E. coli-Produced Monophosphoryl Lipid a Significantly Enhances Protective Immunity of Pandemic H1N1 Vaccine
Abstract
1. Introduction
2. Materials and Methods
2.1. Mice and Cells
2.2. Preparation of Adjuvants
2.3. Preparation of Influenza Virus
2.4. Cell Viability Assay
2.5. In Vitro Activation and Antigen Processing of BMDCs
2.6. Western Blotting
2.7. Immunizations and Viral Challenge
2.8. HI Assay
2.9. Enzyme-Linked Immunosorbent Assay (ELISA)
2.10. Enzyme-Linked Immunospot (ELISPOT) Assay
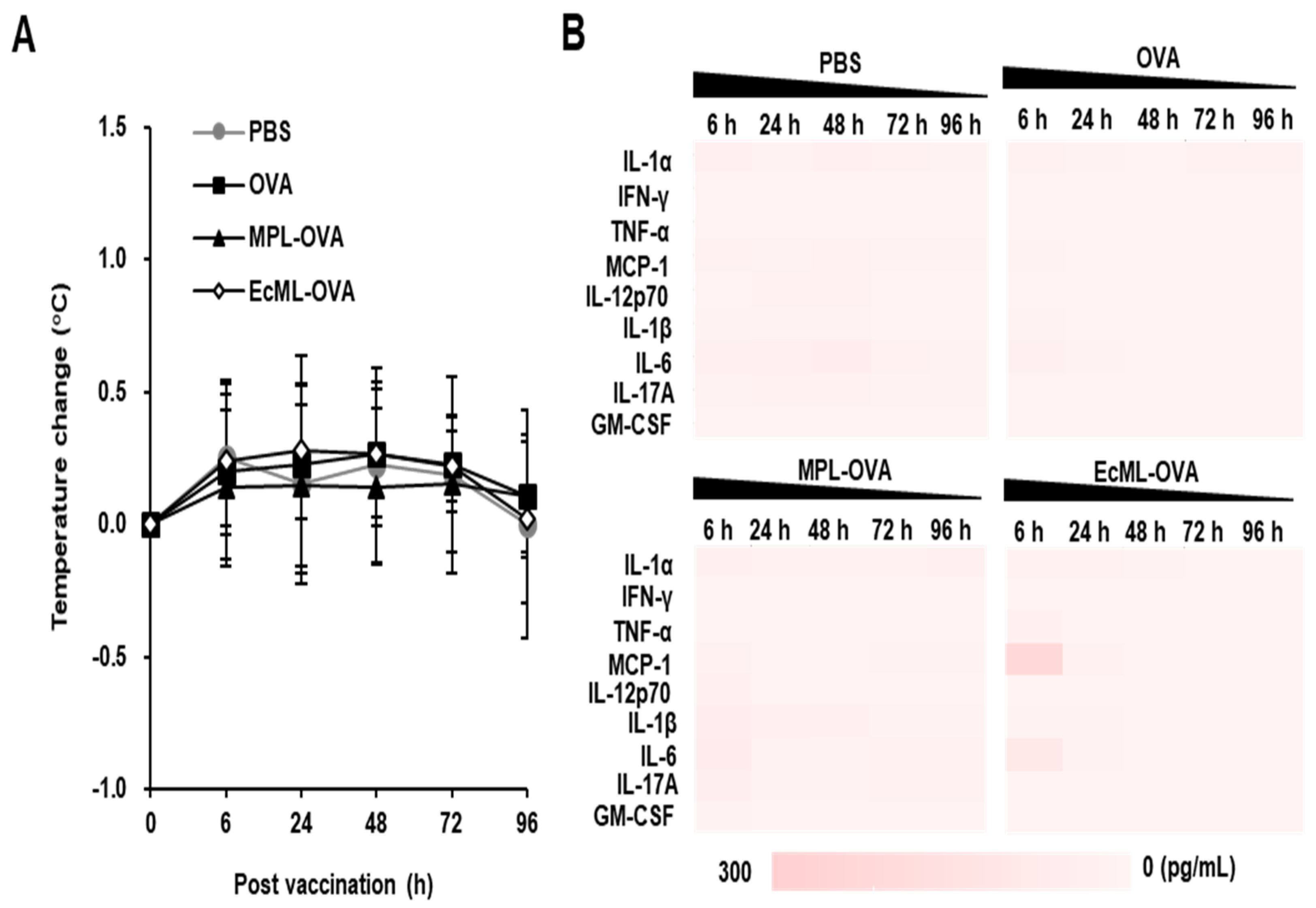
2.11. Systemic Inflammatory Responses after Vaccination
2.12. Statistical Analysis
3. Results
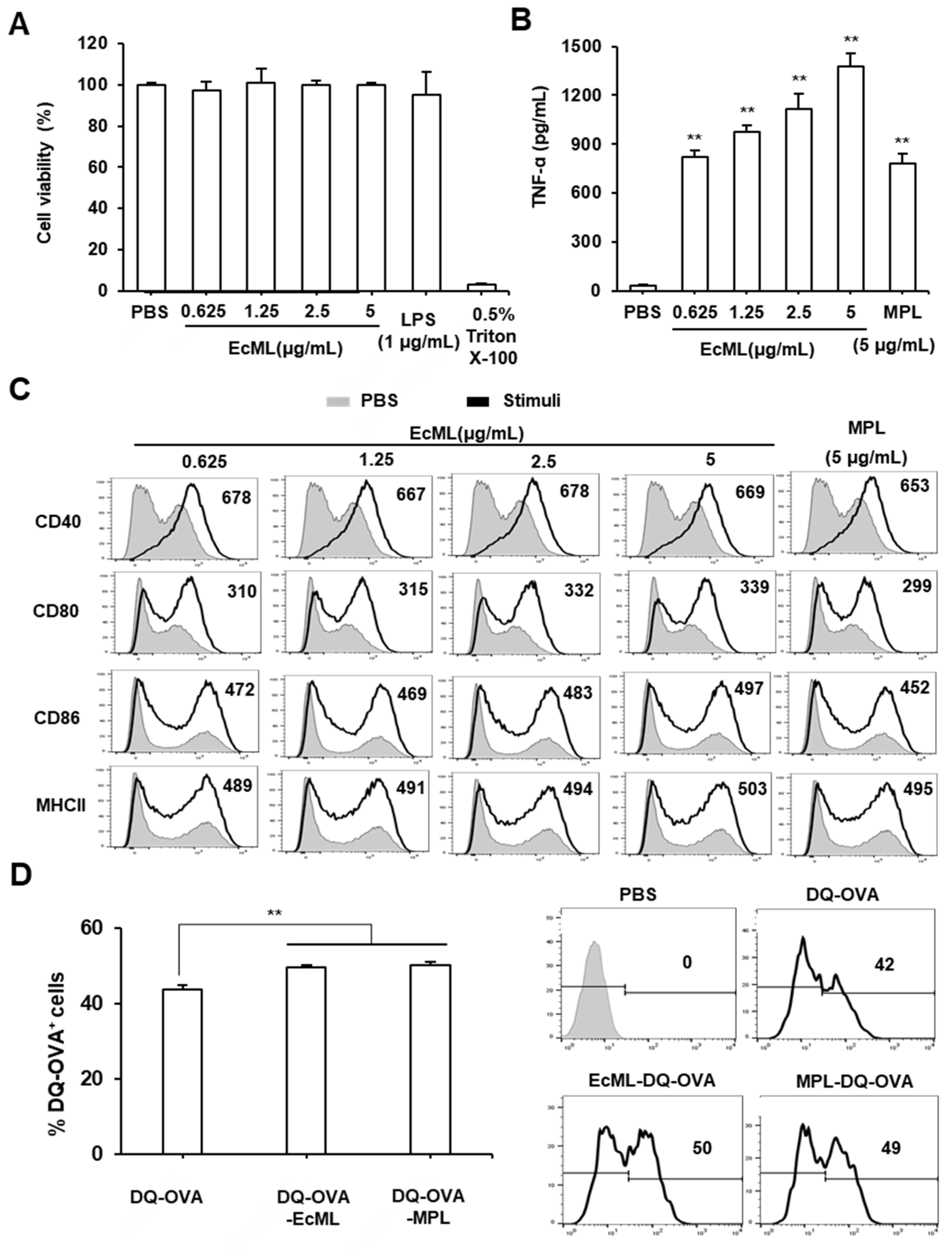
3.1. EcML Enhances Activation and Antigen Processing of Dendritic Cells (DCs) In Vitro
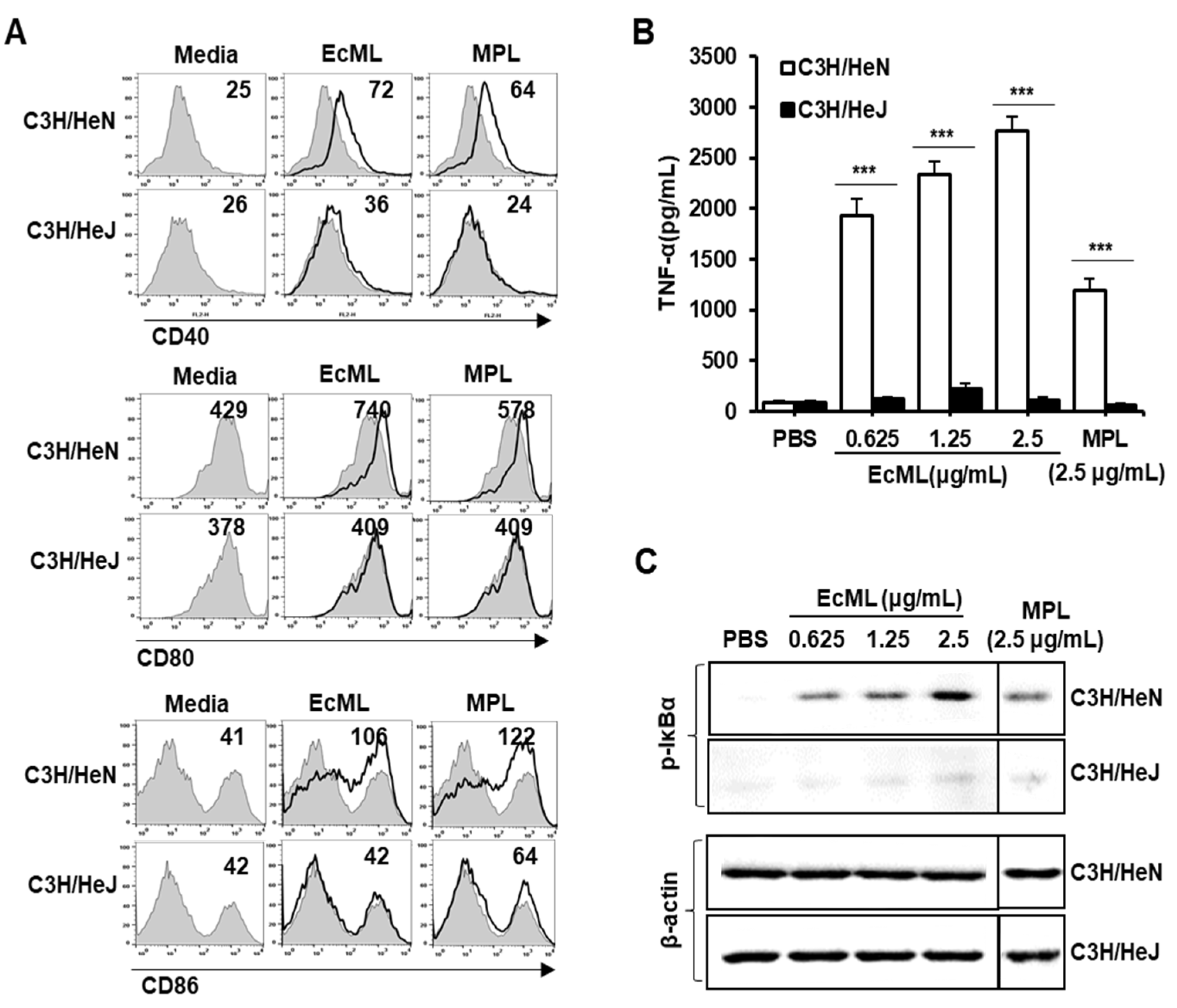
3.2. EcML-Induced DC Activation is Mediated by TLR4
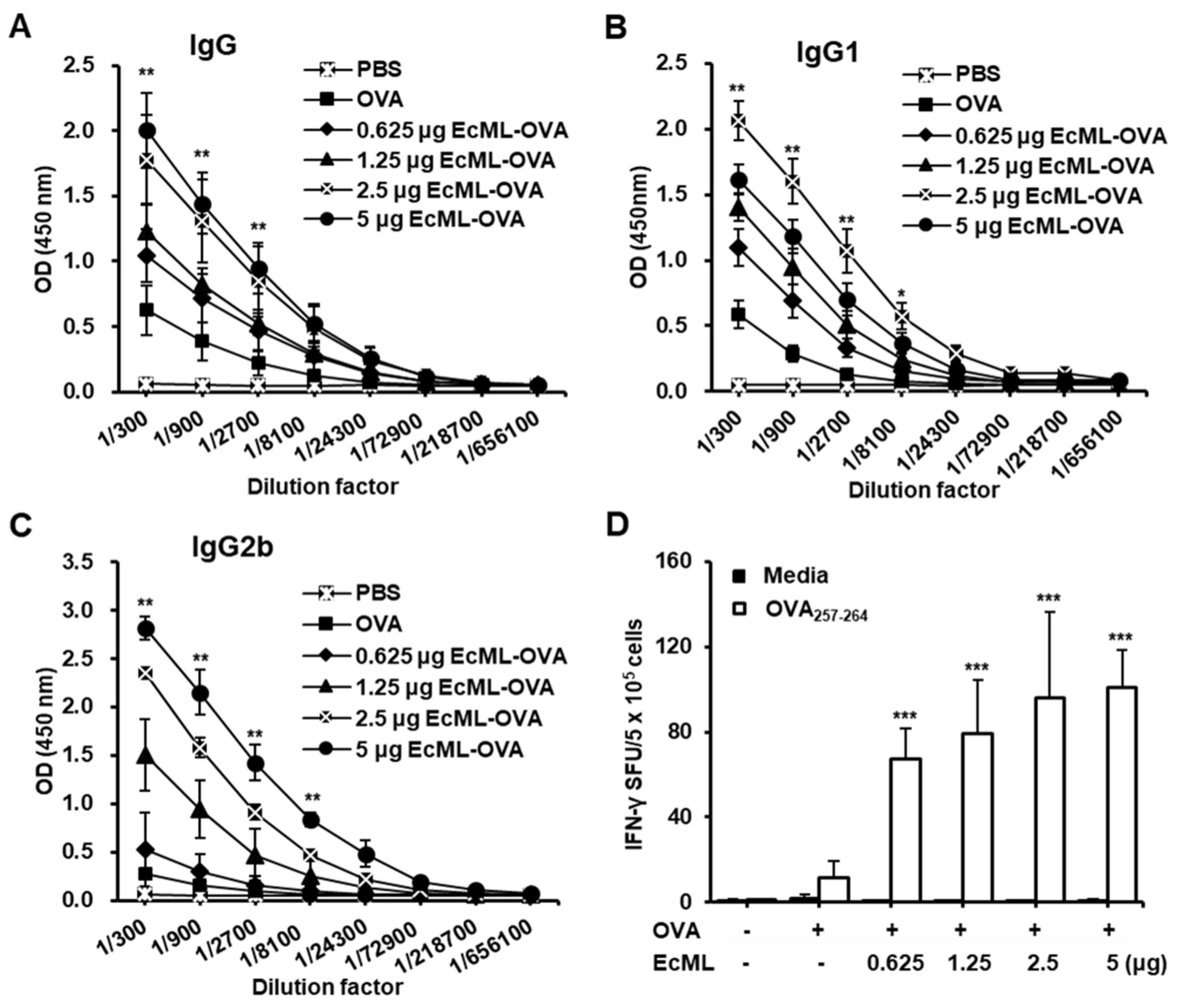
3.3. EcML Treatment Results in Dose-Dependent Enhancement of Antigen-Specific Humoral and Cellular Immunity
3.4. EcML Significantly Enhances OVA-Specific Cellular Immune Responses Compared to Alum
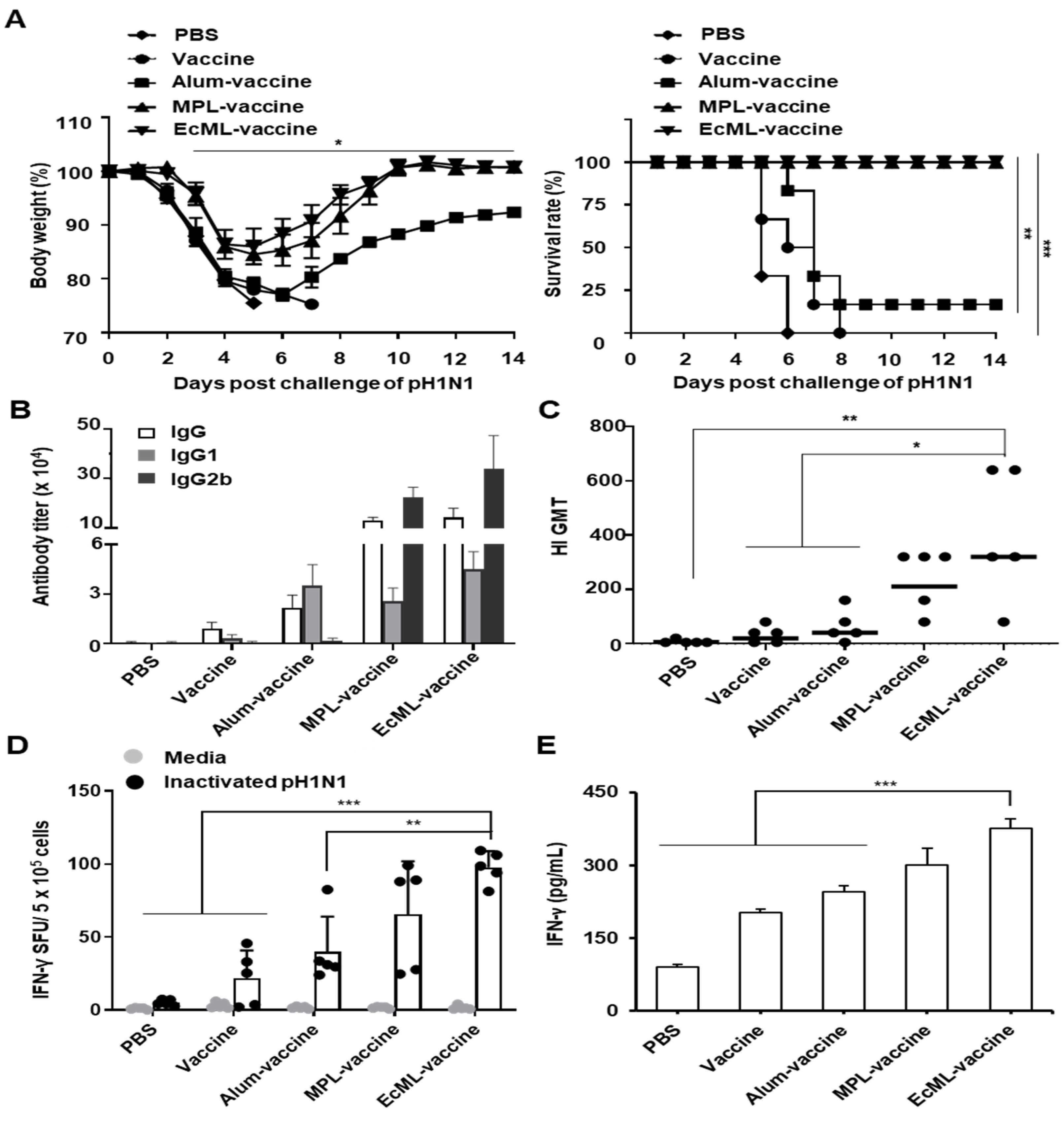
3.5. EcML Enhances the Protective Efficacy of Influenza Vaccine Antigen
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Bar-On, Y.; Glasner, A.; Meningher, T.; Achdout, H.; Gur, C.; Lankry, D.; Vitenshtein, A.; Meyers, A.F.; Mandelboim, M.; Mandelboim, O. Neuraminidase-mediated, nkp46-dependent immune-evasion mechanism of influenza viruses. Cell Rep. 2013, 3, 1044–1050. [Google Scholar] [CrossRef] [PubMed]
