Activation of OX40 and CD27 Costimulatory Signalling in Sheep through Recombinant Ovine Ligands
Abstract
1. Introduction
2. Materials and Methods
2.1. Cells and Viruses
2.2. Cloning and Generation of Recombinant Viruses
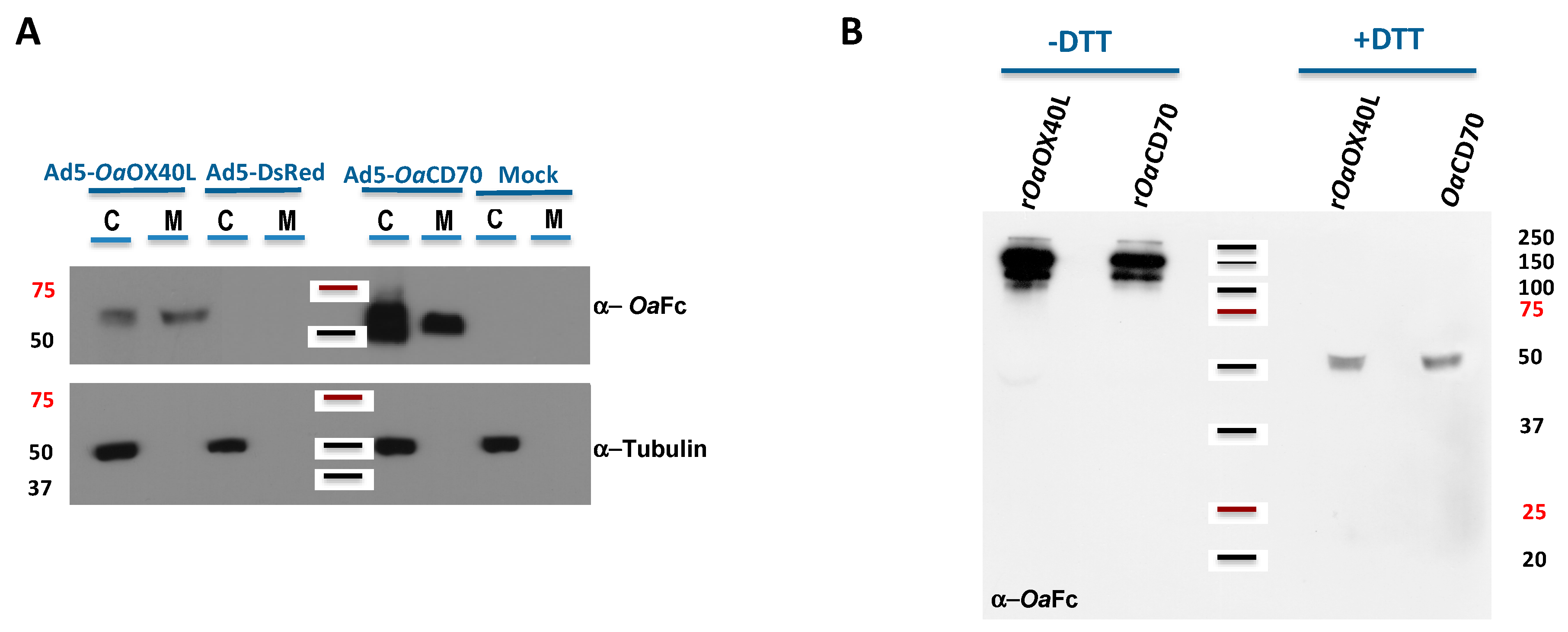
2.3. Protein Detection by Western Blot
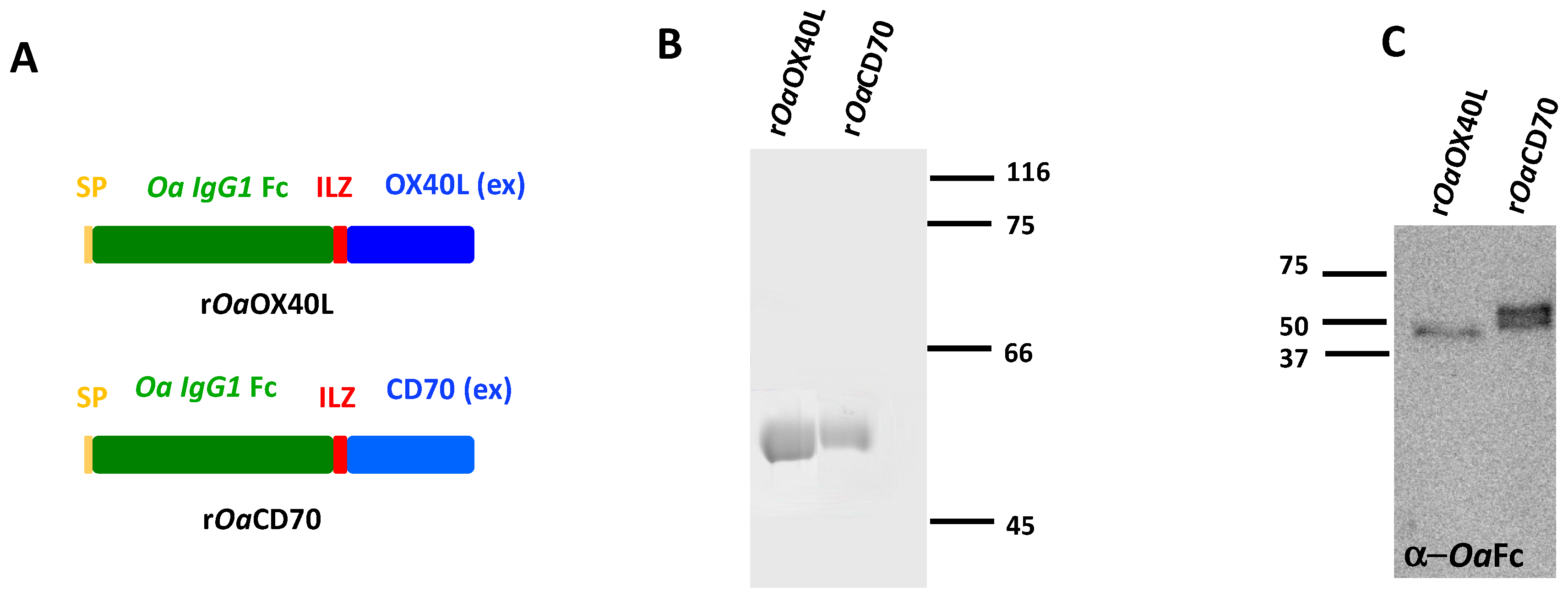
2.4. Purification of Recombinant Ovine CD70 (rOaCD70) and OX40L (rOaOX40L) Proteins
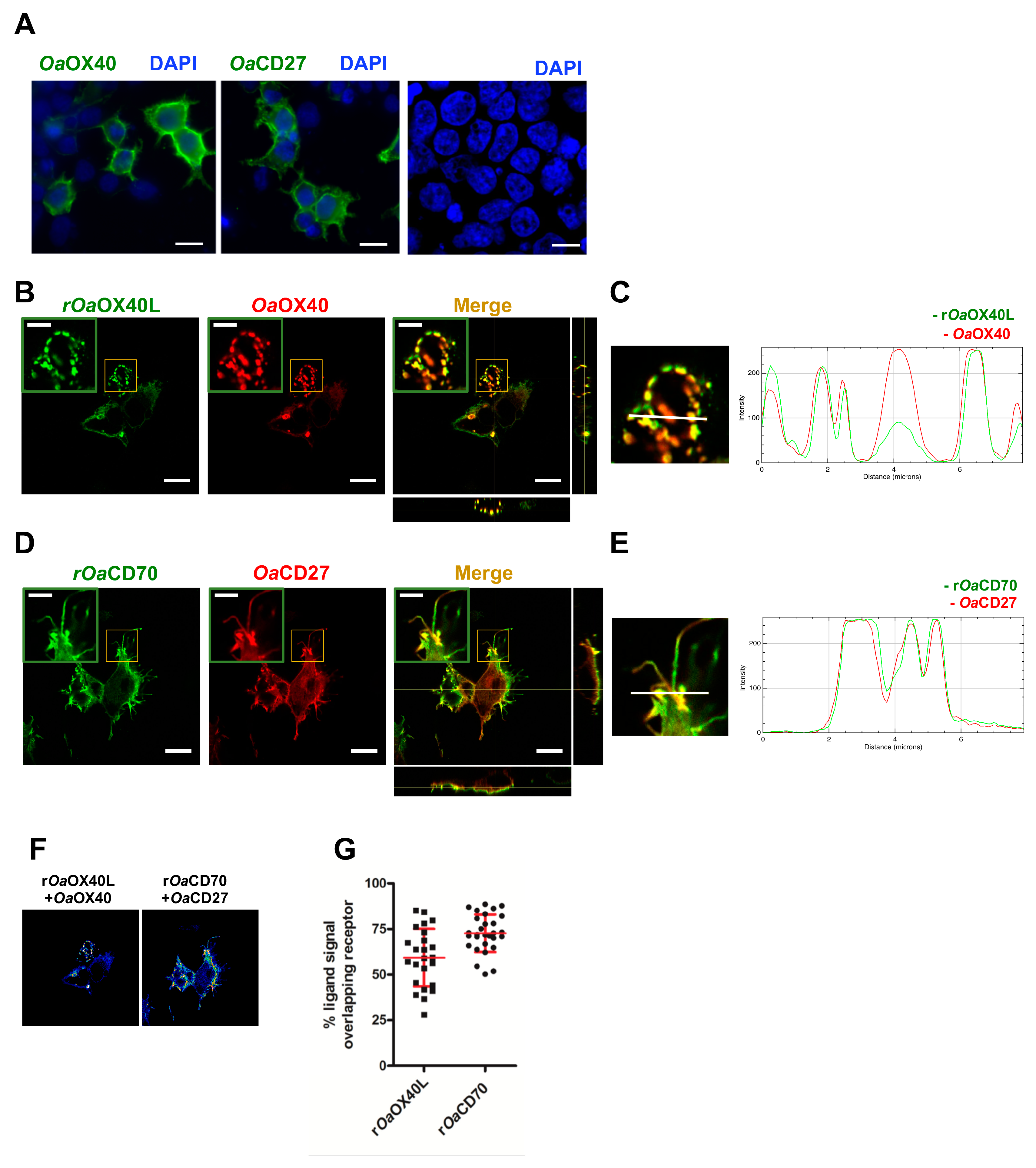
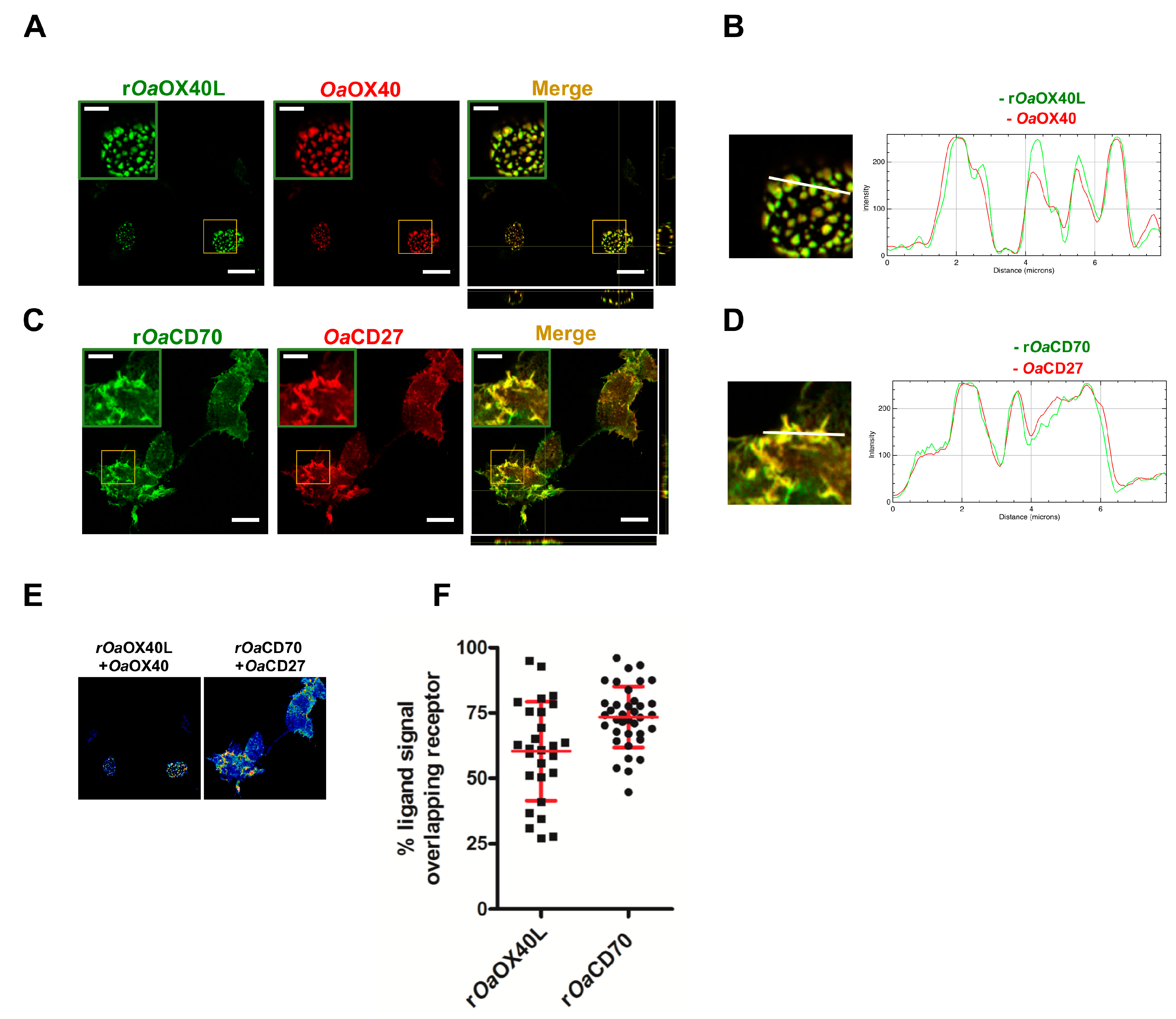
2.5. Immunofluorescence Microscopy
2.6. PBMC Isolation and T Cell Enrichment
2.7. CFSE Proliferation Assays
2.8. Intracellular Cytokine Staining and Flow Cytometry
2.9. Statistical Analyses
2.10. Ethics Statement
3. Results
3.1. Purification of Soluble Secreted Recombinant Ovine Proteins, rOaOX40L and rOaCD70
3.2. The rOaOX40L and rOaCD70 Proteins Bind to Cells Expressing Their Predicted Cognate Receptors
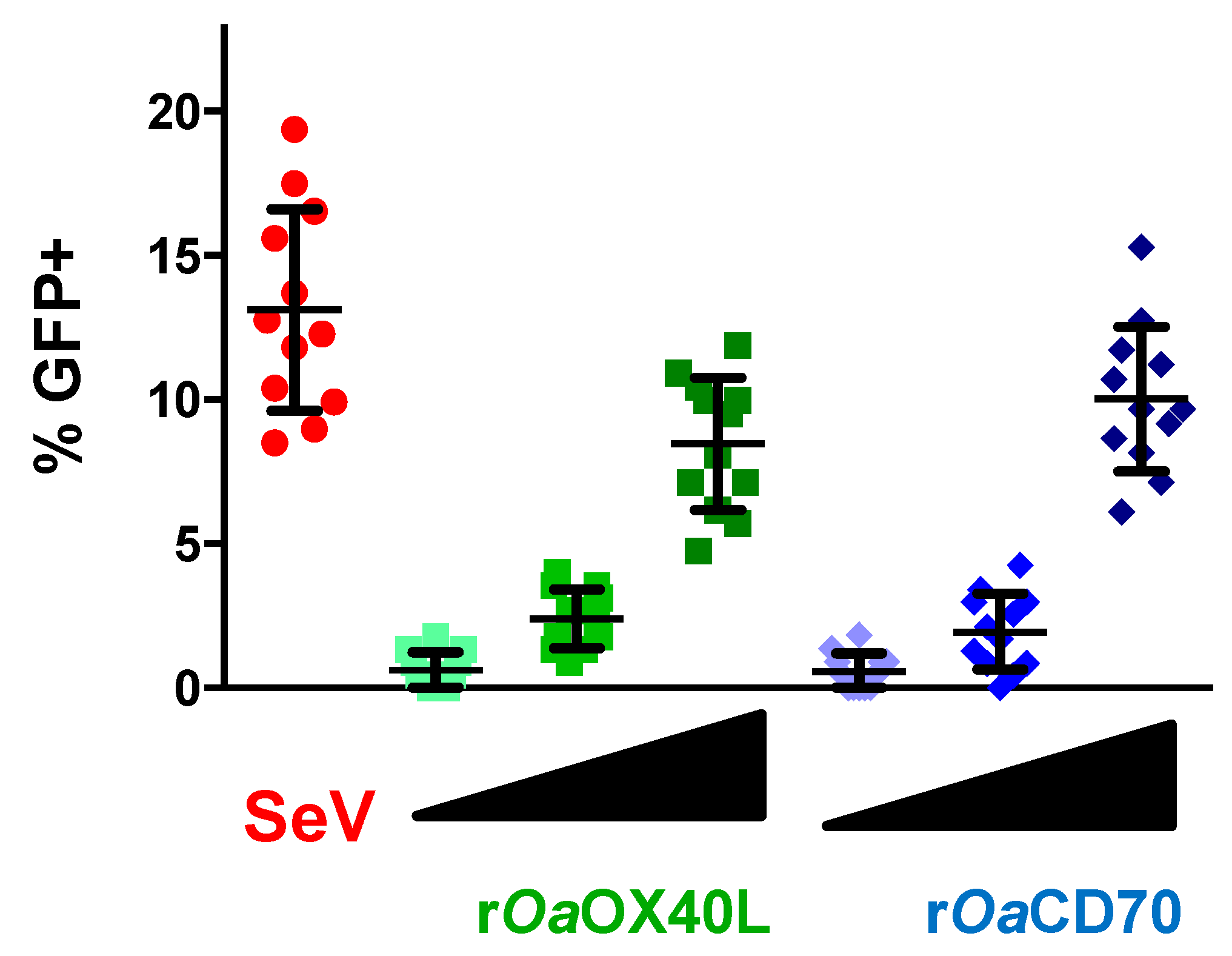
3.3. Recombinant Adenoviruses Express and Secrete rOaOX40L and rOaCD70 Proteins
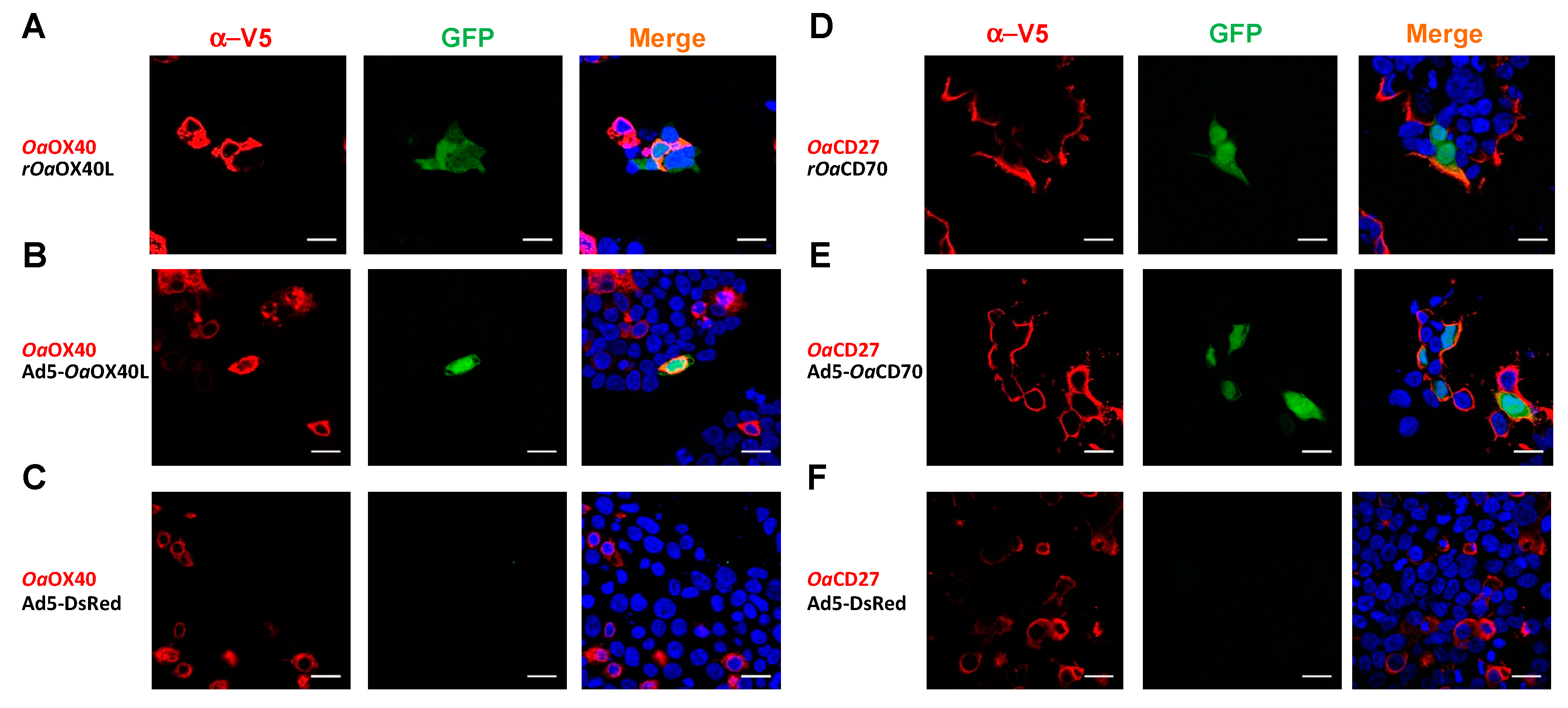
3.4. Ovine OaOX40 and OaCD27 Act as Cognate Signalling Receptors for rOaOX40L and rOaCD70
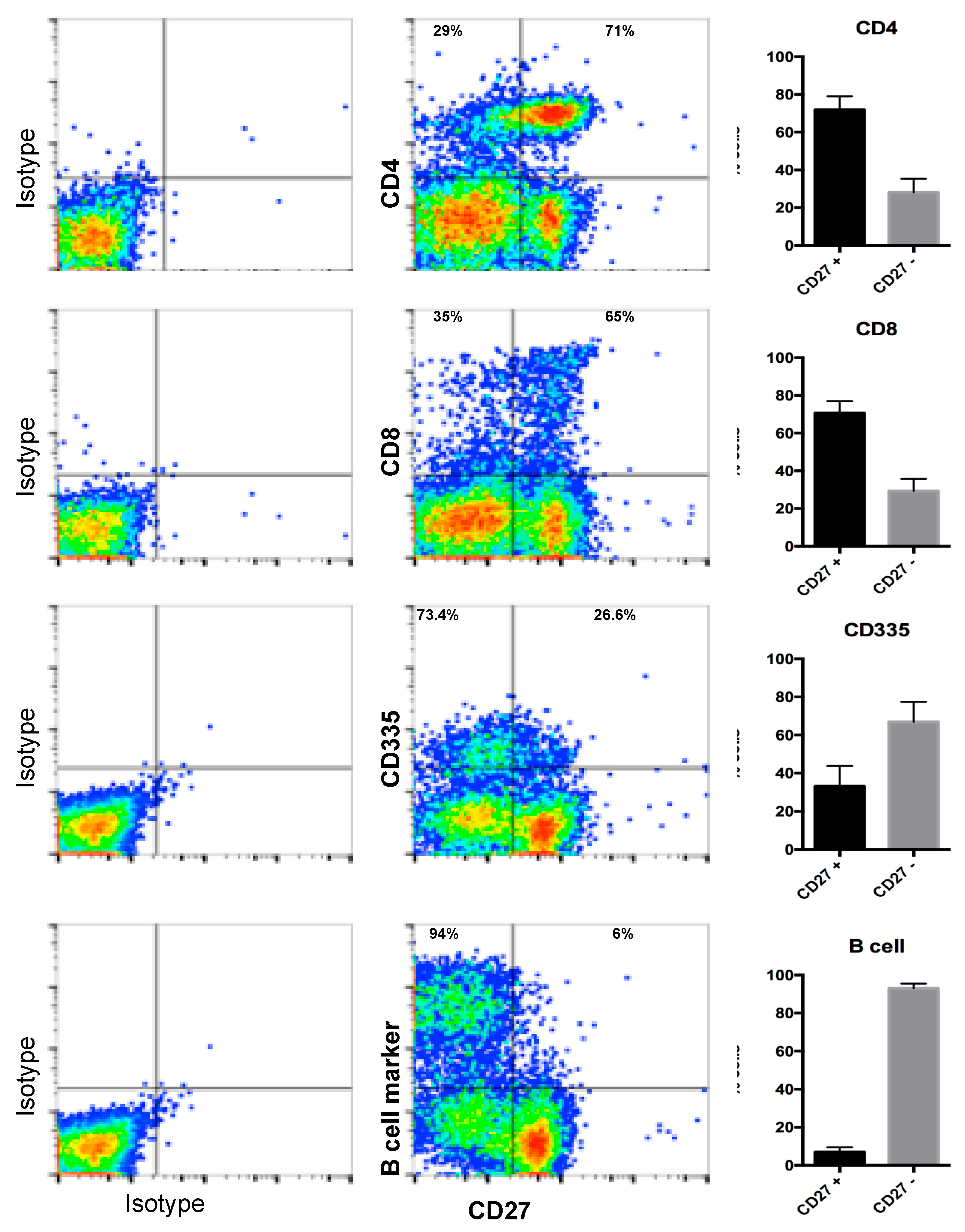
3.5. Detection of Endogenous CD27 Protein in Ovine Peripheral Blood Mononuclear Cells
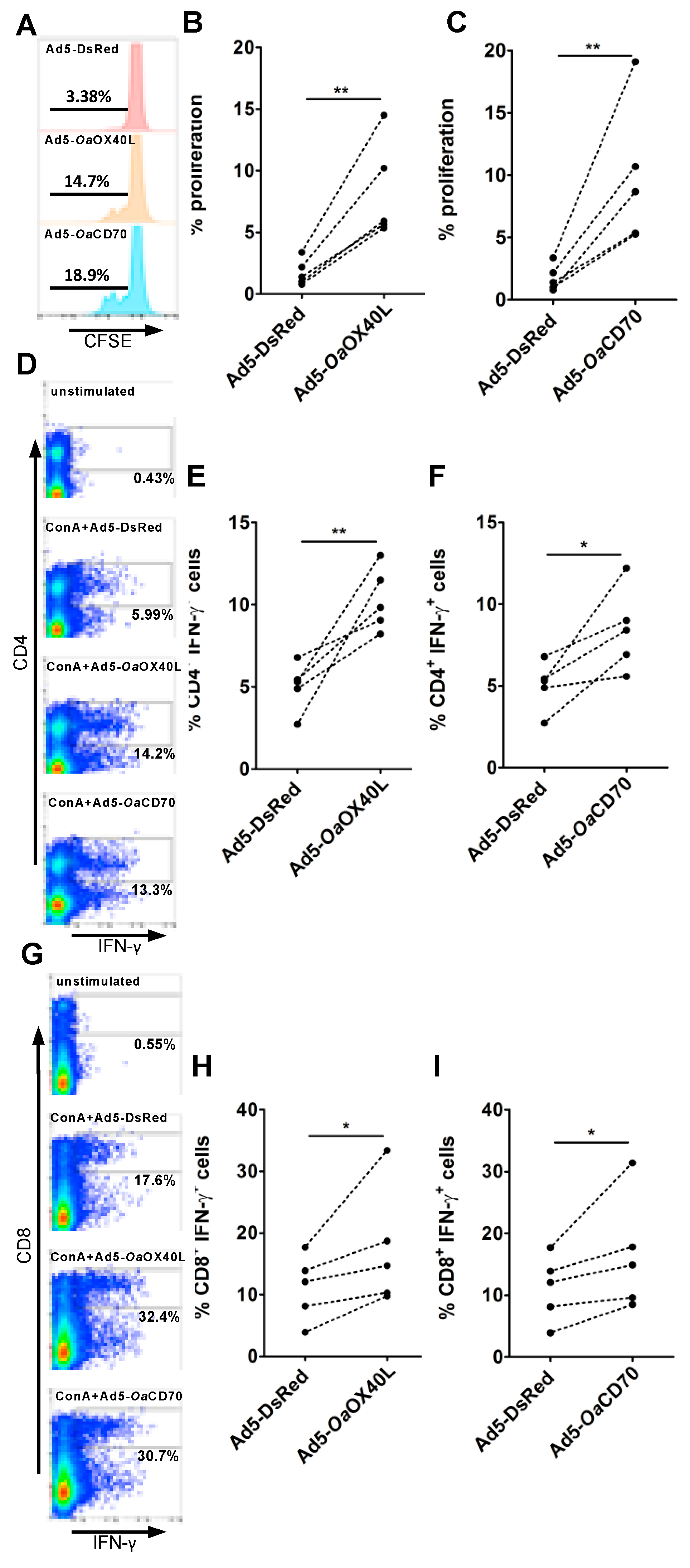
3.6. OaOX40L and OaCD70 Stimulate Ovine T Cell Proliferation and IFN-γ Production
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Croft, M. Costimulation of T cells by OX40, 4-1BB, and CD27. Cytokine Growth Factor Rev. 2003, 14, 265–273. [Google Scholar] [CrossRef]
- Bishop, G.A.; Hostager, B.S. Signaling by CD40 and its mimics in B cell activation. Immunol. Res. 2001. [Google Scholar] [CrossRef]
- Wortzman, M.E.; Clouthier, D.L.; Mcpherson, A.J.; Lin, G.H.Y.; Watts, T.H. The contextual role of TNFR family members in CD8+ T-cell control of viral infections. Immunol. Rev. 2013, 255, 125–148. [Google Scholar] [CrossRef]
