Egg-Independent Influenza Vaccines and Vaccine Candidates
Abstract
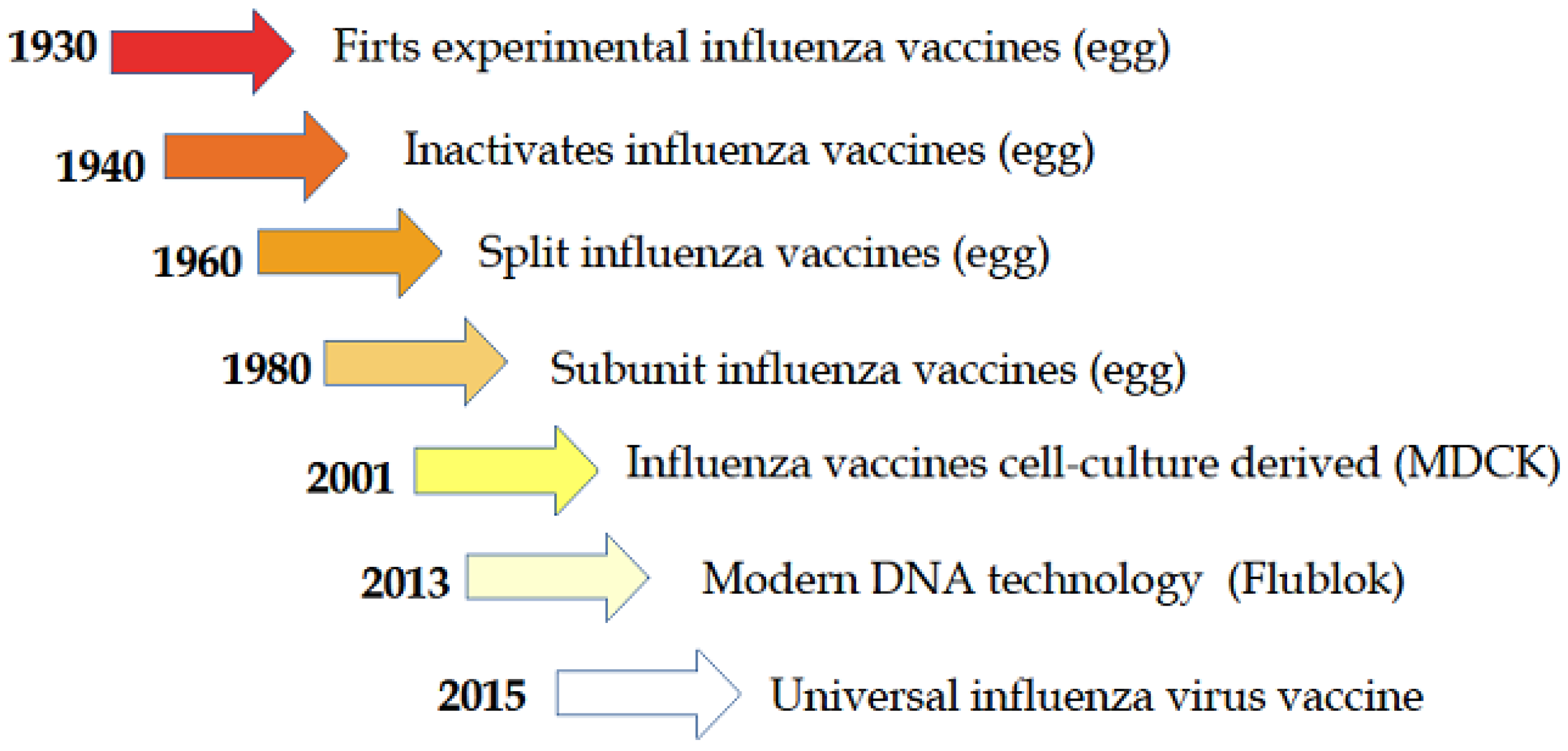
:1. Introduction
2. Cell Culture-Derived Vaccines and Vaccine Candidates
3. New Technologies for New Influenza Vaccines
4. Conclusions
Conflicts of Interest
References
- World Health Organization. Influenza Web Page. Available online: http://www.who.int/mediacentre/factsheets/fs211/en/ (accessed on 5 September 2016).
- World Health Organization (WHO). Weekly epidemiological record Relevé épidémiologique hebdomadaire. ANNÉE 2014, 89, 245–256. [Google Scholar]
- WHO. Global Epidemiological Surveillance Standards for Influenza. Available online: http://www.who.int/influenza/resources/documents/influenza_surveillance_manual/en/ (accessed on 24 May 2017).
- Salomon, R.; Webster, R.G. The influenza virus enigma. Cell 2009, 3, 402–410. [Google Scholar] [CrossRef] [PubMed]
- Hilleman, M.R. Realities and enigmas of human viral influenza: Pathogenesis, epidemiology and control. Vaccine 2002, 20, 3068–3087. [Google Scholar] [CrossRef]
- European Centre for Disease Prevention and Control. Available online: http://ecdc.europa.eu/en/healthtopics/seasonal_influenza/vaccines/Pages/influenza_vaccination.aspx (accessed on 22 May 2017).
