Immunogenicity of Alternative Dosing Schedules for HPV Vaccines among Adolescent Girls and Young Women: A Systematic Review and Meta-Analysis
Abstract
1. Introduction
2. Materials and Methods
2.1. Literature Search and Study Selection
2.2. Data Extraction
2.3. Definitions
2.3.1. Vaccines
2.3.2. Age
2.3.3. Dose
2.3.4. Analysis
3. Results
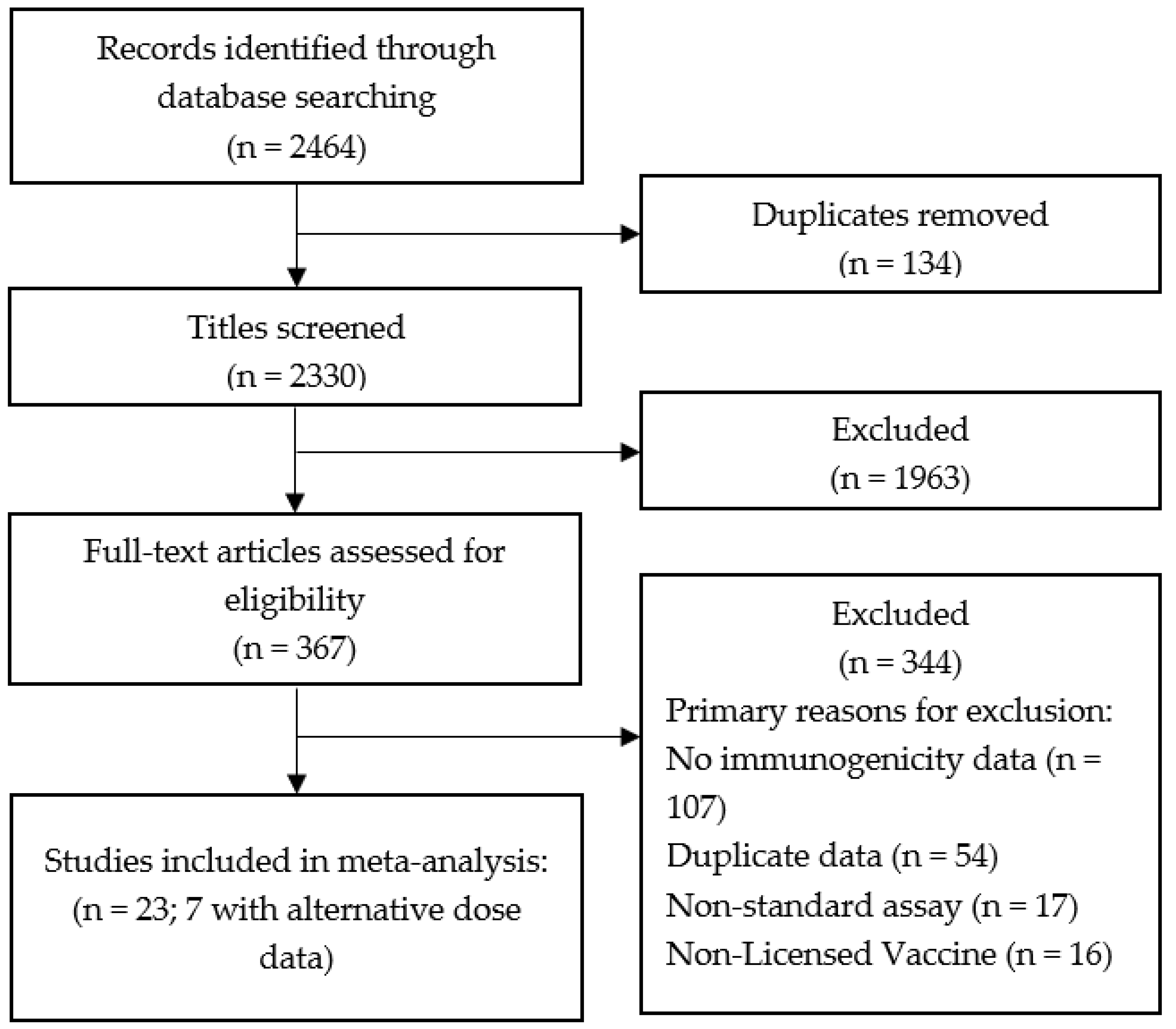
3.1. Study Selection
3.2. Study Characteristics
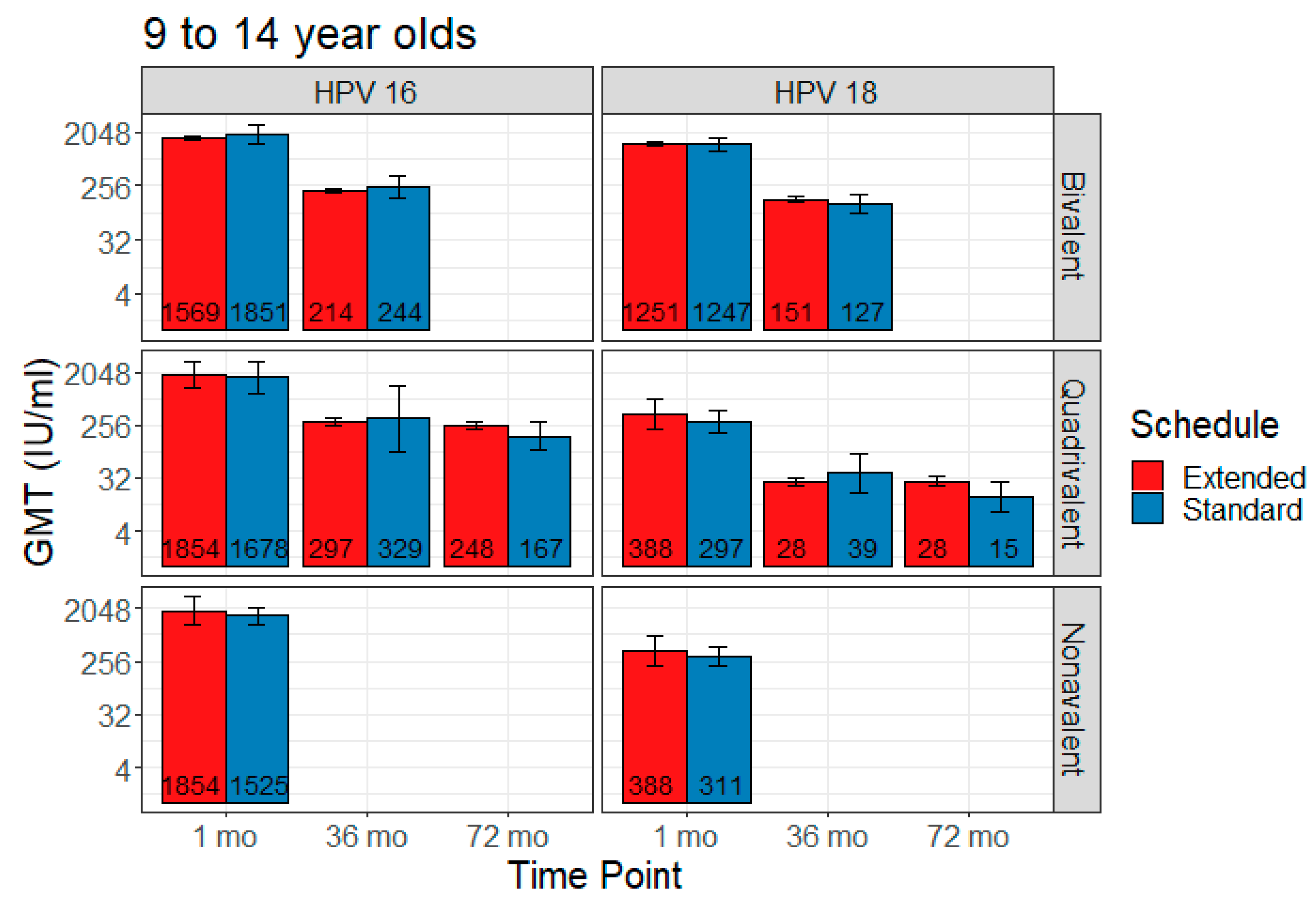
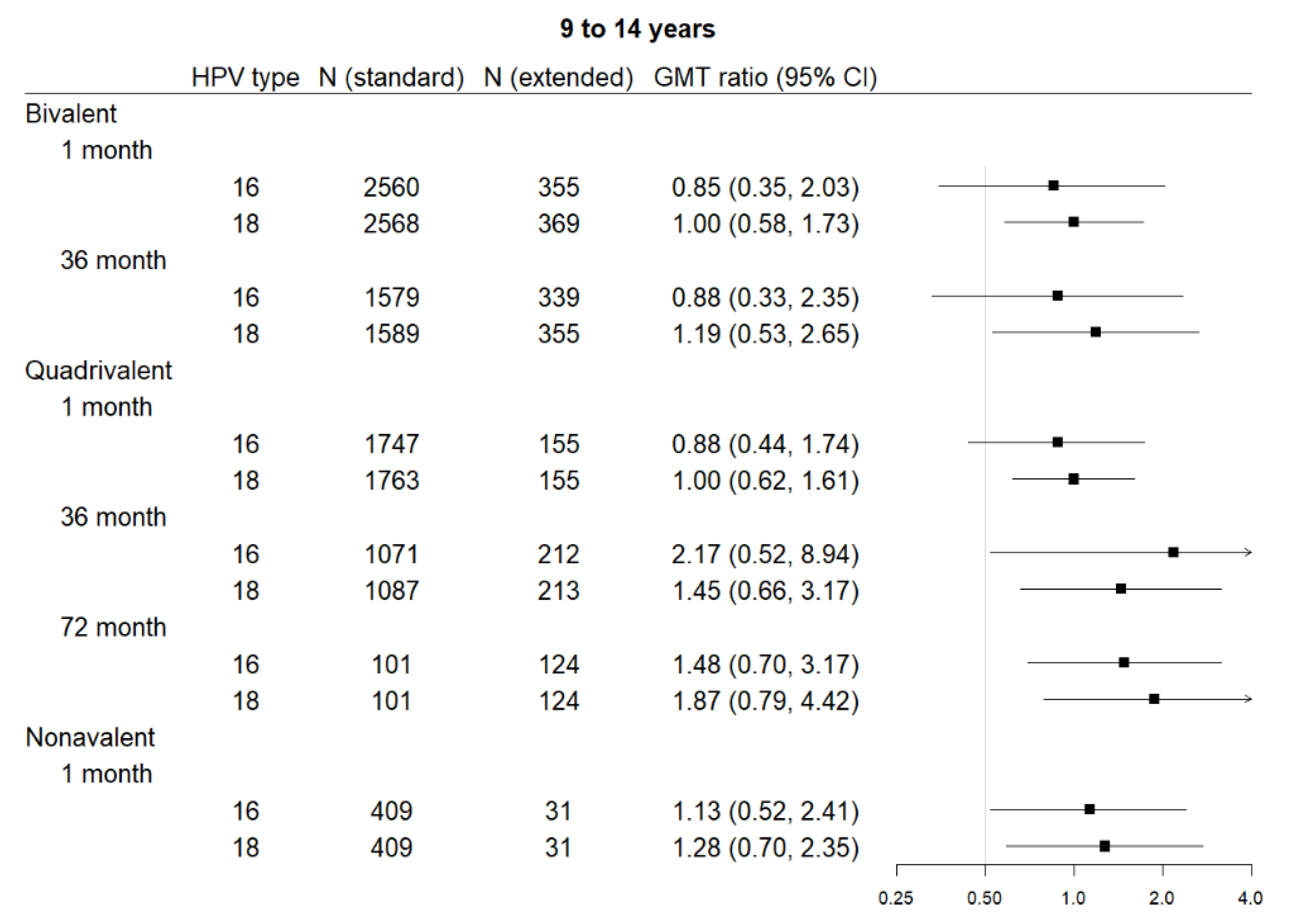
3.3. Extended Interval
