Antigen-Specific IFN-γ/IL-17-Co-Producing CD4+ T-Cells are the Determinants for Protective Efficacy of Tuberculosis Subunit Vaccine
Abstract
1. Introduction
2. Materials and Methods
2.1. Ethics Statement
2.2. Mice
2.3. Preparations of Mtb Strains and Antigens
2.4. Immunisation and Mtb Infection in Mice
2.5. Intracellular Cytokine Staining
2.6. Bacterial Counts and Histopathological Analysis
2.7. Quantification of Cytokines
2.8. Cell Culture
2.9. In Vitro T-Cell Proliferation and Polarisation Assay
2.10. Colocalisation of Phagosomes and Phagolysosomes
2.11. Statistical Analysis
3. Results
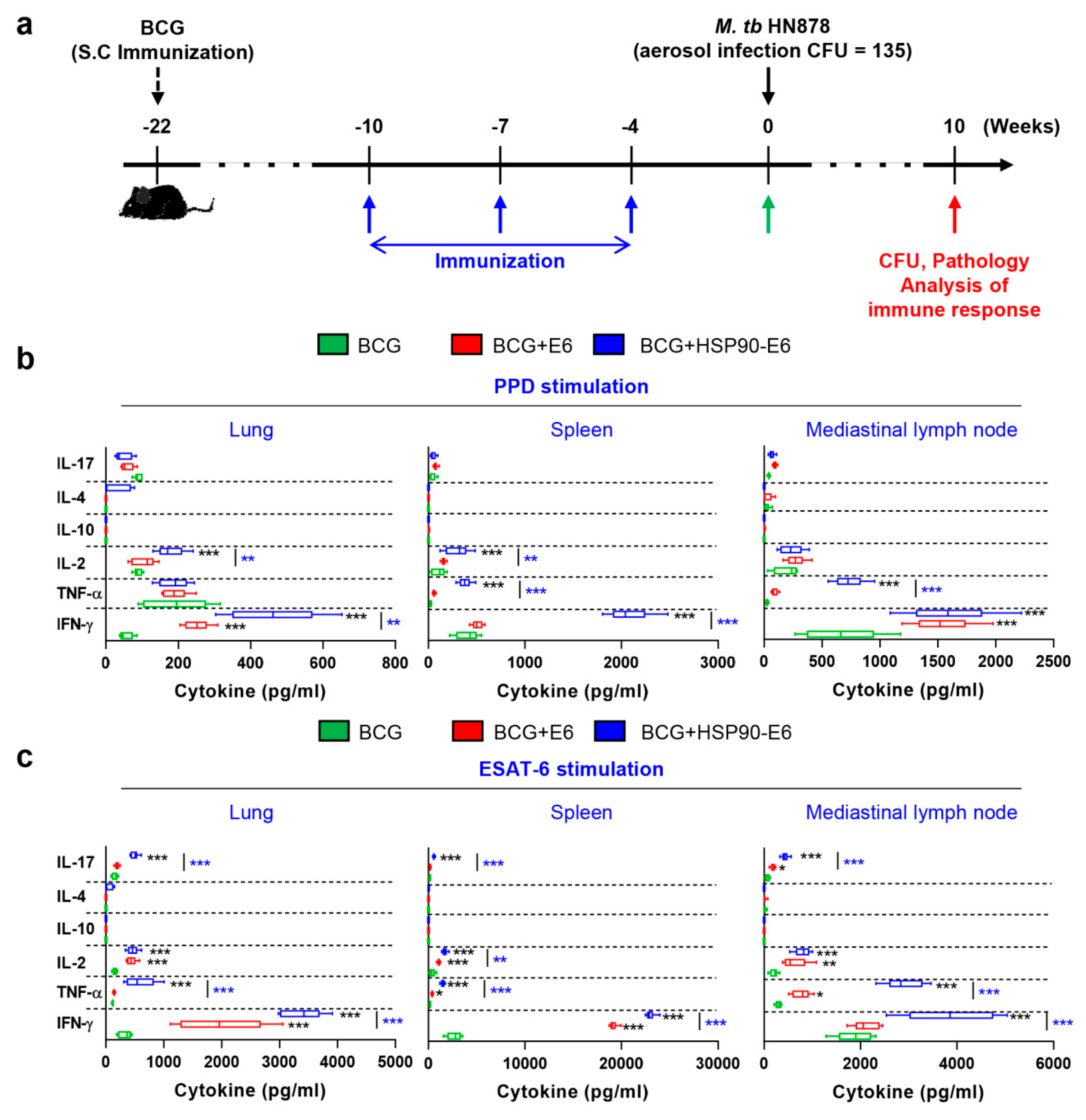
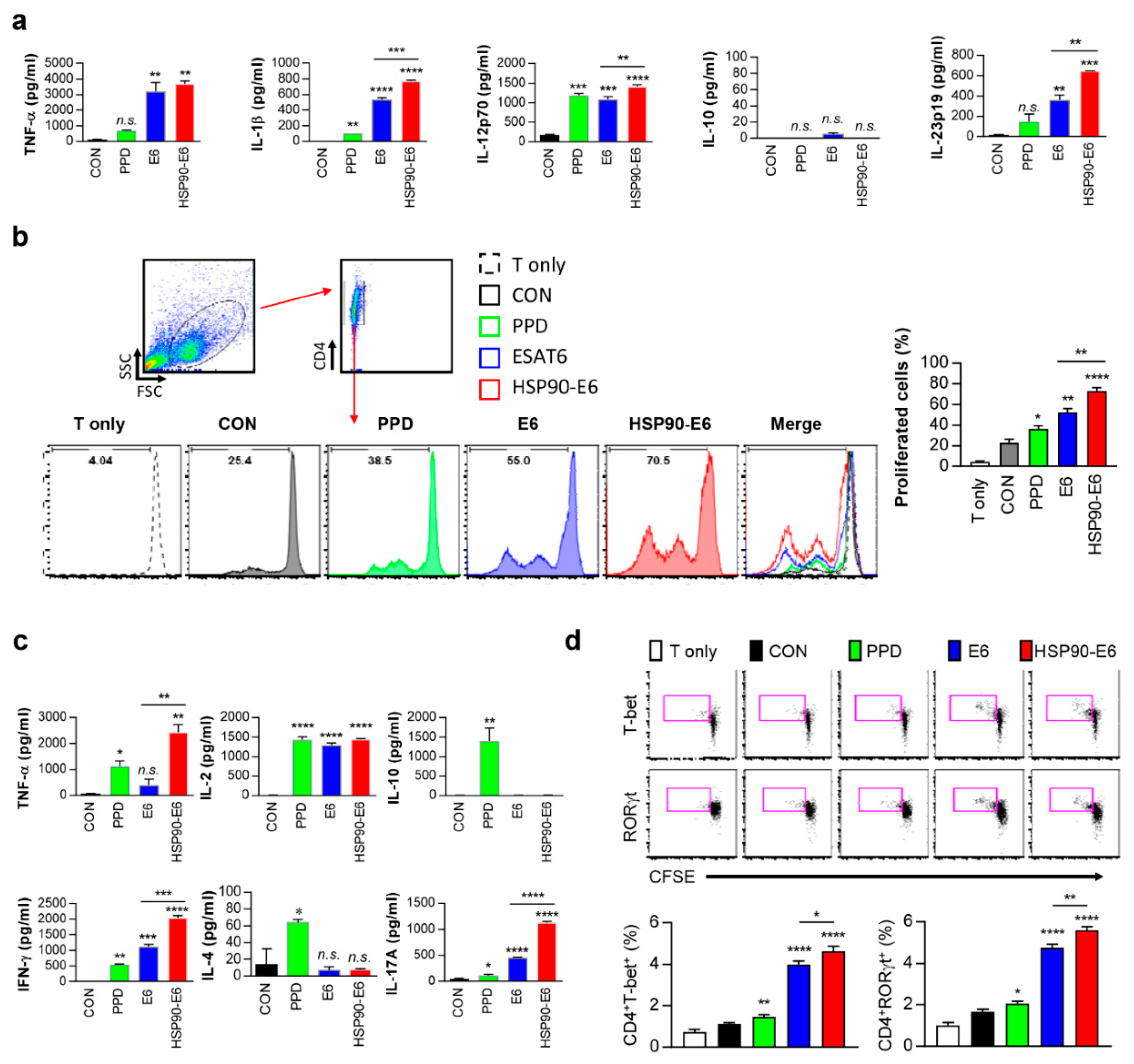
3.1. Characterisation of the Immune Responses induced by HSP90-E6
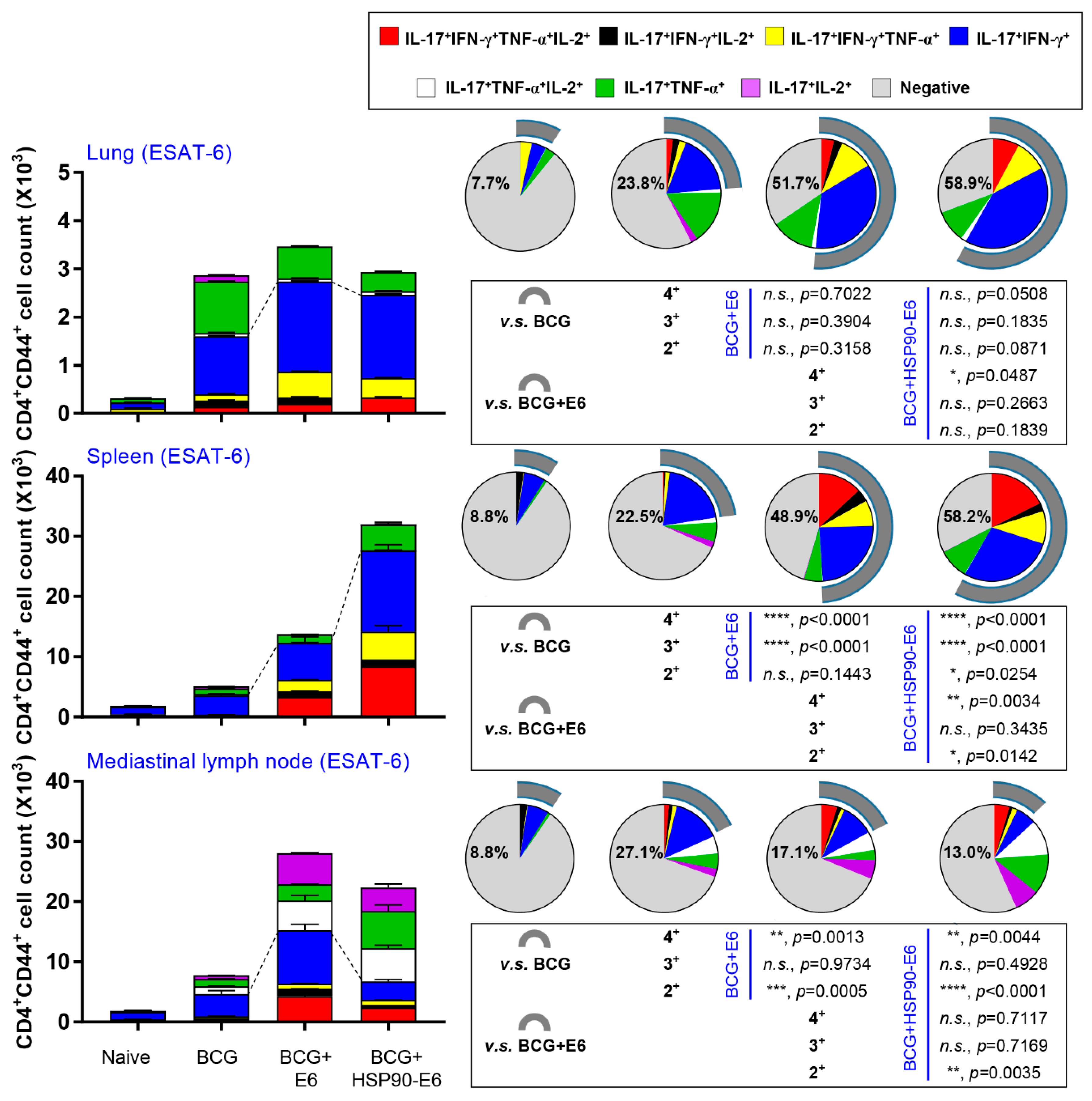
3.2. HSP90-E6/CIA05 induces Ag-Specific Multifunctional T-Cells
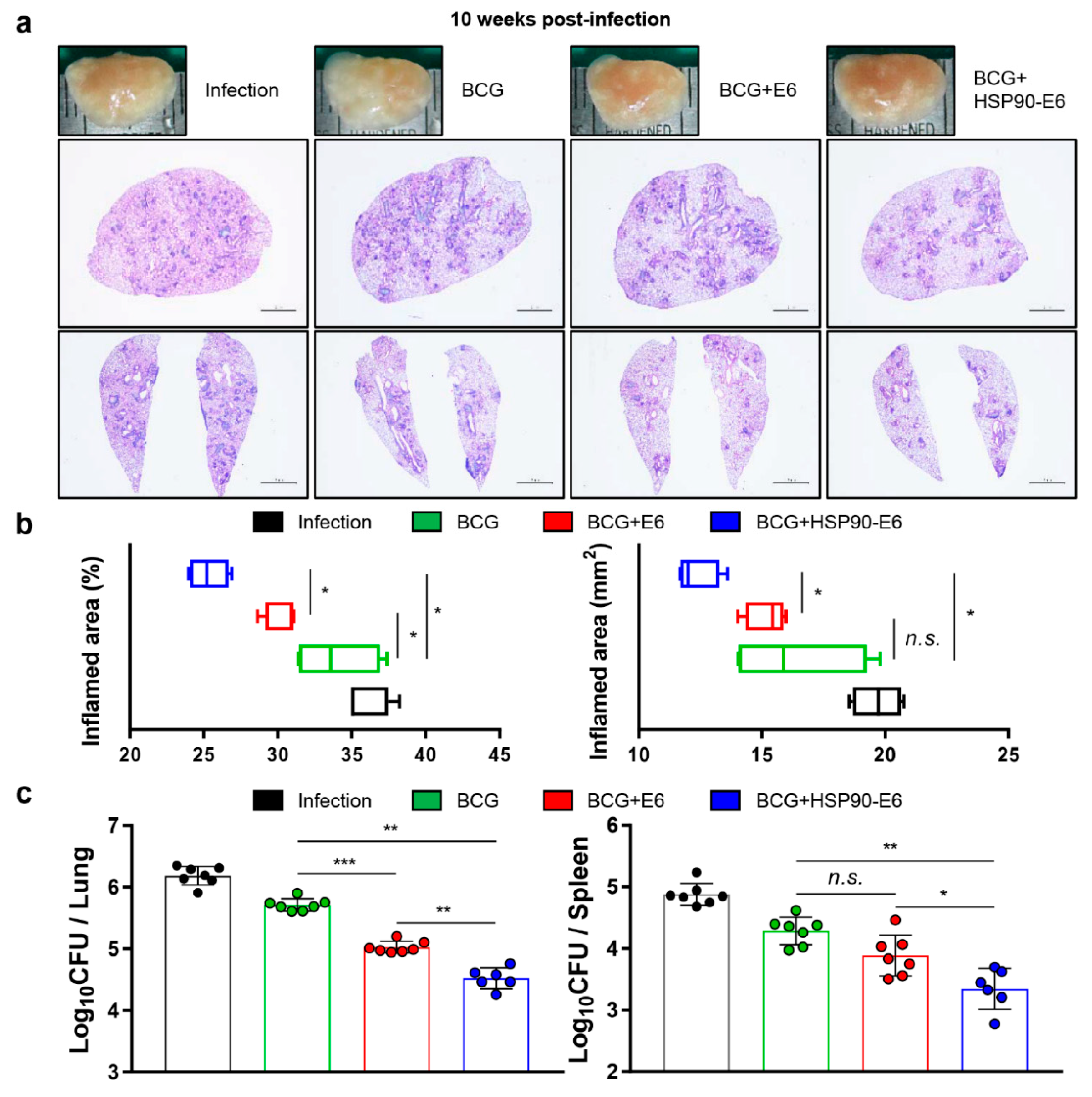
3.3. Protective Efficacy of HSP90-E6 Boosting against Hypervirulent Mtb HN878
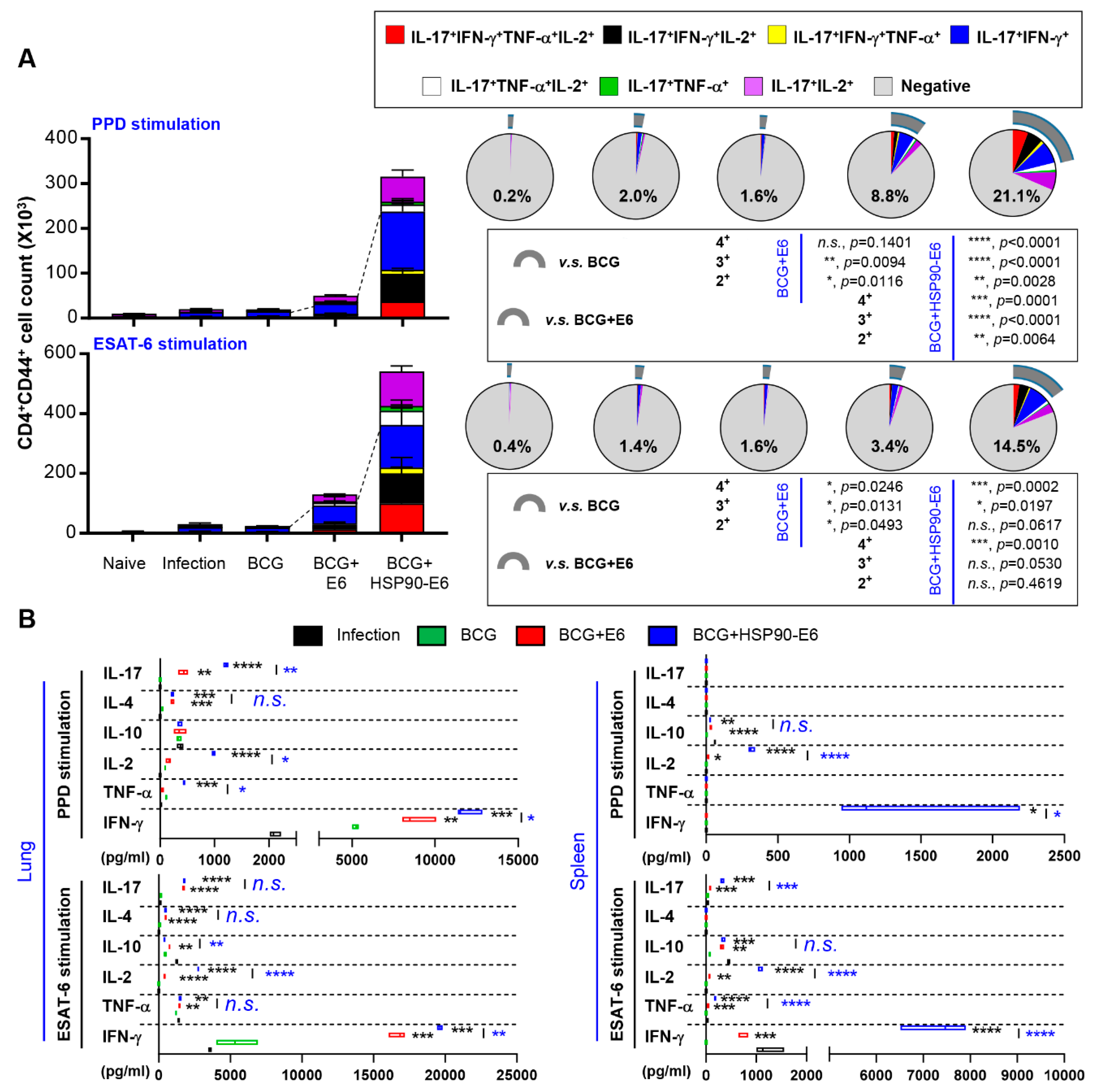
3.4. Multifunctional T-Cells and Cytokine Profiles in Mice Immunised with BCG Prime HSP90-E6/CIA05 Boosting after Challenge with Mtb HN878
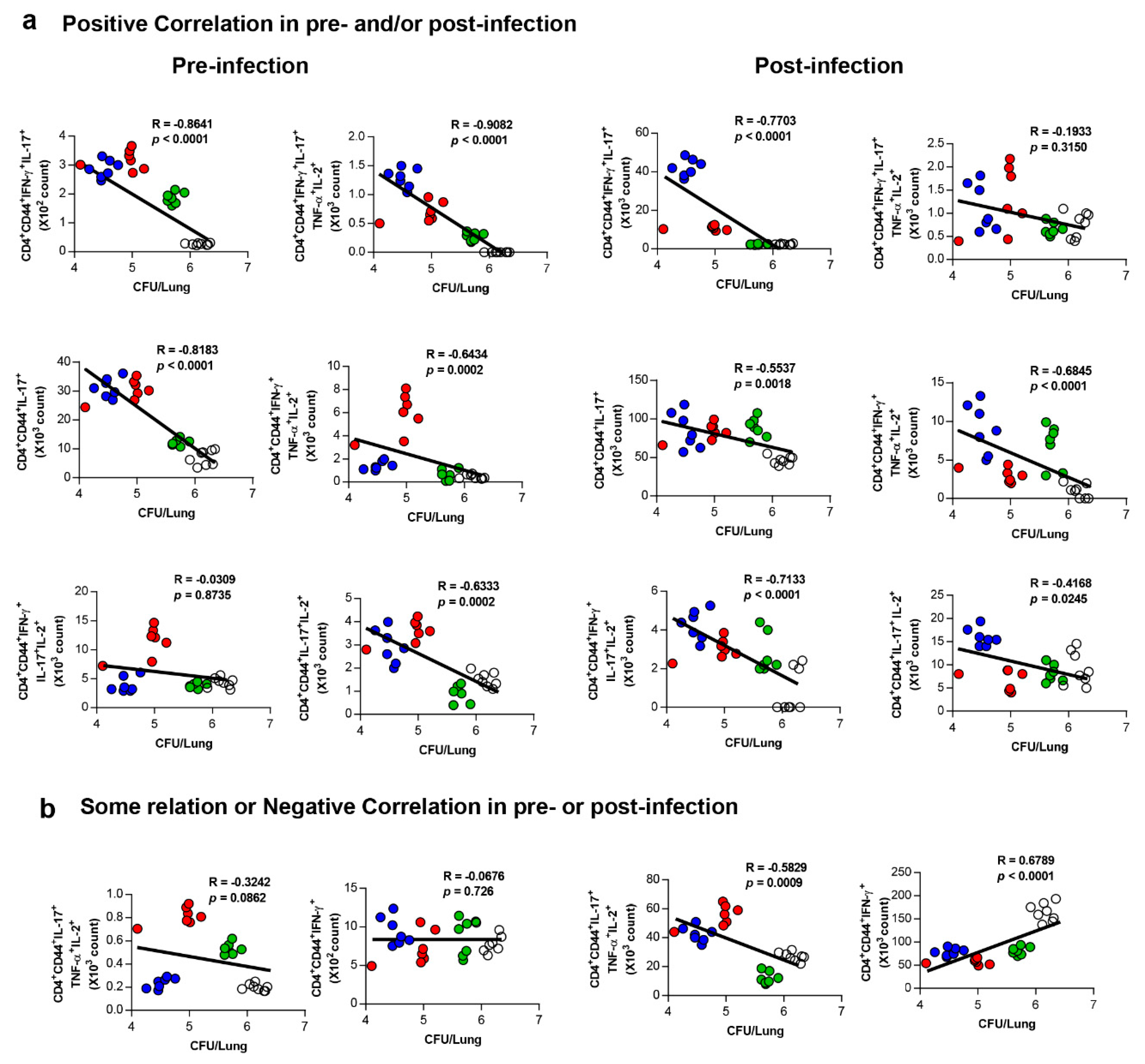
3.5. Correlates of Protection for Improved BCG Boosting Efficacy of BCG-HSP90-E6/CIA05 in the Lungs
3.6. Th1- and Th17-Related Responses are Simultaneously induced in DCs and Co-Cultured CD4+ T-Cells upon Stimulation with HSP90-E6
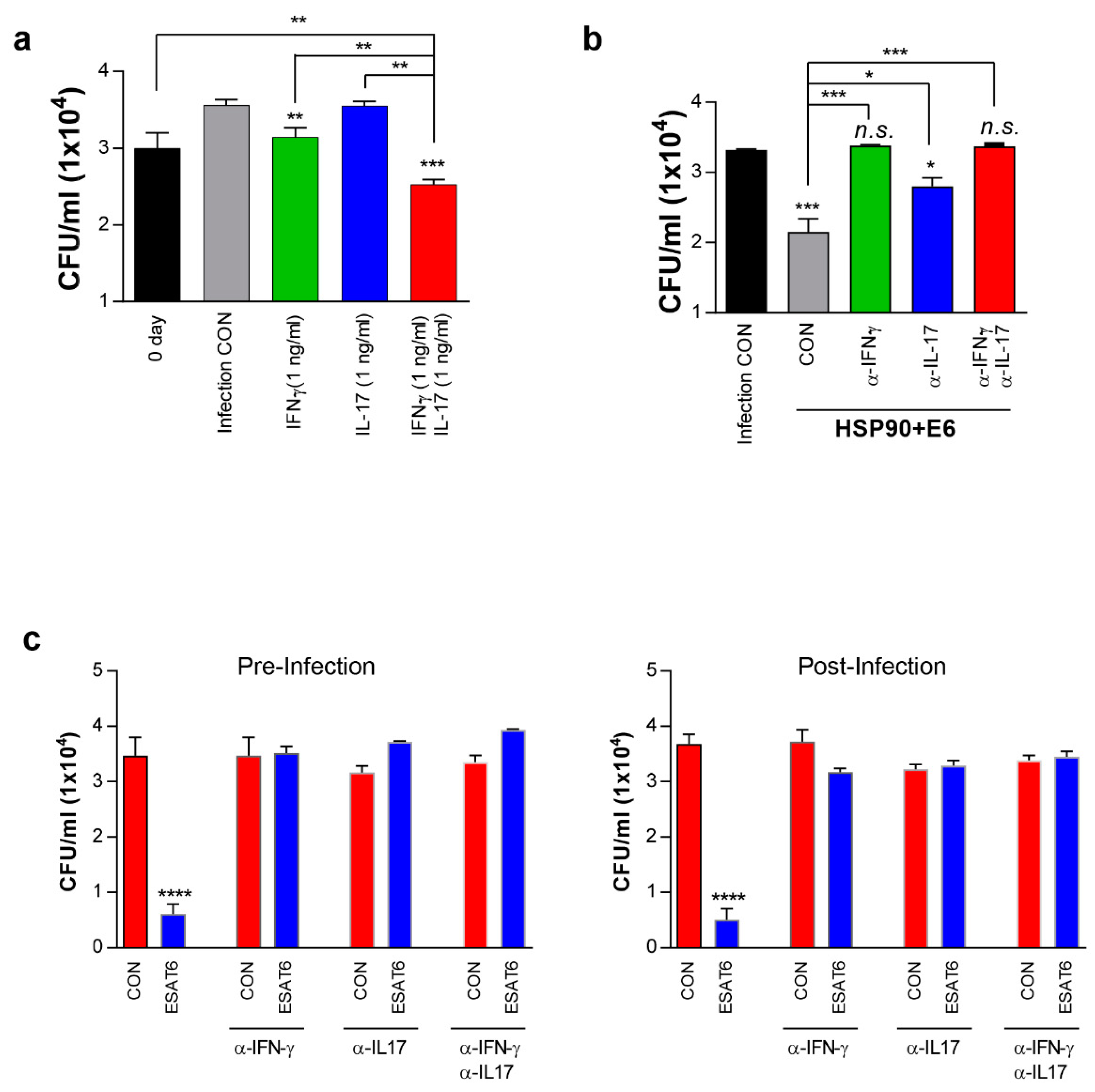
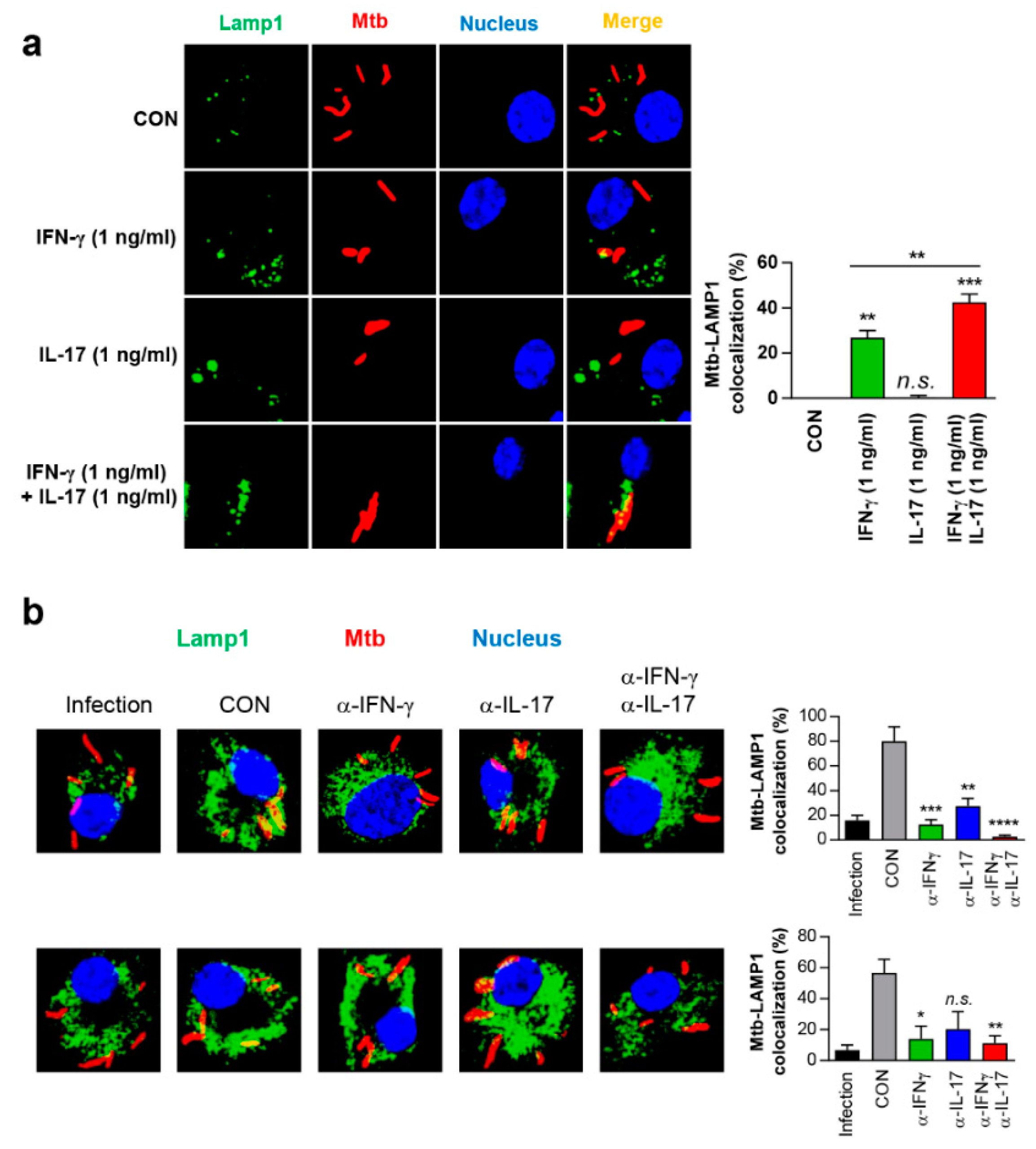
3.7. IFN-γ and IL-17 Synergistically Exert Anti-Mycobacterial Activity Via Enhanced Phagolysosomal Maturation
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Bloom, B.R.; Atun, R.; Cohen, T.; Dye, C.; Fraser, H.; Gomez, G.B.; Knight, G.; Murray, M.; Nardell, E.; Rubin, E.; et al. Tuberculosis. In Major Infectious Diseases, 3rd ed.; Holmes, K.K., Bertozzi, S., Bloom, B.R., Jha, P., Eds.; The World Bank: Washington, DC, USA, 2017. [Google Scholar] [CrossRef]
- Zumla, A.; George, A.; Sharma, V.; Herbert, R.H.; Oxley, A.; Oliver, M. The WHO 2014 global tuberculosis report—Further to go. Lancet Glob. Health 2015, 3, e10–e12. [Google Scholar] [CrossRef]
- Brandt, L.; Feino Cunha, J.; Weinreich Olsen, A.; Chilima, B.; Hirsch, P.; Appelberg, R.; Andersen, P. Failure of the Mycobacterium bovis BCG vaccine: Some species of environmental mycobacteria block multiplication of BCG and induction of protective immunity to tuberculosis. Infect. Immun. 2002, 70, 672–678. [Google Scholar] [CrossRef] [PubMed]