- Glezen, W.P. Emerging infections: Pandemic influenza. Epidemiol. Rev. 1996, 18, 64–76. [Google Scholar] [CrossRef] [PubMed]
- Dawood, F.S.; Iuliano, A.D.; Reed, C.; Meltzer, M.I.; Shay, D.K.; Cheng, P.Y.; Bandaranayake, D.; Breiman, R.F.; Brooks, W.A.; Buchy, P.; et al. Estimated global mortality associated with the first 12 months of 2009 pandemic influenza a h1n1 virus circulation: A modelling study. Lancet Infect. Dis. 2012, 12, 687–695. [Google Scholar] [CrossRef]
- Tregoning, J.S.; Russell, R.F.; Kinnear, E. Adjuvanted influenza vaccines. Hum. Vaccines Immunother. 2018, 14, 550–564. [Google Scholar] [CrossRef]
- Marrack, P.; McKee, A.S.; Munks, M.W. Towards an understanding of the adjuvant action of aluminium. Nat. Rev. Immunol. 2009, 9, 287–293. [Google Scholar] [CrossRef] [PubMed]
- Sivakumar, S.M.; Safhi, M.M.; Kannadasan, M.; Sukumaran, N. Vaccine adjuvants-current status and prospects on controlled release adjuvancity. Saudi. Pharm. J. 2011, 19, 197–206. [Google Scholar] [CrossRef]
- Miller, E.; Andrews, N.; Stellitano, L.; Stowe, J.; Winstone, A.M.; Shneerson, J.; Verity, C. Risk of narcolepsy in children and young people receiving as03 adjuvanted pandemic a/h1n1 2009 influenza vaccine: Retrospective analysis. BMJ 2013, 346, f794. [Google Scholar] [CrossRef]
- Nohynek, H.; Jokinen, J.; Partinen, M.; Vaarala, O.; Kirjavainen, T.; Sundman, J.; Himanen, S.L.; Hublin, C.; Julkunen, I.; Olsen, P.; et al. As03 adjuvanted ah1n1 vaccine associated with an abrupt increase in the incidence of childhood narcolepsy in finland. PLoS ONE 2012, 7, e33536. [Google Scholar] [CrossRef]