3.3.1. Bivalent
3.3.2. Quadrivalent
3.3.3. Nonavalent
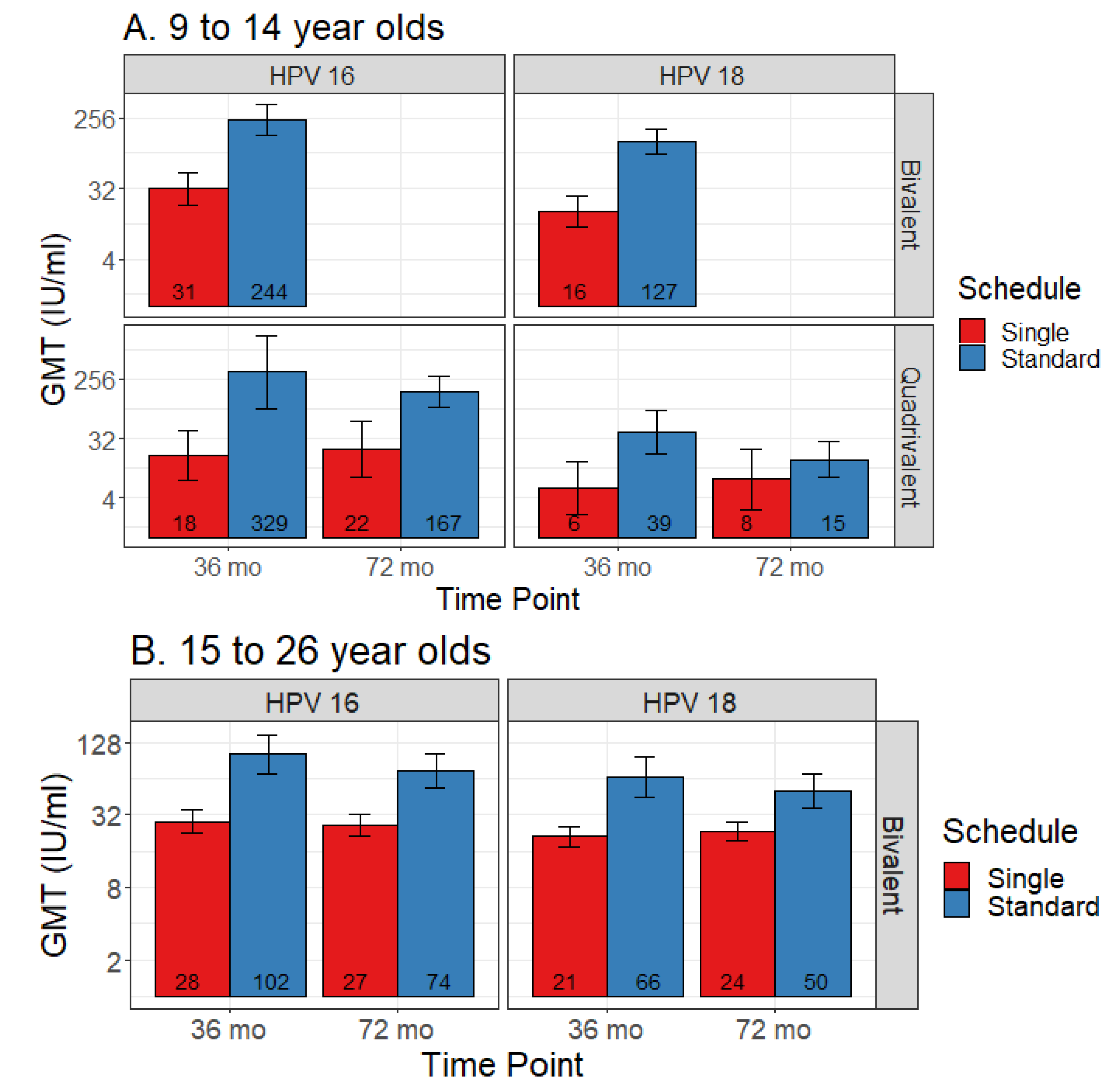
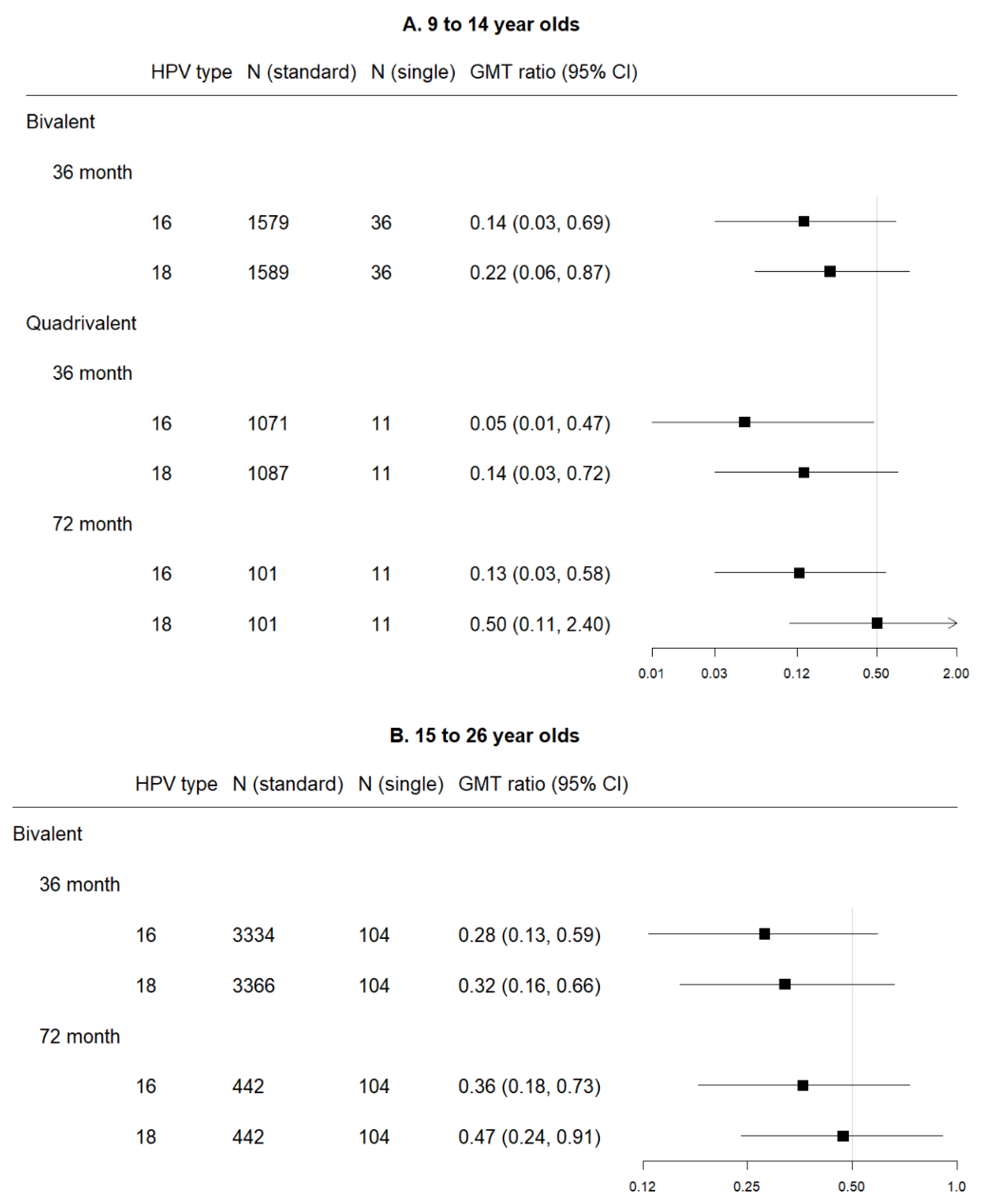
3.4. Single Dose
3.4.1. Bivalent
3.4.2. Quadrivalent
3.4.3. Nonavalent
4. Discussion
Limitations
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
Data Availability
References
- Ferlay, J.; Shin, H.-R.; Bray, F.; Forman, D.; Mathers, C.; Parkin, D.M. Estimates of worldwide burden of cancer in 2008: GLOBOCAN 2008. Int. J. Cancer 2010, 127, 2893–2917. [Google Scholar] [CrossRef] [PubMed]
