Immunization Status against Measles, Mumps, Rubella and Varicella in a Large Population of Internationally Adopted Children Referred to Meyer Children’s University Hospital from 2009 to 2018
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Population
2.2. Study Design
2.3. Seroprevalence of Antibody Protection against Measles, Mumps, Rubella, and Varicella
2.4. Concordance between Vaccine Documentation and Serotesting Results
2.5. Statistical Analysis
3. Results
3.1. Characteristics of the Study Population
3.2. Evaluation of the Immunological Coverage by Means of Serological Tests
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- European Centre for Disease Prevention and Control. Public health guidance on screening and vaccination for infectious diseases in newly arrived migrants within the EU/EEA. Available online: https://www.ecdc.europa.eu/sites/portal/files/documents/Public%20health%20guidance%20on%20screening%20and%20vaccination%20of%20migrants%20in%20the%20EU%20EEA.pdf (accessed on 1 August 2019).
- World Health Organization. Measles no longer endemic in 79% of the WHO European Region. Available online: http://www.euro.who.int/en/media-centre/sections/press-releases/2017/measles-no-longer-endemic-in-79-of-the-who-european-region (accessed on 2 September 2019).
- World Health Organization. European Region loses ground in effort to eliminate measles. Available online: http://www.euro.who.int/en/media-centre/sections/press-releases/2019/european-region-loses-ground-in-effort-to-eliminate-measles (accessed on 2 September 2019).
- European Centres for Disease Prevention and Control. Measles. Available online: https://www.ecdc.europa.eu/en/measles (accessed on 15 August 2019).
- The Portal of Epidemiology for Public Health by the Italian National Institute of Health. Latest updates. Available online: https://www.epicentro.iss.it/morbillo/aggiornamenti (accessed on 14 January 2020).
- Dixit, D.; Rajapakse, N.; Kuhn, S. Caring for kids new to Canada Immunizations: Bringing Newcomer Children Up-to-date. 2018. Available online: https://www.kidsnewtocanada.ca/screening/immunizations (accessed on 15 August 2019).
- Ministry of Health. National Vaccine Prevention Plan (PNPV) 2017–2019. Available online: https://www.gazzettaufficiale.it/eli/id/2017/02/18/17A01195/sg (accessed on 3 January 2020).
- Ministry of Health. Vaccinations of the Pediatric and Adolescent age–Vaccination Coverage. Available online: http://www.salute.gov.it/portale/documentazione/p6_2_8_3_1.jsp?lingua=italiano&id=20) (accessed on 3 January 2020).
- Nardone, A.; de Ory, F.; Carton, M.; Cohen, D.; van Damme, P.; Davidkin, I.; Rota, M.C.; de Melker, H.; Mossong, J.; Slacikova, M. The comparative sero-epidemiology of varicella zoster virus in 11 countries in the European region. Vaccine 2007, 25, 7866–7872. [Google Scholar] [CrossRef] [PubMed]
- Bechini, A.; Boccalini, S.; Baldo, V.; Cocchio, S.; Castiglia, P.; Gallo, T.; Giuffrida, S.; Locuratolo, F.; Tafuri, S.; Martinelli, D. Impact of universal vaccination against varicella in Italy. Hum. Vaccin. Immunother. 2015, 11, 63–71. [Google Scholar] [CrossRef] [PubMed]
- Boccalini, S.; Bonanni, P.; Bechini, A. Preparing to introduce the varicella vaccine into the Italian immunisation programme: Varicella-related hospitalisations in Tuscany, 2004–2012. Euro Surveill. 2016, 21. [Google Scholar] [CrossRef] [PubMed]
- Pieri, L.; Porchia, B.R.; Pieralli, F.; Varone, O.; Niccolai, G.; Roselli, A.; Boccalini, S.; Bonanni, P.; Bechini, A. Working Group of Tuscan LHU’s; Working Group of Tuscan LHU’s. Assessment of the effectiveness of the universal varicella vaccination program in Toscana (Italy), in the period 2010–2013. Epidemiol. Prev. 2015, 39, 119–123. [Google Scholar]
- Commission for Intercountry Adoptions. Data and Perspectives in Intercountry Adoptions. Summary Report Dossiers from 2016 to 2017. Available online: http://www.commissioneadozioni.it/media/1564/57b8c_summary_report_cai_en.pdf (accessed on 3 September 2019).