- Pitt, J.M.; Blankley, S.; McShane, H.; O’Garra, A. Vaccination against tuberculosis: How can we better BCG? Microb. Pathog. 2013, 58, 2–16. [Google Scholar] [CrossRef] [PubMed]
- Nemes, E.; Geldenhuys, H.; Rozot, V.; Rutkowski, K.T.; Ratangee, F.; Bilek, N.; Mabwe, S.; Makhethe, L.; Erasmus, M.; Toefy, A.; et al. Prevention of M. tuberculosis infection with H4:IC31 vaccine or BCG revaccination. N. Engl. J. Med. 2018, 379, 138–149. [Google Scholar] [CrossRef]
- Van Der Meeren, O.; Hatherill, M.; Nduba, V.; Wilkinson, R.J.; Muyoyeta, M.; Van Brakel, E.; Ayles, H.M.; Henostroza, G.; Thienemann, F.; Scriba, T.J.; et al. Phase 2b controlled trial of M72/AS01E vaccine to prevent tuberculosis. N. Engl. J. Med. 2018, 379, 1621–1634. [Google Scholar] [CrossRef] [PubMed]
- Penn-Nicholson, A.; Tameris, M.; Smit, E.; Day, T.A.; Musvosvi, M.; Jayashankar, L.; Vergara, J.; Mabwe, S.; Bilek, N.; Geldenhuys, H.; et al. Safety and immunogenicity of the novel tuberculosis vaccine ID93 + GLA-SE in BCG-vaccinated healthy adults in South Africa: A randomised, double-blind, placebo-controlled phase 1 trial. Lancet Respir. Med. 2018, 6, 287–298. [Google Scholar] [CrossRef]
- Reed, S.G.; Coler, R.N.; Dalemans, W.; Tan, E.V.; DeLa Cruz, E.C.; Basaraba, R.J.; Orme, I.M.; Skeiky, Y.A.; Alderson, M.R.; Cowgill, K.D.; et al. Defined tuberculosis vaccine, Mtb72F/AS02A, evidence of protection in cynomolgus monkeys. Proc. Natl. Acad. Sci. USA 2009, 106, 2301–2306. [Google Scholar] [CrossRef]
- Billeskov, R.; Elvang, T.T.; Andersen, P.L.; Dietrich, J. The HyVac4 subunit vaccine efficiently boosts BCG-primed anti-mycobacterial protective immunity. PLoS ONE 2012, 7, e39909. [Google Scholar] [CrossRef]
- Choi, H.G.; Choi, S.; Back, Y.W.; Paik, S.; Park, H.S.; Kim, W.S.; Kim, H.; Cha, S.B.; Choi, C.H.; Shin, S.J.; et al. Rv2299c, a novel dendritic cell-activating antigen of Mycobacterium tuberculosis, fused-ESAT-6 subunit vaccine confers improved and durable protection against the hypervirulent strain HN878 in mice. Oncotarget 2017, 8, 19947–19967. [Google Scholar] [CrossRef] [PubMed]
- Wui, S.R.; Kim, H.K.; Han, J.E.; Kim, J.M.; Kim, Y.H.; Chun, J.H.; Cho, Y.J.; Lee, N.G. A combination of the TLR4 agonist CIA05 and alum promotes the immune responses to Bacillus anthracis protective antigen in mice. Int. Immunopharmacol. 2011, 11, 1195–1204. [Google Scholar] [CrossRef]
