Immunologic Dose-Response to Adenovirus-Vectored Vaccines in Animals and Humans: A Systematic Review of Dose-Response Studies of Replication Incompetent Adenoviral Vaccine Vectors when Given via an Intramuscular or Subcutaneous Route
Abstract
1. Introduction
- Assess the number of available papers, including adenoviral dose-response studies, and the distribution of host species and adenoviral serotypes within these papers.
- Assess which immunological responses dose-response data were available.
- Assess the dosing strategies used in adenoviral dose-ranging studies, including number and magnitude of dose levels.
2. Materials and Methods
2.1. Study Types, Study Design, Population, Intervention and Outcome Measures
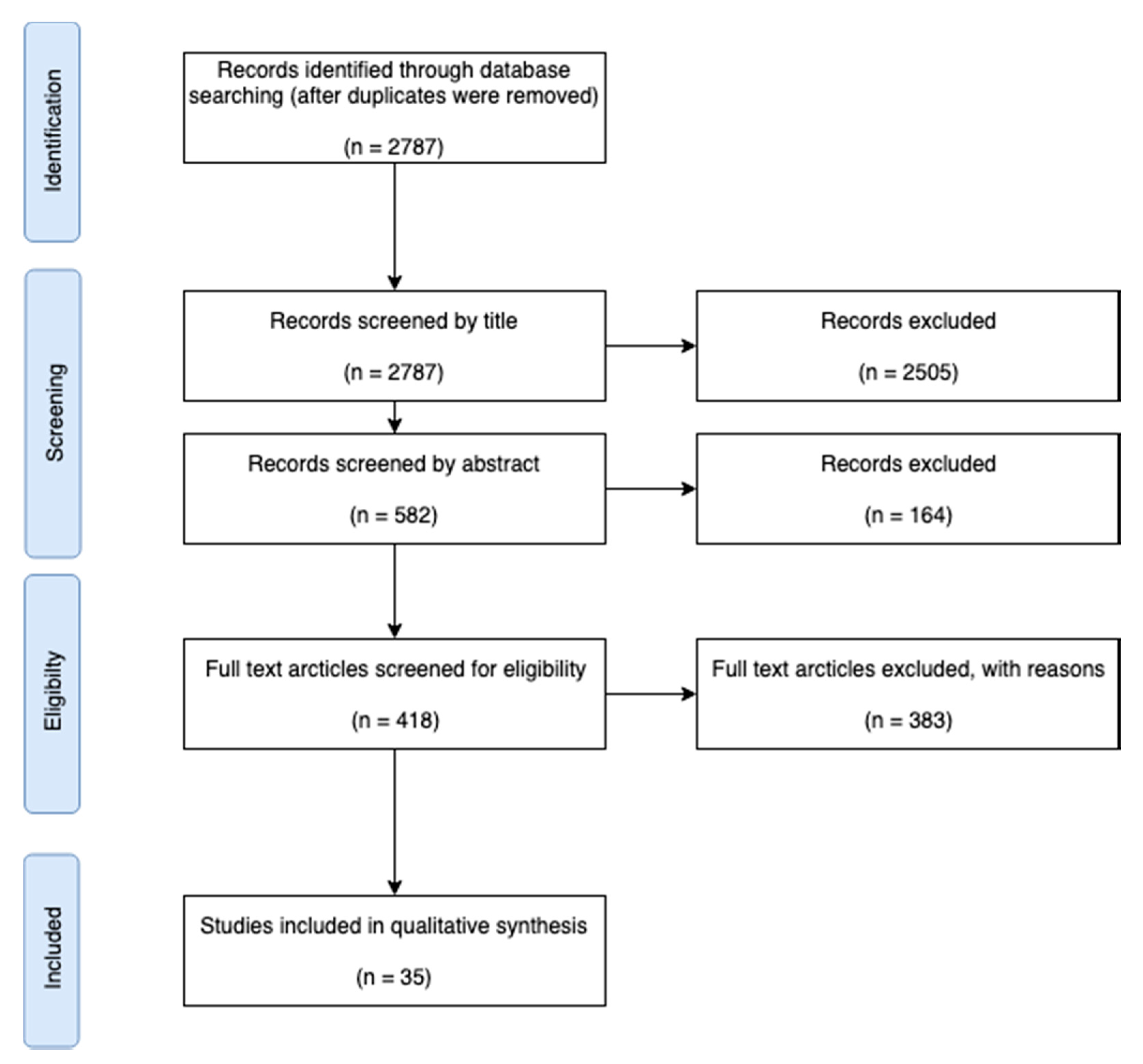
2.2. Search Strategy
2.3. Paper Selection (Inclusion/Exclusion Criteria)
2.4. Data Extraction
2.5. Assessment of Methodological Quality
2.6. Comparing Doses
3. Results
3.1. Objective 1: Assess the Number of Available Papers Including Adenoviral Dose-Response Studies, and the Distribution of Host Species and Adenoviral Serotypes within These Papers
3.2. Objective 2: Assess for Which Immunological Responses Dose-Response Data Were Available
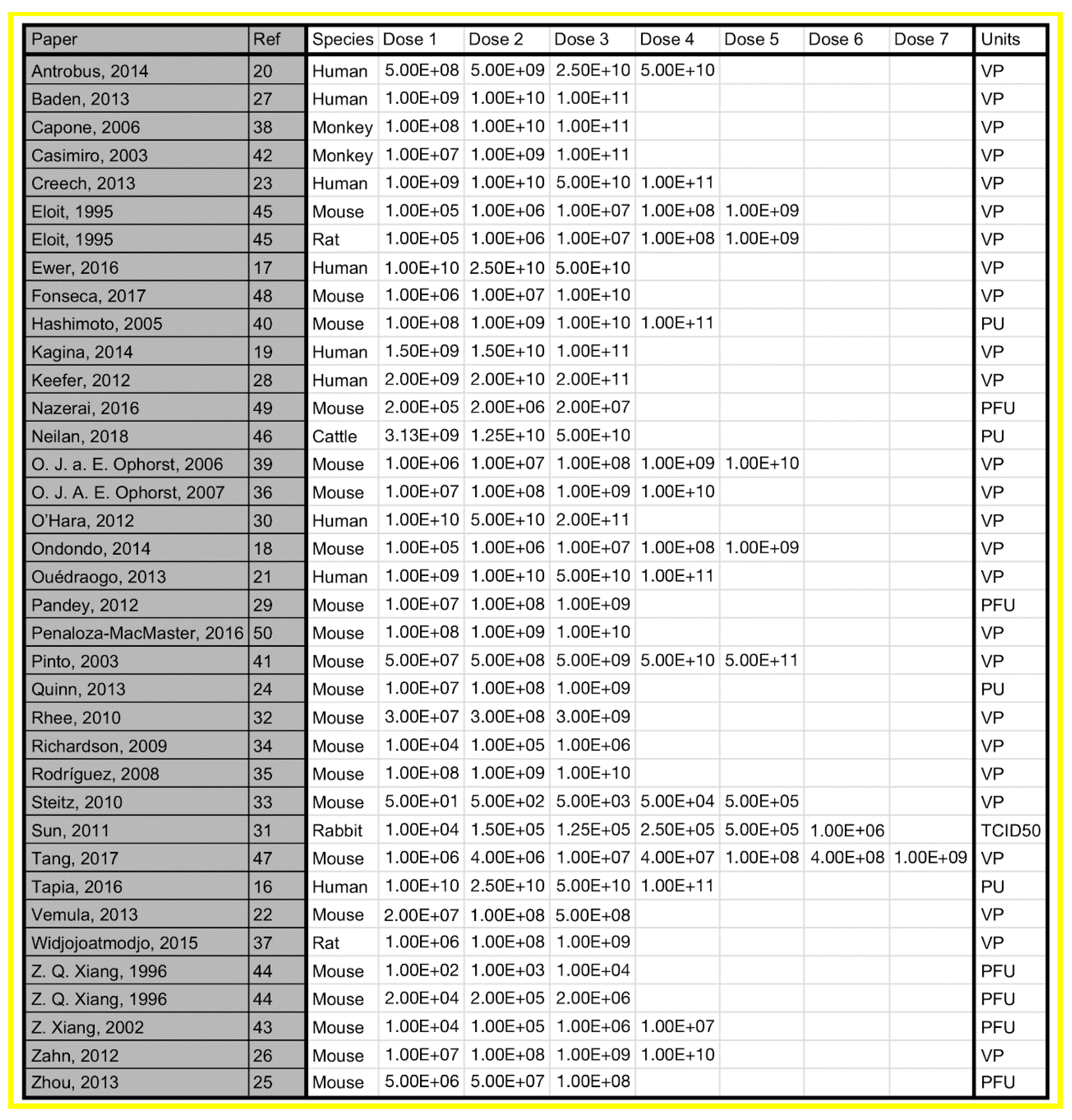
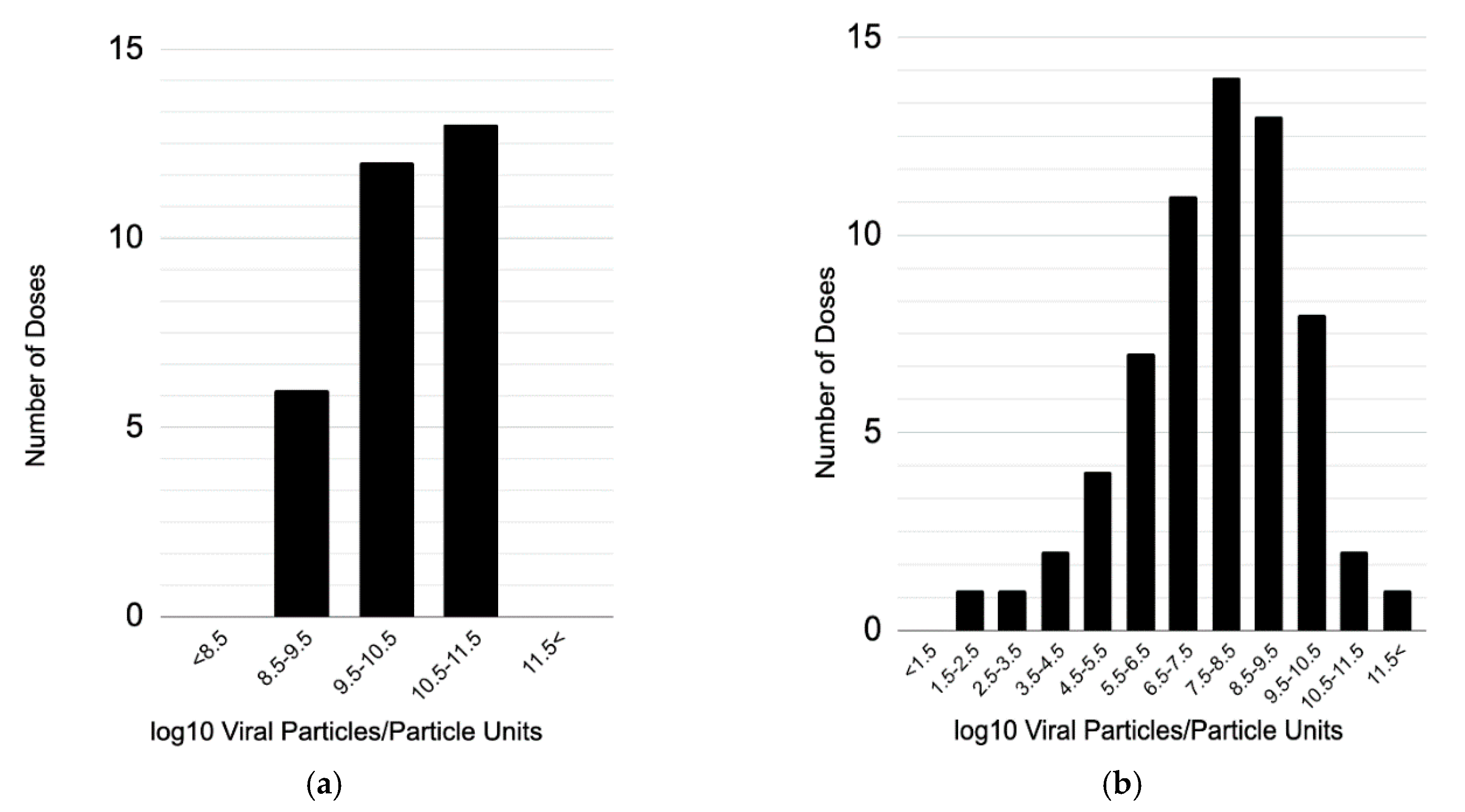
3.3. Objective 3: Assess the Dosing Strategies Used in Adenoviral Dose-Ranging Studies, Including Number and Magnitude of Dose Levels
3.3.1. Number of Dose Levels
3.3.2. Magnitude of Dose Levels
4. Discussion
Author Contributions
Funding
Conflicts of Interest
Appendix A
Criteria A1. Search Terms, PubMed
| Strategy | PUBMED Search | # |
| Concept 1 Adenovirus | adenovirus OR adenoviral OR adenovector OR adenovectors OR adenoviridae | 1 |
| Concept 2 Dose | dose OR doses OR dosage OR dosages OR dosing OR dosed OR dose response OR dose-response OR dose responses OR dose-responses OR dose response relationship OR dose-response relationship | 2 |
| Concept 3 Immune response | immunity OR immune OR immune-response OR immune response OR immune responses OR immune-responses OR immunostimulation OR immunodynamic OR immunodynamics OR immunisation OR immunisations OR immunization OR immunizations OR immunise OR immunises OR immunize OR immunizes OR immunised OR immunized OR immunising OR immunizing OR immunogenecity OR immunogenic OR immunology | 3 |
| Combine with AND | #1 AND #2 AND #3 | |
| Add filter: | Humans, Other Animals, English |
Criteria A2. Exclusion Criteria
References
- Davis, H.L. Novel vaccines and adjuvant systems: The utility of animal models for predicting immunogenicity in humans. Hum. Vaccines 2008, 4, 246–250. [Google Scholar] [CrossRef] [PubMed]
- Nair, A.B.; Jacob, S. A simple practice guide for dose conversion between animals and human. J. Basic Clin. Pharm. 2016, 7, 27–31. [Google Scholar] [CrossRef] [PubMed]