- Giordano, D.; Provenzano, S.; Santangelo, O.; Piazza, D.; Ferraro, D. Active immunization status against measles, mumps, rubella, hepatitis B in internationally adopted children, surveyed at the university hospital of Palermo, Sicily. Ann. Ig. 2018, 30, 431–435. [Google Scholar]
- De Monléon, J.; Regnier, F.; Ajana, F.; Baptiste, C.; Callamand, P. Mise à jour des vaccinations de l ’ enfant arrivant de l ’ étranger ( adopté, réfugié ou migrant ) en France Catch-up vaccination of worldwide newcoming ( adopted, refugee or migrant ) children in France. Arch. Pediatr. 2014, 21, 329–334. [Google Scholar] [CrossRef]
- Schulte, J.; Maloney, S.; Aronson, J.; Gabriel, P.; Zhou, J.; Saiman, L. Evaluating Acceptability and Completeness of Overseas Immunization Records of Internationally Adopted Children. Pediatrics 2002, 109, 2–5. [Google Scholar] [CrossRef]
- Centers for Disease Control and Prevention. CDC Yellow Book 2020: Health information for international Travel. International Adoption. Available online: https://wwwnc.cdc.gov/travel/yellowbook/2020/family-travel/international-adoption (accessed on 8 September 2019).
- World Health Organization. Vaccine-Preventable Diseases: Morning System 2019 Global Summary. 2019. Available online: http://apps.who.int/immunization_monitoring/globalsummary/diseases (accessed on 15 August 2019).
- Sollai, S.; Ghetti, F.; Bianchi, L.; De Martino, M.; Galli, L.; Chiappini, E. Infectious diseases prevalence, vaccination coverage, and diagnostic challenges in a population of internationally adopted children referred to a Tertiary Care Children’s Hospital from 2009 to 2015. Medicine 2017, 96, 1–10. [Google Scholar] [CrossRef]
- Viviano, E.; Cataldo, F.; Accomando, S.; Firenze, A.; Maria, R.; Romano, N. Immunization status of internationally adopted children in Italy. Vaccine 2006, 24, 4138–4143. [Google Scholar] [CrossRef]
- Giambi, C.; Del Manso, M.; Marchetti, G.; Olsson, K.; Adel Ali, K.; Declich, S.; Venice Survey Working Group. Immunisation of migrants in EU/EEA countries: Policies and practices. Vaccine 2019, 37, 5439–5451. [Google Scholar] [CrossRef]
- Schulpen, T.; van Seventer, A.; Rümke, H.; van Loon, A. Immunisation status of children adopted from China. Lancet 2001, 358, 2131–2132. [Google Scholar] [CrossRef]
- Miller, L.C.; Tseng, B.; Tirella, L.G.; Chan, W.; Feig, E. Health of Children Adopted from Ethiopia. Matern. Child Health J. 2008, 100, 599–605. [Google Scholar] [CrossRef] [PubMed]
- Cilleruelo, M.; de Ory, F.; Ruiz-Contrerasc, J.; González-González, R.; Mellado, M.; Garcia-Hortelanoa, M.; Herruzoe, R. Internationally adopted children: What vaccines should they receive. Vaccine 2008, 26, 5784–5790. [Google Scholar] [CrossRef] [PubMed]
