Advances in the Development of Anti-Haemonchus contortus Vaccines: Challenges, Opportunities, and Perspectives
Abstract
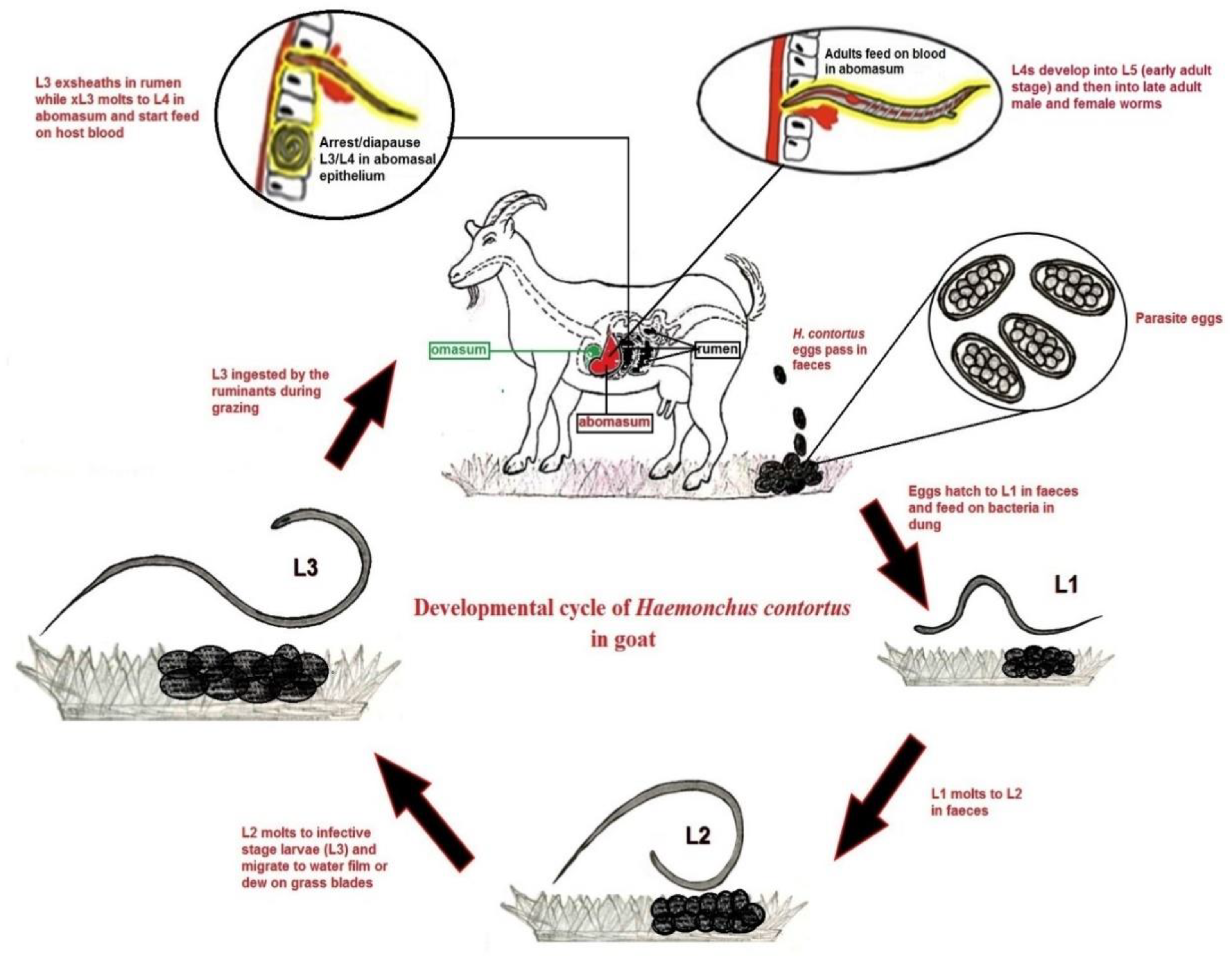
1. Introduction
2. Importance of Anti-H. contortus Vaccine
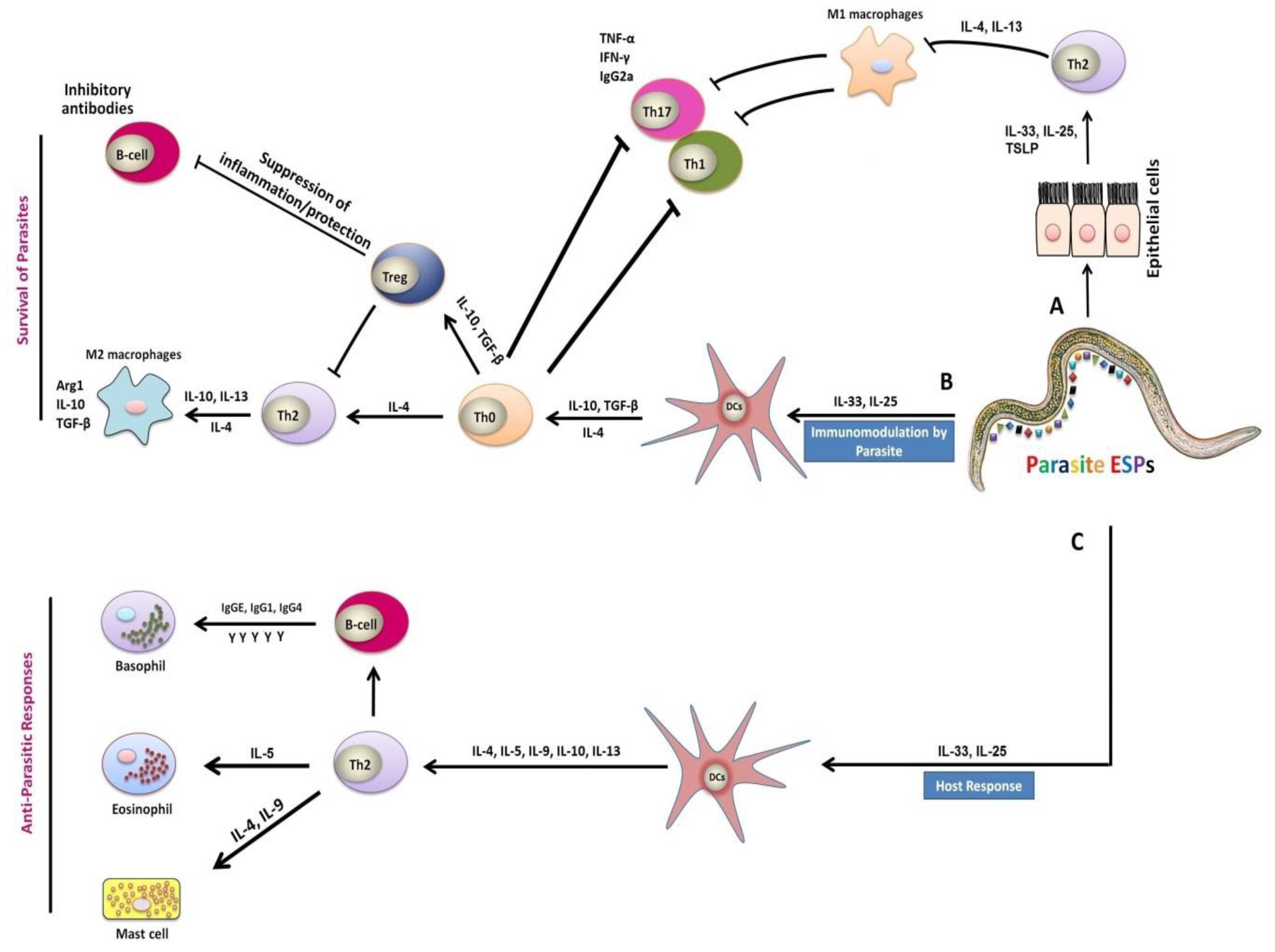
3. Mechanisms of Helminths-Associated Immunity
4. Genomic and Proteomic Profile Exploited from H. contortus
5. Historical Account of H. contortus Vaccines
5.1. Vaccinations with Gut-Derived/Hidden Antigens
5.2. DNA-Based Vaccination
5.3. Protein-Based Vaccination
6. Binding Proteins as Vaccine Antigens and Their Immunological Aspects
7. Challenges to the Development of Effective Vaccines
7.1. Diversity Within H. contortus Parasite
7.2. Host-Related Genetic Diversity
7.3. Multistage Complexity of Parasite
7.4. Composition of Protective Vaccine
8. Future Perspectives
9. Concluding Remarks
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- O’Connor, L.J.; Walkden-Brown, S.W.; Kahn, L.P. Ecology of the free-living stages of major trichostrongylid parasites of sheep. Vet. Parasitol. 2006, 142, 1–15. [Google Scholar] [CrossRef]
- Roeber, F.; Jex, A.R.; Gasser, R.B. Impact of gastrointestinal parasitic nematodes of sheep, and the role of advanced molecular tools for exploring epidemiology and drug resistance—An Australian perspective. Parasites Vectors 2013, 6, 153. [Google Scholar] [CrossRef]
- Emery, D.L.; Hunt, P.W.; Le Jambre, L.F. Haemonchus contortus: The then and now, and where to from here? Int. J. Parasitol. 2016, 46, 755–769. [Google Scholar] [CrossRef]
- Peter, J.W.; Chandrawathani, P. Haemonchus contortus: Parasite problem No. 1 from tropics—Polar Circle. Problems and prospects for control based on epidemiology. Trop. Biomed. 2005, 22, 131–137. [Google Scholar]
- Roos, M.H. Drug Resistance in the sheep nematode parasite Haemonchus contortus, mechanisms and clinical perspectives. In Antimicrobial Drug Resistance: Clinical and Epidemiological Aspects; Humana Press: Totowa, NJ, USA, 2009; pp. 1127–1132. [Google Scholar] [CrossRef]
- Saddiqi, H.A.; Riaz, N.; Sarwar, M.; Iqbal, Z.; Muhammad, G.; Nisa, M.; Shahzad, A. Small ruminant resistance against gastrointestinal nematodes: A case of Haemonchus contortus. Parasitol. Res. 2011, 109, 1483–1500. [Google Scholar] [CrossRef]
- Kaplan, R.M.; Vidyashankar, A.N. An inconvenient truth: Global worming and anthelmintic resistance. Vet. Parasitol. 2012, 186, 70–78. [Google Scholar] [CrossRef]
- Kaplan, R.M. Drug resistance in nematodes of veterinary importance: A status report. Trends Parasitol. 2004, 20, 477–481. [Google Scholar] [CrossRef]
- Prichard, R. Drug resistance in nematodes. In Antimicrobial Drug Resistance: Mechanisms of Drug Resistance; Mc Gill University: Sainte-Anne-de-Bellevue, QC, Canada, 2009; pp. 621–628. [Google Scholar] [CrossRef]
- Nisbet, A.J.; Meeusen, E.N.T.; González, J.; Piedrafita, D. Immunity to Haemonchus contortus and vaccine development. Adv. Parasitol. 2016, 93, 353–396. [Google Scholar] [CrossRef]
- Michel, J. Arrested development of nematodes and some related phenomena. Adv. Parasitol. 1974, 12, 279–366. [Google Scholar] [CrossRef]
- Waller, P.; Rudby-Martin, L.; Ljungström, B.; Rydzik, A. The epidemiology of abomasal nematodes of sheep in Sweden, with particular reference to over-winter survival strategies. Vet. Parasitol. 2004, 122, 207–220. [Google Scholar] [CrossRef]
- Knox, D.P.; Redmond, D.L.; Newlands, G.F.; Skuce, P.J.; Pettit, D.; Smith, W.D. The nature and prospects for gut membrane proteins as vaccine candidates for Haemonchus contortus and other ruminant trichostrongyloids. Int. J. Parasitol. 2003, 33, 1129–1137. [Google Scholar] [CrossRef]
- Tak, I.R.; Dar, J.S.; Dar, S.A.; Ganai, B.A.; Chishti, M.Z.; Ahmad, F. A comparative analysis of various antigenic proteins found in Haemonchus contortus—A review. Mol. Biol. 2015, 49, 789–795. [Google Scholar] [CrossRef]
- Wang, C.; Li, F.; Zhang, Z.; Yang, X.; Ahmad, A.A.; Li, X.; Du, A.; Hu, M. Recent research progress in China on Haemonchus contortus. Front. Microbiol. 2017, 8. [Google Scholar] [CrossRef]
