Impact of the Respiratory Microbiome on Host Responses to Respiratory Viral Infection
Abstract
:1. Introduction
1.1. Burden of Acute Respiratory Infections (ARI)
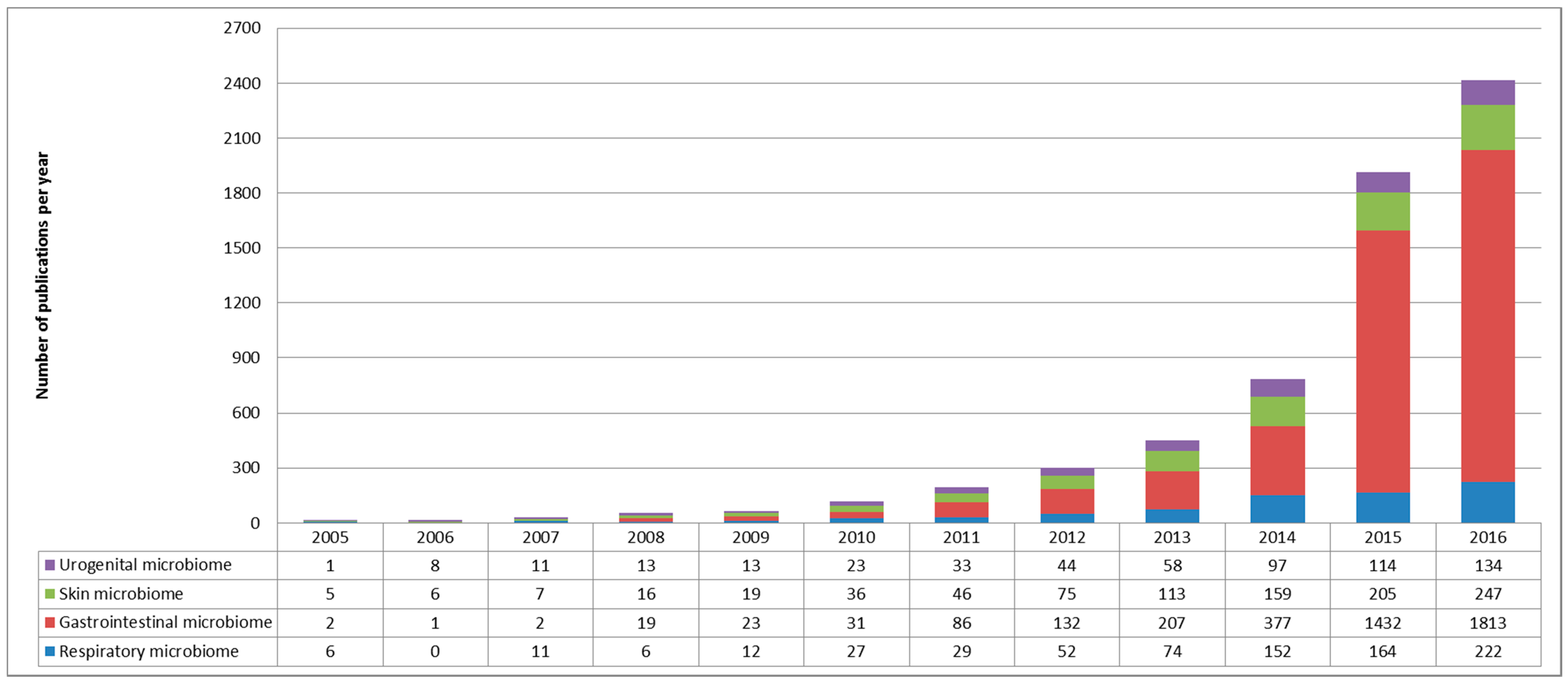
1.2. The Respiratory Microbiome: Still a Lot of Unknowns
2. Impact of the Respiratory Microbiota on Viral Infection Pathogenesis