- Kumar, H.; Kawai, T.; Akira, S. Toll-like receptors and innate immunity. Biochem. Biophys. Res. Commun. 2009, 388, 621–625. [Google Scholar] [CrossRef]
- Reed, S.G.; Hsu, F.C.; Carter, D.; Orr, M.T. The science of vaccine adjuvants: Advances in tlr4 ligand adjuvants. Curr. Opin. Immunol. 2016, 41, 85–90. [Google Scholar] [CrossRef]
- Gregg, K.A.; Harberts, E.; Gardner, F.M.; Pelletier, M.R.; Cayatte, C.; Yu, L.; McCarthy, M.P.; Marshall, J.D.; Ernst, R.K. Rationally designed tlr4 ligands for vaccine adjuvant discovery. MBio 2017, 8, e00492-17. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Shim, S.M.; Nguyen, T.Q.; Kim, E.H.; Kim, K.; Lim, Y.T.; Sung, M.H.; Webby, R.; Poo, H. Poly-gamma-glutamic acid/chitosan nanogel greatly enhances the efficacy and heterosubtypic cross-reactivity of h1n1 pandemic influenza vaccine. Sci. Rep. 2017, 7, 44839. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, Q.T.; Kwak, C.; Lee, W.S.; Kim, J.; Jeong, J.; Sung, M.H.; Yang, J.; Poo, H. Poly-gamma-glutamic acid complexed with alum induces cross-protective immunity of pandemic h1n1 vaccine. Front. Immunol. 2019, 10, 1604. [Google Scholar] [CrossRef] [PubMed]
- Steinhagen, F.; Kinjo, T.; Bode, C.; Klinman, D.M. Tlr-based immune adjuvants. Vaccine 2011, 29, 3341–3355. [Google Scholar] [CrossRef] [PubMed]
- Paavonen, J.; Jenkins, D.; Bosch, F.X.; Naud, P.; Salmeron, J.; Wheeler, C.M.; Chow, S.N.; Apter, D.L.; Kitchener, H.C.; Castellsague, X.; et al. Efficacy of a prophylactic adjuvanted bivalent l1 virus-like-particle vaccine against infection with human papillomavirus types 16 and 18 in young women: An interim analysis of a phase iii double-blind, randomised controlled trial. Lancet 2007, 369, 2161–2170. [Google Scholar] [CrossRef]