- Arbyn, M.; Weiderpass, E.; Bruni, L.; De Sanjosé, S.; Saraiya, M.; Ferlay, J.; Bray, F. Estimates of incidence and mortality of cervical cancer in 2018: A worldwide analysis. Lancet Glob. Health 2020, 8, e191–e203. [Google Scholar] [CrossRef]
- Burger, E.A.; Campos, N.G.; Sy, S.; Regan, C.; Kim, J.J. Health and economic benefits of single-dose HPV vaccination in a Gavi-eligible country. Vaccine 2018, 36, 4823–4829. [Google Scholar] [CrossRef] [PubMed]
- Weaver, B.A. Epidemiology and natural history of HPV. J. Am. Osteopath. Assoc. 2006, 106, S2–S7. [Google Scholar]
- Forman, D.; de Martel, C.; Lacey, C.J.; Soerjomatarama, I.; Lortet-Tieulent, J.; Bruni, L.; Vignat, J.; Ferlay, J.; Bray, F.; Plummer, M.; et al. Global burden of human papillomavirus and related diseases. Vaccine 2012, 30S, F12–F23. [Google Scholar] [CrossRef]
- World Health Organization (WHO). Cervical Cancer [Internet]. Available online: https://www.who.int/cancer/prevention/diagnosis-screening/cervical-cancer/en/ (accessed on 25 November 2019).
