Vaccination with Combination DNA and Virus-Like Particles Enhances Humoral and Cellular Immune Responses upon Boost with Recombinant Modified Vaccinia Virus Ankara Expressing Human Immunodeficiency Virus Envelope Proteins
Abstract
1. Introduction
2. Materials and Methods
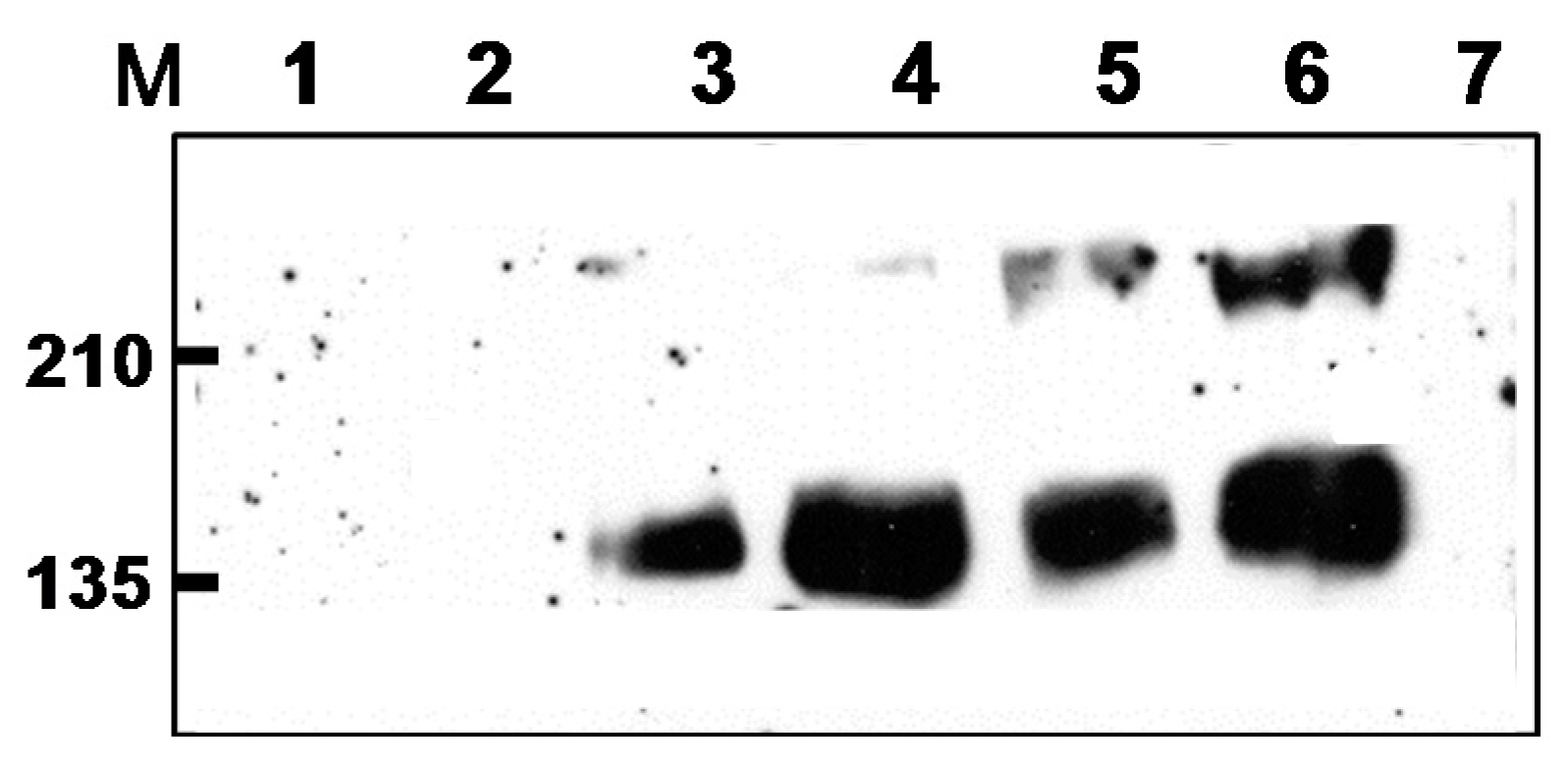
2.1. Preparation of DNA, rMVA Expressing tPAhFLex-gp120, and VLP Vaccines
2.2. Mice and Immunizations
2.3. Preparation of Peripheral Blood Mononuclear Cells (PBMCs) and Intracellular Cytokine Staining (ICC) Assay
2.4. DC Preparation and CD4+ T Cell Isolation
2.5. Lymphoproliferation and Cytokine Production In Vitro
2.6. ELISA (Enzyme-Linked Immunosorbent Assay)
2.7. ELISPOT (Enzyme-Linked ImmunoSpot)
2.8. Statistical Analysis
3. Results
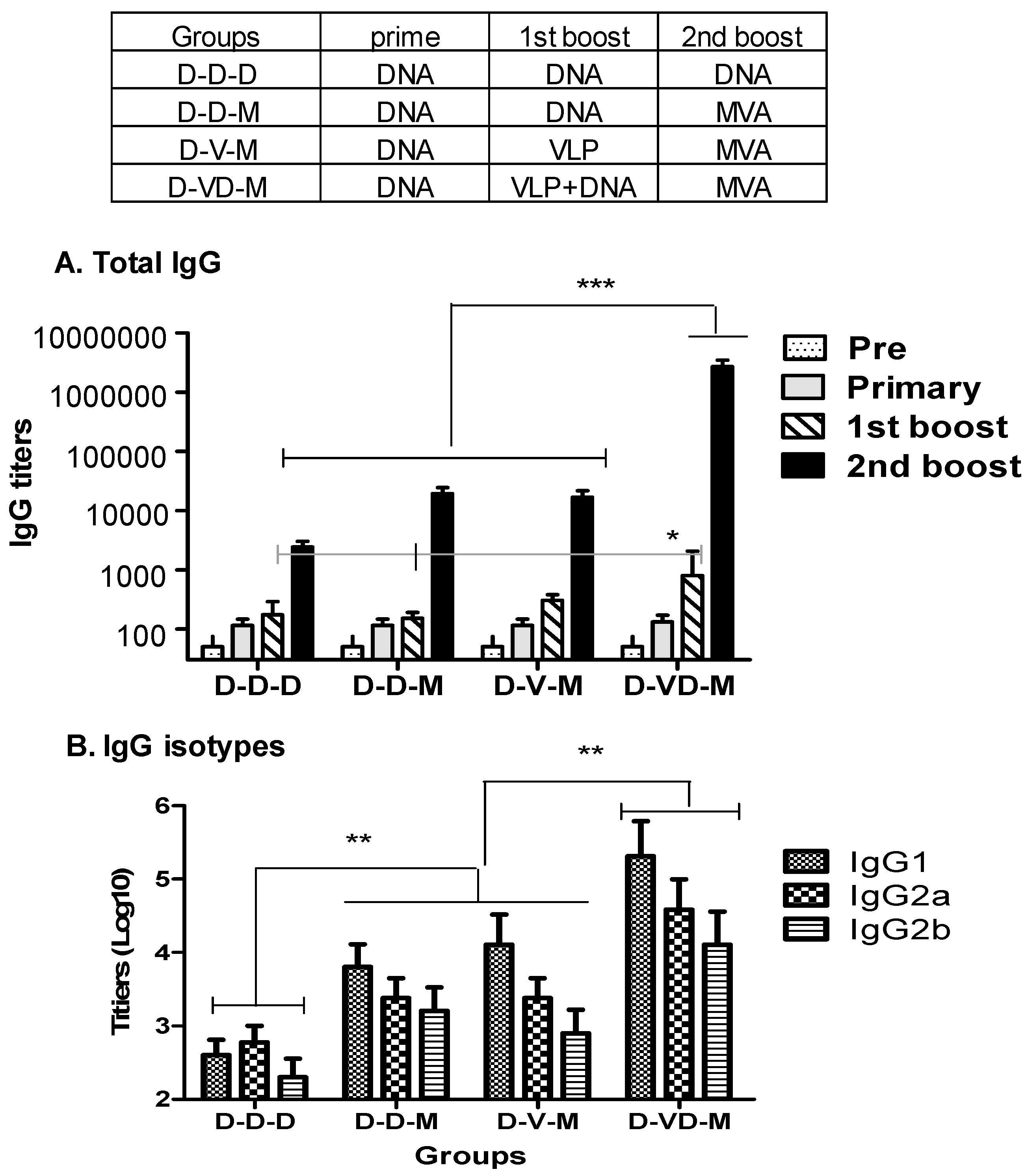
3.1. Boost with Combination VLP+DNA Vaccines Prior to rMVA Enhances HIV Env gp120-Specific Antibody Responses
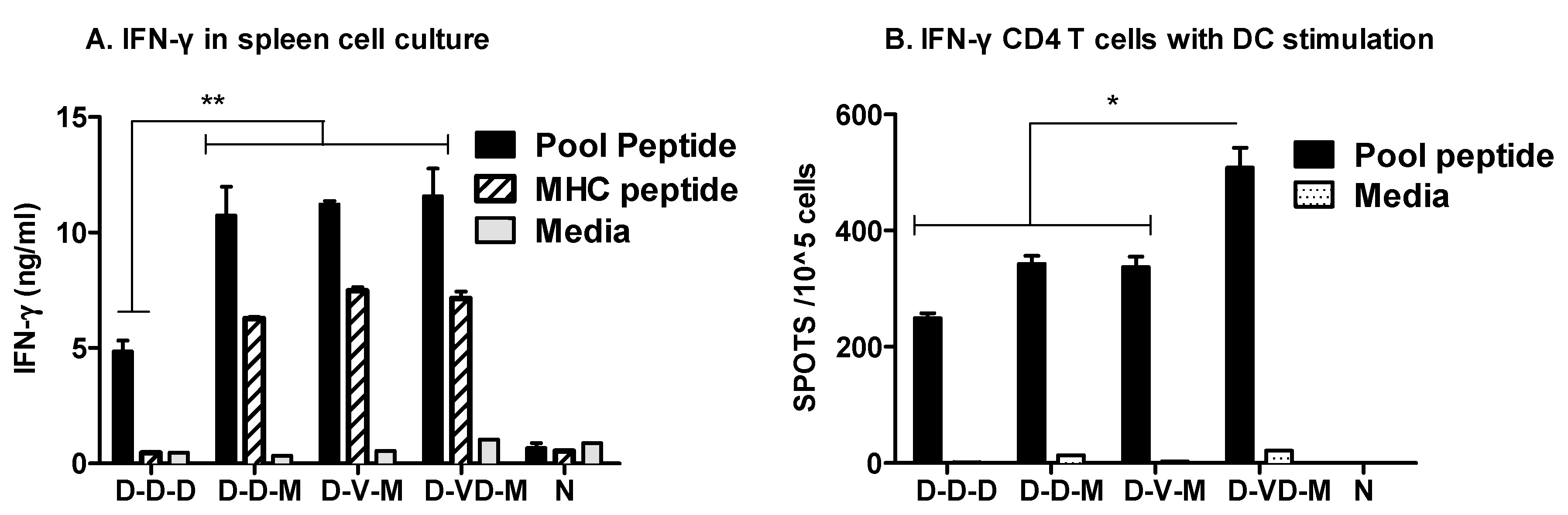
3.2. Boosting with Combination VLP + DNA Vaccines Prior to rMVA Enhances IFN-γ CD8 T Cells in Spleens
3.3. Boost with a Combination of VLP and DNA Vaccine Prior to rMVA Boost Effectively Induces IFN-γ CD4+ T Cells
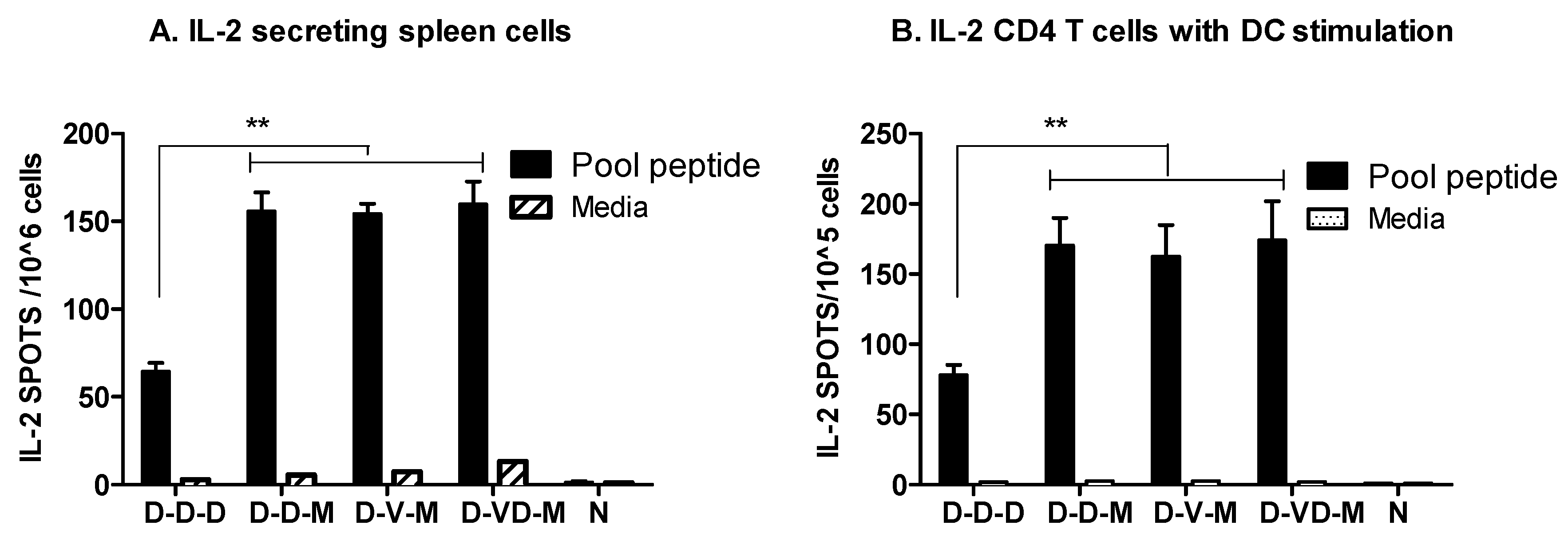
3.4. Combination of VLP and DNA Vaccination Effectively Induces Splenocyte Proliferation Responses
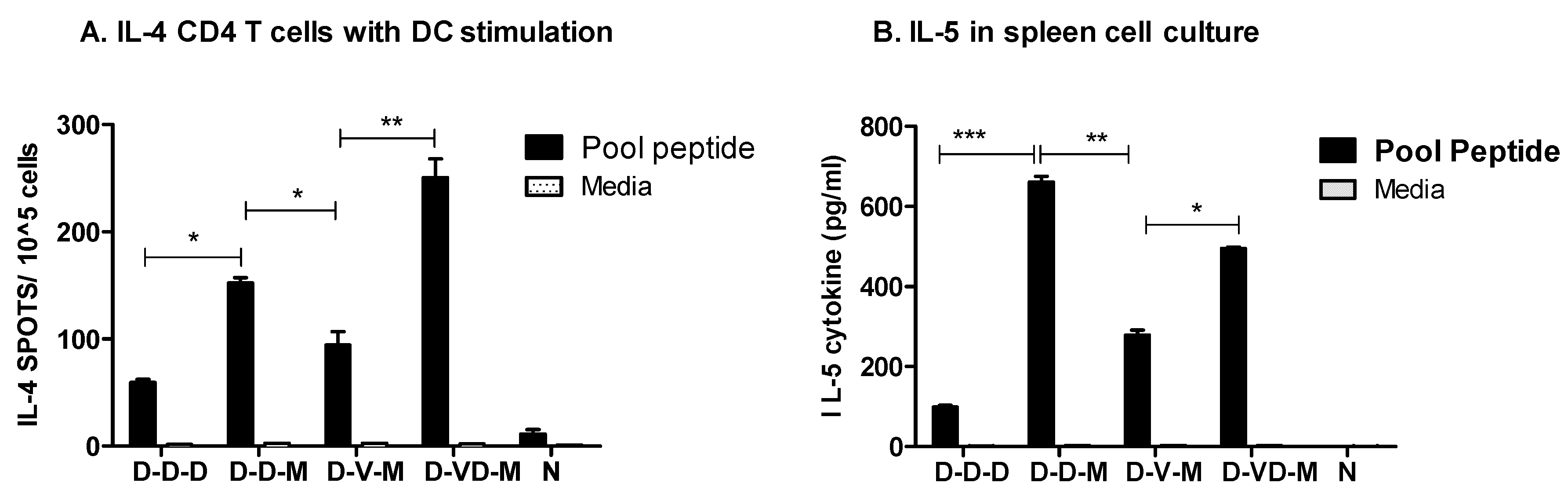
3.5. Boost with Combination VLP + DNA Vaccines Prior to rMVA Is Effective in Induceing IL-4 CD4+ T Cells
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Desrosiers, R.C. Prospects for an AIDS vaccine. Nat. Med. 2004, 10, 221–223. [Google Scholar] [CrossRef] [PubMed]
- Baba, T.W.; Liska, V.; Khimani, A.H.; Ray, N.B.; Dailey, P.J.; Penninck, D.; Bronson, R.; Greene, M.F.; McClure, H.M.; Martin, L.N.; et al. Live attenuated, multiply deleted simian immunodeficiency virus causes AIDS in infant and adult macaques. Nat. Med. 1999, 5, 194–203. [Google Scholar] [CrossRef] [PubMed]
- Sekaly, R.P. The failed HIV Merck vaccine study: A step back or a launching point for future vaccine development? J. Exp. Med. 2008, 205, 7–12. [Google Scholar] [CrossRef] [PubMed]