- Staat, M.A.; Stadler, L.P.; Donauer, S.; Trehan, I.; Rice, M.; Salisbury, S. Serologic testing to verify the immune status of internationally adopted children against vaccine preventable diseases. Bone 2010, 23, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Chiappini, E.; Zaffaroni, M.; Bianconi, M.; Veneruso, G.; Grasso, N.; Garazzino, S.; Bona, G. Italian multicentre study found infectious and vaccine-preventable diseases in children adopted from Africa and recommends prompt medical screening. Acta Paediatr. 2018, 107, 1581–1586. [Google Scholar] [CrossRef] [PubMed]
- Stadler, L.; Mezoff, A.; Staat, M. Hepatitis B Virus Screening for Internationally Adopted Children. Pediatrics 2008, 122, 1223–1228. [Google Scholar] [CrossRef]
- Jones, V.F.; Committee On Early Childhood, Adoption, And Dependent Care. Comprehensive health evaluation of the newly adopted child. Pediatrics 2012, 129, e214–e223. [Google Scholar] [CrossRef]
- Verla-tebit, E.; Zhu, X.; Holsinger, E.; Mandalakas, A.M. Predictive Value of Immunization Records and Risk Factors for Immunization Failure in Internationally Adopted Children. Arch. Pediatr. Adolesc. Med. 2009, 163, 473–479. [Google Scholar] [CrossRef]
- Miller, L.; Chan, W.; Comfort, K.; Tirella, L. Health of Children Adopted From Guatemala: Comparison of Orphanage and Foster Care. Pediatrics 2005, 115, 710–717. [Google Scholar] [CrossRef]
- Italian Pediatric Society. New indications of the GLNBI-SIP (Working group for the migrant child of the Italian Pediatric Society) for the health care of the migrant minor. In Proceedings of the Consensus Conference GLNBI-SIP, Rome, Italy, 30 November 2013. [Google Scholar]
- Venturini, E.; Piccini, P.; Tersigni, C.; Chiappini, E.; Galli, L. Systematic review shows that immunising internationally adopted children is a major challenge for primary health care. Acta Paediatr. 2019, 108, 411–418. [Google Scholar] [CrossRef]
- Woudenberg, T.; van Binnendijk, R.; Veldhuijzen, I.; Woonink, F.; Ruijs, H.; van der Klis, F.; Kerkhof, J.; de Melker, H.; de Swart, R.; Hahné, S. Additional Evidence on Serological Correlates of Protection against Measles: An Observational Cohort Study among Once Vaccinated Children Exposed to Measles. Vaccines 2019, 7, 158. [Google Scholar] [CrossRef] [PubMed]
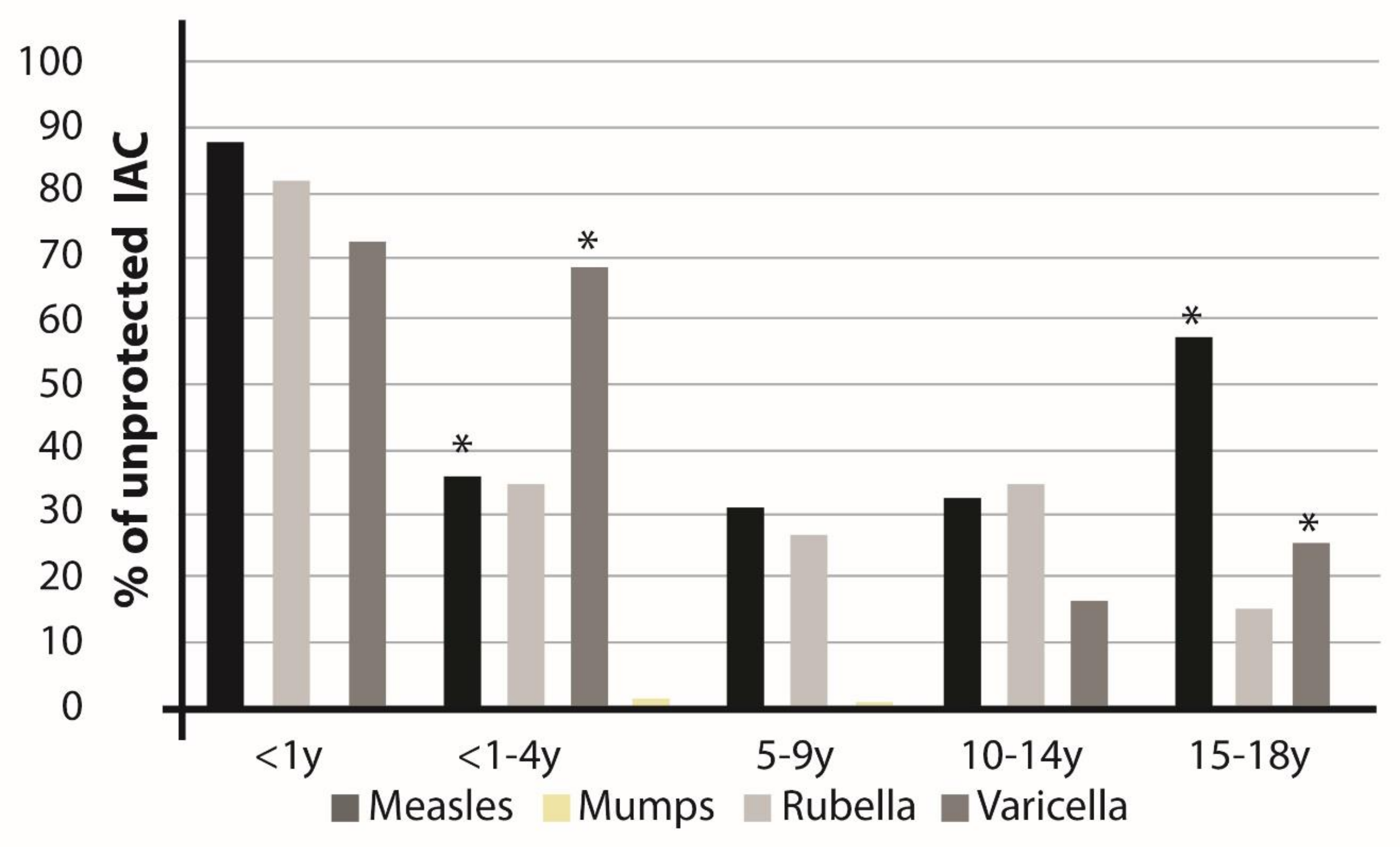
| Africa | Americas | Asia | Europe | Total | |
|---|---|---|---|---|---|
| Population (n) | 347 (18%) | 419 (21.7%) | 384 (19.9%) | 777 (40.3%) | 1927 (100%) |
| Median age (y) | 5.19 | 7.37 | 4.81 | 6.19 | 5.99 |
| IQR | 2.66–6.79 | 5.30–9.34 | 2.01–7.06 | 3.73–8.26 | 3.33–8.21 |
| Females (n) | 145 (41.8%) | 193 (46.1%) | 175 (45.6%) | 260 (33.5%) | 773 (40.1%) |