- Campi-Azevedo, A.C.; de Almeida Estevam, P.; Coelho-dos-Reis, J.G.; Peruhype-Magalhães, V.; Villela-Rezende, G.; Quaresma, P.F.; de Lourdes Sousa Maia, M.; Farias, R.H.G.; Camacho, L.A.B.; da Silva Freire, M.; et al. Subdoses of 17DD yellow fever vaccine elicit equivalent virological/immunological kinetics timeline. BMC Infect. Dis. 2014, 14, 391. [Google Scholar] [CrossRef] [PubMed]
- Guerin, P.J.; Naess, L.M.; Fogg, C.; Rosenqvist, E.; Pinoges, L.; Bajunirwe, F.; Nabasumba, C.; Borrow, R.; Frøholm, L.O.; Ghabri, S.; et al. Immunogenicity of fractional doses of tetravalent a/c/y/w135 meningococcal polysaccharide vaccine: Results from a randomized non-inferiority controlled trial in Uganda. PLoS ONE Negl. Trop. Dis. 2008, 2, e342. [Google Scholar] [CrossRef]
- Martins, R.M.; Maia, M.D.L.S.; Farias, R.H.G.; Camacho, L.A.B.; Freire, M.S.; Galler, R.; Yamamura, A.M.Y.; Almeida, L.F.C.; Lima, S.M.B.; Nogueira, R.M.R.; et al. 17DD yellow fever vaccine: A double blind, randomized clinical trial of immunogenicity and safety on a dose-response study. Hum. Vaccines Immunother. 2013, 9, 879–888. [Google Scholar] [CrossRef] [PubMed]
- Regules, J.A.; Cicatelli, S.B.; Bennett, J.W.; Paolino, K.M.; Twomey, P.S.; Moon, J.E.; Kathcart, A.K.; Hauns, K.D.; Komisar, J.L.; Qabar, A.N.; et al. Fractional Third and Fourth Dose of RTS,S/AS01 Malaria Candidate Vaccine: A Phase 2a Controlled Human Malaria Parasite Infection and Immunogenicity Study. J. Infect. Dis. 2016, 214, 762–771. [Google Scholar] [CrossRef]
- Rhodes, S.J.; Zelmer, A.; Knight, G.M.; Prabowo, S.A.; Stockdale, L.; Evans, T.G.; Lindenstrøm, T.; White, R.G.; Fletcher, H. The TB vaccine H56 + IC31 dose-response curve is peaked not saturating: Data generation for new mathematical modelling methods to inform vaccine dose decisions. Vaccine 2016, 34, 6285–6291. [Google Scholar] [CrossRef]
- Rhodes, S.J.; Knight, G.M.; Kirschner, D.E.; White, R.G.; Evans, T.G. Dose finding for new vaccines: The role for immunostimulation/immunodynamic modelling. arXiv 2018, arXiv:1811.04024. [Google Scholar] [CrossRef]
- Rhodes, S.J.; Guedj, J.; Fletcher, H.A.; Lindenstrøm, T.; Scriba, T.J.; Evans, T.G.; Knight, G.M.; White, R.G. Using vaccine Immunostimulation/Immunodynamic modelling methods to inform vaccine dose decision-making. Npj Vaccines 2018, 3, 36. [Google Scholar] [CrossRef]
- Rhodes, S.J.; Sarfas, C.; Knight, G.M.; White, A.; Pathan, A.A.; McShane, H.; Evans, T.G.; Fletcher, H.; Sharpe, S.; White, R.G. Using Data from Macaques to Predict Gamma Interferon Responses after Mycobacterium bovis BCG Vaccination in Humans: A Proof-of-Concept Study of Immunostimulation/Immunodynamic Modeling Methods. Clin. Vaccine Immunol. 2017, 24. [Google Scholar] [CrossRef]