2.1. Bacterial Microbiota Direct Impact on the Respiratory Tract Environment
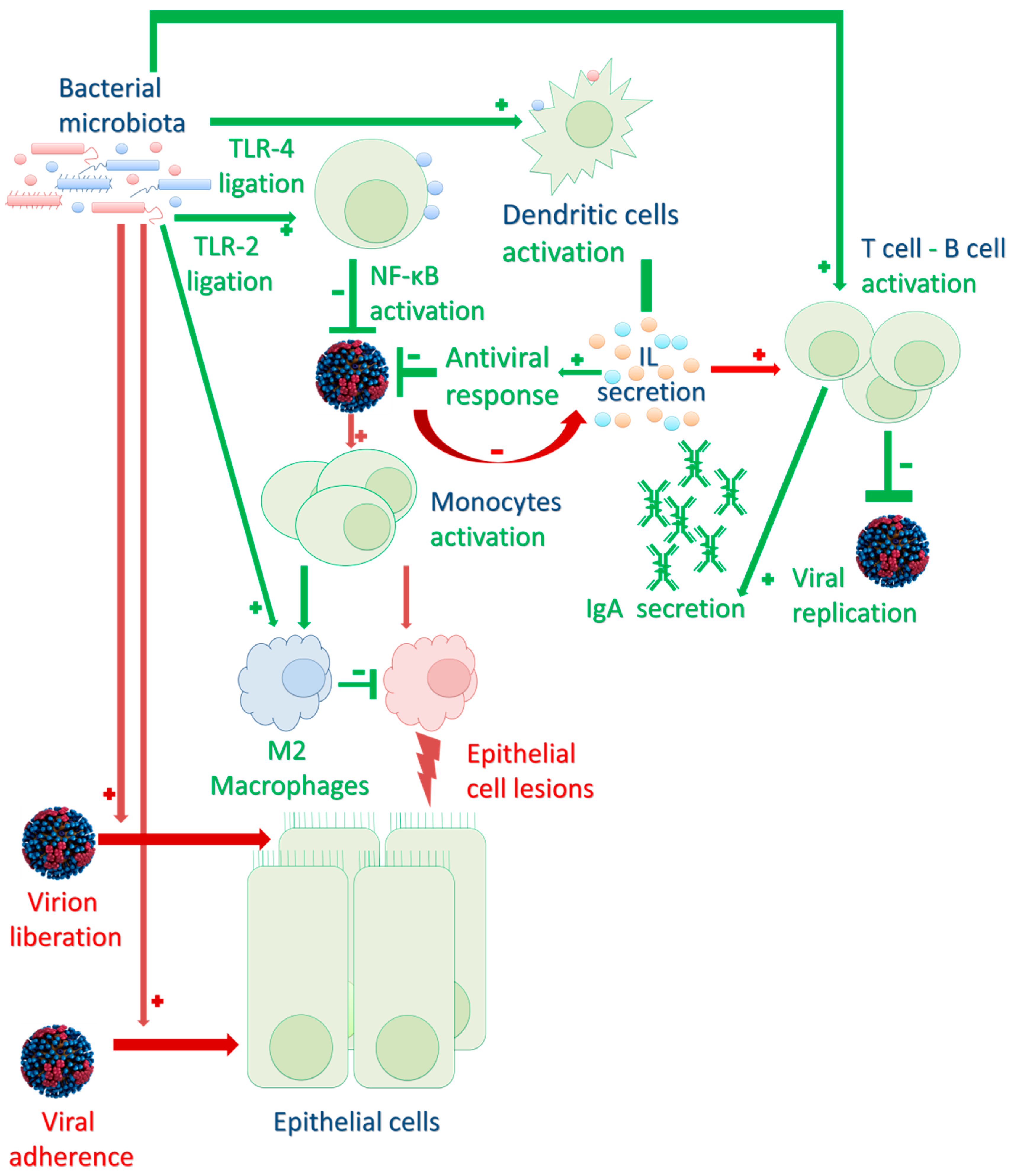
2.2. Impact on Innate and Adaptive Immune Responses to Infection
3. Clinical Use of the Respiratory Microbiome Impact during Viral Infections
3.1. Respiratory Microbiome Characterization: Usable as a Prognostic Biomarker?
3.2. Respiratory Microbiome: A Therapeutic Target?