- Croft, M. Control of Immunity by the TNFR-Related Molecule OX40 (CD134). Annu. Rev. Immunol. 2010, 28, 57–78. [Google Scholar] [CrossRef]
- Gramaglia, I.; Jember, A.; Pippig, S.D.; Weinberg, A.D.; Killeen, N.; Croft, M. The OX40 Costimulatory Receptor Determines the Development of CD4 Memory by Regulating Primary Clonal Expansion. J. Immunol. 2000, 165, 3043–3050. [Google Scholar] [CrossRef]
- Weinberg, A.D.; Evans, D.E.; Thalhofer, C.; Shi, T.; Prell, R.A. The generation of T cell memory: A review describing the molecular and cellular events following OX40 (CD134) engagement. J. Leukoc. Biol. 2004, 75, 962–972. [Google Scholar] [CrossRef]
- Al-Shamkhani, A.; Birkeland, M.L.; Puklavec, M.; Brown, M.H.; James, W.; Barclay, A.N. OX40 is differentially expressed on activated rat and mouse T cells and is the sole receptor for the OX40 ligand. Eur. J. Immunol. 1996. [Google Scholar] [CrossRef] [PubMed]
- Godfrey, W.R.; Fagnoni, F.F.; Harara, M.A.; Buck, D.; Engleman, E.G. Identification of a human ox-40 ligand, a costimulator of cd4+ t cells with homology to tumor necrosis factor. J. Exp. Med. 1994. [Google Scholar] [CrossRef] [PubMed]
- Compaan, D.M.; Hymowitz, S.G. The Crystal Structure of the Costimulatory OX40-OX40L Complex. Structure 2006, 14, 1321–1330. [Google Scholar] [CrossRef] [PubMed]
- Vanamee, É.S.; Faustman, D.L. Structural principles of tumor necrosis factor superfamily signaling. Sci. Signal. 2018, 11, 1–12. [Google Scholar] [CrossRef]
- Grant, E.J.; Nüssing, S.; Sant, S.; Clemens, E.B.; Kedzierska, K. The role of CD27 in anti-viral T-cell immunity. Curr. Opin. Virol. 2017, 22, 77–88. [Google Scholar] [CrossRef] [PubMed]
- Akiba, H.; Nakano, H.; Nishinaka, S.; Shindo, M.; Kobata, T.; Atsuta, M.; Morimoto, C.; Ware, C.F.; Malinin, N.L.; Wallach, D.; et al. CD27, a member of the tumor necrosis factor receptor superfamily, activates NF-κB and stress-activated protein kinase/c-Jun N-terminal kinase via TRAF2, TRAF5, and NF-κB-inducing kinase. J. Biol. Chem. 1998, 273, 13353–13358. [Google Scholar] [CrossRef] [PubMed]