- Rhodes, S.J.; Knight, G.M.; Kirschner, D.E.; White, R.G.; Evans, T.G. Dose finding for new vaccines: The role for immunostimulation/immunodynamic modelling. J. Theor. Biol. 2019, 465, 51–55. [Google Scholar] [CrossRef] [PubMed]
- Handel, A.; Li, Y.; McKay, B.; Pawelek, K.A.; Zarnitsyna, V.; Antia, R. Exploring the impact of inoculum dose on host immunity and morbidity to inform model-based vaccine design. PLoS ONE Comput. Biol. 2018, 14, e1006505. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Zhou, D. Adenoviral vector-based strategies against infectious disease and cancer. Hum. Vaccines Immunother. 2016, 12, 2064–2074. [Google Scholar] [CrossRef] [PubMed]
- Sheets, R.L.; Stein, J.; Bailer, R.T.; Koup, R.A.; Andrews, C.; Nason, M.; He, B.; Koo, E.; Trotter, H.; Duffy, C.; et al. Biodistribution and Toxicological Safety of Adenovirus Type 5 and Type 35 Vectored Vaccines Against Human Immunodeficiency Virus-1 (HIV-1), Ebola, or Marburg Are Similar Despite Differing Adenovirus Serotype Vector, Manufacturer’s Construct, or Gene Inserts. J. Immunotoxicol. 2008, 5, 315–335. [Google Scholar] [PubMed]
- Klasse, P.J. Molecular Determinants of the Ratio of Inert to Infectious Virus Particles. Prog. Mol. Biol. Transl. Sci. 2015, 129, 285–326. [Google Scholar] [PubMed]
- Tapia, M.D.; Sow, S.O.; Lyke, K.E.; Haidara, F.C.; Diallo, F.; Doumbia, M.; Traore, A.; Coulibaly, F.; Kodio, M.; Onwuchekwa, U.; et al. Use of ChAd3-EBO-Z Ebola virus vaccine in Malian and US adults, and boosting of Malian adults with MVA-BN-Filo: A phase 1, single-blind, randomised trial, a phase 1b, open-label and double-blind, dose-escalation trial, and a nested, randomised, double-blind, placebo-controlled trial. Lancet Infect. Dis. 2016, 16, 31–42. [Google Scholar]
- Ewer, K.; Rampling, T.; Venkatraman, N.; Bowyer, G.; Wright, D.; Lambe, T.; Imoukhuede, E.B.; Payne, R.; Fehling, S.K.; Strecker, T.; et al. A Monovalent Chimpanzee Adenovirus Ebola Vaccine Boosted with MVA. N. Engl. J. Med. 2016, 374, 1635–1646. [Google Scholar] [CrossRef]
- Ondondo, B.; Abdul-Jawad, S.; Bridgeman, A.; Hanke, T. Characterization of T-Cell Responses to Conserved Regions of the HIV-1 Proteome in BALB/c Mice. Clin. Vaccine Immunol. CVI 2014, 21, 1565–1572. [Google Scholar] [CrossRef]
- Kagina, B.M.N.; Tameris, M.D.; Geldenhuys, H.; Hatherill, M.; Abel, B.; Hussey, G.D.; Scriba, T.J.; Mahomed, H.; Sadoff, J.C.; Hanekom, W.A.; et al. The novel tuberculosis vaccine, AERAS-402, is safe in healthy infants previously vaccinated with BCG, and induces dose-dependent CD4 and CD8T cell responses. Vaccine 2014, 32, 5908–5917. [Google Scholar] [CrossRef]
- Antrobus, R.D.; Coughlan, L.; Berthoud, T.K.; Dicks, M.D.; Hill, A.V.; Lambe, T.; Gilbert, S.C. Clinical assessment of a novel recombinant simian adenovirus ChAdOx1 as a vectored vaccine expressing conserved Influenza A antigens. Mol. Ther. J. Am. Soc. Gene Ther. 2014, 22, 668–674. [Google Scholar] [CrossRef]