- Boisvenue, R.J.; Stiff, M.I.; Tonkinson, L.V.; Cox, G.N.; Hageman, R. Fibrinogen-degrading proteins from Haemonchus contortus used to vaccinate sheep. Am. J. Vet. Res. 1992, 53, 1263–1265. [Google Scholar]
- Smith, T.S.; Graham, M.; Munn, E.A.; Newton, S.E.; Knox, D.P.; Coadwell, W.; McMichael-Phillips, D.; Smith, H.; Smith, W.; Oliver, J.J. Cloning and characterization of a microsomal aminopeptidase from the intestine of the nematode Haemonchus contortus. Biochimica Biophysica Acta (BBA) 1997, 1338, 295–306. [Google Scholar] [CrossRef]
- Smith, W.D.; Smith, S.K.; Murray, J.M. Protection studies with integral membrane fractions of Haemonchus contortus. Parasite Immunol. 1994, 16, 231–241. [Google Scholar] [CrossRef]
- Redmond, D.L.; Knox, D.P.; Newlands, G.; Smith, W.D. Molecular cloning and characterisation of a developmentally regulated putative metallopeptidase present in a host protective extract of Haemonchus contortus. Mol. Biochem. Parasitol. 1997, 85, 77–87. [Google Scholar] [CrossRef]
- Longbottom, D.; Redmond, D.L.; Russell, M.; Liddell, S.; Smith, W.D.; Knox, D.P. Molecular cloning and characterisation of a putative aspartate proteinase associated with a gut membrane protein complex from adult Haemonchus contortus. Mol. Biochem. Parasitol. 1997, 88, 63–72. [Google Scholar] [CrossRef]
- Vanhoy, G.; Carman, M.; Habing, G.; Lakritz, J.; Hinds, C.A.; Niehaus, A.; Kaplan, R.M.; Marsh†, A.E. Safety and serologic response to a Haemonchus contortus vaccine in alpacas. Vet. Parasitol. 2018, 252, 180–186. [Google Scholar] [CrossRef]
- Bassetto, C.; Almeida, F.; Newlands, G.F.J.; Smith, W.; Castilhos, A.; Fernandes, S.; Siqueira, E.; Amarante, A. Trials with the Haemonchus vaccine, Barbervax®, in ewes and lambs in a tropical environment: Nutrient supplementation improves protection in periparturient ewes. Vet. Parasitol. 2018, 264, 52–57. [Google Scholar] [CrossRef]
- Hewitson, J.P.; Maizels, R.M. Vaccination against helminth parasite infections. Expert Rev. Vaccines 2014, 13, 473–487. [Google Scholar] [CrossRef]
- Lacroux, C.; Nguyen, T.H.C.; Andréoletti, O.; Prévot, F.; Grisez, C.; Bergeaud, J.-P.; Gruner, L.; Brunel, J.-C.; Francois, D.; Dorchies, P.; et al. Haemonchus contortus (Nematoda: Trichostrongylidae) infection in lambs elicits an unequivocal Th2 immune response. Vet. Res. 2006, 37, 607–622. [Google Scholar] [CrossRef]
- Shakya, K.; Miller, J.; Lomax, L.; Burnett, D. Evaluation of immune response to artificial infections of Haemonchus contortus in Gulf Coast Native compared with Suffolk lambs. Vet. Parasitol. 2011, 181, 239–247. [Google Scholar] [CrossRef]
- Balic, A.; Cunningham, C.P.; Meeusen, E.N.T. Eosinophil interactions with Haemonchus contortus larvae in the ovine gastrointestinal tract. Parasite Immunol. 2006, 28, 107–115. [Google Scholar] [CrossRef]
- Meeusen, E.N.T.; Balic, A.; Bowles, V.M. Cells, cytokines and other molecules associated with rejection of gastrointestinal nematode parasites. Vet. Immunol. Immunopathol. 2005, 108, 121–125. [Google Scholar] [CrossRef]
- Jones, W.; Emery, D.; McClure, S.; Wagland, B. Changes in inflammatory mediators and larval inhibitory activity in intestinal contents and mucus during primary and challenge infections of sheep with Trichostrongylus colubriformis. Int. J. Parasitol. 1994, 24, 519–525. [Google Scholar] [CrossRef]
- Emery, D. Vaccination against worm parasites of animals. Vet. Parasitol. 1996, 64, 31–45. [Google Scholar] [CrossRef]
- Anthony, R.M.; Urban, J.F.; Alem, F.; Hamed, H.A.; Rozo, C.T.; Boucher, J.-L.; Van Rooijen, N.; Gause, W.C. Memory TH2 cells induce alternatively activated macrophages to mediate protection against nematode parasites. Nat. Med. 2006, 12, 955–960. [Google Scholar] [CrossRef]
- Jackson, J.A.; Friberg, I.M.; Little, S.; Bradley, J.E. Review series on helminths, immune modulation and the hygiene hypothesis: Immunity against helminths and immunological phenomena in modern human populations: Coevolutionary legacies? Immunology 2009, 126, 18–27. [Google Scholar] [CrossRef]
- Maizels, R.M.; Yazdanbakhsh, M. Immune Regulation by helminth parasites: Cellular and molecular mechanisms. Nat. Rev. Immunol. 2003, 3, 733–744. [Google Scholar] [CrossRef]