- Valentine, L.E.; Watkins, D.I. Relevance of studying T cell responses in SIV-infected rhesus macaques. Trends Microbiol. 2008, 16, 605–611. [Google Scholar] [CrossRef] [PubMed]
- Watkins, D.I.; Burton, D.R.; Kallas, E.G.; Moore, J.P.; Koff, W.C. Nonhuman primate models and the failure of the Merck HIV-1 vaccine in humans. Nat. Med. 2008, 14, 617–621. [Google Scholar] [CrossRef] [PubMed]
- Rerks-Ngarm, S.; Pitisuttithum, P.; Nitayaphan, S.; Kaewkungwal, J.; Chiu, J.; Paris, R.; Premsri, N.; Namwat, C.; de Souza, M.; Adams, E.; et al. Vaccination with ALVAC and AIDSVAX to prevent HIV-1 infection in Thailand. N. Engl. J. Med. 2009, 361, 2209–2220. [Google Scholar] [CrossRef] [PubMed]
- Blumberg, R.S.; Paradis, T.; Hartshorn, K.L.; Vogt, M.; Ho, D.D.; Hirsch, M.S.; Leban, J.; Sato, V.L.; Schooley, R.T. Antibody-dependent cell-mediated cytotoxicity against cells infected with the human immunodeficiency virus. J. Infect. Dis. 1987, 156, 878–884. [Google Scholar] [CrossRef] [PubMed]
- Evans, L.A.; Thomson-Honnebier, G.; Steimer, K.; Paoletti, E.; Perkus, M.E.; Hollander, H.; Levy, J.A. Antibody-dependent cellular cytotoxicity is directed against both the gp120 and gp41 envelope proteins of HIV. AIDS 1989, 3, 273–276. [Google Scholar] [CrossRef] [PubMed]
- Blue, C.E.; Spiller, O.B.; Blackbourn, D.J. The relevance of complement to virus biology. Virology 2004, 319, 176–184. [Google Scholar] [CrossRef] [PubMed]
- Jayasekera, J.P.; Moseman, E.A.; Carroll, M.C. Natural antibody and complement mediate neutralization of influenza virus in the absence of prior immunity. J. Virol. 2007, 81, 3487–3494. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Zhang, C.; Jia, L.; Wen, C.; Liu, H.; Wang, Y.; Sun, Y.; Huang, L.; Zhou, Y.; Song, H. A novel approach to inhibit HIV-1 infection and enhance lysis of HIV by a targeted activator of complement. Virol. J. 2009, 6. [Google Scholar] [CrossRef] [PubMed]
- Iyer, S.S.; Amara, R.R. DNA/MVA Vaccines for HIV/AIDS. Vaccines (Basel) 2014, 2, 160–178. [Google Scholar] [CrossRef] [PubMed]
- Hanke, T.; McMichael, A.J.; Mwau, M.; Wee, E.G.; Ceberej, I.; Patel, S.; Sutton, J.; Tomlinson, M.; Samuel, R.V. Development of a DNA-MVA/HIVA vaccine for Kenya. Vaccine 2002, 20, 1995–1998. [Google Scholar] [CrossRef]