- Ouédraogo, A.; Tiono, A.B.; Kargougou, D.; Yaro, J.B.; Ouédraogo, E.; Kaboré, Y.; Kangoye, D.; Bougouma, E.C.; Gansane, A.; Henri, N.; et al. A phase 1b randomized, controlled, double-blinded dosage-escalation trial to evaluate the safety, reactogenicity and immunogenicity of an adenovirus type 35 based circumsporozoite malaria vaccine in Burkinabe healthy adults 18 to 45 years of age. PLoS ONE 2013, 8, e78679. [Google Scholar] [CrossRef] [PubMed]
- Vemula, S.V.; Amen, O.; Katz, J.M.; Donis, R.; Sambhara, S.; Mittal, S.K. Beta-defensin 2 enhances immunogenicity and protection of an adenovirus-based H5N1 influenza vaccine at an early time. Virus Res. 2013, 178, 398–403. [Google Scholar] [CrossRef] [PubMed]
- Creech, C.B.; Dekker, C.L.; Ho, D.; Phillips, S.; Mackey, S.; Murray-Krezan, C.; Grazia Pau, M.; Hendriks, J.; Brown, V.; Dally, L.G.; et al. Randomized, placebo-controlled trial to assess the safety and immunogenicity of an adenovirus type 35-based circumsporozoite malaria vaccine in healthy adults. Hum. Vaccines Immunother. 2013, 9, 2548–2557. [Google Scholar] [CrossRef] [PubMed]
- Quinn, K.M.; Da Costa, A.; Yamamoto, A.; Berry, D.; Lindsay, R.W.B.; Darrah, P.A.; Wang, L.; Cheng, C.; Kong, W.-P.; Gall, J.G.D.; et al. Comparative analysis of the magnitude, quality, phenotype, and protective capacity of simian immunodeficiency virus gag-specific CD8+ T cells following human-, simian-, and chimpanzee-derived recombinant adenoviral vector immunization. J. Immunol. Baltim. Md 1950 2013, 190, 2720–2735. [Google Scholar] [CrossRef] [PubMed]
- Zhou, G.; Wang, H.; Wang, F.; Yu, L. Recombinant adenovirus expressing type Asia1 foot-and-mouth disease virus capsid proteins induces protective immunity against homologous virus challenge in mice. Res. Vet. Sci. 2013, 94, 796–802. [Google Scholar] [CrossRef]
- Zahn, R.; Gillisen, G.; Roos, A.; Koning, M.; van der Helm, E.; Spek, D.; Weijtens, M.; Grazia Pau, M.; Radošević, K.; Weverling, G.J.; et al. Ad35 and ad26 vaccine vectors induce potent and cross-reactive antibody and T-cell responses to multiple filovirus species. PLoS ONE 2012, 7, e44115. [Google Scholar] [CrossRef]
- Baden, L.R.; Walsh, S.R.; Seaman, M.S.; Tucker, R.P.; Krause, K.H.; Patel, A.; Johnson, J.A.; Kleinjan, J.; Yanosick, K.E.; Perry, J.; et al. First-in-human evaluation of the safety and immunogenicity of a recombinant adenovirus serotype 26 HIV-1 Env vaccine (IPCAVD 001). J. Infect. Dis. 2013, 207, 240–247. [Google Scholar] [CrossRef]
- Keefer, M.C.; Gilmour, J.; Hayes, P.; Gill, D.; Kopycinski, J.; Cheeseman, H.; Cashin-Cox, M.; Naarding, M.; Clark, L.; Fernandez, N.; et al. A phase I double blind, placebo-controlled, randomized study of a multigenic HIV-1 adenovirus subtype 35 vector vaccine in healthy uninfected adults. PLoS ONE 2012, 7, e41936. [Google Scholar] [CrossRef]