- Wang, L.J.; Cao, Y.; Shi, H.N. Helminth infections and intestinal inflammation. World J. Gastroenterol. 2008, 14, 5125–5132. [Google Scholar] [CrossRef] [PubMed]
- Ditgen, D.; Anandarajah, E.M.; Meissner, K.A.; Brattig, N.; Wrenger, C.; Liebau, E. Harnessing the helminth secretome for therapeutic immunomodulators. Biomed Res. Int. 2014, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Anthony, R.M.; Rutitzky, L.I.; Urban, J.F.; Stadecker, M.J.; Gause, W.C. Protective immune mechanisms in helminth infection. Nat. Rev. Immunol. 2007, 7, 975–987. [Google Scholar] [CrossRef] [PubMed]
- Bricarello, P.; Gennari, S.M.; Oliveira-Sequeira, T.; Vaz, C.; De Gonçalves, I.G.; Echevarria, F. Worm burden and immunological responses in Corriedale and Crioula Lanada sheep following natural infection with Haemonchus contortus. Small Rumin. Res. 2004, 51, 75–83. [Google Scholar] [CrossRef]
- Wilkes, C.P.; Bleay, C.; Paterson, S.; Viney, M. The immune response during a Strongyloides ratti infection of rats. Parasite Immunol. 2007, 29, 339–346. [Google Scholar] [CrossRef] [PubMed]
- Paterson, S.; Wilkes, C.; Bleay, C.; Viney, M.E. Immunological responses elicited by different infection regimes with Strongyloides ratti. PLoS ONE 2008, 3, e2509. [Google Scholar] [CrossRef]
- Eschbach, M.-L.; Klemm, U.; Kolbaum, J.; Blankenhaus, B.; Brattig, N.; Breloer, M. Strongyloides ratti infection induces transient nematode-specific Th2 response and reciprocal suppression of IFN-γ production in mice. Parasite Immunol. 2010, 32, 370–383. [Google Scholar] [CrossRef]
- Ferreira, I.; Smyth, D.J.; Gaze, S.; Aziz, A.; Giacomin, P.R.; Ruyssers, N.; Artis, D.; Laha, T.; Navarro, S.; Loukas, A.; et al. Hookworm excretory/secretory products induce interleukin-4 (IL-4)+IL-10+CD4+T cell responses and suppress pathology in a mouse model of colitis. Infect. Immun. 2013, 81, 2104–2111. [Google Scholar] [CrossRef]
- Aranzamendi, C.; Franssen, F.; Rutten, V.P.M.G.; Fransen, F.; Langelaar, M.F.M.; Van Der Ley, P.; Van Putten, J.P.M.; Pinelli, E. Trichinella spiralis—Secreted products modulate DC functionality and expand regulatory T cells in vitro. Parasite Immunol. 2012, 34, 210–223. [Google Scholar] [CrossRef]
- Abdoli, A.; Ardakani, H.M. Helminth infections and immunosenescence: The friend of my enemy. Exp. Gerontol. 2020, 133, 110852. [Google Scholar] [CrossRef]
- Mendlovic, F.; Flisser, A. Dendritic cells in the gut: Interaction with intestinal helminths. J. Biomed. Biotechnol. 2010, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Schwarz, E.M.; Korhonen, P.K.; Campbell, B.E.; Young, N.D.; Jex, A.R.; Riaz, N.; Hall, R.S.; Mondal, A.; Howe, A.; Pell, J.; et al. The genome and developmental transcriptome of the strongylid nematode Haemonchus contortus. Genome Boil. 2013, 14, R89. [Google Scholar] [CrossRef] [PubMed]
- Yatsuda, A.; Krijgsveld, J.; Cornelissen, A.W.C.A.; Heck, A.J.R.; De Vries, E. Comprehensive analysis of the secreted proteins of the parasite Haemonchus contortus reveals extensive sequence variation and differential immune recognition. J. Boil. Chem. 2003, 278, 16941–16951. [Google Scholar] [CrossRef] [PubMed]
- Rhoads, M.; Fetterer, R. Developmentally regulated secretion of cathepsin L-like cysteine proteases by Haemonchus contortus. J. Parasitol. 1995, 81, 505–512. [Google Scholar] [CrossRef]
- Shompole, S.; Jasmer, D.P. Cathepsin B-like cysteine proteases confer intestinal cysteine protease activity in Haemonchus contortus. J. Boil. Chem. 2000, 276, 2928–2934. [Google Scholar] [CrossRef]
- Karanu, F.; Rurangirwa, F.; McGuire, T.; Jasmer, D. Haemonchus contortus: Identification of proteases with diverse characteristics in adult worm excretory-secretory products. Exp. Parasitol. 1993, 77, 362–371. [Google Scholar] [CrossRef]
- Schallig, H.D.; Van Leeuwen, M.A.; Cornelissen, A.W. Protective immunity induced by vaccination with two Haemonchus contortus excretory secretory proteins in sheep. Parasite Immunol. 1997, 19, 447–453. [Google Scholar] [CrossRef]