- Goepfert, P.A.; Elizaga, M.L.; Sato, A.; Qin, L.; Cardinali, M.; Hay, C.M.; Hural, J.; DeRosa, S.C.; DeFawe, O.D.; Tomaras, G.D.; et al. Phase 1 safety and immunogenicity testing of DNA and recombinant modified vaccinia Ankara vaccines expressing HIV-1 virus-like particles. J. Infect. Dis. 2011, 203, 610–619. [Google Scholar] [CrossRef] [PubMed]
- Goulder, P.J.; Rowland-Jones, S.L.; McMichael, A.J.; Walker, B.D. Anti-HIV cellular immunity: Recent advances towards vaccine design. AIDS 1999, 13, S121–S136. [Google Scholar] [PubMed]
- Schmitz, J.E.; Kuroda, M.J.; Santra, S.; Sasseville, V.G.; Simon, M.A.; Lifton, M.A.; Racz, P.; Tenner-Racz, K.; Dalesandro, M.; Scallon, B.J.; et al. Control of viremia in simian immunodeficiency virus infection by CD8+ lymphocytes. Science 1999, 283, 857–860. [Google Scholar] [CrossRef] [PubMed]
- Levy, J.A.; Mackewicz, C.E.; Barker, E. Controlling HIV pathogenesis: The role of the noncytotoxic anti-HIV response of CD8+ T cells. Immunol. Today 1996, 17, 217–224. [Google Scholar] [CrossRef]
- Lai, L.; Vodros, D.; Kozlowski, P.A.; Montefiori, D.C.; Wilson, R.L.; Akerstrom, V.L.; Chennareddi, L.; Yu, T.; Kannanganat, S.; Ofielu, L.; et al. GM-CSF DNA: An adjuvant for higher avidity IgG, rectal IgA, and increased protection against the acute phase of a SHIV-89.6P challenge by a DNA/MVA immunodeficiency virus vaccine. Virology 2007, 369, 153–167. [Google Scholar] [CrossRef] [PubMed]
- Robinson, H.L.; Montefiori, D.C.; Villinger, F.; Robinson, J.E.; Sharma, S.; Wyatt, L.S.; Earl, P.L.; McClure, H.M.; Moss, B.; Amara, R.R. Studies on GM-CSF DNA as an adjuvant for neutralizing Ab elicited by a DNA/MVA immunodeficiency virus vaccine. Virology 2006, 352, 285–294. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Lai, L.; Amara, R.R.; Montefiori, D.C.; Villinger, F.; Chennareddi, L.; Wyatt, L.S.; Moss, B.; Robinson, H.L. Preclinical studies of human immunodeficiency virus/AIDS vaccines: Inverse correlation between avidity of anti-Env antibodies and peak postchallenge viremia. J. Virol. 2009, 83, 4102–4111. [Google Scholar] [CrossRef] [PubMed]