- Cooper, A.M. Cell-mediated immune responses in tuberculosis. Annu. Rev. Immunol. 2009, 27, 393–422. [Google Scholar] [CrossRef]
- Khader, S.A.; Bell, G.K.; Pearl, J.E.; Fountain, J.J.; Rangel-Moreno, J.; Cilley, G.E.; Shen, F.; Eaton, S.M.; Gaffen, S.L.; Swain, S.L.; et al. IL-23 and IL-17 in the establishment of protective pulmonary CD4+ T cell responses after vaccination and during Mycobacterium tuberculosis challenge. Nat. Immunol. 2007, 8, 369–377. [Google Scholar] [CrossRef] [PubMed]
- Cooper, A.M.; Dalton, D.K.; Stewart, T.A.; Griffin, J.P.; Russell, D.G.; Orme, I.M. Disseminated tuberculosis in interferon gamma gene-disrupted mice. J. Exp. Med. 1993, 178, 2243–2247. [Google Scholar] [CrossRef] [PubMed]
- Green, A.M.; Difazio, R.; Flynn, J.L. IFN-gamma from CD4 T cells is essential for host survival and enhances CD8 T cell function during Mycobacterium tuberculosis infection. J. Immunol. 2013, 190, 270–277. [Google Scholar] [CrossRef] [PubMed]
- Goldsack, L.; Kirman, J.R. Half-truths and selective memory: Interferon gamma, CD4(+) T cells and protective memory against tuberculosis. Tuberculosis 2007, 87, 465–473. [Google Scholar] [CrossRef]
- Tchilian, E.Z.; Desel, C.; Forbes, E.K.; Bandermann, S.; Sander, C.R.; Hill, A.V.; McShane, H.; Kaufmann, S.H. Immunogenicity and protective efficacy of prime-boost regimens with recombinant (delta)ureC hly+ Mycobacterium bovis BCG and modified vaccinia virus ankara expressing M. tuberculosis antigen 85A against murine tuberculosis. Infect. Immun. 2009, 77, 622–631. [Google Scholar] [CrossRef]
- Leal, I.S.; Smedegard, B.; Andersen, P.; Appelberg, R. Failure to induce enhanced protection against tuberculosis by increasing T-cell-dependent interferon-gamma generation. Immunology 2001, 104, 157–161. [Google Scholar] [CrossRef]
- Lindenstrom, T.; Agger, E.M.; Korsholm, K.S.; Darrah, P.A.; Aagaard, C.; Seder, R.A.; Rosenkrands, I.; Andersen, P. Tuberculosis subunit vaccination provides long-term protective immunity characterized by multifunctional CD4 memory T cells. J. Immunol. 2009, 182, 8047–8055. [Google Scholar] [CrossRef]
- Aagaard, C.S.; Hoang, T.T.; Vingsbo-Lundberg, C.; Dietrich, J.; Andersen, P. Quality and vaccine efficacy of CD4+ T cell responses directed to dominant and subdominant epitopes in ESAT-6 from Mycobacterium tuberculosis. J. Immunol. 2009, 183, 2659–2668. [Google Scholar] [CrossRef]