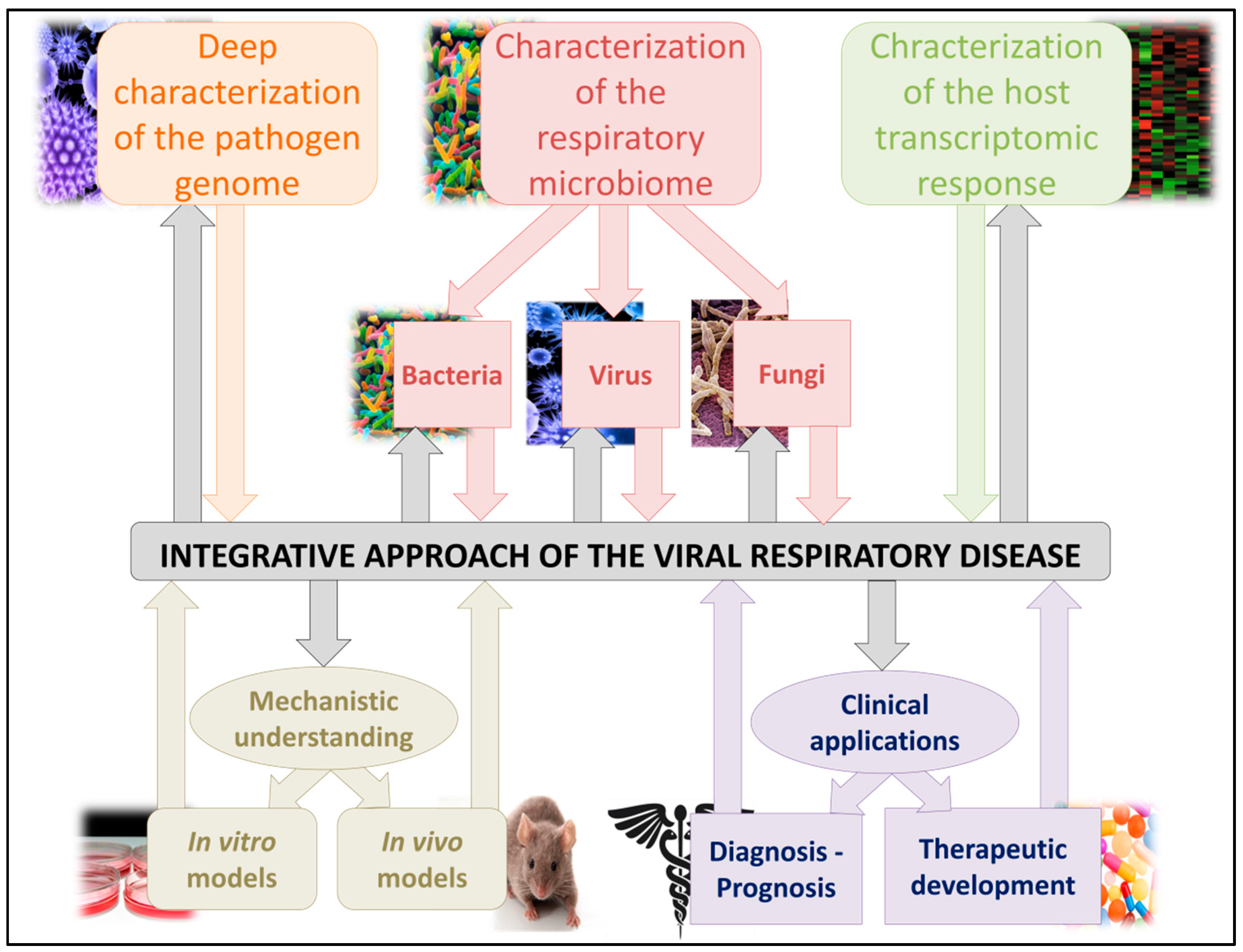
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- WHO Report Prepared for the WHO Annual Consultation on the Composition of Influenza Vaccine for the Northern Hemisphere 2017/18 2 March 2017. Available online: http://www.who.int/influenza/vaccines/virus/recommendations/2017_18_north/en/ (accessed on 1 November 2017).