- Kobata, T.; Jacquot, S.; Kozlowski, S.; Agematsu, K.; Schlossman, S.F.; Morimoto, C. CD27-CD70 interactions regulate B-cell activation by T cells. Proc. Natl. Acad. Sci. USA 1995, 92, 11249–11253. [Google Scholar] [CrossRef] [PubMed]
- Buchan, S.L.; Rogel, A.; Al-Shamkhani, A. The immunobiology of CD27 and OX40 and their potential as targets for cancer immunotherapy. Blood 2018, 131, 39–48. [Google Scholar] [CrossRef] [PubMed]
- Fu, Y.; Lin, Q.; Zhang, Z.; Zhang, L. Therapeutic strategies for the costimulatory molecule OX40 in T-cell-mediated immunity. Acta Pharm. Sin. B 2020, 10, 414–433. [Google Scholar] [CrossRef] [PubMed]
- Duttagupta, P.A.; Boesteanu, A.C.; Katsikis, P.D. Costimulation signals for memory CD8+ T cells during viral infections. Crit. Rev. Immunol. 2009, 29, 469–486. [Google Scholar] [CrossRef]
- Gupta, S.; Termini, J.M.; Kanagavelu, S.; Stone, G.W. Design of vaccine adjuvants incorporating TNF superfamily ligands and TNF superfamily molecular mimics. Immunol. Res. 2013. [Google Scholar] [CrossRef]
- Barr, T.A.; Carlring, J.; Heath, A.W. Co-stimulatory agonists as immunological adjuvants. Vaccine 2006, 24, 3399–3407. [Google Scholar] [CrossRef]
- Kanagavelu, S.K.; Snarsky, V.; Termini, J.M.; Gupta, S.; Barzee, S.; Wright, J.A.; Khan, W.N.; Kornbluth, R.S.; Stone, G.W. Soluble multi-trimeric TNF superfamily ligand adjuvants enhance immune responses to a HIV-1 Gag DNA vaccine. Vaccine 2012, 30, 691–702. [Google Scholar] [CrossRef]
- Claerebout, E.; Geldhof, P. Helminth Vaccines in Ruminants: From Development to Application. Vet. Clin. N. Am.–Food Anim. Pract. 2020, 36, 159–171. [Google Scholar] [CrossRef]
- Lacasta, D.; Ferrer, L.M.; Ramos, J.J.; González, J.M.; Ortín, A.; Fthenakis, G.C. Vaccination schedules in small ruminant farms. Vet. Microbiol. 2015. [Google Scholar] [CrossRef] [PubMed]
- OIE. Manual of Diagnostic Tests and Vaccines for Terrestrial Animals. World Organ. Anim. Heal 2013. [Google Scholar] [CrossRef]