- Kundi, M. New hepatitis b vaccine formulated with an improved adjuvant system. Expert Rev. Vaccines 2007, 6, 133–140. [Google Scholar] [CrossRef]
- Mata-Haro, V.; Cekic, C.; Martin, M.; Chilton, P.M.; Casella, C.R.; Mitchell, T.C. The vaccine adjuvant monophosphoryl lipid a as a trif-biased agonist of tlr4. Science 2007, 316, 1628–1632. [Google Scholar] [CrossRef]
- Casella, C.R.; Mitchell, T.C. Putting endotoxin to work for us: Monophosphoryl lipid a as a safe and effective vaccine adjuvant. Cell Mol. Life Sci. 2008, 65, 3231–3240. [Google Scholar] [CrossRef]
- Reed, S.G.; Carter, D.; Casper, C.; Duthie, M.S.; Fox, C.B. Correlates of gla family adjuvants’ activities. Semin. Immunol. 2018, 39, 22–29. [Google Scholar] [CrossRef]
- Ji, Y.; An, J.; Hwang, D.; Ha, D.H.; Lim, S.M.; Lee, C.; Zhao, J.; Song, H.K.; Yang, E.G.; Zhou, P.; et al. Metabolic engineering of escherichia coli to produce a monophosphoryl lipid a adjuvant. Metab. Eng. 2020, 57, 193–202. [Google Scholar] [CrossRef]
- Zuo, B.; Qi, H.; Lu, Z.; Chen, L.; Sun, B.; Yang, R.; Zhang, Y.; Liu, Z.; Gao, X.; You, A.; et al. Alarmin-painted exosomes elicit persistent antitumor immunity in large established tumors in mice. Nat. Commun. 2020, 11, 1790. [Google Scholar] [CrossRef]
- Matheu, M.P.; Sen, D.; Cahalan, M.D.; Parker, I. Generation of bone marrow derived murine dendritic cells for use in 2-photon imaging. J. Vis. Exp. 2008, 17, e773. [Google Scholar] [CrossRef] [PubMed]