- Pandey, A.; Singh, N.; Vemula, S.V.; Couëtil, L.; Katz, J.M.; Donis, R.; Sambhara, S.; Mittal, S.K. Impact of preexisting adenovirus vector immunity on immunogenicity and protection conferred with an adenovirus-based H5N1 influenza vaccine. PLoS ONE 2012, 7, e33428. [Google Scholar] [CrossRef]
- O’Hara, G.A.; Duncan, C.J.A.; Ewer, K.J.; Collins, K.A.; Elias, S.C.; Halstead, F.D.; Goodman, A.L.; Edwards, N.J.; Reyes-Sandoval, A.; Bird, P.; et al. Clinical assessment of a recombinant simian adenovirus ChAd63: A potent new vaccine vector. J. Infect. Dis. 2012, 205, 772–781. [Google Scholar] [CrossRef]
- Sun, Y.; Li, H.-Y.; Tian, D.-Y.; Han, Q.-Y.; Zhang, X.; Li, N.; Qiu, H.-J. A novel alphavirus replicon-vectored vaccine delivered by adenovirus induces sterile immunity against classical swine fever. Vaccine 2011, 29, 8364–8372. [Google Scholar] [CrossRef] [PubMed]
- Rhee, E.G.; Kelley, R.P.; Agarwal, I.; Lynch, D.M.; Porte, A.L.; Simmons, N.L.; Clark, S.L.; Barouch, D.H. TLR4 Ligands Augment Antigen-Specific CD8+ T Lymphocyte Responses Elicited by a Viral Vaccine Vector. J. Virol. 2010, 84, 10413–10419. [Google Scholar] [CrossRef] [PubMed]
- Steitz, J.; Barlow, P.G.; Hossain, J.; Kim, E.; Okada, K.; Kenniston, T.; Rea, S.; Donis, R.O.; Gambotto, A. A Candidate H1N1 Pandemic Influenza Vaccine Elicits Protective Immunity in Mice. PLoS ONE 2010, 5. [Google Scholar] [CrossRef] [PubMed]
- Richardson, J.S.; Yao, M.K.; Tran, K.N.; Croyle, M.A.; Strong, J.E.; Feldmann, H.; Kobinger, G.P. Enhanced protection against Ebola virus mediated by an improved adenovirus-based vaccine. PLoS ONE 2009, 4, e5308. [Google Scholar] [CrossRef]
- Rodrigues, E.G.; Zavala, F.; Eichinger, D.; Wilson, J.M.; Tsuji, M. Single immunizing dose of recombinant adenovirus efficiently induces CD8+ T cell-mediated protective immunity against malaria. J. Immunol. Baltim. Md 1950 1997, 158, 1268–1274. [Google Scholar]
- Ophorst, O.J.A.E.; Radosević, K.; Klap, J.M.; Sijtsma, J.; Gillissen, G.; Mintardjo, R.; van Ooij, M.J.M.; Holterman, L.; Companjen, A.; Goudsmit, J.; et al. Increased immunogenicity of recombinant Ad35-based malaria vaccine through formulation with aluminium phosphate adjuvant. Vaccine 2007, 25, 6501–6510. [Google Scholar] [CrossRef]
- Widjojoatmodjo, M.N.; Bogaert, L.; Meek, B.; Zahn, R.; Vellinga, J.; Custers, J.; Serroyen, J.; Radošević, K.; Schuitemaker, H. Recombinant low-seroprevalent adenoviral vectors Ad26 and Ad35 expressing the respiratory syncytial virus (RSV) fusion protein induce protective immunity against RSV infection in cotton rats. Vaccine 2015, 33, 5406–5414. [Google Scholar] [CrossRef]