- Scriba, T.J.; Tameris, M.; Mansoor, N.; Smit, E.; van der Merwe, L.; Isaacs, F.; Keyser, A.; Moyo, S.; Brittain, N.; Lawrie, A.; et al. Modified vaccinia Ankara-expressing Ag85A, a novel tuberculosis vaccine, is safe in adolescents and children, and induces polyfunctional CD4+ T cells. Eur. J. Immunol. 2010, 40, 279–290. [Google Scholar] [CrossRef]
- Abel, B.; Tameris, M.; Mansoor, N.; Gelderbloem, S.; Hughes, J.; Abrahams, D.; Makhethe, L.; Erasmus, M.; de Kock, M.; van der Merwe, L.; et al. The novel tuberculosis vaccine, AERAS-402, induces robust and polyfunctional CD4+ and CD8+ T cells in adults. Am. J. Respir. Crit. Care Med. 2010, 181, 1407–1417. [Google Scholar] [CrossRef] [PubMed]
- Wozniak, T.M.; Saunders, B.M.; Ryan, A.A.; Britton, W.J. Mycobacterium bovis BCG-specific Th17 cells confer partial protection against Mycobacterium tuberculosis infection in the absence of gamma interferon. Infect. Immun. 2010, 78, 4187–4194. [Google Scholar] [CrossRef] [PubMed]
- Scriba, T.J.; Kalsdorf, B.; Abrahams, D.A.; Isaacs, F.; Hofmeister, J.; Black, G.; Hassan, H.Y.; Wilkinson, R.J.; Walzl, G.; Gelderbloem, S.J.; et al. Distinct, specific IL-17- and IL-22-producing CD4+ T cell subsets contribute to the human anti-mycobacterial immune response. J. Immunol. 2008, 180, 1962–1970. [Google Scholar] [CrossRef] [PubMed]
- Okamoto Yoshida, Y.; Umemura, M.; Yahagi, A.; O’Brien, R.L.; Ikuta, K.; Kishihara, K.; Hara, H.; Nakae, S.; Iwakura, Y.; Matsuzaki, G. Essential role of IL-17A in the formation of a mycobacterial infection-induced granuloma in the lung. J. Immunol. 2010, 184, 4414–4422. [Google Scholar] [CrossRef] [PubMed]
- Khader, S.A.; Guglani, L.; Rangel-Moreno, J.; Gopal, R.; Junecko, B.A.; Fountain, J.J.; Martino, C.; Pearl, J.E.; Tighe, M.; Lin, Y.Y.; et al. IL-23 is required for long-term control of Mycobacterium tuberculosis and B cell follicle formation in the infected lung. J. Immunol. 2011, 187, 5402–5407. [Google Scholar] [CrossRef] [PubMed]
- Freches, D.; Korf, H.; Denis, O.; Havaux, X.; Huygen, K.; Romano, M. Mice genetically inactivated in interleukin-17A receptor are defective in long-term control of Mycobacterium tuberculosis infection. Immunology 2013, 140, 220–231. [Google Scholar] [CrossRef]
