Feasibility Analysis of Interleukin-13 as a Target for a Therapeutic Vaccine
Abstract
:1. Introduction
2. Non-Clinical Data on IL-13 and Susceptibility to Disease
2.1. Schistosoma and Helminth Susceptibility
2.2. Leishmaniasis
2.3. Viral Infections
2.4. Other Infections
2.5. Septic Shock
2.6. Malignancies
3. Safety Signals from Clinical Trials and Post-Marketing Surveillance
4. Evidence for IL13 Related Phenotypes from Large-Scale Genetic Databases
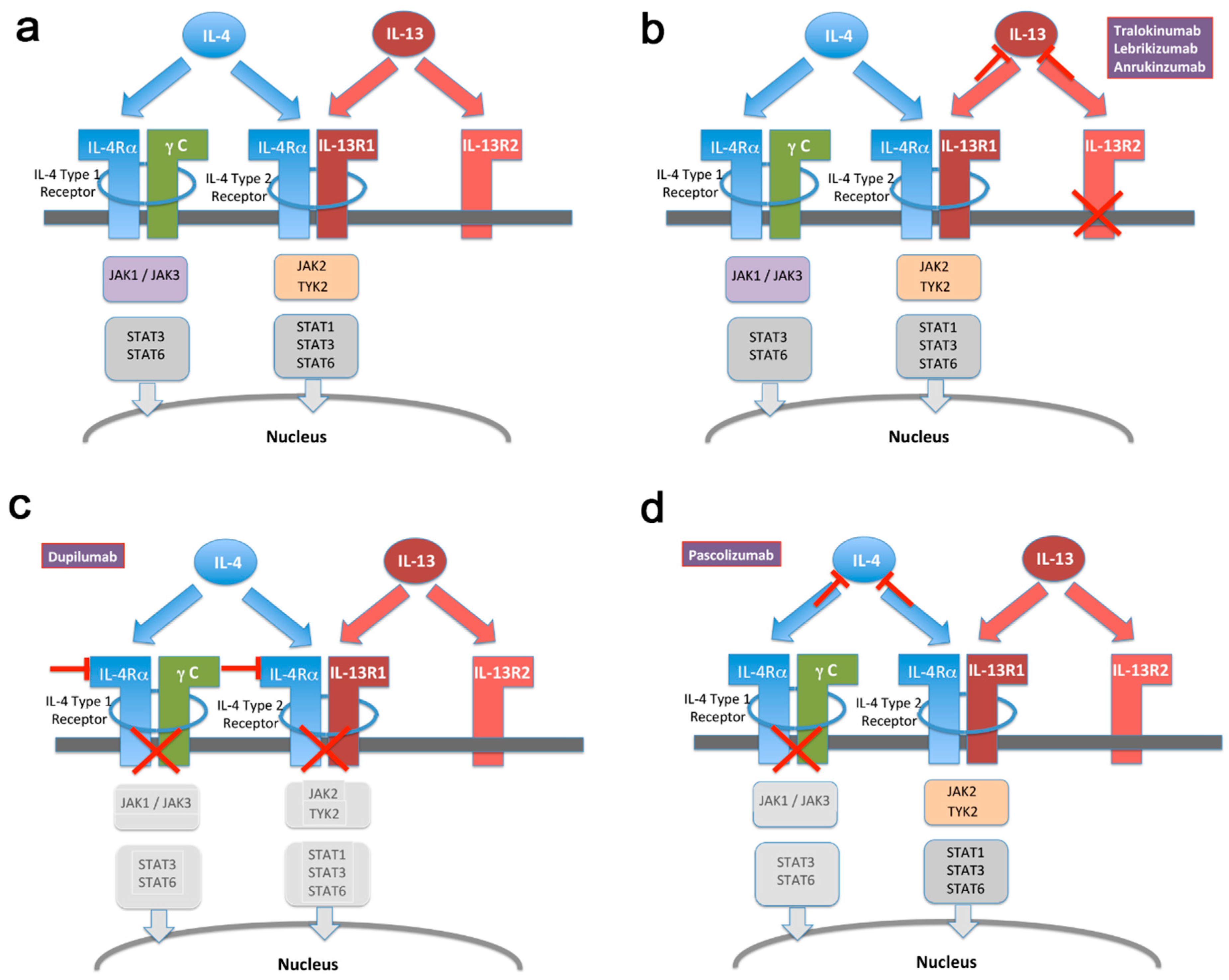
5. IL-13 on Its Own Represents a More Selective Target Than IL-4/IL-13 in Combination for Treatment of Atopic Conditions
6. Additional Potential Effects of an IL-13 Vaccine
6.1. Improvement of Fibrosis
6.2. Glucose Metabolism
6.3. Atherosclerosis
7. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Borman, Z.A.; Côté-Daigneault, J.; Colombel, J.-F. The risk for opportunistic infections in inflammatory bowel disease with biologics: An update. Expert Rev. Gastroenterol. Hepatol. 2018, 12, 1101–1108. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.F.; Mo, Y.Q.; Jing, J.; Ma, J.D.; Zheng, D.H.; Dai, L. Short-course tocilizumab increases risk of hepatitis B virus reactivation in patients with rheumatoid arthritis: a prospective clinical observation. Int. J. Rheum. Dis. 2017, 20, 859–869. [Google Scholar] [CrossRef] [PubMed]
- Kouriba, B.; Chevillard, C.; Bream, J.H.; Argiro, L.; Dessein, H.; Arnaud, V.; Sangare, L.; Dabo, A.; Beavogui, A.H.; Arama, C.; et al. Analysis of the 5q31-q33 locus shows an association between IL13-1055C/T IL-13-591A/G polymorphisms and Schistosoma haematobium infections. J. Immunol. 2005, 174, 6274–6281. [Google Scholar] [CrossRef] [PubMed]
- Grant, A.V.; Araujo, M.I.; Ponte, E.V.; Oliveira, R.R.; Gao, P.; Cruz, A.A.; Barnes, K.C.; Beaty, T.H. Functional polymorphisms in IL13 are protective against high Schistosoma mansoni infection intensity in a Brazilian population. PLoS ONE 2012, 7, e35863. [Google Scholar] [CrossRef] [PubMed]
- Isnard, A.; Kouriba, B.; Doumbo, O.; Chevillard, C. Association of rs7719175, located in the IL13 gene promoter, with Schistosoma haematobium infection levels and identification of a susceptibility haplotype. Genes Immun. 2011, 12, 31–39. [Google Scholar] [CrossRef] [PubMed]
- Ellis, M.K.; Zhao, Z.Z.; Chen, H.G.; Montgomery, G.W.; Li, Y.S.; McManus, D.P. Analysis of the 5q31 33 locus shows an association between single nucleotide polymorphism variants in the IL-5 gene and symptomatic infection with the human blood fluke, Schistosoma japonicum. J. Immunol. 2007, 179, 8366–8371. [Google Scholar] [CrossRef] [PubMed]
- Chiaramonte, M.G.; Schopf, L.R.; Neben, T.Y.; Cheever, A.W.; Donaldson, D.D.; Wynn, T.A. IL-13 Is a Key Regulatory Cytokine for Th2 Cell-Mediated Pulmonary Granuloma Formation and IgE Responses Induced by Schistosoma mansoni Eggs. J. Immunol. 1999, 162, 920–930. [Google Scholar]
- Fallon, P.G.; Richardson, E.J.; McKenzie, G.J.; McKenzie, A.N. Schistosome infection of transgenic mice defines distinct and contrasting pathogenic roles for IL-4 and IL-13: IL-13 is a profibrotic agent. J. Immunol. 2000, 164, 2585–2591. [Google Scholar] [CrossRef]