- Sailaja, G.; Husain, S.; Nayak, B.P.; Jabbar, A.M. Long-term maintenance of gp120-specific immune responses by genetic vaccination with the HIV-1 envelope genes linked to the gene encoding Flt-3 ligand. J. Immunol. 2003, 170, 2496–2507. [Google Scholar] [CrossRef] [PubMed]
- Kwa, S.; Sadagopal, S.; Shen, X.; Hong, J.J.; Gangadhara, S.; Basu, R.; Victor, B.; Iyer, S.S.; LaBranche, C.C.; Montefiori, D.C.; et al. CD40L-adjuvanted DNA/modified vaccinia virus Ankara simian immunodeficiency virus (SIV) vaccine enhances protection against neutralization-resistant mucosal SIV infection. J. Virol. 2015, 89, 4690–4695. [Google Scholar] [CrossRef] [PubMed]
- Kannanganat, S.; Wyatt, L.S.; Gangadhara, S.; Chamcha, V.; Chea, L.S.; Kozlowski, P.A.; LaBranche, C.C.; Chennareddi, L.; Lawson, B.; Reddy, P.B.; et al. High Doses of GM-CSF Inhibit Antibody Responses in Rectal Secretions and Diminish Modified Vaccinia Ankara/Simian Immunodeficiency Virus Vaccine Protection in TRIM5alpha-Restrictive Macaques. J. Immunol. 2016, 197, 3586–3596. [Google Scholar] [CrossRef] [PubMed]
- Iyer, S.S.; Gangadhara, S.; Victor, B.; Shen, X.; Chen, X.; Nabi, R.; Kasturi, S.P.; Sabula, M.J.; Labranche, C.C.; Reddy, P.B.; et al. Virus-Like Particles Displaying Trimeric Simian Immunodeficiency Virus (SIV) Envelope gp160 Enhance the Breadth of DNA/Modified Vaccinia Virus Ankara SIV Vaccine-Induced Antibody Responses in Rhesus Macaques. J. Virol. 2016, 90, 8842–8854. [Google Scholar] [CrossRef] [PubMed]
- Quan, F.S.; Sailaja, G.; Skountzou, I.; Huang, C.; Vzorov, A.; Compans, R.W.; Kang, S.M. Immunogenicity of virus-like particles containing modified human immunodeficiency virus envelope proteins. Vaccine 2007, 25, 3841–3850. [Google Scholar] [CrossRef] [PubMed]
- Sailaja, G.; Skountzou, I.; Quan, F.S.; Compans, R.W.; Kang, S.M. Human immunodeficiency virus-like particles activate multiple types of immune cells. Virology 2007, 362, 331–341. [Google Scholar] [CrossRef] [PubMed]
- Belyakov, I.M.; Wyatt, L.S.; Ahlers, J.D.; Earl, P.; Pendleton, C.D.; Kelsall, B.L.; Strober, W.; Moss, B.; Berzofsky, J.A. Induction of a mucosal cytotoxic T-lymphocyte response by intrarectal immunization with a replication-deficient recombinant vaccinia virus expressing human immunodeficiency virus 89.6 envelope protein. J. Virol. 1998, 72, 8264–8272. [Google Scholar] [PubMed]