- Diaz-San Segundo, F.; Medina, G.N.; Stenfeldt, C.; Arzt, J.; de los Santos, T. Foot-and-mouth disease vaccines. Vet. Microbiol. 2017. [Google Scholar] [CrossRef] [PubMed]
- Faburay, B.; LaBeaud, A.D.; McVey, D.S.; Wilson, W.C.; Richt, J.A. Current status of rift valley fever vaccine development. Vaccines 2017, 5, 29. [Google Scholar] [CrossRef]
- Kumar, N.; Barua, S.; Riyesh, T.; Tripathi, B.N. Advances in peste des petits ruminants vaccines. Vet. Microbiol. 2017. [Google Scholar] [CrossRef]
- van Rijn, P.A. Prospects of Next-Generation Vaccines for Bluetongue. Front. Vet. Sci. 2019, 6, 406. [Google Scholar] [CrossRef]
- Crystal, R.G. Adenovirus: The first effective in vivo gene delivery vector. Hum. Gene Ther. 2014, 25, 3–11. [Google Scholar] [CrossRef]
- Martín, V.; Pascual, E.; Avia, M.; Peña, L.; Valcárcel, F.; Sevilla, N. Protective efficacy in sheep of adenovirus-vectored vaccines against bluetongue virus is associated with specific T cell responses. PLoS ONE 2015, 10, e0143273. [Google Scholar] [CrossRef]
- Rojas, J.M.; Moreno, H.; Valcárcel, F.; Peña, L.; Sevilla, N.; Martín, V. Vaccination with recombinant adenoviruses expressing the peste des petits ruminants virus F or H proteins overcomes viral immunosuppression and induces protective immunity against PPRV challenge in sheep. PLoS ONE 2014, 9, e0101226. [Google Scholar] [CrossRef]
- Rojas, M.J.; Sevilla, N.; Martín, V. Adenovirus as Tools in Animal Health. In Adenoviruses; IntechOpen Limited: London, UK, 2019. [Google Scholar]
- Rojas, J.M.; Moreno, H.; García, A.; Ramírez, J.C.; Sevilla, N.; Martín, V. Two replication-defective adenoviral vaccine vectors for the induction of immune responses to PPRV. Vaccine 2014, 32, 393–400. [Google Scholar] [CrossRef]
- Harbury, P.B.; Kim, P.S.; Alber, T. Crystal structure of an isoleucine-zipper trimer. Nature 1994, 371, 80–83. [Google Scholar] [CrossRef] [PubMed]