- Bao, K.; Reinhardt, R.L. The differential expression of IL-4 and IL-13 and its impact on type-2 immunity. Cytokine 2015, 75, 25–37. [Google Scholar] [CrossRef]
- Bancroft, A.J.; McKenzie, A.N.; Grencis, R.K. A critical role for IL-13 in resistance to intestinal nematode infection. J. Immunol. 1998, 160, 3453–3461. [Google Scholar]
- Sorobetea, D.; Svensson-Frej, M.; Grencis, R. Immunity to gastrointestinal nematode infections. Mucosal Immunol. 2018, 11, 304–315. [Google Scholar] [CrossRef] [PubMed]
- Hurdayal, R.; Brombacher, F. Interleukin-4 Receptor Alpha: From Innate to Adaptive Immunity in Murine Models of Cutaneous Leishmaniasis. Front. Immunol. 2017, 8, 1354. [Google Scholar] [CrossRef]
- Alexander, J.; Brombacher, F.; McGachy, H.A.; McKenzie, A.N.J.; Walker, W.; Carter, K.C. An essential role for IL-13 in maintaining a non-healing response following Leishmania mexicana infection. Eur. J. Immunol. 2002, 32, 2923–2933. [Google Scholar] [CrossRef]
- Doherty, T.M.; Kastelein, R.; Menon, S.; Andrade, S.; Coffman, R.L.; McKenzie, A.N.J. Modulation of murine macrophage function by IL-13. J. Immunol. 1993, 151, 7151–7160. [Google Scholar] [PubMed]
- Swihart, K.; Fruth, U.; Messmer, N.; Hug, K.; Behin, R.; Huang, S.; Del Giudice, G.; Aguet, M.; Louis, J.A. Mice from a genetically resistant background lacking the interferon gamma receptor are susceptible to infection with Leishmania major but mount a polarized T helper cell 1-type CD4+ T cell response. J. Exp. Med. 1995, 181, 961–971. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.E.; Reiner, S.L.; Zheng, S.; Dalton, D.K.; Locksley, R.M. CD4+ effector cells default to the Th2 pathway in interferon gamma-deficient mice infected with Leishmania major. J. Exp. Med. 1994, 179, 1367–1371. [Google Scholar] [CrossRef] [PubMed]
- Mattner, F.; Magram, J.; Ferrante, J.; Launois, P.; Di Padova, K.; Behin, R.; Gately, M.K.; Louis, J.A.; Alber, G. Genetically resistant mice lacking interleukin-12 are susceptible to infection withLeishmania major and mount a polarized Th2 cell response. Eur. J. Immunol. 1996, 26, 1553–1559. [Google Scholar] [CrossRef] [PubMed]
- Scharton-Kersten, T.; Afonso, L.C.; Wysocka, M.; Trinchieri, G.; Scott, P. IL-12 is required for natural killer cell activation and subsequent T helper 1 cell development in experimental leishmaniasis. J. Immunol. 1995, 154, 5320–5330. [Google Scholar]
- Donovan, C.; Bourke, J.E.; Vlahos, R. Targeting the IL-33/IL-13 Axis for Respiratory Viral Infections. Trends Pharmacol. Sci. 2016, 37, 252–261. [Google Scholar] [CrossRef]
- Busse, W.W.; Lemanske, R.F. Asthma. N. Engl. J. Med 2001, 344, 350–362. [Google Scholar] [CrossRef]
- Schwarze, J.; Hamelmann, E.; Bradley, K.L.; Takeda, K.; Gelfand, E.W. Respiratory syncytial virus infection results in airway hyperresponsiveness and enhanced airway sensitization to allergen. J. Clin. Investig. 1997, 100, 226–233. [Google Scholar] [CrossRef] [PubMed]