- Takahashi, H.; Cohen, J.; Hosmalin, A.; Cease, K.B.; Houghten, R.; Cornette, J.L.; DeLisi, C.; Moss, B.; Germain, R.N.; Berzofsky, J.A. An immunodominant epitope of the human immunodeficiency virus envelope glycoprotein gp160 recognized by class I major histocompatibility complex molecule-restricted murine cytotoxic T lymphocytes. Proc. Natl. Acad. Sci. USA 1988, 85, 3105–3109. [Google Scholar] [CrossRef] [PubMed]
- Chamcha, V.; Kannanganat, S.; Gangadhara, S.; Nabi, R.; Kozlowski, P.A.; Montefiori, D.C.; LaBranche, C.C.; Wrammert, J.; Keele, B.F.; Balachandran, H.; et al. Strong, but Age-Dependent, Protection Elicited by a Deoxyribonucleic Acid/Modified Vaccinia Ankara Simian Immunodeficiency Virus Vaccine. Open Forum Infect. Dis. 2016, 3. [Google Scholar] [CrossRef] [PubMed]
- Nigam, P.; Earl, P.L.; Americo, J.L.; Sharma, S.; Wyatt, L.S.; Edghill-Spano, Y.; Chennareddi, L.S.; Silvera, P.; Moss, B.; Robinson, H.L.; et al. DNA/MVA HIV-1/AIDS vaccine elicits long-lived vaccinia virus-specific immunity and confers protection against a lethal monkeypox challenge. Virology 2007, 366, 73–83. [Google Scholar] [CrossRef] [PubMed]
- Smith, J.M.; Amara, R.R.; Campbell, D.; Xu, Y.; Patel, M.; Sharma, S.; Butera, S.T.; Ellenberger, D.L.; Yi, H.; Chennareddi, L.; et al. DNA/MVA vaccine for HIV type 1: Effects of codon-optimization and the expression of aggregates or virus-like particles on the immunogenicity of the DNA prime. AIDS Res. Hum. Retrovir. 2004, 20, 1335–1347. [Google Scholar] [CrossRef] [PubMed]
- Amara, R.R.; Villinger, F.; Staprans, S.I.; Altman, J.D.; Montefiori, D.C.; Kozyr, N.L.; Xu, Y.; Wyatt, L.S.; Earl, P.L.; Herndon, J.G.; et al. Different patterns of immune responses but similar control of a simian-human immunodeficiency virus 89.6P mucosal challenge by modified vaccinia virus Ankara (MVA) and DNA/MVA vaccines. J. Virol. 2002, 76, 7625–7631. [Google Scholar] [CrossRef] [PubMed]