- Capone, S.; Meola, A.; Ercole, B.B.; Vitelli, A.; Pezzanera, M.; Ruggeri, L.; Davies, M.E.; Tafi, R.; Santini, C.; Luzzago, A.; et al. A novel adenovirus type 6 (Ad6)-based hepatitis C virus vector that overcomes preexisting anti-ad5 immunity and induces potent and broad cellular immune responses in rhesus macaques. J. Virol. 2006, 80, 1688–1699. [Google Scholar] [CrossRef]
- Ophorst, O.J.A.E.; Radošević, K.; Havenga, M.J.E.; Pau, M.G.; Holterman, L.; Berkhout, B.; Goudsmit, J.; Tsuji, M. Immunogenicity and Protection of a Recombinant Human Adenovirus Serotype 35-Based Malaria Vaccine against Plasmodium yoelii in Mice. Infect. Immun. 2006, 74, 313–320. [Google Scholar] [CrossRef]
- Hashimoto, M.; Boyer, J.L.; Hackett, N.R.; Wilson, J.M.; Crystal, R.G. Induction of Protective Immunity to Anthrax Lethal Toxin with a Nonhuman Primate Adenovirus-Based Vaccine in the Presence of Preexisting Anti-Human Adenovirus Immunity. Infect. Immun. 2005, 73, 6885–6891. [Google Scholar] [CrossRef]
- Pinto, A.R.; Fitzgerald, J.C.; Giles-Davis, W.; Gao, G.P.; Wilson, J.M.; Ertl, H.C.J. Induction of CD8+ T cells to an HIV-1 antigen through a prime boost regimen with heterologous E1-deleted adenoviral vaccine carriers. J. Immunol. Baltim. Md 1950 2003, 171, 6774–6779. [Google Scholar] [CrossRef] [PubMed]
- Casimiro, D.R.; Chen, L.; Fu, T.-M.; Evans, R.K.; Caulfield, M.J.; Davies, M.-E.; Tang, A.; Chen, M.; Huang, L.; Harris, V.; et al. Comparative immunogenicity in rhesus monkeys of DNA plasmid, recombinant vaccinia virus, and replication-defective adenovirus vectors expressing a human immunodeficiency virus type 1 gag gene. J. Virol. 2003, 77, 6305–6313. [Google Scholar] [CrossRef] [PubMed]
- Xiang, Z.; Gao, G.; Reyes-Sandoval, A.; Cohen, C.J.; Li, Y.; Bergelson, J.M.; Wilson, J.M.; Ertl, H.C.J. Novel, Chimpanzee Serotype 68-Based Adenoviral Vaccine Carrier for Induction of Antibodies to a Transgene Product. J. Virol. 2002, 76, 2667–2675. [Google Scholar] [CrossRef] [PubMed]
- Xiang, Z.Q.; Yang, Y.; Wilson, J.M.; Ertl, H.C. A replication-defective human adenovirus recombinant serves as a highly efficacious vaccine carrier. Virology 1996, 219, 220–227. [Google Scholar] [CrossRef]
- Eloit, M.; Adam, M. Isogenic adenoviruses type 5 expressing or not expressing the E1A gene: Efficiency as virus vectors in the vaccination of permissive and non-permissive species. J. Gen. Virol. 1995, 76, 1583–1589. [Google Scholar] [CrossRef] [PubMed]
- Neilan, J.G.; Schutta, C.; Barrera, J.; Pisano, M.; Zsak, L.; Hartwig, E.; Rasmussen, M.V.; Kamicker, B.J.; Ettyreddy, D.; Brough, D.E.; et al. Efficacy of an adenovirus-vectored foot-and-mouth disease virus serotype A subunit vaccine in cattle using a direct contact transmission model. BMC Vet. Res. 2018, 14. [Google Scholar] [CrossRef]