- Howell, M.D.; Gao, P.; Kim, B.E.; Lesley, L.J.; Streib, J.E.; Taylor, P.A.; Zaccaro, D.J.; Boguniewicz, M.; Beck, L.A.; Hanifin, J.M.; et al. The signal transducer and activator of transcription 6 gene (STAT6) increases the propensity of patients with atopic dermatitis toward disseminated viral skin infections. J. Allergy Clin. Immunol. 2011, 128, 1006–1014. [Google Scholar] [CrossRef] [PubMed]
- Contoli, M.; Ito, K.; Padovani, A.; Poletti, D.; Marku, B.; Edwards, M.R.; Stanciu, L.A.; Gnesini, G.; Pastore, A.; Spanevello, A.; et al. Th2 cytokines impair innate immune responses to rhinovirus in respiratory epithelial cells. Allergy 2015, 70, 910–920. [Google Scholar] [CrossRef] [PubMed]
- Edwards, M.R.; Bartlett, N.W.; Hussell, T.; Openshaw, P.; Johnston, S.L. The microbiology of asthma. Nat. Rev. Microbiol. 2012, 10, 459–471. [Google Scholar] [CrossRef] [PubMed]
- Letvin, A.N.; Wu, K.; Holtzman, M.J.; Keeler, S.P.; Agapov, E.V.; Hinojosa, M.E. Influenza A Virus Infection Causes Chronic Lung Disease Linked to Sites of Active Viral RNA Remnants. J. Immunol. 2018, 201. [Google Scholar] [CrossRef]
- Nguyen-Van-Tam, J.S.; Openshaw, P.J.M.; Hashim, A.; Gadd, E.M.; Lim, W.S.; Semple, M.G.; Read, R.C.; Taylor, B.L.; Brett, S.J.; McMenamin, J.; et al. Risk factors for hospitalisation and poor outcome with pandemic A/H1N1 influenza: United Kingdom first wave (May-September 2009). Thorax 2010, 65, 645–651. [Google Scholar] [CrossRef] [PubMed]
- Caballero, M.T.; Hijano, D.R.; Acosta, P.L.; Mateu, C.G.; Marcone, D.N.; Linder, J.E.; Talarico, L.B.; Elder, J.M.; Echavarria, M.; Miller, E.K.; et al. Interleukin-13 associates with life-threatening rhinovirus infections in infants and young children. Pediatr. Pulmonol. 2018, 53, 787–795. [Google Scholar] [CrossRef]
- Baraldo, S.; Contoli, M.; Bazzan, E.; Turato, G.; Padovani, A.; Marku, B.; Calabrese, F.; Caramori, G.; Ballarin, A.; Snijders, D.; et al. Deficient antiviral immune responses in childhood: Distinct roles of atopy and asthma. J. Allergy Clin. Immunol. 2012, 130, 1307–1314. [Google Scholar] [CrossRef]
- Lachowicz-Scroggins, M.E.; Boushey, H.A.; Finkbeiner, W.E.; Widdicombe, J.H. Interleukin-13-induced mucous metaplasia increases susceptibility of human airway epithelium to rhinovirus infection. Am. J. Respir. Cell Mol. Biol. 2010, 43, 652–661. [Google Scholar] [CrossRef]
- Dakhama, A.; Park, J.-W.; Taube, C.; Joetham, A.; Balhorn, A.; Miyahara, N.; Takeda, K.; Gelfand, E.W. The enhancement or prevention of airway hyperresponsiveness during reinfection with respiratory syncytial virus is critically dependent on the age at first infection and IL-13 production. J. Immunol. 2005, 175, 1876–1883. [Google Scholar] [CrossRef]