- World Health Organization (WHO). Human papillomavirus vaccines: WHO position paper. Wkly. Epidemiol. Rec. 2017, 92, 241–268. [Google Scholar]
- Tan, N.; Sharma, M.; Winer, R.; Galloway, D.; Rees, H.; Barnabas, R. Model-estimated effectiveness of single dose 9-valent HPV vaccination for HIV-positive and HIV-negative females in South Africa. Vaccine 2018, 36, 4830–4836. [Google Scholar] [CrossRef]
- Hu, Y.M.; Guo, M.; Li, C.G.; Chu, K.; He, W.G.; Zhang, J.; Gu, J.-X.; Li, J.; Zhao, H.; Wu, X.-H.; et al. Immunogenicity noninferiority study of 2 doses and 3 doses of an Escherichia coli-produced HPV bivalent vaccine in girls vs. 3 doses in young women. Sci. China Life Sci. 2020, 63, 582–591. [Google Scholar] [CrossRef]
- Safaeian, M.; Sampson, J.N.; Pan, Y.; Porras, C.; Kemp, T.J.; Herrero, R.; Quint, W.; Van Doorn, L.-J.; Schussler, J.; Lowy, D.R.; et al. Durability of Protection Afforded by Fewer Doses of the HPV16/18 Vaccine: The CVT Trial. J. Natl. Cancer Inst. 2017, 110, 205–212. [Google Scholar] [CrossRef]
- McInnes, M.D.F.; Moher, D.; Thombs, B.D.; McGrath, T.A.; Bossuyt, P.M.; Clifford, T.; Cohen, J.F.; Deeks, J.J.; Gatsonis, C.; Hooft, L.; et al. Preferred Reporting Items for a Systematic Review and Meta-analysis of Diagnostic Test Accuracy Studies The PRISMA-DTA Statement. Jama J. Am. Med. Assoc. 2018, 319, 388–396. [Google Scholar] [CrossRef]
- Brown, D.; Müller, M.; Sehr, P.; Pawlita, M.; Seitz, H.; Rubio, I.; Antonello, J.; Radley, D.; Roberts, C.C.; Saah, A. Concordance assessment between a multiplexed competitive Luminex immunoassay, a multiplexed IgG Luminex immunoassay, and a pseudovirion-based neutralization assay for detection of human papillomaviruse types 16 and 18. Vaccine 2014, 32, 5880–5887. [Google Scholar] [CrossRef] [PubMed]
- Pinto, L.A.; Wilkin, T.J.; Kemp, T.J.; Abrahamsen, M.; Isaacs-Soriano, K.; Pan, Y.; Webster-Cyriaque, J.; Palefsky, J.M.; Giuliano, A.R. Oral and systemic HPV antibody kinetics post-vaccination among HIV-positive and HIV-negative men. Vaccine 2019, 37, 2502–2510. [Google Scholar] [CrossRef] [PubMed]
- Kjaer, S.K.; Sigurdsson, K.; Iversen, O.E.; Hernandez-Avila, M.; Wheeler, C.M.; Perez, G.; Brown, D.R.; Koutsky, L.A.; Tay, E.H.; García, P.; et al. A pooled analysis of continued prophylactic efficacy of quadrivalent human papillomavirus (types 6/11/16/18) vaccine against high-grade cervical and external genital lesions. Cancer Prev. Res. 2009, 2, 868–878. [Google Scholar] [CrossRef] [PubMed]
- Garland, S.M.; Kjaer, S.K.; Muñoz, N.; Block, S.L.; Brown, D.R.; Dinubile, M.J.; Lindsay, B.R.; Kuter, B.J.; Perez, G.; Dominiak-Felden, G.; et al. Impact and effectiveness of the quadrivalent human papillomavirus vaccine: A systematic review of 10 years of real-world experience. Clin. Infect. Dis. 2016, 63, 519–527. [Google Scholar] [CrossRef] [PubMed]
- Lehtinen, M.; Paavonen, J.; Wheeler, C.M.; Jaisamrarn, U.; Garland, S.M.; Castellsagué, X.; Skinner, S.R.; Apter, D.; Naud, P.; Salmerón, J.; et al. Overall efficacy of HPV-16/18 AS04-adjuvanted vaccine against grade 3 or greater cervical intraepithelial neoplasia: 4-year end-of-study analysis of the randomised, double-blind PATRICIA trial. Lancet Oncol. 2012, 13, 89–99. [Google Scholar] [CrossRef]
- Hildesheim, A.; Wacholder, S.; Catteau, G.; Struyf, F.; Dubin, G.; Herrero, R. Efficacy of the HPV-16/18 vaccine: Final according to protocol results from the blinded phase of the randomized Costa Rica HPV-16/18 vaccine trial. Vaccine 2014, 32, 5087–5097. [Google Scholar] [CrossRef]
- Joura, E.A.; Giuliano, A.R.; Iversen, O.E.; Bouchard, C.; Mao, C.; Mehlsen, J.; Moreira, E.D.; Ngan, Y.; Petersen, L.K.; Lazcano-Ponce, E.; et al. A 9-valent HPV vaccine against infection and intraepithelial neoplasia in women. N. Engl. J. Med. 2014, 372, 711–723. [Google Scholar] [CrossRef]