- Mulens-Arias, V.; Rojas, J.M.; Pérez-Yagüe, S.; Morales, M.P.; Barber, D.F. Polyethylenimine-coated SPIONs trigger macrophage activation through TLR-4 signaling and ROS production and modulate podosome dynamics. Biomaterials 2015. [Google Scholar] [CrossRef] [PubMed]
- Hathcock, K.S. Depletion of Accessory Cells by Adherence to Sephadex G-10. In Current Protocols in Immunology; John Wiley & Sons, Inc. New Jersey: Somerset, NJ, USA, 2001. [Google Scholar]
- Rojas, J.M.; Rodríguez-Martín, D.; Avia, M.; Martín, V.; Sevilla, N. Peste des petits ruminants virus fusion and hemagglutinin proteins trigger antibody-dependent cell-mediated cytotoxicity in infected cells. Front. Immunol. 2019. [Google Scholar] [CrossRef] [PubMed]
- Rojas, J.M.; Rodríguez-Calvo, T.; Peña, L.; Sevilla, N. T cell responses to bluetongue virus are directed against multiple and identical CD4 + and CD8 + T cell epitopes from the VP7 core protein in mouse and sheep. Vaccine 2011. [Google Scholar] [CrossRef]
- Rojas, J.M.; Rodríguez-Calvo, T.; Sevilla, N. Recall T cell responses to bluetongue virus produce a narrowing of the T cell repertoire. Vet. Res. 2017. [Google Scholar] [CrossRef]
- Denoeud, J.; Moser, M. Role of CD27/CD70 pathway of activation in immunity and tolerance. J. Leukoc. Biol. 2011, 89, 195–203. [Google Scholar] [CrossRef]
- Tesselaar, K.; Gravestein, L.A.; Van Schijndel, G.M.W.; Borst, J.; Van Lier, R.A.W. Characterization of Murine CD70, the Ligand of the TNF Receptor Family Member CD27. J. Immunol. 1997, 159, 4959–4965. [Google Scholar]
- Croft, M. The role of TNF superfamily members in T-cell function and diseases. Nat. Rev. Immunol. 2009, 9, 271–285. [Google Scholar] [CrossRef]
- Kawamata, S.; Hori, T.; Imura, A.; Takaori-Kondo, A.; Uchiyama, T. Activation of OX40 signal transduction pathways leads to tumor necrosis factor receptor-associated factor (TRAF) 2- and TRAF5-mediated NF-κB activation. J. Biol. Chem. 1998, 273, 5808–5814. [Google Scholar] [CrossRef]