- Castilow, E.M.; Meyerholz, D.K.; Varga, S.M. IL-13 Is Required for Eosinophil Entry into the Lung during Respiratory Syncytial Virus Vaccine-Enhanced Disease. J. Immunol. 2008, 180, 2376–2384. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Wu, J.; Qi, F.; Zeng, S.; Xu, L.; Hu, H.; Wang, D.; Liu, B. Natural helper cells contribute to pulmonary eosinophilia by producing IL-13 via IL-33/ST2 pathway in a murine model of respiratory syncytial virus infection. Int. Immunopharmacol. 2015, 28, 337–343. [Google Scholar] [CrossRef]
- Halim, T.Y.F.; Krauß, R.H.; Sun, A.C.; Takei, F. Lung Natural Helper Cells Are a Critical Source of Th2 Cell-Type Cytokines in Protease Allergen-Induced Airway Inflammation. Immunity 2012, 36, 451–463. [Google Scholar] [CrossRef]
- Forton, J.T.; Rowlands, K.; Rockett, K.; Hanchard, N.; Herbert, M.; Kwiatkowski, D.P.; Hull, J. Genetic association study for RSV bronchiolitis in infancy at the 5q31 cytokine cluster. Thorax 2009, 64, 345–352. [Google Scholar] [CrossRef]
- Wang, M.; Wang, S.; Song, Z.; Ji, X.; Zhang, Z.; Zhou, J.; Ni, C. Associations of IL-4, IL-4R, and IL-13 gene polymorphisms in coal workers′ pneumoconiosis in China: A case-control study. PLoS ONE 2011, 6, e22624. [Google Scholar] [CrossRef] [PubMed]
- Asquith, K.L.; Horvat, J.C.; Kaiko, G.E.; Carey, A.J.; Beagley, K.W.; Hansbro, P.M.; Foster, P.S. Interleukin-13 promotes susceptibility to chlamydial infection of the respiratory and genital tracts. PLoS Pathog. 2011, 7, e1001339. [Google Scholar] [CrossRef] [PubMed]
- Starkey, M.R.; Essilfie, A.T.; Horvat, J.C.; Kim, R.Y.; Nguyen, D.H.; Beagley, K.W.; Mattes, J.; Foster, P.S.; Hansbro, P.M. Constitutive production of IL-13 promotes early-life Chlamydia respiratory infection and allergic airway disease. Mucosal Immunol. 2013, 6, 569–579. [Google Scholar] [CrossRef] [PubMed]
- Blanco-Quiros, A.; Casado-Flores, J.; Garrote Adrados, J.A.; Moro, M.N.; Antón, J.A.; Sanz, E.A. Interleukin-13 is involved in the survival of children with sepsis. Acta Paediatr. 2005, 94, 1828–1831. [Google Scholar] [CrossRef]
- Collighan, N.; Giannoudis, P.V.; Kourgeraki, O.; Perry, S.L.; Guillou, P.J.; Bellamy, M.C. Interleukin 13 and inflammatory markers in human sepsis. Br. J. Surg. 2004, 91, 762–768. [Google Scholar] [CrossRef]
- Skinnider, B.F.; Kapp, U.; Mak, T.W. Interleukin 13: A growth factor in hodgkin lymphoma. Int. Arch. Allergy Immunol. 2001, 126, 267–276. [Google Scholar] [CrossRef]
- Nakayama, S.; Yokote, T.; Hiraoka, N.; Nishiwaki, U.; Hanafusa, T.; Nishimura, Y.; Tsuji, M. Role of mast cells in fibrosis of classical Hodgkin lymphoma. Int. J. Immunopathol. Pharmacol. 2016, 29, 603–611. [Google Scholar] [CrossRef] [PubMed]
- Kaur, D.; Hollins, F.; Woodman, L.; Yang, W.; Monk, P.; May, R.; Bradding, P.; Brightling, C.E. Mast cells express IL-13Rα1: IL-13 promotes human lung mast cell proliferation and FcɛRI expression. Allergy 2006, 61, 1047–1053. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.; Hwang, R.F.; Logsdon, C.D.; Ullrich, S.E. Dynamic Mast Cell–Stromal Cell Interactions Promote Growth of Pancreatic Cancer. Cancer Res. 2013, 73, 3927–3937. [Google Scholar] [CrossRef] [PubMed]