- Tang, A.; Freed, D.C.; Li, F.; Meschino, S.; Prokop, M.; Bett, A.; Casimiro, D.; Wang, D.; Fu, T.-M. Functionally inactivated dominant viral antigens of human cytomegalovirus delivered in replication incompetent adenovirus type 6 vectors as vaccine candidates. Hum. Vaccines Immunother. 2017, 13, 2763–2771. [Google Scholar] [CrossRef]
- Fonseca, J.A.; McCaffery, J.N.; Kashentseva, E.; Singh, B.; Dmitriev, I.; Curiel, D.T.; Moreno, A. A prime-boost immunization regimen based on a Simian Adenovirus 36 vectored multi-stage malaria vaccine induces protective immunity in mice. Vaccine 2017, 35, 3239–3248. [Google Scholar] [CrossRef]
- Nazerai, L.; Bassi, M.R.; Uddback, I.E.M.; Holst, P.J.; Christensen, J.P.; Thomsen, A.R. Early life vaccination: Generation of adult-quality memory CD8+ T cells in infant mice using non-replicating adenoviral vectors. Sci. Rep. 2016, 6, 38666. [Google Scholar] [CrossRef]
- Penaloza-MacMaster, P.; Alayo, Q.A.; Ra, J.; Provine, N.M.; Larocca, R.; Lee, B.; Barouch, D.H. Inhibitory receptor expression on memory CD8 T cells following Ad vector immunization. Vaccine 2016, 34, 4955–4963. [Google Scholar] [CrossRef]
- Manzoli, L.; Flacco, M.E.; D’Addario, M.; Capasso, L.; Vito, C.D.; Marzuillo, C.; Villari, P.; Ioannidis, J.P.A. Non-publication and delayed publication of randomized trials on vaccines: Survey. BMJ 2014, 348. [Google Scholar] [CrossRef] [PubMed]
- Gallaher, S.D.; Berk, A.J. A rapid Q-PCR titration protocol for adenovirus and helper-dependent adenovirus vectors that produces biologically relevant results. J. Virol. Methods 2013, 192, 28–38. [Google Scholar] [CrossRef] [PubMed]
- Darrah, P.A.; Patel, D.T.; De Luca, P.M.; Lindsay, R.W.B.; Davey, D.F.; Flynn, B.J.; Hoff, S.T.; Andersen, P.; Reed, S.G.; Morris, S.L.; et al. Multifunctional T H 1 cells define a correlate of vaccine-mediated protection against Leishmania major. Nat. Med. 2007, 13, 843–850. [Google Scholar] [CrossRef] [PubMed]
- Billeskov, R.; Wang, Y.; Solaymani-Mohammadi, S.; Frey, B.; Kulkarni, S.; Andersen, P.; Agger, E.M.; Sui, Y.; Berzofsky, J.A. Low Antigen Dose in Adjuvant-Based Vaccination Selectively Induces CD4 T Cells with Enhanced Functional Avidity and Protective Efficacy. J. Immunol. Baltim. Md 1950 2017, 198, 3494–3506. [Google Scholar] [CrossRef]
| Number of Papers (%) | Host | Paper References |
|---|---|---|
| 21 (60%) | Mouse | [18,22,24,25,26,29,32,33,34,35,36,39,40,41,43,44,45,47,48,49,50] |
| 9 (26%) | Human | [16,17,19,20,21,23,27,28,30] |
| 2 (6%) | Monkey | [38,42] |
| 2 (6%) | Rat | [37,45] |
| 1 (3%) | Rabbit | [31] |
| 1 (3%) | Cattle | [46] |
| Human | Non-Human Primates |
|---|---|
| Ad5 (16, 46%) [22,24,25,29,31,33,34,38,39,42,43,44,45,46,49,50] | ChAd63 (3, 9%) [24,30,43] |
| Ad35 (9, 26%) [19,21,23,24,26,28,35,36,39] | ChAd3 (3, 9%) [16,17,24] |
| Ad26 (4, 11%) [26,27,32,37] | AdC6 (1, 3%) [41] |