- Harris, J.-A.S. Infection Control in Pediatric Extended Care Facilities. Infect. Control Hosp. Epidemiol. 2006, 27, 598–603. [Google Scholar] [CrossRef] [PubMed]
- Nair, H.; Nokes, D.J.; Gessner, B.D.; Dherani, M.; Madhi, S.A.; Singleton, R.J.; O’Brien, K.L.; Roca, A.; Wright, P.F.; Bruce, N.; et al. Global Burden of Acute Lower Respiratory Infections due to Respiratory Syncytial Virus in Young Children: A Systematic Review and Meta-Analysis. Lancet 2010, 375, 1545–1555. [Google Scholar] [CrossRef]
- Lederberg, J. ‘Ome Sweet’ Omics—A Genealogical Treasury of Words. Scientist 2001, 15, 8. [Google Scholar]
- Moustafa, A.; Xie, C.; Kirkness, E.; Biggs, W.; Wong, E.; Turpaz, Y.; Bloom, K.; Delwart, E.; Nelson, K.E.; Venter, J.C.; et al. The Blood DNA Virome in 8,000 Humans. PLOS Pathog. 2017, 13, e1006292. [Google Scholar] [CrossRef] [PubMed]
- Sender, R.; Fuchs, S.; Milo, R. Revised Estimates for the Number of Human and Bacteria Cells in the Body. PLOS Biol. 2016, 14, e1002533. [Google Scholar] [CrossRef] [PubMed]
- Luoto, R.; Ruuskanen, O.; Waris, M.; Kalliomäki, M.; Salminen, S.; Isolauri, E. Prebiotic and Probiotic Supplementation Prevents rhinovirus Infections in Preterm infants: A Randomized, Placebo-Controlled Trial. J. Allergy Clin. Immunol. 2014, 133, 405–413. [Google Scholar] [CrossRef] [PubMed]
- Tapiovaara, L.; Kumpu, M.; Mäkivuokko, H.; Waris, M.; Korpela, R.; Pitkäranta, A.; Winther, B. Human Rhinovirus in Experimental Infection after Peroral Lactobacillus Rhamnosus GG Consumption, a Pilot Study. Int. Forum Allergy Rhinol. 2016, 6, 848–853. [Google Scholar] [CrossRef] [PubMed]
- Chertow, D.S.; Memoli, M.J. Bacterial Coinfection in Influenza: A Grand Rounds Review. JAMA 2013, 309, 275–282. [Google Scholar] [CrossRef] [PubMed]
- McCullers, J.A.; Bartmess, K.C. Role of Neuraminidase in Lethal Synergism between Influenza Virus and Streptococcus pneumoniae. J. Infect. Dis. 2003, 187, 1000–1009. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Li, F.; Sun, R.; Gao, X.; Wei, H.; Li, L.-J.; Tian, Z. Bacterial Colonization Dampens Influenza-Mediated Acute Lung Injury via Induction of M2 Alveolar Macrophages. Nat. Commun. 2013, 4. [Google Scholar] [CrossRef] [PubMed]
- Ichinohe, T.; Pang, I.K.; Kumamoto, Y.; Peaper, D.R.; Ho, J.H.; Murray, T.S.; Iwasaki, A. Microbiota Regulates Immune Defense against Respiratory Tract Influenza A Virus Infection. Proc. Natl. Acad. Sci. USA 2011, 108, 5354–5359. [Google Scholar] [CrossRef] [PubMed]
- Langevin, S.; Pichon, M.; Smith, E.; Morrison, J.; Bent, Z.; Green, R.; Baker, K.; Solberg, O.; Gillet, Y.; Javouhey, E.; et al. Early Nasopharyngeal Microbial Signature Associated with Severe Influenza in Children: A Retrospective Pilot Study. J. Gen. Virol. 2017. [Google Scholar] [CrossRef] [PubMed]
- De Paepe, M.; Leclerc, M.; Tinsley, C.R.; Petit, M.-A. Bacteriophages: An Underestimated Role in Human and Animal Health? Front. Cell. Infect. Microbiol. 2014, 4, 39. [Google Scholar] [CrossRef] [PubMed]