- Dicker, A.J.; Inglis, N.F.; Manson, E.D.T.; Subhadra, S.; Illangopathy, M.; Muthusamy, R.; Knox, D.P. Proteomic analysis of Mecistocirrus digitatus and Haemonchus contortus intestinal protein extracts and subsequent efficacy testing in a vaccine trial. PLoS Neglected Trop. Dis. 2014, 8, e2909. [Google Scholar] [CrossRef]
- Nagaraj, S.H.; Harsha, H.; Reverter, A.; Colgrave, M.L.; Sharma, R.; Andronicos, N.; Hunt, P.; Menzies, M.; Lees, M.S.; Sekhar, N.R.; et al. Proteomic analysis of the abomasal mucosal response following infection by the nematode, Haemonchus contortus, in genetically resistant and susceptible sheep. J. Proteom. 2012, 75, 2141–2152. [Google Scholar] [CrossRef]
- Wang, F.; Xu, L.; Song, X.; Li, X.; Yan, R. Identification of differentially expressed proteins between free-living and activated third-stage larvae of Haemonchus contortus. Vet. Parasitol. 2016, 215, 72–77. [Google Scholar] [CrossRef]
- Gadahi, J.A.; Wang, S.; Bo, G.; Ehsan, M.; Yan, R.; Song, X.; Xu, L.; Li, X. Proteomic analysis of the excretory and secretory proteins of Haemonchus contortus (HcESP) binding to goat PBMCs in vivo revealed stage-specific binding profiles. PLoS ONE 2016, 11, e0159796. [Google Scholar] [CrossRef] [PubMed]
- Yan, F.; Xu, L.; Liu, L.; Yan, R.; Song, X.; Li, X. Immunoproteomic analysis of whole proteins from male and female adult Haemonchus contortus. Vet. J. 2010, 185, 174–179. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.; Ma, G.; Ang, C.-S.; Korhonen, P.K.; Xu, R.; Nie, S.; Koehler, A.V.; Simpson, R.J.; Greening, D.W.; Reid, G.E.; et al. Somatic proteome of Haemonchus contortus. Int. J. Parasitol. 2019, 49, 311–320. [Google Scholar] [CrossRef]
- Wang, T.; Ma, G.; Ang, C.-S.; Korhonen, P.K.; Koehler, A.V.; Young, N.D.; Nie, S.; Williamson, N.A.; Gasser, R.B. High throughput LC-MS/MS-based proteomic analysis of excretory-secretory products from short-term in vitro culture of Haemonchus contortus. J. Proteom. 2019, 204, 103375. [Google Scholar] [CrossRef] [PubMed]
- Smith, W.; Zarlenga, D. Developments and hurdles in generating vaccines for controlling helminth parasites of grazing ruminants. Vet. Parasitol. 2006, 139, 347–359. [Google Scholar] [CrossRef]
- Smith, S.K.; Smith, W.D. Immunisation of sheep with an integral membrane glycoprotein complex of Haemonchus contortus and with its major polypeptide components. Res. Vet. Sci. 1996, 60, 1–6. [Google Scholar] [CrossRef]
- Yanming, S.; Ruofeng, Y.; Muleke, C.I.; Guangwei, Z.; Lixin, X.; Li, X. Vaccination of goats with recombinant galectin antigen induces partial protection against Haemonchus contortus infection. Parasite Immunol. 2007, 29, 319–326. [Google Scholar] [CrossRef]
- Roberts, B.; Antonopoulos, A.; Haslam, S.M.; Dicker, A.J.; McNeilly, T.N.; Johnston, S.L.; Dell, A.; Knox, D.; Britton, C. Novel expression of Haemonchus contortus vaccine candidate aminopeptidase H11 using the free-living nematode Caenorhabditis elegans. Vet. Res. 2013, 44, 111. [Google Scholar] [CrossRef]
- Smith, S.; Pettit, D.; Newlands, G.; Redmond, D.; Skuce, P.; Knox, D.; Smith, W. Further immunization and biochemical studies with a protective antigen complex from the microvillar membrane of the intestine of Haemonchus contortus. Parasite Immunol. 1999, 21, 187–199. [Google Scholar] [CrossRef]
- Zhao, G.; Yan, R.; Muleke, C.I.; Sun, Y.; Xu, L.; Li, X. Vaccination of goats with DNA vaccines encoding H11 and IL-2 induces partial protection against Haemonchus contortus infection. Vet. J. 2012, 191, 94–100. [Google Scholar] [CrossRef]
- Sun, W.; Song, X.; Yan, R.; Xu, L.; Li, X. Vaccination of goats with a glutathione peroxidase DNA vaccine induced partial protection against Haemonchus contortus infection. Vet. Parasitol. 2011, 182, 239–247. [Google Scholar] [CrossRef] [PubMed]
- Han, K.; Xu, L.; Yan, R.; Song, X.; Li, X. Vaccination of goats with glyceraldehyde-3-phosphate dehydrogenase DNA vaccine induced partial protection against Haemonchus contortus. Vet. Immunol. Immunopathol. 2012, 149, 177–185. [Google Scholar] [CrossRef] [PubMed]
- Yan, R.; Sun, W.; Song, X.; Xu, L.; Li, X. Vaccination of goats with DNA vaccine encoding Dim-1 induced partial protection against Haemonchus contortus: A preliminary experimental study. Res. Vet. Sci. 2013, 95, 189–199. [Google Scholar] [CrossRef] [PubMed]