- Gopal, R.; Monin, L.; Slight, S.; Uche, U.; Blanchard, E.; Fallert Junecko, B.A.; Ramos-Payan, R.; Stallings, C.L.; Reinhart, T.A.; Kolls, J.K.; et al. Unexpected role for IL-17 in protective immunity against hypervirulent Mycobacterium tuberculosis HN878 infection. PLoS Pathog. 2014, 10, e1004099. [Google Scholar] [CrossRef] [PubMed]
- Cruz, A.; Torrado, E.; Carmona, J.; Fraga, A.G.; Costa, P.; Rodrigues, F.; Appelberg, R.; Correia-Neves, M.; Cooper, A.M.; Saraiva, M.; et al. BCG vaccination-induced long-lasting control of Mycobacterium tuberculosis correlates with the accumulation of a novel population of CD4(+)IL-17(+)TNF(+)IL-2(+) T cells. Vaccine 2015, 33, 85–91. [Google Scholar] [CrossRef] [PubMed]
- Desel, C.; Dorhoi, A.; Bandermann, S.; Grode, L.; Eisele, B.; Kaufmann, S.H. Recombinant BCG DeltaureC hly+ induces superior protection over parental BCG by stimulating a balanced combination of type 1 and type 17 cytokine responses. J. Infect. Dis. 2011, 204, 1573–1584. [Google Scholar] [CrossRef] [PubMed]
- Cha, S.B.; Kim, W.S.; Kim, J.S.; Kim, H.; Kwon, K.W.; Han, S.J.; Eum, S.Y.; Cho, S.N.; Shin, S.J. Repeated aerosolized-boosting with gamma-irradiated Mycobacterium bovis BCG confers improved pulmonary protection against the hypervirulent Mycobacterium tuberculosis strain HN878 in mice. PLoS ONE 2015, 10, e0141577. [Google Scholar] [CrossRef]
- Kwon, K.W.; Choi, H.H.; Han, S.J.; Kim, J.S.; Kim, W.S.; Kim, H.; Kim, L.H.; Kang, S.M.; Park, J.; Shin, S.J. Vaccine efficacy of a Mycobacterium tuberculosis Beijing-specific proline-glutamic acid (PE) antigen against highly virulent outbreak isolates. FASEB J. 2019, 33, 6483–6496. [Google Scholar] [CrossRef]
- Jeong, Y.I.; Kim, S.W.; Jung, I.D.; Lee, J.S.; Chang, J.H.; Lee, C.M.; Chun, S.H.; Yoon, M.S.; Kim, G.T.; Ryu, S.W.; et al. Curcumin suppresses the induction of indoleamine 2,3-dioxygenase by blocking the janus-activated kinase-protein kinase Cdelta-STAT1 signaling pathway in interferon-gamma-stimulated murine dendritic cells. J. Biol. Chem. 2009, 284, 3700–3708. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.I.; Whang, J.; Choi, H.G.; Son, Y.J.; Jeon, H.S.; Back, Y.W.; Park, H.S.; Paik, S.; Park, J.K.; Choi, C.H.; et al. Mycobacterium avium MAV2054 protein induces macrophage apoptosis by targeting mitochondria and reduces intracellular bacterial growth. Sci. Rep. 2016, 6, 37804. [Google Scholar] [CrossRef] [PubMed]