- Thaci, B.; Brown, C.E.; Binello, E.; Werbaneth, K.; Sampath, P.; Sengupta, S. Significance of interleukin-13 receptor alpha 2-targeted glioblastoma therapy. Neuro. Oncol. 2014, 16, 1304–1312. [Google Scholar] [CrossRef] [PubMed]
- Rabe, K.F.; Nair, P.; Brusselle, G.; Maspero, J.F.; Castro, M.; Sher, L.; Zhu, H.; Hamilton, J.D.; Swanson, B.N.; Khan, A.; et al. Efficacy and Safety of Dupilumab in Glucocorticoid-Dependent Severe Asthma. N. Engl. J. Med. 2018, 378, 2475–2485. [Google Scholar] [CrossRef] [PubMed]
- Castro, M.; Corren, J.; Pavord, I.D.; Maspero, J.; Wenzel, S.; Rabe, K.F.; Busse, W.W.; Ford, L.; Sher, L.; FitzGerald, J.M.; et al. Dupilumab Efficacy and Safety in Moderate-to-Severe Uncontrolled Asthma. N. Engl. J. Med. 2018, 378, 2486–2496. [Google Scholar] [CrossRef] [PubMed]
- De Bruin-Weller, M.; Thaci, D.; Smith, C.H.; Reich, K.; Cork, M.J.; Radin, A.; Zhang, Q.; Akinlade, B.; Gadkari, A.; Eckert, L.; et al. Dupilumab with concomitant topical corticosteroid treatment in adults with atopic dermatitis with an inadequate response or intolerance to ciclosporin A or when this treatment is medically inadvisable: a placebo-controlled, randomized phase III clinical trial (LIBERTY AD CAFE). Br. J. Dermatol. 2018, 178, 1083–1101. [Google Scholar] [CrossRef]
- Simpson, E.L. Dupilumab Improves General Health-Related Quality-of-Life in Patients with Moderate-to-Severe Atopic Dermatitis: Pooled Results from Two Randomized, Controlled Phase 3 Clinical Trials. Dermatol. Ther. (Heidelb) 2017, 7, 243–248. [Google Scholar] [CrossRef]
- Home-ClinicalTrials.gov. Available online: https://clinicaltrials.gov/ (accessed on 8 February 2019).
- Wang, F.P.; Tang, X.J.; Wei, C.Q.; Xu, L.R.; Mao, H.; Luo, F.M. Dupilumab treatment in moderate-to-severe atopic dermatitis: A systematic review and meta-analysis. J. Dermatol. Sci. 2018, 90, 190–198. [Google Scholar] [CrossRef]
- Russell, R.J.; Chachi, L.; FitzGerald, J.M.; Russell, R.J.; Chachi, L.; FitzGerald, J.M.; Chaudhuri, R.; Leaker, B.; McGarvey, L.; Siddiqui, S.; et al. Effect of tralokinumab, an interleukin-13 neutralising monoclonal antibody, on eosinophilic airway inflammation in uncontrolled moderate-to-severe asthma (MESOS): a multicentre, double-blind, randomised, placebo-controlled phase 2 trial. Lancet Respir. Med. 2018, 6, 499–510. [Google Scholar] [CrossRef]
- Parker, J.M.; Glaspole, I.N.; Lancaster, L.H.; Haddad, T.J.; She, D.; Roseti, S.L.; Fiening, J.P.; Grant, E.P.; Kell, C.M.; Flaherty, K.R. A Phase 2 Randomized Controlled Study of Tralokinumab in Subjects with Idiopathic Pulmonary Fibrosis. Am. J. Respir. Crit. Care Med. 2018, 197, 94–103. [Google Scholar] [CrossRef] [PubMed]
- Brightling, C.E.; Chanez, P.; Leigh, R.; O′Byrne, P.M.; Korn, S.; She, D.; May, R.D.; Streicher, K.; Ranade, K.; Piper, E. Efficacy and safety of tralokinumab in patients with severe uncontrolled asthma: a randomised, double-blind, placebo-controlled, phase 2b trial. Lancet Respir. Med. 2015, 3, 692–701. [Google Scholar] [CrossRef]