- Rathore, D.; Suchitra, S.; Saini, M.; Singh, B.; Joshi, P. Identification of a 66 kDa Haemonchus contortus excretory/secretory antigen that inhibits host monocytes. Vet. Parasitol. 2006, 138, 291–300. [Google Scholar] [CrossRef]
- Marcilla, A.; Trelis, M.; Cortés, A.; Sotillo, J.; Cantalapiedra, F.; Minguez, M.T.; Valero, M.L.; Del Pino, M.M.S.; Muñoz-Antoli, C.; Toledo, R.; et al. Extracellular vesicles from parasitic helminths contain specific excretory/secretory proteins and are internalized in intestinal host cells. PLoS ONE 2012, 7, e45974. [Google Scholar] [CrossRef]
- Nagaraj, S.H.; Gasser, R.B.; Ranganathan, S. Needles in the EST haystack: Large-scale identification and analysis of Excretory-Secretory (ES) proteins in parasitic nematodes using Expressed Sequence Tags (ESTs). PLoS Neglected Trop. Dis. 2008, 2, e301. [Google Scholar] [CrossRef]
- Munn, E.A. Rational design of nematode vaccines: Hidden antigens. Int. J. Parasitol. 1997, 27, 359–366. [Google Scholar] [CrossRef]
- Schallig, H.D.F.H.; Van Leeuwen, M.A.W. Protective immunity to the blood-feeding nematode Haemonchus contortus induced by vaccination with parasite low molecular weight antigens. Parasitology 1997, 114, 293–299. [Google Scholar] [CrossRef]
- Jasmer, D.P.; McGuire, T.C. Protective immunity to a blood-feeding nematode (Haemonchus contortus) induced by parasite gut antigens. Infect. Immun. 1991, 59, 4412–4417. [Google Scholar] [CrossRef]
- Andrews, S.; Rolph, T.; Munn, E.; Taylort, M. Duration of protective immunity against ovine haemonchosis following vaccination with the nematode gut membrane antigen H11. Res. Vet. Sci. 1997, 62, 223–227. [Google Scholar] [CrossRef]
- Bu, Y.; Jia, C.; Tian, X.; Aimulajiang, K.; Memon, M.A.; Yan, R.; Song, X.; Xu, L.; Li, X. Immunization of goats with recombinant protein 14-3-3 isoform 2(rHcftt-2) induced moderate protection against Haemonchus contortus challenge. Pathogens 2020, 9, 46. [Google Scholar] [CrossRef] [PubMed]
- Cox, G.N.; Pratt, D.; Hageman, R.; Boisvenue, R.J. Molecular cloning and primary sequence of a cysteine protease expressed by Haemonchus contortus adult worms. Mol. Biochem. Parasitol. 1990, 41, 25–34. [Google Scholar] [CrossRef]
- Anbu, K.A.; Joshi, P. Identification of a 55 kDa Haemonchus contortus excretory/secretory glycoprotein as a neutrophil inhibitory factor. Parasite Immunol. 2007, 30, 23–30. [Google Scholar] [CrossRef]
- Reinhardt, S.; Scott, I.; Simpson, H.V. Neutrophil and eosinophil chemotactic factors in the excretory/secretory products of sheep abomasal nematode parasites: NCF and ECF in abomasal nematodes. Parasitol. Res. 2011, 109, 627–635. [Google Scholar] [CrossRef] [PubMed]
- Merkelbach, P.; Scott, I.; Khalaf, S.; Simpson, H.V. Excretory/secretory products of Haemonchus contortus inhibit aminopyrine accumulation by rabbit gastric glands in vitro. Vet. Parasitol. 2002, 104, 217–228. [Google Scholar] [CrossRef]
- Wang, W.; Wang, S.; Zhang, H.; Yuan, C.; Yan, R.; Song, X.; Xu, L.; Li, X. Galectin Hco-gal-m from Haemonchus contortus modulates goat monocytes and T cell function in different patterns. Parasites Vectors 2014, 7, 1–12. [Google Scholar] [CrossRef]
- Sun, Y.; Yan, R.; Muleke, C.I.; Zhao, G.; Xu, L.; Li, X. Recombinant Galectins of Haemonchus contortus parasite induces apoptosis in the peripheral blood lymphocytes of goat. Int. J. Pept. Res. Ther. 2006, 13, 387–392. [Google Scholar] [CrossRef]
- Wang, W.; Yuan, C.; Wang, S.; Song, X.; Xu, L.; Yan, R.; Hasson, I.; Li, X. Transcriptional and proteomic analysis reveal recombinant galectins of Haemonchus contortus down-regulated functions of goat PBMC and modulation of several signaling cascades in vitro. J. Proteom. 2014, 98, 123–137. [Google Scholar] [CrossRef]
- Li, Y.; Yuan, C.; Wang, L.; Lu, M.; Wang, Y.; Wen, Y.; Yan, R.; Xu, L.; Song, X.; Li, X. Transmembrane protein 147 (TMEM147): Another partner protein of Haemonchus contortus galectin on the goat peripheral blood mononuclear cells (PBMC). Parasites Vectors 2016, 9, 355. [Google Scholar] [CrossRef][Green Version]