- Derrick, S.C.; Yabe, I.M.; Yang, A.; Morris, S.L. Vaccine-induced anti-tuberculosis protective immunity in mice correlates with the magnitude and quality of multifunctional CD4 T cells. Vaccine 2011, 29, 2902–2909. [Google Scholar] [CrossRef]
- Gopal, R.; Lin, Y.; Obermajer, N.; Slight, S.; Nuthalapati, N.; Ahmed, M.; Kalinski, P.; Khader, S.A. IL-23-dependent IL-17 drives Th1-cell responses following Mycobacterium bovis BCG vaccination. Eur. J. Immunol. 2012, 42, 364–373. [Google Scholar] [CrossRef] [PubMed]
- Torrado, E.; Cooper, A.M. IL-17 and Th17 cells in tuberculosis. Cytokine Growth Factor Rev. 2010, 21, 455–462. [Google Scholar] [CrossRef] [PubMed]
- Jurado, J.O.; Pasquinelli, V.; Alvarez, I.B.; Pena, D.; Rovetta, A.I.; Tateosian, N.L.; Romeo, H.E.; Musella, R.M.; Palmero, D.; Chuluyan, H.E.; et al. IL-17 and IFN-gamma expression in lymphocytes from patients with active tuberculosis correlates with the severity of the disease. J. Leukoc. Biol. 2012, 91, 991–1002. [Google Scholar] [CrossRef] [PubMed]
- Pitt, J.M.; Stavropoulos, E.; Redford, P.S.; Beebe, A.M.; Bancroft, G.J.; Young, D.B.; O’Garra, A. Blockade of IL-10 signaling during bacillus calmette-guerin vaccination enhances and sustains Th1, Th17, and innate lymphoid IFN-gamma and IL-17 responses and increases protection to Mycobacterium tuberculosis infection. J. Immunol. 2012, 189, 4079–4087. [Google Scholar] [CrossRef] [PubMed]
- Khader, S.A.; Pearl, J.E.; Sakamoto, K.; Gilmartin, L.; Bell, G.K.; Jelley-Gibbs, D.M.; Ghilardi, N.; deSauvage, F.; Cooper, A.M. IL-23 compensates for the absence of IL-12p70 and is essential for the IL-17 response during tuberculosis but is dispensable for protection and antigen-specific IFN-gamma responses if IL-12p70 is available. J. Immunol. 2005, 175, 788–795. [Google Scholar] [CrossRef]
- Lyadova, I.V.; Panteleev, A.V. Th1 and Th17 cells in tuberculosis: Protection, pathology, and biomarkers. Mediat. Inflamm. 2015, 2015, 854507. [Google Scholar] [CrossRef] [PubMed]
- Allie, N.; Grivennikov, S.I.; Keeton, R.; Hsu, N.J.; Bourigault, M.L.; Court, N.; Fremond, C.; Yeremeev, V.; Shebzukhov, Y.; Ryffel, B.; et al. Prominent role for T cell-derived tumour necrosis factor for sustained control of Mycobacterium tuberculosis infection. Sci. Rep. 2013, 3, 1809. [Google Scholar] [CrossRef] [PubMed]
- Segueni, N.; Benmerzoug, S.; Rose, S.; Gauthier, A.; Bourigault, M.L.; Reverchon, F.; Philippeau, A.; Erard, F.; Le Bert, M.; Bouscayrol, H.; et al. Innate myeloid cell TNFR1 mediates first line defence against primary Mycobacterium tuberculosis infection. Sci. Rep. 2016, 6, 22454. [Google Scholar] [CrossRef]