- Lim, Y.T.; Shim, S.M.; Noh, Y.W.; Lee, K.S.; Choi, D.Y.; Uyama, H.; Bae, H.H.; Kim, J.H.; Hong, K.S.; Sung, M.H.; et al. Bioderived polyelectrolyte nanogels for robust antigen loading and vaccine adjuvant effects. Small 2011, 7, 3281–3286. [Google Scholar] [CrossRef] [PubMed]
- Merad, M.; Sathe, P.; Helft, J.; Miller, J.; Mortha, A. The dendritic cell lineage: Ontogeny and function of dendritic cells and their subsets in the steady state and the inflamed setting. Annu. Rev. Immunol. 2013, 31, 563–604. [Google Scholar] [CrossRef] [PubMed]
- Iwasaki, A.; Medzhitov, R. Toll-like receptor control of the adaptive immune responses. Nat. Immunol. 2004, 5, 987–995. [Google Scholar] [CrossRef]
- Jeong, Y.; Kim, G.B.; Ji, Y.; Kwak, G.J.; Nam, G.H.; Hong, Y.; Kim, S.; An, J.; Kim, S.H.; Yang, Y.; et al. Dendritic cell activation by an e. Coli-derived monophosphoryl lipid a enhances the efficacy of pd-1 blockade. Cancer Lett. 2020, 472, 19–28. [Google Scholar] [CrossRef]
- Karin, M. How nf-kappab is activated: The role of the ikappab kinase (ikk) complex. Oncogene 1999, 18, 6867–6874. [Google Scholar] [CrossRef]
- Ko, E.J.; Lee, Y.T.; Lee, Y.; Kim, K.H.; Kang, S.M. Distinct effects of monophosphoryl lipid a, oligodeoxynucleotide cpg, and combination adjuvants on modulating innate and adaptive immune responses to influenza vaccination. Immune Netw. 2017, 17, 326–342. [Google Scholar] [CrossRef]
- Steinman, R.M. Linking innate to adaptive immunity through dendritic cells. Novartis Found. Symp. 2006, 279, 101–109; discussion 109–113, 216–219. [Google Scholar]