- Goepfert, P.A.; Elizaga, M.L.; Seaton, K.; Tomaras, G.D.; Montefiori, D.C.; Sato, A.; Hural, J.; DeRosa, S.C.; Kalams, S.A.; McElrath, M.J.; et al. Specificity and 6-month durability of immune responses induced by DNA and recombinant modified vaccinia Ankara vaccines expressing HIV-1 virus-like particles. J. Infect. Dis. 2014, 210, 99–110. [Google Scholar] [CrossRef] [PubMed]
- Ye, L.; Wen, Z.; Dong, K.; Pan, L.; Bu, Z.; Compans, R.W.; Zhang, H.; Yang, C. Immunization with a Mixture of HIV Env DNA and VLP Vaccines Augments Induction of CD8 T Cell Responses. J. Biomed. Biotechnol. 2010. [Google Scholar] [CrossRef] [PubMed]
- Buonaguro, L.; Devito, C.; Tornesello, M.L.; Schroder, U.; Wahren, B.; Hinkula, J.; Buonaguro, F.M. DNA-VLP prime-boost intra-nasal immunization induces cellular and humoral anti-HIV-1 systemic and mucosal immunity with cross-clade neutralizing activity. Vaccine 2007, 25, 5968–5977. [Google Scholar] [CrossRef] [PubMed]
- Forsell, M.N.; Li, Y.; Sundback, M.; Svehla, K.; Liljestrom, P.; Mascola, J.R.; Wyatt, R.; Karlsson Hedestam, G.B. Biochemical and immunogenic characterization of soluble human immunodeficiency virus type 1 envelope glycoprotein trimers expressed by semliki forest virus. J. Virol. 2005, 79, 10902–10914. [Google Scholar] [CrossRef] [PubMed]
- Vasan, S.; Schlesinger, S.J.; Chen, Z.; Hurley, A.; Lombardo, A.; Than, S.; Adesanya, P.; Bunce, C.; Boaz, M.; Boyle, R.; et al. Phase 1 safety and immunogenicity evaluation of ADMVA, a multigenic, modified vaccinia Ankara-HIV-1 B’/C candidate vaccine. PLoS ONE 2010, 5, e8816. [Google Scholar] [CrossRef] [PubMed]
- Hemachandra, A.; Puls, R.L.; Sirivichayakul, S.; Kerr, S.; Thantiworasit, P.; Ubolyam, S.; Cooper, D.A.; Emery, S.; Phanuphak, P.; Kelleher, A.; et al. An HIV-1 clade A/E DNA prime, recombinant fowlpox virus boost vaccine is safe, but non-immunogenic in a randomized phase 1/11a trial in Thai volunteers at low risk of HIV infection. Hum. Vaccines 2010, 6. [Google Scholar] [CrossRef] [PubMed]
- Quan, F.S.; Kim, Y.C.; Yoo, D.G.; Compans, R.W.; Prausnitz, M.R.; Kang, S.M. Stabilization of influenza vaccine enhances protection by microneedle delivery in the mouse skin. PLoS ONE 2009, 4, e7152. [Google Scholar] [CrossRef] [PubMed]