| Males (n) | 202 (58.2%) | 226 (53.9%) | 209 (54.4%) | 517 (66.5%) | 1154 (59.9%) |
| Age Group Year | Africa % (n = 347) | Americas % (n = 419) | Asia % (n = 384) | Europe % (n = 777) | Total % (n = 1927) |
|---|---|---|---|---|---|
| <1 | 6.3 | 0.2 | 6.3 | 0.5 | 2.6 |
| 1–4 | 47.6 | 21 | 48.2 | 40.8 | 39.2 |
| 5–9 | 38.6 | 62.8 | 41.1 | 46.3 | 47.5 |
| 10–14 | 5.5 | 14.6 | 3.6 | 11.1 | 9.3 |
| 15–18 | 2 | 1.4 | 0.8 | 1.3 | 1.3 |
| total | 18% | 21.7% | 19.9% | 40.3% | 100% |
| UNPROTECTED/Total Number of Children for Each Disease | Africa (n = 347) | Americas (n = 419) | Asia (n = 384) | Europe (n = 777) | |
|---|---|---|---|---|---|
| Measles n/N (%) | 657/1870 (35.1%) | 156/337(46.3%) | 147/409 (35.9%) | 159/377 (42.2%) | 195/747 (26.1%) |
| Rubella n/N (%) | 600/1868 (32.1%) | 139/337 (41.3%) | 89/408 (21.8%) | 136/373 (36.5%) | 236/750 (31.5%) |
| Varicella n/N (%) | 290/631 (46.0%) | 63/120 (52.5%) | 42/134 (31.3%) | 102/170 (60.0%) | 83/207 (40.1%) |
| Mumps n/N (%) | 10/25 (40.0%) | 2/15 (13.3%) | 0/193 (0.0%) | 5/122 (4.1%) | 3/514 (0.6%) |
| VPD | Immunization Status According to Documentation n (%) | Immunization Status According to Serological Test n (%) | Total | ||
|---|---|---|---|---|---|
| Protected | Unprotected | ||||
| Measles | Recorded | 717 (74.4) | 247 (25.6) | 964 | p < 0.001 |
| Not recorded | 496 (54.7) | 410 (45.3) | 906 | ||
| Rubella | Recorded | 674 (75.1) | 223 (24.9) | 897 | p < 0.001 |
| Not recorded | 594 (61.2) | 377 (38.8) | 971 | ||
| Varicella | Recorded | 35 (46.7) | 40 (53.3) | 75 | p = 0.172 |
| Not Recorded | 306 (55.0) | 250 (45.0) | 556 | ||
| Mumps | Recorded | 6 (75.0) | 2 (25.0) | 8 | p = 0.294 |
| Not Recorded | 9 (52.9) | 8 (47.1) | 17 | ||
| Continent | Concordance/Discordance | Measles | Rubella | Varicella | |||
|---|---|---|---|---|---|---|---|
| Africa | Concordance | 168 | 49.85% | 154 | 45.70% | 61 | 50.83% |
| Discordance | 169 | 50.15% | 183 | 54.30% | 59 | 49.17% | |
| Total | 337 | 337 | 120 | ||||
| Americas | Concordance | 214 | 52.32% | 212 | 51.96% | 40 | 29.85% |
| Discordance | 195 | 47.68% | 196 | 48.04% | 94 | 70.15% | |
| Total | 409 | 408 | 134 | ||||
| Asia | Concordance | 245 | 64.99% | 234 | 62.73% | 97 | 57.06% |
| Discordance | 132 | 35.01% | 139 | 37.27% | 73 | 42.94% | |
| Total | 377 | 373 | 170 | ||||
| Europe | Concordance | 500 | 66.93% | 451 | 60.13% | 87 | 42.03% |
| Discordance | 247 | 33.07% | 299 | 39.87% | 120 | 57.97% | |
| Total | 747 | 750 | 207 | ||||
| Age group | |||||||
| <1 yrs | Concordance | 43 | 87.76% | 41 | 80.39% | 8 | 72.73% |
| Discordance | 6 | 12.24% | 10 | 19.61% | 3 | 27.27% | |
| Total | 49 | 51 | 11 | ||||
| 1–4 yrs | Concordance | 455 | 62.24% | 473 | 64.97% | 143 | 67.14% |