- Filipe-Santos, O.; Bustamante, J.; Chapgier, A.; Vogt, G.; de Beaucoudrey, L.; Feinberg, J.; Jouanguy, E.; Boisson-Dupuis, S.; Fieschi, C.; Picard, C.; et al. Inborn errors of IL-12/23- and IFN-gamma-mediated immunity: Molecular, cellular, and clinical features. Semin. Immunol. 2006, 18, 347–361. [Google Scholar] [CrossRef] [PubMed]
- Cooper, A.M.; Kipnis, A.; Turner, J.; Magram, J.; Ferrante, J.; Orme, I.M. Mice lacking bioactive IL-12 can generate protective, antigen-specific cellular responses to mycobacterial infection only if the IL-12 p40 subunit is present. J. Immunol. 2002, 168, 1322–1327. [Google Scholar] [CrossRef] [PubMed]
- Vilaplana, C.; Prats, C.; Marzo, E.; Barril, C.; Vegue, M.; Diaz, J.; Valls, J.; Lopez, D.; Cardona, P.J. To achieve an earlier IFN-gamma response is not sufficient to control Mycobacterium tuberculosis infection in mice. PLoS ONE 2014, 9, e100830. [Google Scholar] [CrossRef] [PubMed]
- Gallegos, A.M.; Van Heijst, J.W.; Samstein, M.; Su, X.; Pamer, E.G.; Glickman, M.S. A gamma interferon independent mechanism of CD4 T cell mediated control of M. tuberculosis infection in vivo. PLoS Pathog. 2011, 7, e1002052. [Google Scholar] [CrossRef] [PubMed]
- Torrado, E.; Cooper, A.M. What do we really know about how CD4 T cells control Mycobacterium tuberculosis? PLoS Pathog. 2011, 7, e1002196. [Google Scholar] [CrossRef] [PubMed]
- Orr, M.T.; Ireton, G.C.; Beebe, E.A.; Huang, P.W.; Reese, V.A.; Argilla, D.; Coler, R.N.; Reed, S.G. Immune subdominant antigens as vaccine candidates against Mycobacterium tuberculosis. J. Immunol. 2014, 193, 2911–2918. [Google Scholar] [CrossRef] [PubMed]
- Aguilo, N.; Alvarez-Arguedas, S.; Uranga, S.; Marinova, D.; Monzon, M.; Badiola, J.; Martin, C. Pulmonary but not subcutaneous delivery of BCG vaccine confers protection to tuberculosis-susceptible mice by an interleukin 17-dependent mechanism. J. Infect. Dis. 2016, 213, 831–839. [Google Scholar] [CrossRef]
- Wareham, A.S.; Tree, J.A.; Marsh, P.D.; Butcher, P.D.; Dennis, M.; Sharpe, S.A. Evidence for a role for interleukin-17, Th17 cells and iron homeostasis in protective immunity against tuberculosis in cynomolgus macaques. PLoS ONE 2014, 9, e88149. [Google Scholar] [CrossRef]