- Song, J.M.; Hossain, J.; Yoo, D.G.; Lipatov, A.S.; Davis, C.T.; Quan, F.S.; Chen, L.M.; Hogan, R.J.; Donis, R.O.; Compans, R.W.; et al. Protective immunity against H5N1 influenza virus by a single dose vaccination with virus-like particles. Virology 2010, 405, 165–175. [Google Scholar] [CrossRef] [PubMed]
- Bosio, C.M.; Moore, B.D.; Warfield, K.L.; Ruthel, G.; Mohamadzadeh, M.; Aman, M.J.; Bavari, S. Ebola and Marburg virus-like particles activate human myeloid dendritic cells. Virology 2004, 326, 280–287. [Google Scholar] [CrossRef] [PubMed]
- Buonaguro, L.; Tornesello, M.L.; Tagliamonte, M.; Gallo, R.C.; Wang, L.X.; Kamin-Lewis, R.; Abdelwahab, S.; Lewis, G.K.; Buonaguro, F.M. Baculovirus-derived human immunodeficiency virus type 1 virus-like particles activate dendritic cells and induce ex vivo T-cell responses. J. Virol. 2006, 80, 9134–9143. [Google Scholar] [CrossRef] [PubMed]
- Lenz, P.; Day, P.M.; Pang, Y.Y.; Frye, S.A.; Jensen, P.N.; Lowy, D.R.; Schiller, J.T. Papillomavirus-like particles induce acute activation of dendritic cells. J. Immunol. 2001, 166, 5346–5355. [Google Scholar] [CrossRef] [PubMed]
- Zhang, R.; Li, M.; Chen, C.; Yao, Q. SHIV virus-like particles bind and activate human dendritic cells. Vaccine 2004, 23, 139–147. [Google Scholar] [CrossRef] [PubMed]
- Moron, V.G.; Rueda, P.; Sedlik, C.; Leclerc, C. In vivo, dendritic cells can cross-present virus-like particles using an endosome-to-cytosol pathway. J. Immunol. 2003, 171, 2242–2250. [Google Scholar] [CrossRef] [PubMed]
- Takamura, S.; Niikura, M.; Li, T.C.; Takeda, N.; Kusagawa, S.; Takebe, Y.; Miyamura, T.; Yasutomi, Y. DNA vaccine-encapsulated virus-like particles derived from an orally transmissible virus stimulate mucosal and systemic immune responses by oral administration. Gene Ther. 2004, 11, 628–635. [Google Scholar] [CrossRef] [PubMed]
- Gurunathan, S.; Klinman, D.M.; Seder, R.A. DNA vaccines: Immunology, application, and optimization. Annu. Rev. Immunol. 2000, 18, 927–974. [Google Scholar] [CrossRef] [PubMed]