- Barr, J.J.; Auro, R.; Furlan, M.; Whiteson, K.L.; Erb, M.L.; Pogliano, J.; Stotland, A.; Wolkowicz, R.; Cutting, A.S.; Doran, K.S.; et al. Bacteriophage Adhering to Mucus Provide a Non-Host-Derived Immunity. Proc. Natl. Acad. Sci. USA 2013, 110, 10771–10776. [Google Scholar] [CrossRef] [PubMed]
- Yun, Y.; Srinivas, G.; Kuenzel, S.; Linnenbrink, M.; Alnahas, S.; Bruce, K.D.; Steinhoff, U.; Baines, J.F.; Schaible, U.E. Environmentally Determined Differences in the Murine Lung Microbiota and Their Relation to Alveolar Architecture. PLoS ONE 2014, 9, e113466. [Google Scholar] [CrossRef] [PubMed]
- Marsland, B.J.; Gollwitzer, E.S. Host-Microorganism Interactions in Lung Diseases. Nat. Rev. Immunol. 2014, 14, 827–835. [Google Scholar] [CrossRef] [PubMed]
- Leung, R.K.-K.; Zhou, J.-W.; Guan, W.; Li, S.-K.; Yang, Z.-F.; Tsui, S.K.-W. Modulation of Potential Respiratory Pathogens by pH1N1 Viral Infection. Clin. Microbiol. Infect. 2013, 19, 930–935. [Google Scholar] [CrossRef] [PubMed]
- Salk, H.M.; Simon, W.L.; Lambert, N.D.; Kennedy, R.B.; Grill, D.E.; Kabat, B.F.; Poland, G.A. Taxa of the Nasal Microbiome are Associated with Influenza-Specific IgA Response to Live Attenuated Influenza Vaccine. PLoS ONE 2016, 11, e0162803. [Google Scholar] [CrossRef] [PubMed]
- Verkaik, N.J.; Nguyen, D.T.; de Vogel, C.P.; Moll, H.A.; Verbrugh, H.A.; Jaddoe, V.W.V.; Hofman, A.; van Wamel, W.J.B.; van den Hoogen, B.G.; Buijs-Offerman, R.M.G.B.; et al. Streptococcus Pneumoniae Exposure is Associated with Human Metapneumovirus Seroconversion and Increased Susceptibility to in Vitro HMPV Infection. Clin. Microbiol. Infect. 2011, 17, 1840–1844. [Google Scholar] [CrossRef] [PubMed]
- De Steenhuijsen Piters, W.A.A.; Heinonen, S.; Hasrat, R.; Bunsow, E.; Smith, B.; Suarez-Arrabal, M.-C.; Chaussabel, D.; Cohen, D.M.; Sanders, E.A.M.; Ramilo, O.; et al. Nasopharyngeal Microbiota, Host Transcriptome and Disease Severity in Children with Respiratory Syncytial Virus Infection. Am. J. Respir. Crit. Care Med. 2016, 194, 1104–1115. [Google Scholar] [CrossRef] [PubMed]
- Teo, S.M.; Mok, D.; Pham, K.; Kusel, M.; Serralha, M.; Troy, N.; Holt, B.J.; Hales, B.J.; Walker, M.L.; Hollams, E.; et al. The Infant Nasopharyngeal Microbiome Impacts Severity of Lower Respiratory Infection and Risk of Asthma Development. Cell Host Microbe 2015, 17, 704–715. [Google Scholar] [CrossRef] [PubMed]
- Beura, L.K.; Hamilton, S.E.; Bi, K.; Schenkel, J.M.; Odumade, O.A.; Casey, K.A.; Thompson, E.A.; Fraser, K.A.; Rosato, P.C.; Filali-Mouhim, A.; et al. Normalizing the Environment Recapitulates Adult Human Immune Traits in Laboratory Mice. Nature 2016, 532, 512–516. [Google Scholar] [CrossRef] [PubMed]
- Furusawa, Y.; Obata, Y.; Fukuda, S.; Endo, T.A.; Nakato, G.; Takahashi, D.; Nakanishi, Y.; Uetake, C.; Kato, K.; Kato, T.; et al. Commensal Microbe-Derived Butyrate Induces the Differentiation of Colonic Regulatory T cells. Nature 2013, 504, 446–450. [Google Scholar] [CrossRef] [PubMed]
- Cagno, V.; Donalisio, M.; Civra, A.; Volante, M.; Veccelli, E.; Oreste, P.; Rusnati, M.; Lembo, D. Highly Sulfated K5 Escherichia Coli Polysaccharide Derivatives Inhibit Respiratory Syncytial Virus Infectivity in Cell Lines and Human Tracheal-Bronchial Histocultures. Antimicrob. Agents Chemother. 2014, 58, 4782–4794. [Google Scholar] [CrossRef] [PubMed]