- Vesikari, T.; Brodszki, N.; Van Damme, P.; Diez-Domingo, J.; Icardi, G.; Petersen, L.K.; Tran, C.; Thomas, S.; Luxembourg, A.; Baudin, M. A Randomized, Double-Blind, Phase III Study of the Immunogenicity and Safety of a 9-Valent Human Papillomavirus L1 Virus-Like Particle Vaccine (V503) Versus Gardasil® in 9–15-Year-Old Girls. Pediatr. Infect. Dis. J. 2015, 34, 992–998. [Google Scholar] [CrossRef]
- Gilca, V.; Sauvageau, C.; Panicker, G.; De Serres, G.; Ouakki, M.; Unger, E.R. Antibody persistence after a single dose of quadrivalent HPV vaccine and the effect of a dose of nonavalent vaccine given 3–8 years later—An exploratory study. Hum. Vaccines Immunother. 2019, 15, 503–507. [Google Scholar] [CrossRef]
- Sow, P.S.; Watson-Jones, D.; Kiviat, N.; Changalucha, J.; Mbaye, K.D.; Brown, J.; Bousso, K.; Kavishe, B.; Andreasen, A.; Toure, M.; et al. Safety and immunogenicity of human papillomavirus-16/18 AS04-adjuvanted vaccine: A randomized trial in 10–25-year-old HIV-seronegative African girls and young women. J. Infect. Dis. 2013, 207, 1753–1763. [Google Scholar] [CrossRef]
- Lazcano-Ponce, E.; Stanley, M.; Muñoz, N.; Torres, L.; Cruz-Valdez, A.; Salmerón, J.; Rojas-Martínez, R.; Herrero, R.; Hernández-Ávila, M. Overcoming barriers to HPV vaccination: Non-inferiority of antibody response to human papillomavirus 16/18 vaccine in adolescents vaccinated with a two-dose vs. a three-dose schedule at 21 months. Vaccine 2014, 32, 725–732. [Google Scholar] [CrossRef] [PubMed]
- Toh, Z.Q.; Russell, F.M.; Reyburn, R.; Fong, J.; Tuivaga, E.; Ratu, T.; Nguyen, C.D.; Devi, R.; Kama, M.; Matanitobua, S.; et al. Sustained antibody responses 6 years following 1, 2, or 3 doses of quadrivalent human papillomavirus (HPV) vaccine in adolescent fijian girls, and subsequent responses to a single dose of bivalent HPV vaccine: A prospective cohort study. Clin. Infect. Dis. 2017, 64, 852–859. [Google Scholar] [PubMed]
- Sankaranarayanan, R.; Prabhu, P.R.; Pawlita, M.; Gheit, T.; Bhatla, N.; Muwonge, R.; Nene, B.M.; Esmy, P.O.; Joshi, S.; Poli, U.R.R.; et al. Immunogenicity and HPV infection after one, two, and three doses of quadrivalent HPV vaccine in girls in India: A multicentre prospective cohort study. Lancet Oncol. 2016, 17, 67–77. [Google Scholar] [CrossRef]
- Hurt, L.; Nsouli-Maktabi, H.; Rohrbeck, P.; Clark, L. Use of Quadrivalent Human Papillomavirus Vaccine and the Prevalence of Antibodies to Vaccine-targeted Strains Among Female Service Members before and after Vaccination. Med. Surveill Mon. Rep. 2016, 23, 6–13. [Google Scholar]
- Huang, L.M.; Puthanakit, T.; Cheng-Hsun, C.; Ren-Bin, T.; Schwarz, T.; Pellegrino, A.; Esposito, S.; Frenette, L.; McNeil, S.; Durando, P.; et al. Sustained immunogenicity of 2-dose human papillomavirus 16/18 AS04-adjuvanted vaccine schedules in girls aged 9–14 years: A randomized trial. J. Infect. Dis. 2017, 215, 1711–1719. [Google Scholar] [CrossRef] [PubMed]
- Lamontagne, D.S.; Thiem, V.D.; Huong, V.M.; Tang, Y.; Neuzil, K.M. Immunogenicity of quadrivalent HPV vaccine among girls 11 to 13 years of age vaccinated using alternative dosing schedules: Results 29 to 32 months after third dose. J. Infect. Dis. 2013, 208, 1325–1334. [Google Scholar] [CrossRef][Green Version]
- Neuzil, K.M.; Canh, D.G.; Thiem, V.D.; Janmohamed, A.; Huong, V.M.; Tang, Y.; Diep, N.T.N.; Tsu, V.; Lamontagne, D.S. Immunogenicity and Reactogenicity of Alternative Schedules of HPV Vaccine in Vietnam. Jama J. Am. Med. Assoc. 2011, 305, 1424. [Google Scholar] [CrossRef]
- Puthanakit, T.; Huang, L.M.; Chiu, C.H.; Tang, R.; Schwarz, T.F.; Esposito, S.; Frenette, L.; Giaquinto, C.; McNeil, S.; Rheault, P.; et al. Randomized open trial comparing 2-dose regimens of the human papillomavirus 16/18 as04-adjuvanted vaccine in girls aged 9–14 years versus a 3-dose regimen in women aged 15–25 years. J. Infect. Dis. 2016, 214, 525–536. [Google Scholar] [CrossRef]