- Quan, F.S.; Yoo, D.G.; Song, J.M.; Clements, J.D.; Compans, R.W.; Kang, S.M. Kinetics of immune responses to influenza virus-like particles and dose-dependence of protection with a single vaccination. J. Virol. 2009, 83, 4489–4497. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Hellerstein, M.; McDonnel, M.; Amara, R.R.; Wyatt, L.S.; Moss, B.; Robinson, H.L. Dose-response studies for the elicitation of CD8 T cells by a DNA vaccine, used alone or as the prime for a modified vaccinia Ankara boost. Vaccine 2007, 25, 2951–2958. [Google Scholar] [CrossRef] [PubMed]


© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gangadhara, S.; Kwon, Y.-M.; Jeeva, S.; Quan, F.-S.; Wang, B.; Moss, B.; Compans, R.W.; Amara, R.R.; Jabbar, M.A.; Kang, S.-M. Vaccination with Combination DNA and Virus-Like Particles Enhances Humoral and Cellular Immune Responses upon Boost with Recombinant Modified Vaccinia Virus Ankara Expressing Human Immunodeficiency Virus Envelope Proteins. Vaccines 2017, 5, 52. https://doi.org/10.3390/vaccines5040052
Gangadhara S, Kwon Y-M, Jeeva S, Quan F-S, Wang B, Moss B, Compans RW, Amara RR, Jabbar MA, Kang S-M. Vaccination with Combination DNA and Virus-Like Particles Enhances Humoral and Cellular Immune Responses upon Boost with Recombinant Modified Vaccinia Virus Ankara Expressing Human Immunodeficiency Virus Envelope Proteins. Vaccines. 2017; 5(4):52. https://doi.org/10.3390/vaccines5040052
Chicago/Turabian StyleGangadhara, Sailaja, Young-Man Kwon, Subbiah Jeeva, Fu-Shi Quan, Baozhong Wang, Bernard Moss, Richard W. Compans, Rama Rao Amara, M. Abdul Jabbar, and Sang-Moo Kang. 2017. "Vaccination with Combination DNA and Virus-Like Particles Enhances Humoral and Cellular Immune Responses upon Boost with Recombinant Modified Vaccinia Virus Ankara Expressing Human Immunodeficiency Virus Envelope Proteins" Vaccines 5, no. 4: 52. https://doi.org/10.3390/vaccines5040052
APA StyleGangadhara, S., Kwon, Y.-M., Jeeva, S., Quan, F.-S., Wang, B., Moss, B., Compans, R. W., Amara, R. R., Jabbar, M. A., & Kang, S.-M. (2017). Vaccination with Combination DNA and Virus-Like Particles Enhances Humoral and Cellular Immune Responses upon Boost with Recombinant Modified Vaccinia Virus Ankara Expressing Human Immunodeficiency Virus Envelope Proteins. Vaccines, 5(4), 52. https://doi.org/10.3390/vaccines5040052





