibody | Advanced Gastric, Colorectal, Pancreatic and Liver cancers | Recruiting for Phase II | 2018 | [98] |
| AR20.5 | DTRPAP and DTnRPAP | MUC1 from an ovarian cancer patient, derived from human fluids and breast cancer cell MCF-7 culture medium |
|
|
| 2004 2018 | [24,99,100] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bose, M.; Mukherjee, P. Potential of Anti-MUC1 Antibodies as a Targeted Therapy for Gastrointestinal Cancers. Vaccines 2020, 8, 659. https://doi.org/10.3390/vaccines8040659
Bose M, Mukherjee P. Potential of Anti-MUC1 Antibodies as a Targeted Therapy for Gastrointestinal Cancers. Vaccines. 2020; 8(4):659. https://doi.org/10.3390/vaccines8040659
Chicago/Turabian StyleBose, Mukulika, and Pinku Mukherjee. 2020. "Potential of Anti-MUC1 Antibodies as a Targeted Therapy for Gastrointestinal Cancers" Vaccines 8, no. 4: 659. https://doi.org/10.3390/vaccines8040659
APA StyleBose, M., & Mukherjee, P. (2020). Potential of Anti-MUC1 Antibodies as a Targeted Therapy for Gastrointestinal Cancers. Vaccines, 8(4), 659. https://doi.org/10.3390/vaccines8040659