- Yuan, C.; Zhang, H.; Wang, W.; Li, Y.; Yan, R.; Xu, L.; Song, X.; Li, X. Transmembrane protein 63A is a partner protein of Haemonchus contortus galectin in the regulation of goat peripheral blood mononuclear cells. Parasites Vectors 2015, 8, 211. [Google Scholar] [CrossRef][Green Version]
- Lu, M.; Tian, X.; Yang, X.; Yuan, C.; Ehsan, M.; Liu, X.; Yan, R.; Xu, L.; Song, X.; Li, X. The N- and C-terminal carbohydrate recognition domains of Haemonchus contortus galectin bind to distinct receptors of goat PBMC and contribute differently to its immunomodulatory functions in host-parasite interactions. Parasites Vectors 2017, 10, 409. [Google Scholar] [CrossRef] [PubMed]
- Gadahi, J.A.; Yongqian, B.; Ehsan, M.; Zhang, Z.C.; Wang, S.; Yan, R.F.; Song, X.K.; Xu, L.X.; Li, X. Haemonchus contortus excretory and secretory proteins (HcESPs) suppress functions of goat PBMCs in vitro. Oncotarget 2016, 7, 35670–35679. [Google Scholar] [CrossRef] [PubMed]
- Gadahi, J.A.; Ehsan, M.; Wang, S.; Zhang, Z.; Wang, Y.; Yan, R.; Song, X.; Xu, L.; Li, X. Recombinant protein of Haemonchus contortus 14-3-3 isoform 2 (rHcftt-2) decreased the production of IL-4 and suppressed the proliferation of goat PBMCs in vitro. Exp. Parasitol. 2016, 171, 57–66. [Google Scholar] [CrossRef] [PubMed]
- Gadahi, J.A.; Li, B.; Ehsan, M.; Wang, S.; Zhang, Z.; Wang, Y.; Hasan, M.W.; Yan, R.; Song, X.; Xu, L.; et al. Recombinant Haemonchus contortus 24 kDa excretory/secretory protein (rHcES-24) modulate the immune functions of goat PBMCs in vitro. Oncotarget 2016, 7, 83926–83937. [Google Scholar] [CrossRef] [PubMed]
- Ehsan, M.; Gadahi, J.A.; Hasan, M.W.; Haseeb, M.; Ali, H.; Yan, R.; Xu, L.; Song, X.; Zhu, X.-Q.; Li, X. Characterization of Haemonchus contortus excretory/secretory antigen (ES-15) and its modulatory functions on goat immune cells in vitro. Pathogens 2020, 9, 162. [Google Scholar] [CrossRef]
- Ehsan, M.; Gao, W.; Gadahi, J.A.; Lu, M.; Liu, X.; Wang, Y.; Yan, R.; Xu, L.; Song, X.; Li, X. Arginine kinase from Haemonchus contortus decreased the proliferation and increased the apoptosis of goat PBMCs in vitro. Parasites Vectors 2017, 10, 311. [Google Scholar] [CrossRef]
- Ehsan, M.; Wang, W.; Gadahi, J.A.; Hasan, M.W.; Lu, M.; Wang, Y.; Liu, X.; Haseeb, M.; Yan, R.; Xu, L.; et al. The serine/threonine-protein phosphatase 1 from Haemonchus contortus is actively involved in suppressive regulatory roles on immune functions of goat peripheral blood mononuclear cells. Front. Immunol. 2018, 9. [Google Scholar] [CrossRef]
- Wen, Y.; Wang, Y.; Wang, W.; Lu, M.; Ehsan, M.; Tian, X.; Yan, R.; Song, X.; Xu, L.; Li, X. Recombinant Miro domain-containing protein of Haemonchus contortus (rMiro-1) activates goat peripheral blood mononuclear cells in vitro. Vet. Parasitol. 2017, 243, 100–104. [Google Scholar] [CrossRef]
- Gadahi, J.A.; Ehsan, M.; Wang, S.; Zhang, Z.; Yan, R.; Song, X.; Xu, L.; Li, X. Recombinant protein of Haemonchus contortus small GTPase ADP-ribosylation factor 1 (HcARF1) modulate the cell mediated immune response in vitro. Oncotarget 2017, 8, 112211–112221. [Google Scholar] [CrossRef]
- Ehsan, M.; Gadahi, J.A.; Lu, M.; Yan, R.; Xu, L.; Song, X.; Zhu, X.-Q.; Du, A.; Hu, M.; Li, X. Recombinant elongation factor 1 alpha of Haemonchus contortus affects the functions of goat PBMCs. Parasite Immunol. 2020, 42, e12703. [Google Scholar] [CrossRef]
- Ehsan, M.; Haseeb, M.; Hu, R.-S.; Ali, H.; Memon, M.A.; Yan, R.; Xu, L.; Song, X.; Zhu, X.-Q.; Li, X. Tropomyosin: An excretory/secretory protein from Haemonchus contortus mediates the immuno-suppressive potential of goat peripheral blood mononuclear cells in vitro. Vaccines 2020, 8, 109. [Google Scholar] [CrossRef] [PubMed]
- Ehsan, M.; Gadahi, J.A.; Liu, T.; Lu, M.; Wang, Y.; Hasan, M.W.; Haseeb, M.; Yan, R.; Xu, L.; Song, X.; et al. Identification of a novel methyltransferase-type 12 protein from Haemonchus contortus and its effects on functions of goat PBMCs. Parasites Vectors 2020, 13, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Wu, L.; Liu, X.; Wang, S.; Ehsan, M.; Yan, R.; Song, X.; Xu, L.; Li, X. Characterization of a secreted cystatin of the parasitic nematode Haemonchus contortus and its immune-modulatory effect on goat monocytes. Parasites Vectors 2017, 10, 425. [Google Scholar] [CrossRef] [PubMed]