- Sajjan, U.S.; Jia, Y.; Newcomb, D.C.; Bentley, J.K.; Lukacs, N.W.; LiPuma, J.; Hershenson, M.B.H. Influenzae Potentiates Airway Epithelial Cell Responses to Rhinovirus by Increasing ICAM-1 and TLR3 Expression. FASEB J. 2006, 20, 2121–2123. [Google Scholar] [CrossRef] [PubMed]
- Pfeiffer, J.K.; Virgin, H.W. Viral Immunity. Transkingdom Control of Viral Infection and Immunity in the Mammalian Intestine. Science 2016, 351. [Google Scholar] [CrossRef] [PubMed]
- Kuss, S.K.; Best, G.T.; Etheredge, C.A.; Pruijssers, A.J.; Frierson, J.M.; Hooper, L.V.; Dermody, T.S.; Pfeiffer, J.K. Intestinal Microbiota Promote Enteric Virus Replication and Systemic Pathogenesis. Science 2011, 334, 249–252. [Google Scholar] [CrossRef] [PubMed]
- Rondina, M.T.; Tatsumi, K.; Bastarache, J.A.; Mackman, N. Microvesicle Tissue Factor Activity and Interleukin-8 Levels are Associated with Mortality in Patients with Influenza A/H1N1 Infection. Crit. Care Med. 2016, 44, 574–578. [Google Scholar] [CrossRef] [PubMed]
- Oshansky, C.M.; Gartland, A.J.; Wong, S.-S.; Jeevan, T.; Wang, D.; Roddam, P.L.; Caniza, M.A.; Hertz, T.; DeVincenzo, J.P.; Webby, R.J.; et al. Mucosal Immune Responses Predict Clinical Outcomes during Influenza Infection Independently of Age and Viral Load. Am. J. Respir. Crit. Care Med. 2014, 189, 449–462. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.; Jiang, Z.-Y.; Sun, Y.-F.; Yu, B.; Chen, J.; Dai, C.-Q.; Wu, X.-L.; Tang, X.-L.; Chen, X.-Y. Microbiota Regulates the TLR7 Signaling Pathway Against Respiratory Tract Influenza A Virus Infection. Curr. Microbiol. 2013, 67, 414–422. [Google Scholar] [CrossRef] [PubMed]
- Abt, M.C.; Osborne, L.C.; Monticelli, L.A.; Doering, T.A.; Alenghat, T.; Sonnenberg, G.F.; Paley, M.A.; Antenus, M.; Williams, K.L.; Erikson, J.; et al. Commensal Bacteria Calibrate the Activation Threshold of Innate Antiviral Immunity. Immunity 2012, 37, 158–170. [Google Scholar] [CrossRef] [PubMed]
- Arpaia, N.; Campbell, C.; Fan, X.; Dikiy, S.; van der Veeken, J.; deRoos, P.; Liu, H.; Cross, J.R.; Pfeffer, K.; Coffer, P.J.; et al. Metabolites Produced by Commensal Bacteria Promote Peripheral Regulatory T-cell Generation. Nature 2013, 504, 451–455. [Google Scholar] [CrossRef] [PubMed]
- Mayer-Hamblett, N.; Rosenfeld, M.; Gibson, R.L.; Ramsey, B.W.; Kulasekara, H.D.; Retsch-Bogart, G.Z.; Morgan, W.; Wolter, D.J.; Pope, C.E.; Houston, L.S.; et al. Pseudomonas Aeruginosa in Vitro Phenotypes Distinguish Cystic Fibrosis Infection Stages and Outcomes. Am. J. Respir. Crit. Care Med. 2014, 190, 289–297. [Google Scholar] [CrossRef] [PubMed]
- Biesbroek, G.; Wang, X.; Keijser, B.J.F.; Eijkemans, R.M.; Trzciński, K.; Rots, N.Y.; Veenhoven, R.H.; Sanders, E.A.M.; Bogaert, D. Seven-Valent Pneumococcal Conjugate Vaccine and Nasopharyngeal Microbiota in Healthy Children. Emerg. Infect. Dis. 2014, 20, 201–210. [Google Scholar] [CrossRef] [PubMed]