- LaMontagne, D.S.; Mugisha, E.; Pan, Y.; Kumakech, E.; Ssemaganda, A.; Kemp, T.J.; Cover, J.; Pinto, L.A.; Safaeian, M. Immunogenicity of bivalent HPV vaccine among partially vaccinated young adolescent girls in Uganda. Vaccine 2014, 32, 6303–6311. [Google Scholar] [CrossRef]
- Brophy, J.; Bitnun, A.; Alimenti, A.; Lapointe, N.; Samson, L.; Read, S.; Karatzios, C.; Dobson, S.; Moses, E.; Blitz, S.; et al. Immunogenicity and safety of the quadrivalent human papillomavirus vaccine in girls living with HIV. Pediatr. Infect. Dis. J. 2018, 37, 595–597. [Google Scholar] [CrossRef]
- Dobson, S.R.M.; McNeil, S.; Dionne, M.; Dawar, M.; Ogilvie, G.; Krajden, M.; Sauvageau, C.; Scheifele, D.W.; Kollmann, T.R.; Halperin, S.A.; et al. Immunogenicity of 2 doses of HPV vaccine in younger adolescents vs. 3 doses in young women: A randomized clinical trial. Jama J. Am. Med. Assoc. 2013, 309, 1793–1802. [Google Scholar] [CrossRef] [PubMed]
- Herrin, D.M.; Coates, E.E.; Costner, P.J.; Kemp, T.J.; Nason, M.C.; Saharia, K.K.; Pan, Y.; Sarwar, U.N.; Holman, L.; Yamshchikov, G.; et al. Comparison of adaptive and innate immune responses induced by licensed vaccines for human papillomavirus. Hum. Vaccines Immunother. 2014, 10, 3446–3454. [Google Scholar] [CrossRef]
- Iversen, O.E.; Miranda, M.J.; Ulied, A.; Soerdal, T.; Lazarus, E.; Chokephaibulkit, K.; Block, S.L.; Skrivanek, A.; Azurah, A.G.N.; Fong, S.M.; et al. Immunogenicity of the 9-valent HPV Vaccine Using 2-Dose Regimens in Girls and Boys vs. A 3-Dose Regimen in Women. Jama J. Am. Med. Assoc. 2016, 316, 2411–2421. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.J.; Kim, K.T.; Kim, J.H.; Cha, S.; Kim, J.W.; Bae, D.S.; Nam, J.H.; Ahn, W.S.; Choi, H.S. Vaccination with a human papillomavirus (HPV)-16/18 AS04-adjuvanted cervical cancer vaccine in Korean girls aged 10–14 years. J. Korean Med. Sci. 2010, 25, 1197–1204. [Google Scholar] [CrossRef] [PubMed]
- Lazcano-Ponce, E.; Torres-Ibarra, L.; Cruz-Valdez, A.; Salmerón, J.; Barrientos-Gutiérrez, T.; Prado-Galbarro, J.; Stanley, M.; Muñoz, N.; Herrero, R.; Hernández-Ávila, M. Persistence of immunity when using different human papillomavirus vaccination schedules and booster-dose effects 5 years after primary vaccination. J. Infect. Dis. 2019, 219, 41–49. [Google Scholar] [CrossRef] [PubMed]
- Leung, T.F.; Liu, A.P.Y.; Lim, F.S.; Thollot, F.; Oh, H.M.L.; Lee, B.W.; Rombo, L.; Tan, N.C.; Rouzier, R.; Friel, D.; et al. Comparative immunogenicity and safety of human papillomavirus (HPV)-16/18 AS04-adjuvanted vaccine and HPV-6/11/16/18 vaccine administered according to 2- and 3-dose schedules in girls aged 9–14 years: Results to month 12 from a randomized trial. Hum. Vaccines Immunother. 2015, 11, 1689–1702. [Google Scholar] [CrossRef] [PubMed]
- Leung, T.F.; Liu, A.P.Y.; Lim, F.S.; Thollot, F.; Oh, H.M.L.; Lee, B.W.; Rombo, L.; Tan, N.C.; Rouzier, R.; De Simoni, S.; et al. Comparative immunogenicity and safety of human papillomavirus (HPV)-16/18 AS04-adjuvanted vaccine and 4vHPV vaccine administered according to two- or three-dose schedules in girls aged 9–14 years: Results to month 36 from a randomized trial. Vaccine 2018, 36, 98–106. [Google Scholar] [CrossRef]
- Ogilvie, G.; Sauvageau, C.; Dionne, M.; McNeil, S.; Krajden, M.; Money, D.; Dobson, S. Immunogenicity of 2 vs. 3 Doses of the Quadrivalent Human Papillomavirus Vaccine in Girls Aged 9 to 13 Years after 60 Months. Jama J. Am. Med. Assoc. 2017, 317, 1687–1688. [Google Scholar] [CrossRef][Green Version]
- Petäjä, T.; Pedersen, C.; Poder, A.; Strauss, G.; Catteau, G.; Thomas, F.; Lehtinen, M.; Descamps, D. Long-term persistence of systemic and mucosal immune response to HPV-16/18 AS04-adjuvanted vaccine in preteen/adolescent girls and young women. Int. J. Cancer 2011, 129, 2147–2157. [Google Scholar] [CrossRef]