- Li, B.; Gadahi, J.A.; Gao, W.; Zhang, Z.; Ehsan, M.; Xu, L.; Song, X.; Li, X.; Yan, R. Characterization of a novel aspartyl protease inhibitor from Haemonchus contortus. Parasites Vectors 2017, 10, 191. [Google Scholar] [CrossRef]
- Charlesworth, B. Effective population size and patterns of molecular evolution and variation. Nat. Rev. Genet. 2009, 10, 195–205. [Google Scholar] [CrossRef]
- Gilleard, J.S.; Redman, E. Genetic diversity and population structure of Haemonchus contortus. Adv. Parasitol. 2016, 93, 31–68. [Google Scholar] [CrossRef]
- Redman, E.; Packard, E.; Grillo, V.; Smith, J.; Jackson, F.; Gilleard, J.S. Microsatellite analysis reveals marked genetic differentiation between Haemonchus contortus laboratory isolates and provides a rapid system of genetic fingerprinting. Int. J. Parasitol. 2008, 38, 111–122. [Google Scholar] [CrossRef]
- Hunt, P.; Knox, M.; Le Jambre, L.; McNally, J.; Anderson, L. Genetic and phenotypic differences between isolates of Haemonchus contortus in Australia. Int. J. Parasitol. 2008, 38, 885–900. [Google Scholar] [CrossRef]
- Poeschel, G.P.; Todd, A.C. Disease-producing capacity of Haemonchus contortus isolates in sheep. Am. J. Vet. Res. 1972, 33, 2207–2213. [Google Scholar]
- Maizels, R.M.; Kurniawan, A. Variation and polymorphism in helminth parasites. Parasitology 2002, 125, S25–S37. [Google Scholar] [CrossRef]
- Willadsen, P. Antigen cocktails: Valid hypothesis or unsubstantiated hope? Trends Parasitol. 2008, 24, 164–167. [Google Scholar] [CrossRef] [PubMed]
- Lightowlers, M.W.; Colebrook, A.; Gauci, C.G.; Gauci, S.; Kyngdon, C.; Monkhouse, J.; Rodriquez, C.V.; Read, A.; Rolfe, R.; Sato, C. Vaccination against cestode parasites: Anti-helminth vaccines that work and why. Vet. Parasitol. 2003, 115, 83–123. [Google Scholar] [CrossRef]
- Barnes, E.; Dobson, R.; Barger, I. Worm control and anthelmintic resistance: Adventures with a model. Parasitol. Today 1995, 11, 56–63. [Google Scholar] [CrossRef]
- Ekroth, A.K.E.; Rafaluk-Mohr, C.; King, K.C. Host genetic diversity limits parasite success beyond agricultural systems: A meta-analysis. Proc. R. Soc. B Biol. Sci. 2019, 286, 20191811. [Google Scholar] [CrossRef] [PubMed]
- Whiteman, N.K.; Matson, K.D.; Bollmer, J.L.; Parker, P.G. Disease ecology in the Galápagos Hawk (Buteo galapagoensis): Host genetic diversity, parasite load and natural antibodies. Proc. R. Soc. Lond. B Boil. Sci. 2005, 273, 797–804. [Google Scholar] [CrossRef]
- Hoffmann, A.A.; Sgro, C.M. Climate change and evolutionary adaptation. Nature 2011, 470, 479–485. [Google Scholar] [CrossRef]
- Chauhan, N.; Tiwari, S.; Iype, T.; Jain, U. An overview of adjuvants utilized in prophylactic vaccine formulation as immunomodulators. Expert Rev. Vaccines 2017, 16, 491–502. [Google Scholar] [CrossRef]
- Reed, S.G.; Orr, M.T.; Fox, C.B. Key roles of adjuvants in modern vaccines. Nat. Med. 2013, 19, 1597–1608. [Google Scholar] [CrossRef]
- Stutzer, C.; Richards, S.A.; Ferreira, M.; Baron, S.; Maritz-Olivier, C. Metazoan parasite vaccines: Present status and future prospects. Front. Microbiol. 2018, 8, 67. [Google Scholar] [CrossRef]
- Cauwelaert, N.D.; Desbien, A.L.; Hudson, T.E.; Pine, S.O.; Reed, S.G.; Coler, R.N.; Orr, M.T. The TLR4 agonist vaccine adjuvant, GLA-SE, requires canonical and atypical mechanisms of action for TH1 induction. PLoS ONE 2016, 11, e0146372. [Google Scholar] [CrossRef]
- Petermann, J.; Bonnefond, R.; Mermoud, I.; Rantoen, D.; Meynard, L.; Munro, C.; Hüe, T.; Lua, L.H.L. Evaluation of three adjuvants with respect to both adverse effects and the efficacy of antibody production to the Bm86 protein. Exp. Appl. Acarol. 2017, 72, 303–315. [Google Scholar] [CrossRef] [PubMed]
- Himly, M.; Mills-Goodlet, R.; Geppert, M.; Duschl, A. Nanomaterials in the context of type 2 immune responses—Fears and potentials. Front. Immunol. 2017, 8, 471. [Google Scholar] [CrossRef] [PubMed]