- Holler, N.; Tardivel, A.; Kovacsovics-Bankowski, M.; Hertig, S.; Gaide, O.; Martinon, F.; Tinel, A.; Deperthes, D.; Calderara, S.; Schulthess, T.; et al. Two Adjacent Trimeric Fas Ligands Are Required for Fas Signaling and Formation of a Death-Inducing Signaling Complex. Mol. Cell. Biol. 2003, 23, 1428–1440. [Google Scholar] [CrossRef]
- Morris, N.P.; Peters, C.; Montler, R.; Hu, H.M.; Curti, B.D.; Urba, W.J.; Weinberg, A.D. Development and characterization of recombinant human Fc:OX40L fusion protein linked via a coiled-coil trimerization domain. Mol. Immunol. 2007, 44, 3112–3121. [Google Scholar] [CrossRef] [PubMed]
- Harbury, P.; Zhang, T.; Kim, P.; Alber, T. and Four-Stranded Coiled Coils in GCN4 Leucine Zipper Mutants. Science 1993, 262, 26. [Google Scholar] [CrossRef] [PubMed]
- Morris, A.E.; Remmele, R.L.; Klinke, R.; Macduff, B.M.; Fanslow, W.C.; Armitage, R.J. Incorporation of an isoleucine zipper motif enhances the biological activity of soluble CD40L (CD154). J. Biol. Chem. 1999, 274, 418–423. [Google Scholar] [CrossRef] [PubMed]
- Rozanov, D.V.; Savinov, A.Y.; Golubkov, V.S.; Rozanova, O.L.; Postnova, T.I.; Sergienko, E.A.; Vasile, S.; Aleshin, A.E.; Rega, M.F.; Pellecchia, M.; et al. Engineering a leucine zipper-TRAIL homotrimer with improved cytotoxicity in tumor cells. Mol. Cancer Ther. 2009, 8, 1515–1525. [Google Scholar] [CrossRef]
- Appaiahgari, M.B.; Vrati, S. Adenoviruses as gene/vaccine delivery vectors: Promises and pitfalls. Expert Opin. Biol. Ther. 2015, 15, 337–351. [Google Scholar] [CrossRef]
- Fausther-Bovendo, H.; Kobinger, G.P. Pre-existing immunity against Ad vectors: Humoral, cellular, and innate response, what’s important? Hum. Vaccines Immunother. 2014, 10, 2875–2884. [Google Scholar] [CrossRef]
- Nanda, S.K.; Baron, J.; Royall, E.; Robinson, L.; Falciani, F.; Baron, M.D. Infection of bovine dendritic cells by rinderpest or measles viruses induces different changes in host transcription. Virology 2009, 395, 223–231. [Google Scholar] [CrossRef]