| Ad6 (2, 6%) [38,47] | AdC7 (1, 3%) [40] |
| Ad28 (1, 3%) [24] | sAd11 (1, 3%) [24] |
| sAd16 (1, 3%) [24] | |
| sAdv-36 (1, 3%) [48] | |
| ChAdOx1 (1, 3%) [20] |
| Number of Papers (%) | Response Type | Paper References |
|---|---|---|
| 18 (51%) | Antibody | [16,17,20,21,22,23,25,26,27,28,31,33,36,39,40,42,45,48] |
| 12 (34%) | T cell count | [16,20,21,26,27,28,30,32,36,38,42,47] |
| 12 (34%) | CD8+ T cell count | [19,22,24,32,34,35,36,38,39,48,49,50] |
| 11 (31%) | Virus Neutralisation Titre | [22,25,27,29,30,34,36,37,43,44,46] |
| 4 (12%) | CD4+ T cell count | [19,32,35,38] |
| 3 (9%) | CD8+ T Cell, IFN-y+ Percentage | [19,21,41] |
| 3 (9%) | CD4+ T Cell, IFN-y+ Percentage | [19,21] |
| 2 (6%) | CD4+ T Cell, TNF-a+ Percentage | [19,21] |
| 2 (6%) | CD8+ T Cell, TNF-a+ Percentage | [21] |
| 2 (6%) | CD4+ T Cell, IL-2+ Percentage | [19,21] |
| 2 (6%) | CD8+ T Cell, Il-2+ Percentage | [21] |
| 1 (3%) | CD4+ T Cell, Il-17+ Percentage | [19] |
| Number of Papers (%) | Number of Dose Levels | Paper References |
|---|---|---|
| 21 (60%) | 3 | [17,19,22,24,25,27,28,29,30,32,34,35,37,38,42,44,46,48,49,40] |
| 8 (23%) | 4 | [16,20,21,23,26,36,40,43] |
| 5 (14%) | 5 | [18,33,39,41,45] |
| 1 (3%) | 6 | [31] |
| 1 (3%) | 7 | [47] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Afrough, S.; Rhodes, S.; Evans, T.; White, R.; Benest, J. Immunologic Dose-Response to Adenovirus-Vectored Vaccines in Animals and Humans: A Systematic Review of Dose-Response Studies of Replication Incompetent Adenoviral Vaccine Vectors when Given via an Intramuscular or Subcutaneous Route. Vaccines 2020, 8, 131. https://doi.org/10.3390/vaccines8010131
Afrough S, Rhodes S, Evans T, White R, Benest J. Immunologic Dose-Response to Adenovirus-Vectored Vaccines in Animals and Humans: A Systematic Review of Dose-Response Studies of Replication Incompetent Adenoviral Vaccine Vectors when Given via an Intramuscular or Subcutaneous Route. Vaccines. 2020; 8(1):131. https://doi.org/10.3390/vaccines8010131
Chicago/Turabian StyleAfrough, Sara, Sophie Rhodes, Thomas Evans, Richard White, and John Benest. 2020. "Immunologic Dose-Response to Adenovirus-Vectored Vaccines in Animals and Humans: A Systematic Review of Dose-Response Studies of Replication Incompetent Adenoviral Vaccine Vectors when Given via an Intramuscular or Subcutaneous Route" Vaccines 8, no. 1: 131. https://doi.org/10.3390/vaccines8010131
APA StyleAfrough, S., Rhodes, S., Evans, T., White, R., & Benest, J. (2020). Immunologic Dose-Response to Adenovirus-Vectored Vaccines in Animals and Humans: A Systematic Review of Dose-Response Studies of Replication Incompetent Adenoviral Vaccine Vectors when Given via an Intramuscular or Subcutaneous Route. Vaccines, 8(1), 131. https://doi.org/10.3390/vaccines8010131