- Fawzi, E.M.; González-Sánchez, M.E.; Corral, M.J.; Cuquerella, M.; Alunda, J.M. Vaccination of lambs against Haemonchus contortus infection with a somatic protein (Hc23) from adult helminths. Int. J. Parasitol. 2014, 44, 429–436. [Google Scholar] [CrossRef] [PubMed]
- Fawzi, E.M.; González-Sánchez, M.E.; Corral, M.J.; Alunda, J.M.; Cuquerella, M. Vaccination of lambs with the recombinant protein rHc23 elicits significant protection against Haemonchus contortus challenge. Vet. Parasitol. 2015, 211, 54–59. [Google Scholar] [CrossRef]
- González-Sánchez, M.E.; Cuquerella, M.; Alunda, J.M. Vaccination of lambs against Haemonchus contortus with the recombinant rHc23. Effect of adjuvant and antigen dose. PLoS ONE 2018, 13, e0193118. [Google Scholar] [CrossRef]
- Sheerin, D.; Openshaw, P.J.M.; Pollard, A.J. Issues in vaccinology: Present challenges and future directions. Eur. J. Immunol. 2017, 47, 2017–2025. [Google Scholar] [CrossRef]
| Antigen Name | Database ID | Life Stage | Host Immune Cells | Immune Functional Roles ↑(Increase), ↓(Decrease) | Ref. |
|---|---|---|---|---|---|
| rHco-gal-m | AY253330 | L4 to adult | Monocytes | ↑IL-10, ↓TNF-α, ↓MHCII, ↓apoptosis | [78] |
| T cells | ↓IL-6, ↓IL-10, ↓TNF-α, ↓proliferation, ↑apoptosis | ||||
| rHco-gal-m/f | AY253330/AY253331 | L4 to adult | PBMCs | ↓IL-2, ↓IL-4, ↓IL-6, ↓IL-8, ↑IL-10, ↓IFN-γ, ↓TNF-α, ↑TGF-β1, ↓migration | [80] |
| HcESPs | Whole proteins | L4 to adult | PBMCs | ↓IL-4, ↑IL-10, ↑IL-17, ↓IFN-γ, ↓proliferation, ↓NO, ↑migration | [84] |
| rHcES-15 | AY821552.1 | L4 to adult | PBMCs | ↑IL-4, ↑IL-10, ↑IL-17, ↓TGF-β1, ↓IFN-γ, ↑NO, ↑apoptosis, ↓migration, ↓proliferation | [87] |
| rHcES-24 | AY821551.1 | L4 to adult | PBMCs | ↑IL-4, ↑IL-10, ↑IL-17, ↓IFN-γ, ↑migration, ↓proliferation, ↓NO | [86] |
| rHcftt-2 | CDJ94531 | L4 to adult | PBMCs | ↓IL-4, ↑IL-10, ↑IL-17, ↑IFN-γ, ↑migration, ↓NO, ↓proliferation | [85] |
| rHc-AK | JX422018.1 | L4 to adult | PBMCs | ↑IL-4, ↑IL-10, ↑IL-17, ↑IFN-γ, ↓TGF-β1, ↑NO,↑apoptosis,↓migration, ↓proliferation | [88] |
| rHcSTP-1 | GQ280010.1 | Adults | PBMCs | ↑IL-2, ↓IL-10, ↑TGF-β1, ↑IFN-γ, ↑IL-17, ↑migration, ↑apoptosis, ↑NO, ↓proliferation | [89] |
| rHcARF1 | HF964523.1 | L4 to adult | PBMCs | ↑IL-4, ↑IL-10, ↑IL-17,↓IFN-γ, ↑NO, ↑migration, ↓proliferation | [91] |
| rMiro-1 | CDJ96345.1 | Adults | PBMCs | ↑IL-2, ↑IL-4, ↑IL-17, ↑migration, ↑NO, ↑proliferation | [90] |
| rHCcyst-3 | CDJ92568.1 | Eggs to adults | Monocytes | ↑IL-10, ↑TGF-β1, ↓TNF-α, ↓IL-1β, ↓IL-12p40, ↑NO, ↓phagocytosis, ↓MHCII | [95] |
| rHcAPI | KY284864.1 | Eggs to adults | PBMCs | ↑IL-4, ↑IL-10, ↑IFN-γ | [96] |
| rHcEF-1α | HF960353.1 | L4+L5 | PBMCs | ↑IL-4, ↑IL-17, ↑TGF-β1, ↑IFN-γ, ↓IL-10, ↑migration, ↑apoptosis, ↓NO, ↑proliferation | [92] |
| HcMT-12 | CDJ87424.1 | L4+L5 | PBMCs | ↑IL-2, ↑IL-4, ↓IL-10, ↑IFN-γ, ↓TGF-β1, ↑migration, ↑NO, ↓proliferation | [94] |
| rHc-TpMy | HF965396 | L4+L5 | PBMCs | ↓IL-4, ↓IFN-γ, ↑IL-10, ↑IL-17, ↑TGF-β1, ↓NO, ↓migration, ↑apoptosis, ↓proliferation | [93] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ehsan, M.; Hu, R.-S.; Liang, Q.-L.; Hou, J.-L.; Song, X.; Yan, R.; Zhu, X.-Q.; Li, X. Advances in the Development of Anti-Haemonchus contortus Vaccines: Challenges, Opportunities, and Perspectives. Vaccines 2020, 8, 555. https://doi.org/10.3390/vaccines8030555
Ehsan M, Hu R-S, Liang Q-L, Hou J-L, Song X, Yan R, Zhu X-Q, Li X. Advances in the Development of Anti-Haemonchus contortus Vaccines: Challenges, Opportunities, and Perspectives. Vaccines. 2020; 8(3):555. https://doi.org/10.3390/vaccines8030555
Chicago/Turabian StyleEhsan, Muhammad, Rui-Si Hu, Qin-Li Liang, Jun-Ling Hou, Xiaokai Song, Ruofeng Yan, Xing-Quan Zhu, and Xiangrui Li. 2020. "Advances in the Development of Anti-Haemonchus contortus Vaccines: Challenges, Opportunities, and Perspectives" Vaccines 8, no. 3: 555. https://doi.org/10.3390/vaccines8030555
APA StyleEhsan, M., Hu, R.-S., Liang, Q.-L., Hou, J.-L., Song, X., Yan, R., Zhu, X.-Q., & Li, X. (2020). Advances in the Development of Anti-Haemonchus contortus Vaccines: Challenges, Opportunities, and Perspectives. Vaccines, 8(3), 555. https://doi.org/10.3390/vaccines8030555