- Bessis, N.; GarciaCozar, F.J.; Boissier, M.C. Immune responses to gene therapy vectors: Influence on vector function and effector mechanisms. Gene Ther. 2004, 11, S10–S17. [Google Scholar] [CrossRef]
- Ndi, O.L.; Barton, M.D.; Vanniasinkam, T. Adenoviral Vectors in Veterinary Vaccine Development: Potential for Further Development. World J. Vaccines 2013, 03, 111–121. [Google Scholar] [CrossRef]
- Ferreira, T.B.; Alves, P.M.; Aunins, J.G.; Carrondo, M.J.T. Use of adenoviral vectors as veterinary vaccines. Gene Ther. 2005, 12, S73–S83. [Google Scholar] [CrossRef]
- Perez-Martin, E.; Weiss, M.; Diaz-San Segundo, F.; Pacheco, J.M.; Arzt, J.; Grubman, M.J.; de los Santos, T. Bovine Type III Interferon Significantly Delays and Reduces the Severity of Foot-and-Mouth Disease in Cattle. J. Virol. 2012, 86, 4477–4487. [Google Scholar] [CrossRef] [PubMed]
- Rothel, J.S.; Seow, H.F.; Lightowlers, M.W.; Parry, B.W.; Gauci, C.; Hurst, L.; Mucha, M.J.; Corner, L.A.; Wood, P.R. The use of recombinant ovine IL-1β and TNF-α as natural adjuvants and their physiological effects in vivo. Immunol. Cell Biol. 1998, 76, 167–172. [Google Scholar] [CrossRef] [PubMed]
- Lofthouse, S.A.; Andrews, A.E.; Elhay, M.J.; Bowles, V.M.; Meeusen, E.N.T.; Nash, A.D. Cytokines as adjuvants for ruminant vaccines. Int. J. Parasitol. 1996, 26, 835–842. [Google Scholar] [CrossRef]
- Kanagavelu, S.; Termini, J.M.; Gupta, S.; Raffa, F.N.; Fuller, K.A.; Rivas, Y.; Philip, S.; Kornbluth, R.S.; Stone, G.W. HIV-1 adenoviral vector vaccines expressing multi-trimeric BAFF and 4-1BBL enhance t cell mediated anti-viral immunity. PLoS ONE 2014, 9. [Google Scholar] [CrossRef]
- Salek-Ardakani, S.; Moutaftsi, M.; Crotty, S.; Sette, A.; Croft, M. OX40 Drives Protective Vaccinia Virus-Specific CD8 T Cells. J. Immunol. 2008. [Google Scholar] [CrossRef]
- Salek-Ardakani, S.; Moutaftsi, M.; Sette, A.; Croft, M. Targeting OX40 Promotes Lung-Resident Memory CD8 T Cell Populations That Protect against Respiratory Poxvirus Infection. J. Virol. 2011, 85, 9051–9059. [Google Scholar] [CrossRef]