- Kistner, O.; Barrett, P.N.; Mundt, W.; Reiter, M.; Schober-Bendixen, S.; Dorner, F. Development of a mammalian cell (Vero) derived candidate influenza virus vaccine. Vaccine 1998, 16, 960–968. [Google Scholar] [CrossRef]
- Pandey, A.; Singh, N.; Sambhara, S.; Mittal, S.K. Egg-independent vaccine strategies for highly pathogenic H5N1 influenza viruses. Hum. Vaccin 2010, 6, 178–188. [Google Scholar] [CrossRef] [PubMed]
- Centers for Disease Control and Prevention. How Influenza (Flu) Vaccines Are Made. Available online: https://www.cdc.gov/flu/protect/vaccine/how-fluvaccine-made.htm (accessed on 24 June 2017).
- Centers for Disease Control and Prevention. Seasonal Influenza Vaccine Total Doses Distributed. Available online: https://www.cdc.gov/flu/professionals/vaccination/vaccinesupply.htm (accessed on 24 June 2017).
- Barberis, I.; Myles, P.; Ault, S.K.; Bragazzi, N.L.; Martini, M. History and evolution of influenza control through vaccination: From the first monovalent vaccine to universal vaccines. J. Prev. Med. Hyg. 2016, 57, 115–120. [Google Scholar]
- World Health Organization (WHO). Cell culture as a substrate for the production of influenza vaccines: memorandum from a WHO meeting. Bull. World Health Organ. 1995, 73, 431–435. [Google Scholar]
- Barrett, P.N.; Mundt, W.; Kistner, O.; Howard, M.K. Vero cell platform in vaccine production: Moving towards cell culture-based viral vaccines. Expert Rev. Vaccines 2009, 8, 607–618. [Google Scholar] [CrossRef] [PubMed]
- Chan, C.Y.; Tambyah, P.A. Preflucel®: A Vero-cell culture-derived trivalent influenza vaccine. Expert Rev. Vaccines 2012, 11, 759–773. [Google Scholar] [CrossRef] [PubMed]
- Szymczakiewicz-Multanowska, A.; Groth, N.; Bugarini, R.; Lattanzi, M.; Casula, D.; Hilbert, A.; Tsai, T.; Podda, A. Safety and immunogenicity of a novel influenza subunit vaccine produced in mammalian cell culture. J. Infect. Dis. 2009, 200, 841–848. [Google Scholar] [CrossRef] [PubMed]
- Onions, D.; Egan, W.; Jarrett, R.; Novicki, D.; Gregersen, J.P. Validation of the safety of MDCK cells as a substrate for the production of a cell-derived influenza vaccine. Biologicals 2010, 38, 544–551. [Google Scholar] [CrossRef] [PubMed]
- Robertson, J.S.; Cook, P.; Attwell, A.M.; Williams, S.P. Replicative advantage in tissue culture of egg-adapted influenza virus over tissue-culture derived virus: Implications for vaccine manufacture. Vaccine 1995, 13, 1583–1588. [Google Scholar] [CrossRef]
- European Medicines Agency. Optaflu Influenza Vaccine (Surface Antigen, Inactivated, Prepared in Cell Cultures). Available online: www.ema.europa.eu/docs/en_GB/document_library/EPAR_Summary_for_the_public/human/000758/WC500046952.pdf (accessed on 14 September 2016).
- Manini, I.; Domnich, A.; Amicizia, D.; Rossi, S.; Pozzi, T.; Gasparini, R.; Panatto, D.; Montomoli, E. Flucelvax (Optaflu) for seasonal influenza. Expert Rev. Vaccines 2015, 14, 789–804. [Google Scholar] [CrossRef] [PubMed]
- Doroshenko, A.; Halperin, S.A. Trivalent MDCK cell culture-derived influenza vaccine Optaflu (Novartis Vaccines). Expert Rev. Vaccines 2009, 8, 679–688. [Google Scholar] [CrossRef] [PubMed]
- U.S. Food & Drug Administration. Vaccines, Blood & Biologics. Available online: www.fda.gov/BiologicsBloodVaccines/Vaccines/ApprovedProducts/ucm328684.htm (accessed on 24 May 2017).
- Clark, T.W.; Pareek, M.; Hoschler, K.; Dillon, H.; Nicholson, K.G.; Groth, N.; Stephenson, I. Trial of 2009 influenza A (H1N1) monovalent MF59-adjuvanted vaccine. N. Engl. J. Med. 2009, 361, 2424–2435. [Google Scholar] [CrossRef] [PubMed]
- Reynales, H.; Astudillo, P.; de Vallière, S.; Hatz, C.; Schlagenhauf, P.; Rath, B.; Velentgas, P.; Fariña, A.; Sales-Carmona, V.; Groth, N. A prospective observational safety study on MF59(®) adjuvanted cell culture-derived vaccine, Celtura(®) during the A/H1N1 (2009) influenza pandemic. Vaccine 2012, 30, 6436–6443. [Google Scholar] [CrossRef] [PubMed]
- Celvapan––H1N1 Pandemic Influenza Vaccine. Available online: www.drugdevelopment-echnology.com/projects/celvapan/ (accessed on 25 May 2017).