- Seubert, A.; Monaci, E.; Pizza, M.; O’Hagan, D.T.; Wack, A. The adjuvants aluminum hydroxide and mf59 induce monocyte and granulocyte chemoattractants and enhance monocyte differentiation toward dendritic cells. J. Immunol. 2008, 180, 5402–5412. [Google Scholar] [CrossRef]
- Levitz, S.M.; Golenbock, D.T. Beyond empiricism: Informing vaccine development through innate immunity research. Cell 2012, 148, 1284–1292. [Google Scholar] [CrossRef] [PubMed]
- Mosaheb, M.M.; Reiser, M.L.; Wetzler, L.M. Toll-like receptor ligand-based vaccine adjuvants require intact myd88 signaling in antigen-presenting cells for germinal center formation and antibody production. Front. Immunol. 2017, 8, 225. [Google Scholar] [CrossRef] [PubMed]
- Didierlaurent, A.M.; Laupeze, B.; Di Pasquale, A.; Hergli, N.; Collignon, C.; Garcon, N. Adjuvant system as01: Helping to overcome the challenges of modern vaccines. Expert Rev. Vaccines 2017, 16, 55–63. [Google Scholar] [CrossRef] [PubMed]
- Badovinac, V.P.; Messingham, K.A.; Jabbari, A.; Haring, J.S.; Harty, J.T. Accelerated cd8+ t-cell memory and prime-boost response after dendritic-cell vaccination. Nat. Med. 2005, 11, 748–756. [Google Scholar] [CrossRef]
- Black, S.; Della Cioppa, G.; Malfroot, A.; Nacci, P.; Nicolay, U.; Pellegrini, M.; Sokal, E.; Vertruyen, A. Safety of mf59-adjuvanted versus non-adjuvanted influenza vaccines in children and adolescents: An integrated analysis. Vaccine 2010, 28, 7331–7336. [Google Scholar] [CrossRef]
- Stassijns, J.; Bollaerts, K.; Baay, M.; Verstraeten, T. A systematic review and meta-analysis on the safety of newly adjuvanted vaccines among children. Vaccine 2016, 34, 714–722. [Google Scholar] [CrossRef]