- Piper, E.; Brightling, C.; Niven, R.; Oh, C.; Faggioni, R.; Poon, K.; She, D.; Kell, C.; May, R.D.; Geba, G.P.; et al. A phase II placebo-controlled study of tralokinumab in moderate-to-severe asthma. Eur. Respir. J. 2013, 41, 330–338. [Google Scholar] [CrossRef] [PubMed]
- Hanania, N.A.; Korenblat, P.; Chapman, K.R.; Bateman, E.D.; Kopecky, P.; Paggiaro, P.; Yokoyama, A.; Olsson, J.; Gray, S.; Holweg, C.T.; et al. Efficacy and safety of lebrikizumab in patients with uncontrolled asthma (LAVOLTA I and LAVOLTA II): Replicate, phase 3, randomised, double-blind, placebo-controlled trials. Lancet Respir. Med. 2016, 4, 781–796. [Google Scholar] [CrossRef]
- Korenblat, P.; Kerwin, E.; Leshchenko, I.; Yen, K.; Holweg, C.T.J.; Anzures-Cabrera, J.; Martin, C.; Putnam, W.S.; Governale, L.; Olsson, J.; et al. Efficacy and safety of lebrikizumab in adult patients with mild-to-moderate asthma not receiving inhaled corticosteroids. Respir. Med. 2018, 134, 143–149. [Google Scholar] [CrossRef]
- Simpson, E.L.; Flohr, C.; Eichenfield, L.F.; Bieber, T.; Sofen, H.; Taïeb, A.; Owen, R.; Putnam, W.; Castro, M.; DeBusk, K.; et al. Efficacy and safety of lebrikizumab (an anti-IL-13 monoclonal antibody) in adults with moderate-to-severe atopic dermatitis inadequately controlled by topical corticosteroids: A randomized, placebo-controlled phase II trial (TREBLE). J. Am. Acad. Dermatol. 2018, 78, 863–871. [Google Scholar] [CrossRef]
- Reinisch, W.; Panes, J.; Khurana, S.; Toth, G.; Hua, F.; Comer, G.M.; Hinz, M.; Page, K.; O′Toole, M.; Moorehead, T.M.; et al. Anrukinzumab, an anti-interleukin 13 monoclonal antibody, in active UC: Efficacy and safety from a phase IIa randomised multicentre study. Gut 2015, 64, 894–900. [Google Scholar] [CrossRef]
- Braddock, M.; Hanania, N.A.; Sharafkhaneh, A.; Colice, G.; Carlsson, M. Potential Risks Related to Modulating Interleukin-13 and Interleukin-4 Signalling: A Systematic Review. Drug Saf. 2018, 41, 489–509. [Google Scholar] [CrossRef]
- Wei, C.L.; Cheung, W.; Heng, C.K.; Arty, N.; Chong, S.S.; Lee, B.W.; Puah, K.L.; Yap, H.K. Interleukin-13 genetic polymorphisms in Singapore Chinese children correlate with long-term outcome of minimal-change disease. Nephrol. Dial. Transplant 2005, 20, 728–734. [Google Scholar] [CrossRef]
- Tenbrock, K.; Schubert, A.; Stapenhorst, L.; Kemper, M.J.; Gellermann, J.; Timmermann, K.; Müller-Wiefel, D.E.; Querfeld, U.; Hoppe, B.; Michalk, D. Type I IgE receptor, interleukin 4 receptor and interleukin 13 polymorphisms in children with nephrotic syndrome. Clin. Sci. (Lond) 2002, 102, 507–512. [Google Scholar] [CrossRef]
- Barik, S.; Miller, M.; Cattin-Roy, A.; Ukah, T.; Zaghouani, H. A distinct dendritic cell population arises in the thymus of IL-13Ralpha1-sufficient but not IL-13Ralpha1-deficient mice. Cell. Immunol. 2018, 331, 130–136. [Google Scholar] [CrossRef] [PubMed]
- ExAC Browser. Available online: http://exac.broadinstitute.org/ (accessed on 8 February 2019).
- NHLBI Grand Opportunity Exome Sequencing Project (ESP). Available online: https://esp.gs.washington.edu/drupal/ (accessed on 8 February 2019).