- Dijkman, K.; Sombroek, C.C.; Vervenne, R.A.W.; Hofman, S.O.; Boot, C.; Remarque, E.J.; Kocken, C.H.M.; Ottenhoff, T.H.M.; Kondova, I.; Khayum, M.A.; et al. Prevention of tuberculosis infection and disease by local BCG in repeatedly exposed rhesus macaques. Nat. Med. 2019, 25, 255–262. [Google Scholar] [CrossRef] [PubMed]
- Shen, H.; Chen, Z.W. The crucial roles of Th17-related cytokines/signal pathways in M. tuberculosis infection. Cell. Mol. Immunol. 2018, 15, 216–225. [Google Scholar] [CrossRef] [PubMed]
- Mourik, B.C.; Lubberts, E.; De Steenwinkel, J.E.M.; Ottenhoff, T.H.M.; Leenen, P.J.M. Interactions between Type 1 Interferons and the Th17 Response in Tuberculosis: Lessons Learned from Autoimmune Diseases. Front. Immunol. 2017, 8, 294. [Google Scholar] [CrossRef]
- Scriba, T.J.; Penn-Nicholson, A.; Shankar, S.; Hraha, T.; Thompson, E.G.; Sterling, D.; Nemes, E.; Darboe, F.; Suliman, S.; Amon, L.M.; et al. Sequential inflammatory processes define human progression from M. tuberculosis infection to tuberculosis disease. PLoS Pathog. 2017, 13, e1006687. [Google Scholar] [CrossRef] [PubMed]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Choi, H.-G.; Kwon, K.W.; Choi, S.; Back, Y.W.; Park, H.-S.; Kang, S.M.; Choi, E.; Shin, S.J.; Kim, H.-J. Antigen-Specific IFN-γ/IL-17-Co-Producing CD4+ T-Cells are the Determinants for Protective Efficacy of Tuberculosis Subunit Vaccine. Vaccines 2020, 8, 300. https://doi.org/10.3390/vaccines8020300
Choi H-G, Kwon KW, Choi S, Back YW, Park H-S, Kang SM, Choi E, Shin SJ, Kim H-J. Antigen-Specific IFN-γ/IL-17-Co-Producing CD4+ T-Cells are the Determinants for Protective Efficacy of Tuberculosis Subunit Vaccine. Vaccines. 2020; 8(2):300. https://doi.org/10.3390/vaccines8020300
Chicago/Turabian StyleChoi, Han-Gyu, Kee Woong Kwon, Seunga Choi, Yong Woo Back, Hye-Soo Park, Soon Myung Kang, Eunsol Choi, Sung Jae Shin, and Hwa-Jung Kim. 2020. "Antigen-Specific IFN-γ/IL-17-Co-Producing CD4+ T-Cells are the Determinants for Protective Efficacy of Tuberculosis Subunit Vaccine" Vaccines 8, no. 2: 300. https://doi.org/10.3390/vaccines8020300
APA StyleChoi, H.-G., Kwon, K. W., Choi, S., Back, Y. W., Park, H.-S., Kang, S. M., Choi, E., Shin, S. J., & Kim, H.-J. (2020). Antigen-Specific IFN-γ/IL-17-Co-Producing CD4+ T-Cells are the Determinants for Protective Efficacy of Tuberculosis Subunit Vaccine. Vaccines, 8(2), 300. https://doi.org/10.3390/vaccines8020300