- Mei, J.; Riedel, N.; Grittner, U.; Endres, M.; Banneke, S.; Emmrich, J.V. Body temperature measurement in mice during acute illness: Implantable temperature transponder versus surface infrared thermometry. Sci. Rep. 2018, 8, 3526. [Google Scholar] [CrossRef]
- Olejnik, J.; Hume, A.J.; Muhlberger, E. Toll-like receptor 4 in acute viral infection: Too much of a good thing. PLoS Pathog. 2018, 14, e1007390. [Google Scholar] [CrossRef]
- Simon, W.L.; Salk, H.M.; Ovsyannikova, I.G.; Kennedy, R.B.; Poland, G.A. Cytokine production associated with smallpox vaccine responses. Immunotherapy 2014, 6, 1097–1112. [Google Scholar] [CrossRef]
- Christian, L.M.; Porter, K.; Karlsson, E.; Schultz-Cherry, S. Proinflammatory cytokine responses correspond with subjective side effects after influenza virus vaccination. Vaccine 2015, 33, 3360–3366. [Google Scholar] [CrossRef]
- Amanna, I.J.; Slifka, M.K. Contributions of humoral and cellular immunity to vaccine-induced protection in humans. Virology 2011, 411, 206–215. [Google Scholar] [CrossRef] [PubMed]
- Sokolovska, A.; Hem, S.L.; HogenEsch, H. Activation of dendritic cells and induction of cd4(+) t cell differentiation by aluminum-containing adjuvants. Vaccine 2007, 25, 4575–4585. [Google Scholar] [CrossRef]
- Didierlaurent, A.M.; Collignon, C.; Bourguignon, P.; Wouters, S.; Fierens, K.; Fochesato, M.; Dendouga, N.; Langlet, C.; Malissen, B.; Lambrecht, B.N.; et al. Enhancement of adaptive immunity by the human vaccine adjuvant as01 depends on activated dendritic cells. J. Immunol. 2014, 193, 1920–1930. [Google Scholar] [CrossRef] [PubMed]