- Tarabichi, Y.; Li, K.; Hu, S.; Nguyen, C.; Wang, X.; Elashoff, D.; Saira, K.; Frank, B.; Bihan, M.; Ghedin, E.; et al. The Administration of Intranasal Live Attenuated Influenza Vaccine Induces Changes in the Nasal Microbiota and Nasal Epithelium Gene Expression Profiles. Microbiome 2015, 3. [Google Scholar] [CrossRef] [PubMed]
- Tuvim, M.J.; Evans, S.E.; Clement, C.G.; Dickey, B.F.; Gilbert, B. Augmented Lung Inflammation Protects Against Influenza A Pneumonia. PLoS ONE 2009, 4, e4176. [Google Scholar] [CrossRef] [PubMed]
- Liu, Q.; Zhou, Y.-H.; Yang, Z.-Q. The Cytokine Storm of Severe Influenza and Development of Immunomodulatory Therapy. Cell. Mol. Immunol. 2016, 13, 3–10. [Google Scholar] [CrossRef] [PubMed]
- Monneret, G.; Venet, F. Sepsis-Induced Immune Alterations Monitoring by Flow Cytometry as a Promising Tool for Individualized Therapy. Cytom. B Clin. Cytom. 2016, 90, 376–386. [Google Scholar] [CrossRef] [PubMed]
- Drake, M.G.; Evans, S.E.; Dickey, B.F.; Fryer, A.D.; Jacoby, D.B. Toll-like Receptor-2/6 and Toll-like Receptor-9 Agonists Suppress Viral Replication but not Airway Hyperreactivity in Guinea Pigs. Am. J. Respir. Cell Mol. Biol. 2013, 48, 790–796. [Google Scholar] [CrossRef] [PubMed]
- Williams, A.E.; Edwards, L.; Humphreys, I.R.; Snelgrove, R.; Rae, A.; Rappuoli, R.; Hussell, T. Innate Imprinting by the Modified Heat-Labile Toxin of Escherichia Coli (LTK63) Provides Generic Protection against Lung Infectious Disease. J. Immunol. 2004, 173, 7435–7443. [Google Scholar] [CrossRef] [PubMed]
- Norton, E.B.; Clements, J.D.; Voss, T.G.; Cárdenas-Freytag, L. Prophylactic Administration of Bacterially Derived Immunomodulators Improves the Outcome of Influenza Virus Infection in a Murine Model. J. Virol. 2010, 84, 2983–2995. [Google Scholar] [CrossRef] [PubMed]
- Chiba, E.; Tomosada, Y.; Vizoso-Pinto, M.G.; Salva, S.; Takahashi, T.; Tsukida, K.; Kitazawa, H.; Alvarez, S.; Villena, J. Immunobiotic Lactobacillus Rhamnosus Improves Resistance of Infant Mice against Respiratory Syncytial Virus Infection. Int. Immunopharmacol. 2013, 17, 373–382. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, T.; Yamaya, M.; Sekizawa, K.; Hosoda, M.; Yamada, N.; Ishizuka, S.; Yoshino, A.; Yasuda, H.; Takahashi, H.; Nishimura, H.; et al. Erythromycin Inhibits Rhinovirus Infection in Cultured Human Tracheal Epithelial Cells. Am. J. Respir. Crit. Care Med. 2002, 165, 1113–1118. [Google Scholar] [CrossRef] [PubMed]
| Virus | Model | Sample Site | Bacteria | Consequences | Outcome | Reference |
|---|---|---|---|---|---|---|
| IV | Human clinical samples | Induced sputum | Acinetobacter baumanii | Bacteria species increasing during A(H1N1) infection | No modification | Leung et al. [18] |
| Bacillus spp. | ||||||
| Pseudomonas aeruginosa | ||||||
| Human clinical samples | Induced sputum | Neisseria spp. | Bacteria species decreasing during A(H1N1) infection | No modification | Leung et al. [18] | |
| Prevotella spp. | ||||||
| Mouse model | Lung homogenates | Streptococcus pneumoniae | Increased severity | Complication | McCullers et al. [10] | |