| Discordance | 276 | 37.76% | 255 | 35.03% | 70 | 32.86% | |
| Total | 731 | 728 | 213 | ||||
| 5–9 yrs | Concordance | 514 | 58.08% | 444 | 50.17% | 111 | 36.27% |
| Discordance | 371 | 41.92% | 441 | 49.83% | 195 | 63.73% | |
| Total | 885 | 885 | 306 | ||||
| 10–14 yrs | Concordance | 102 | 56.98% | 88 | 49.44% | 17 | 21.79% |
| Discordance | 57 | 31.84% | 90 | 50.56% | 61 | 78.21% | |
| Total | 179 | 178 | 78 | ||||
| 15–18 yrs | Concordance | 13 | 50.00% | 5 | 19.23% | 6 | 26.09% |
| Discordance | 13 | 50.00% | 21 | 80.77% | 17 | 73.91% | |
| Total | 26 | 26 | 23 | ||||
| Age Group | 0 Dose | 1 Dose | 2 Doses | |
|---|---|---|---|---|
| Measles (n = 1870) | <1 y | 45 | 4 | 0 |
| 1–4 y | 368 | 318 | 45 | |
| 5–9 y | 446 | 246 | 193 | |
| 10–14 y | 93 | 33 | 53 | |
| 15–18 y | 20 | 3 | 3 | |
| Rubella (n = 1868) | <1 y | 50 | 1 | 0 |
| 1–4 y | 407 | 296 | 25 | |
| 5–9 y | 455 | 258 | 172 | |
| 10–14 y | 102 | 32 | 44 | |
| 15–18 y | 23 | 2 | 1 | |
| Varicella (n = 631) | <1 y | 11 | 0 | 0 |
| 1–4 y | 186 | 23 | 4 | |
| 5–9 y | 273 | 25 | 8 | |
| 10–14 y | 75 | 2 | 1 | |
| 15–18 y | 23 | 0 | 0 | |
| Mumps (n = 844) | <1 y | 3 | 0 | 0 |
| 1–4 y | 6 | 286 | 23 | |
| 5–9 y | 7 | 258 | 178 | |
| 10–14 y | 1 | 33 | 45 | |
| 15–18 y | 0 | 3 | 1 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bechini, A.; Boccalini, S.; Alimenti, C.M.; Bonanni, P.; Galli, L.; Chiappini, E. Immunization Status against Measles, Mumps, Rubella and Varicella in a Large Population of Internationally Adopted Children Referred to Meyer Children’s University Hospital from 2009 to 2018. Vaccines 2020, 8, 51. https://doi.org/10.3390/vaccines8010051
Bechini A, Boccalini S, Alimenti CM, Bonanni P, Galli L, Chiappini E. Immunization Status against Measles, Mumps, Rubella and Varicella in a Large Population of Internationally Adopted Children Referred to Meyer Children’s University Hospital from 2009 to 2018. Vaccines. 2020; 8(1):51. https://doi.org/10.3390/vaccines8010051
Chicago/Turabian StyleBechini, Angela, Sara Boccalini, Cecilia Maria Alimenti, Paolo Bonanni, Luisa Galli, and Elena Chiappini. 2020. "Immunization Status against Measles, Mumps, Rubella and Varicella in a Large Population of Internationally Adopted Children Referred to Meyer Children’s University Hospital from 2009 to 2018" Vaccines 8, no. 1: 51. https://doi.org/10.3390/vaccines8010051
APA StyleBechini, A., Boccalini, S., Alimenti, C. M., Bonanni, P., Galli, L., & Chiappini, E. (2020). Immunization Status against Measles, Mumps, Rubella and Varicella in a Large Population of Internationally Adopted Children Referred to Meyer Children’s University Hospital from 2009 to 2018. Vaccines, 8(1), 51. https://doi.org/10.3390/vaccines8010051