- Didierlaurent, A.M.; Morel, S.; Lockman, L.; Giannini, S.L.; Bisteau, M.; Carlsen, H.; Kielland, A.; Vosters, O.; Vanderheyde, N.; Schiavetti, F.; et al. As04, an aluminum salt- and tlr4 agonist-based adjuvant system, induces a transient localized innate immune response leading to enhanced adaptive immunity. J. Immunol. 2009, 183, 6186–6197. [Google Scholar] [CrossRef] [PubMed]

© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nguyen, Q.T.; Kim, E.; Yang, J.; Lee, C.; Ha, D.H.; Lee, C.G.; Lee, Y.R.; Poo, H. E. coli-Produced Monophosphoryl Lipid a Significantly Enhances Protective Immunity of Pandemic H1N1 Vaccine. Vaccines 2020, 8, 306. https://doi.org/10.3390/vaccines8020306
Nguyen QT, Kim E, Yang J, Lee C, Ha DH, Lee CG, Lee YR, Poo H. E. coli-Produced Monophosphoryl Lipid a Significantly Enhances Protective Immunity of Pandemic H1N1 Vaccine. Vaccines. 2020; 8(2):306. https://doi.org/10.3390/vaccines8020306
Chicago/Turabian StyleNguyen, Quyen Thi, Eunjin Kim, Jihyun Yang, Chankyu Lee, Da Hui Ha, Choon Geun Lee, Ye Ram Lee, and Haryoung Poo. 2020. "E. coli-Produced Monophosphoryl Lipid a Significantly Enhances Protective Immunity of Pandemic H1N1 Vaccine" Vaccines 8, no. 2: 306. https://doi.org/10.3390/vaccines8020306
APA StyleNguyen, Q. T., Kim, E., Yang, J., Lee, C., Ha, D. H., Lee, C. G., Lee, Y. R., & Poo, H. (2020). E. coli-Produced Monophosphoryl Lipid a Significantly Enhances Protective Immunity of Pandemic H1N1 Vaccine. Vaccines, 8(2), 306. https://doi.org/10.3390/vaccines8020306