| Human clinical samples | Nasopharyngeal samples | Streptobacillus spp. | Predict a severe form of influenza in children | Complication | Langevin et al. [13] | |
| Porphyromonas spp. | ||||||
| Granulicatella spp. | ||||||
| Fusobacterium spp. | ||||||
| Lachnospiracea spp. | ||||||
| Haemophilus spp. | ||||||
| Human clinical samples | Nasopharyngeal samples | Staphylococcus aureus | Predict a benign form of influenza in children | Protection | Langevin et al. [13] | |
| Human clinical samples | Nasal swabs | Lactobacillus helveticus | Correlated with higher H1 IgA antibody response | Protection | Salk et al. [19] | |
| Bacteroides ovatus | ||||||
| Human clinical samples | Nasopharyngeal samples Nasal swabs | Veillonella dispar | Predict a severe form of influenza in children Correlated with lower H1 IgA antibody response | Protection | Langevin et al. [13] Salk et al. [19] | |
| Human clinical samples | Nasopharyngeal samples Nasal swabs | Prevotella melaninogenica | Predict a severe form of influenza in children Correlated with Higher H1 IgA antibody response | Complication—Protection | Langevin et al. [13] Salk et al. [19] | |
| Human clinical samples | Nasal swabs | Streptococcus infantis | Correlated with Higher H1 IgA antibody response | Complication | Salk et al. [19] | |
| MPV | Cellular model | Cellular model | Streptococcus pneumoniae | Increased seroconversion rate during challenge | Protection | Verkaik et al. [20] |
| RSV | Human clinical samples | Nasopharyngeal samples | Haemophilus influenzae Streptococcus spp. | Increased hospitalization rate | Complication | De Steenhuijsen Piters et al. [21] |
| Mice model—Human clinical samples | Nasopharyngeal samples | Moraxella catarrhalis | Increased severity | Complication | Teo et al. [22] Beura et al. [23] Furusawa et al. [24] | |
| Mice model Human clinical samples | Nasopharyngeal samples Lung homogenates | Staphylococcus aureus | Decreased complications | Protection | De Steenhuijsen Piters et al. [21] Wang et al. [11] | |
| Cellular model | Cellular model | Escherichia coli | Increased protection against infection | Protection | Cagno et al. [25] | |
| RV | Cellular model | Cellular model | Haemophilus influenzae | Entry point production via ICAM-1 and TLR-3 production | Complication | Sajjan et al. [26] |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pichon, M.; Lina, B.; Josset, L. Impact of the Respiratory Microbiome on Host Responses to Respiratory Viral Infection. Vaccines 2017, 5, 40. https://doi.org/10.3390/vaccines5040040
Pichon M, Lina B, Josset L. Impact of the Respiratory Microbiome on Host Responses to Respiratory Viral Infection. Vaccines. 2017; 5(4):40. https://doi.org/10.3390/vaccines5040040
Chicago/Turabian StylePichon, Maxime, Bruno Lina, and Laurence Josset. 2017. "Impact of the Respiratory Microbiome on Host Responses to Respiratory Viral Infection" Vaccines 5, no. 4: 40. https://doi.org/10.3390/vaccines5040040
APA StylePichon, M., Lina, B., & Josset, L. (2017). Impact of the Respiratory Microbiome on Host Responses to Respiratory Viral Infection. Vaccines, 5(4), 40. https://doi.org/10.3390/vaccines5040040