- Schwarz, T.F.; Huang, L.M.; Medina, D.M.R.; Valencia, A.; Lin, T.Y.; Behre, U.; Catteau, G.; Thomas, F.; Descamps, D. Four-year follow-up of the immunogenicity and safety of the HPV-16/18 AS04-adjuvanted vaccine when administered to adolescent girls aged 1014 years. J. Adolesc. Health 2012, 50, 187–194. [Google Scholar] [CrossRef]
- Schwarz, T.; Spaczynski, M.; Kaufmann, A.; Wysocki, J.; Gałaj, A.; Schulze, K.; Suryakiran, P.; Thomas, F.; Descamps, D. Persistence of immune responses to the HPV-16/18 AS04-adjuvanted vaccine in women aged 15–55 years and first-time modelling of antibody responses in mature women: Results from an open-label 6-year follow-up study. BJOG Int. J. Obs. Gynaecol. 2015, 122, 107–118. [Google Scholar] [CrossRef] [PubMed]
- Bruni, L.; Diaz, M.; Barrionuevo-Rosas, L.; Herrero, R.; Bray, F.; Bosch, F.X.; Sanjose, S.; Castellsague, X. Global estimates of human papillomavirus vaccination coverage by region and income level: A pooled analysis. Lancet Glob. Health 2016, 4, e453–e463. [Google Scholar] [CrossRef]
- Gallagher, K.E.; Howard, N.; Kabakama, S.; Mounier-Jack, S.; Burchett, H.E.D.; LaMontagne, D.S.; Watson-Jones, D. Human papillomavirus (HPV) vaccine coverage achievements in low and middle-income countries 2007–2016. Papillomavirus Res. 2017, 4, 72–78. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization (WHO). Weekly Epidemiological Record; World Health Organization (WHO): Geneva, Switzerland, 2019; Volume 47, pp. 541–560. [Google Scholar]
| Author, Year | Alternative Schedules | Dose Schedules (Months) | Study Design * | Age Range(s) | N ** | Vaccine(s) | Region(s) | ||
|---|---|---|---|---|---|---|---|---|---|
| Extended | Single | 9–14 | 15–26 | ||||||
| Gilca 2019 [20] | x | x | Single; (0,36–48); (0,60–72); (0,84–96) | NRS | x | 31 | Quadrivalent, nonavalent | N. America | |
| Huang 2017 [26] | x | (0,1,6); (0,12) | RCT | x | x | 669 | Bivalent | Asia, Europe, N. America | |
| LaMontagne 2013 [27] | x | (0,2,6); (0,12,24) | RCT | x | 223 | Quadrivalent | Asia | ||
| Lamontagne 2014 [30] | x | Single; (0,2,6) | Cross-sectional | x | 231 | Bivalent | Africa | ||
| Neuzil 2011 [28] | x | (0,2,6); (0,12,24) | RCT | x | 418 | Quadrivalent | Asia | ||
| Puthanakit 2016 [29] | x | (0,1,6); (0,6); (0,12) | RCT | x | x | 1195 | Bivalent | Asia, Europe, N. America | |
| Safaeian 2018 [10] | x | Single; (0,1,6); (0,6) | RCT | x | 330 | Bivalent | N. America | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Secor, A.M.; Driver, M.; Kharono, B.; Hergott, D.; Liu, G.; Barnabas, R.V.; Dull, P.; Hawes, S.E.; Drain, P.K. Immunogenicity of Alternative Dosing Schedules for HPV Vaccines among Adolescent Girls and Young Women: A Systematic Review and Meta-Analysis. Vaccines 2020, 8, 618. https://doi.org/10.3390/vaccines8040618
Secor AM, Driver M, Kharono B, Hergott D, Liu G, Barnabas RV, Dull P, Hawes SE, Drain PK. Immunogenicity of Alternative Dosing Schedules for HPV Vaccines among Adolescent Girls and Young Women: A Systematic Review and Meta-Analysis. Vaccines. 2020; 8(4):618. https://doi.org/10.3390/vaccines8040618
Chicago/Turabian StyleSecor, Andrew M., Matthew Driver, Brenda Kharono, Dianna Hergott, Gui Liu, Ruanne V. Barnabas, Peter Dull, Stephen E. Hawes, and Paul K. Drain. 2020. "Immunogenicity of Alternative Dosing Schedules for HPV Vaccines among Adolescent Girls and Young Women: A Systematic Review and Meta-Analysis" Vaccines 8, no. 4: 618. https://doi.org/10.3390/vaccines8040618
APA StyleSecor, A. M., Driver, M., Kharono, B., Hergott, D., Liu, G., Barnabas, R. V., Dull, P., Hawes, S. E., & Drain, P. K. (2020). Immunogenicity of Alternative Dosing Schedules for HPV Vaccines among Adolescent Girls and Young Women: A Systematic Review and Meta-Analysis. Vaccines, 8(4), 618. https://doi.org/10.3390/vaccines8040618