- Izawa, K.; Martin, E.; Soudais, C.; Bruneau, J.; Boutboul, D.; Rodriguez, R.; Lenoir, C.; Hislop, A.D.; Besson, C.; Touzot, F.; et al. Inherited CD70 deficiency in humans reveals a critical role for the CD70-CD27 pathway in immunity to Epstein-Barr virus infection. J. Exp. Med. 2017, 214, 73–89. [Google Scholar] [CrossRef]
- Ahrends, T.; Babała, N.; Xiao, Y.; Yagita, H.; Van Eenennaam, H.; Borst, J. CD27 Agonism Plus PD-1 Blockade Recapitulates CD4+ T-cell Help in Therapeutic Anticancer Vaccination. Cancer Res. 2016, 76, 2921–2931. [Google Scholar] [CrossRef]
- Rojas, J.M.; Avia, M.; Pascual, E.; Sevilla, N.; Martín, V. Vaccination with recombinant adenovirus expressing peste des petits ruminants virus-F or -H proteins elicits T cell responses to epitopes that arises during PPRV infection. Vet. Res. 2017, 48, 79. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rojas, J.M.; Alejo, A.; Avia, J.M.; Rodríguez-Martín, D.; Sánchez, C.; Alcamí, A.; Sevilla, N.; Martín, V. Activation of OX40 and CD27 Costimulatory Signalling in Sheep through Recombinant Ovine Ligands. Vaccines 2020, 8, 333. https://doi.org/10.3390/vaccines8020333
Rojas JM, Alejo A, Avia JM, Rodríguez-Martín D, Sánchez C, Alcamí A, Sevilla N, Martín V. Activation of OX40 and CD27 Costimulatory Signalling in Sheep through Recombinant Ovine Ligands. Vaccines. 2020; 8(2):333. https://doi.org/10.3390/vaccines8020333
Chicago/Turabian StyleRojas, José Manuel, Alí Alejo, Jose Miguel Avia, Daniel Rodríguez-Martín, Carolina Sánchez, Antonio Alcamí, Noemí Sevilla, and Verónica Martín. 2020. "Activation of OX40 and CD27 Costimulatory Signalling in Sheep through Recombinant Ovine Ligands" Vaccines 8, no. 2: 333. https://doi.org/10.3390/vaccines8020333
APA StyleRojas, J. M., Alejo, A., Avia, J. M., Rodríguez-Martín, D., Sánchez, C., Alcamí, A., Sevilla, N., & Martín, V. (2020). Activation of OX40 and CD27 Costimulatory Signalling in Sheep through Recombinant Ovine Ligands. Vaccines, 8(2), 333. https://doi.org/10.3390/vaccines8020333