- Long, A.A. Monoclonal antibodies and other biologic agents in the treatment of asthma. MAbs 2009, 1, 237–246. [Google Scholar] [CrossRef]
- Vale, K. Targeting the JAK-STAT pathway in the treatment of ‘Th2-high’ severe asthma. Future Med. Chem. 2016, 8, 405–419. [Google Scholar] [CrossRef] [PubMed]
- Jiang, H.; Harris, M.B.; Rothman, P. IL-4/IL-13 signaling beyond JAK/STAT. J. Allergy Clin. Immunol. 2000, 105, 1063–1070. [Google Scholar] [CrossRef] [PubMed]
- Figueiredo, A.L.; Domingues, A.L.; Melo, W.G.; Tashiro, T.; de Lorena, V.M.; Montenegro, S.M.; Morais, C.N. Receptor Antagonist of IL-13 Exerts a Potential Negative Regulation During Early Infection of Human Schistosomiasis. Scand. J. Immunol. 2016, 84, 284–290. [Google Scholar] [CrossRef] [PubMed]
- Su, S.; Zhao, Q.; He, C.; Huang, D.; Liu, J.; Chen, F.; Chen, J.; Liao, J.Y.; Cui, X.; Zeng, Y.; et al. miR-142-5p and miR-130a-3p are regulated by IL-4 and IL-13 and control profibrogenic macrophage program. Nat. Commun. 2015, 6, 8523. [Google Scholar] [CrossRef] [PubMed]
- Hammerich, L.; Tacke, F. Interleukins in chronic liver disease: Lessons learned from experimental mouse models. Clin. Exp. Gastroenterol. 2014, 7, 297–306. [Google Scholar] [CrossRef] [PubMed]
- Gieseck, R.L., 3rd; Ramalingam, T.R.; Hart, K.M.; Vannella, K.M.; Cantu, D.A.; Lu, W.Y.; Ferreira-González, S.; Forbes, S.J.; Vallier, L.; Wynn, T.A. Interleukin-13 Activates Distinct Cellular Pathways Leading to Ductular Reaction, Steatosis, and Fibrosis. Immunity 2016, 45, 145–158. [Google Scholar] [CrossRef]
- Stanya, K.J.; Jacobi, D.; Liu, S.; Bhargava, P.; Dai, L.; Gangl, M.R.; Inouye, K.; Barlow, J.L.; Ji, Y.; Mizgerd, J.P.; et al. Direct control of hepatic glucose production by interleukin-13 in mice. J. Clin. Investig. 2013, 123, 261–271. [Google Scholar] [CrossRef]
- Martinez-Reyes, C.P.; Gomez-Arauz, A.Y.; Torres-Castro, I.; Manjarrez-Reyna, A.N.; Palomera, L.F.; Olivos-García, A.; Mendoza-Tenorio, E.; Sánchez-Medina, G.A.; Islas-Andrade, S.; Melendez-Mier, G.; et al. Serum Levels of Interleukin-13 Increase in Subjects with Insulin Resistance but Do Not Correlate with Markers of Low-Grade Systemic Inflammation. J. Diabetes Res. 2018, 2018. [Google Scholar] [CrossRef] [PubMed]
- Rachmin, I.; O′Meara, C.C.; Ricci-Blair, E.M.; Feng, Y.; Christensen, E.M.; Duffy, J.F.; Zitting, K.M.; Czeisler, C.A.; Pancoast, J.R.; Cannon, C.P.; et al. Soluble interleukin-13ralpha1: A circulating regulator of glucose. Am. J. Physiol. Endocrinol. Metab. 2017, 313, E663–E671. [Google Scholar] [CrossRef] [PubMed]
- Zha, L.-F.; Nie, S.-F.; Chen, Q.-W.; Liao, Y.-H.; Zhang, H.-S.; Dong, J.-T.; Xie, T.; Wang, F.; Tang, T.-T.; Xia, N.; et al. IL-13 may be involved in the development of CAD via different mechanisms under different conditions in a Chinese Han population. Sci. Rep. 2018, 8, 6182. [Google Scholar] [CrossRef] [PubMed]
- Jiang, L.Q.; Franck, N.; Egan, B.; Sjögren, R.J.O.; Katayama, M.; Duque-Guimaraes, D.; Arner, P.; Zierath, J.R.; Krook, A. Autocrine role of interleukin-13 on skeletal muscle glucose metabolism in type 2 diabetic patients involves microRNA let-7. Am. J. Physiol. Metab. 2013, 305, E1359–E1366. [Google Scholar] [CrossRef] [PubMed]
- Kretowski, A.; Mysliwiec, J.; Kinalska, I. In vitro interleukin-13 production by peripheral blood in patients with newly diagnosed insulin-dependent diabetes mellitus and their first degree relatives. Scand. J. Immunol. 2000, 51, 321–325. [Google Scholar] [CrossRef] [PubMed]
- Cardilo-Reis, L.; Gruber, S.; Schreier, S.M.; Drechsler, M.; Papac-Milicevic, N.; Weber, C.; Wagner, O.; Stangl, H.; Soehnlein, O.; Binder, C.J. Interleukin-13 protects from atherosclerosis and modulates plaque composition by skewing the macrophage phenotype. EMBO Mol. Med. 2012, 4, 1072–1086. [Google Scholar] [CrossRef] [PubMed]
- Yakubenko, V.P.; Hsi, L.C.; Cathcart, M.K.; Bhattacharjee, A. From macrophage interleukin-13 receptor to foam cell formation: Mechanisms for αMβ2 integrin interference. J. Biol. Chem. 2013, 288, 2778–2788. [Google Scholar] [CrossRef] [PubMed]
- Kleemann, R.; Zadelaar, S.; Kooistra, T. Cytokines and atherosclerosis: A comprehensive review of studies in mice. Cardiovasc. Res. 2008, 79, 360–376. [Google Scholar] [CrossRef]
- Wu, D.; Molofsky, A.B.; Liang, H.-E.; Ricardo-Gonzalez, R.R.; Jouihan, H.A.; Bando, J.K.; Chawla, A.; Locksley, R.M. Eosinophils sustain adipose alternatively activated macrophages associated with glucose homeostasis. Science 2011, 332. [Google Scholar] [CrossRef]
- Rahaman, S.O.; Lennon, D.J.; Febbraio, M.; Podrez, E.A.; Hazen, S.L.; Silverstein, R.L. A CD36-dependent signaling cascade is necessary for macrophage foam cell formation. Cell Metab. 2006, 4, 211–221. [Google Scholar] [CrossRef]
- Mallat, Z.; Ait-Oufella, H.; Tedgui, A. Regulatory T cell responses: Potential role in the control of atherosclerosis. Curr. Opin. Lipidol. 2005, 16, 518–524. [Google Scholar] [CrossRef]
- Foks, A.C.; Lichtman, A.H.; Kuiper, J. Treating Atherosclerosis With Regulatory T Cells. Arterioscler. Thromb. Vasc. Biol. 2015, 35, 280–287. [Google Scholar] [CrossRef] [PubMed]
| Gene | Type of Mutation | Expected (n) 2 | Observed (n) 2 | Metric (z/pLI) 3 |
|---|---|---|---|---|
| IL-13 | Synonymous | 25.9 | 26 | −0.01 |
| Missense | 48.4 | 50 | −0.11 | |
| LoF | 5.4 | 3 | 0.03 | |
| IL-13Rα1 | Synonymous | 33.5 | 25 | 0.91 |
| Missense | 79.2 | 80 | −0.04 | |
| LoF | 11.6 | 0 | 0.98 | |
| IL-13Rα2 | Synonymous | 33.3 | 30 | 0.35 |
| Missense | 77.8 | 72 | 0.32 | |
| LoF | 12.0 | 8 | 0.00 | |
| IL-4Rα | Synonymous | 138.2 | 146 | −0.41 |
| Missense | 283.0 | 296 | −0.38 | |
| LoF | 23.0 | 7 | 0.03 | |
| IL-4 | Synonymous | 29.7 | 29 | 0.08 |
| Missense | 57.3 | 49 | 0.54 | |
| LoF | 6.0 | 2 | 0.17 |
| Gene | MAF European 2 | MAF African 2 | Change | rs ID 3 |
|---|---|---|---|---|
| IL-13 | 0.000 | 0.023 | P60L | rs146770163 |
| 0.023 | 0.000 | W68R | rs143660447 | |
| 0.047 | 0.000 | L72P | rs148077750 | |
| 0.023 | 0.000 | V87G | rs199513811 | |
| 0.000 | 0.023 | T96N | rs374668631 | |
| 0.000 | 0.114 | V125M | rs140196099 | |
| IL-4 | 0.000 | 0.023 | R109W | rs373334025 |
| 0.023 | 0.000 | A118V | rs149950065 | |
| 0.000 | 0.023 | K150M | rs149147538 | |
| IL-4R | 0.012 | 0.000 | S6C | rs151179009 |
| 0.000 | 0.273 | V85M | rs141983833 | |
| 0.000 | 0.023 | P169L | rs145473866 | |
| 0.000 | 0.023 | R200W | rs370524692 | |
| IL-13Rα1 | 0.000 | 0.026 | S153R | rs368784033 |
| 0.000 | 0.052 | N157K | rs138018049 | |
| 0.119 | 0.000 | R340C | rs139927088 | |
| 0.000 | 0.261 | H398Y | rs145848479 | |
| 0.000 | 0.157 | D403N | rs149035999 | |
| 0.000 | 0.026 | D414N | rs142037578 | |
| IL-13Rα2 | 0.000 | 0.052 | R343C | rs375241228 |
| 0.000 | 0.026 | R248W | rs369836103 | |
| 0.000 | 0.026 | T107M | rs368005707 | |
| 0.000 | 0.026 | R74Q | rs377163094 | |
| 0.000 | 0.026 | P55L | rs370152207 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Foerster, J.; Molęda, A. Feasibility Analysis of Interleukin-13 as a Target for a Therapeutic Vaccine. Vaccines 2019, 7, 20. https://doi.org/10.3390/vaccines7010020
Foerster J, Molęda A. Feasibility Analysis of Interleukin-13 as a Target for a Therapeutic Vaccine. Vaccines. 2019; 7(1):20. https://doi.org/10.3390/vaccines7010020
Chicago/Turabian StyleFoerster, John, and Aleksandra Molęda. 2019. "Feasibility Analysis of Interleukin-13 as a Target for a Therapeutic Vaccine" Vaccines 7, no. 1: 20. https://doi.org/10.3390/vaccines7010020
APA StyleFoerster, J., & Molęda, A. (2019). Feasibility Analysis of Interleukin-13 as a Target for a Therapeutic Vaccine. Vaccines, 7(1), 20. https://doi.org/10.3390/vaccines7010020