- Ehrlich, H.J.; Berezuk, G.; Fritsch, S.; Aichinger, G.; Singer, J.; Portsmouth, D.; Hart, M.K.; El-Amin, W.; Kistner, O.; Barrett, P.N. Clinical development of a Vero cell culture-derived seasonal influenza vaccine. Vaccine 2012, 30, 4377–4386. [Google Scholar] [CrossRef] [PubMed]
- Barrett, P.N.; Berezuk, G.; Fritsch, S.; Aichinger, G.; Hart, M.K.; El-Amin, W.; Kistner, O.; Ehrlich, H.J. Efficacy, safety, and immunogenicity of a Vero-cell-culture-derived trivalent influenza vaccine: A multicentre, double-blind, randomised, placebo-controlled trial. Lancet 2011, 377, 751–759. [Google Scholar] [CrossRef]
- European Medicines Agency. Questions and Answers on the Review of Preflucel and Associated Names (Influenza Vaccine, Purified Antigen). Available online: http://www.ema.europa.eu/docs/en_GB/document_library/Referrals_document/Preflucel/WC500130157.pdf (accessed on 14 September 2016).
- Cox, R.J.; Madhun, A.S.; Hauge, S.; Sjursen, H.; Major, D.; Kuhne, M.; Höschler, K.; Saville, M.; Vogel, F.R.; Barclay, W.; et al. A phase I clinical trial of a PER.C6 cell grown influenza H7 virus vaccine. Vaccine 2009, 27, 1889–1897. [Google Scholar] [CrossRef] [PubMed]
- Meghrous, J.; Mahmoud, W.; Jacob, D.; Chubet, R.; Cox, M.; Kamen, A.A. Development of a simple and high-yielding fed-batch process for the production of influenza vaccines. Vaccine 2009, 28, 309–316. [Google Scholar] [CrossRef] [PubMed]
- Buckland, B.; Boulanger, R.; Fino, M.; Srivastava, I.; Holtz, K.; Khramtsov, N.; McPherson, C.; Meghrous, J.; Kubera, P.; Cox, M.M. Technology transfer and scale-up of the Flublok recombinant hemagglutinin (HA) influenza vaccine manufacturing process. Vaccine 2014, 32, 5496–5502. [Google Scholar] [CrossRef] [PubMed]
- Mukhopadhyay, T.K. Vaccines Europe 2009. Expert Rev. Vaccines 2010, 9, 125–128. [Google Scholar] [CrossRef] [PubMed]
- Shoji, Y.; Chichester, J.A.; Jones, M.; Manceva, S.D.; Damon, E.; Mett, V.; Musiychuk, K.; Bi, H.; Farrance, C.; Shamloul, M.; et al. Plant-based rapid production of recombinant subunit hemagglutinin vaccines targeting H1N1 and H5N1 influenza. Hum. Vaccines 2011, 7, 41–50. [Google Scholar] [CrossRef]
- Cummings, J.F.; Guerrero, M.L.; Moon, J.E.; Waterman, P.; Nielsen, R.K.; Jefferson, S.; Gross, F.L.; Hancock, K.; Katz, J.M.; Yusibov, V. Safety and immunogenicity of a plant-produced recombinant monomer hemagglutinin-based influenza vaccine derived from influenza A (H1N1)pdm09 virus: A Phase 1 dose-escalation study in healthy adults. Vaccine 2014, 32, 2251–2259. [Google Scholar] [CrossRef] [PubMed]
- Kreijtz, J.H.; Osterhaus, A.D.; Rimmelzwaan, G.F. Vaccination strategies and vaccine formulation for epidemic and pandemic influenza control. Hum. Vaccines 2009, 5, 126–135. [Google Scholar] [CrossRef]
- Kreijtz, J.H.; Goeijenbier, M.; Moesker, F.M.; van den Dries, L.; Goeijenbier, S.; De Gruyter, H.L.; Lehmann, M.H.; Mutsert, G.D.; van de Vijver, D.A.; Volz, A.; et al. Safety and immunogenicity of a modified-vaccinia-virus-Ankara-based influenza A/H5N1 vaccine: A randomised, double-blind phase 1/2a clinical trial. Lancet Infect. Dis. 2014, 14, 1196–1207. [Google Scholar] [CrossRef]
- Ulmer, J.B.; Donnelly, J.J.; Parker, S.E.; Rhodes, G.H.; Felgner, P.L.; Dwarki, V.J.; Gromkowski, S.H.; Deck, R.R.; DeWitt, C.M.; Friedman, A.; et al. Heterologous protection against influenza by injection of DNA encoding viral protein. Science 1993, 259, 1745–1749. [Google Scholar] [CrossRef] [PubMed]
- Krammer, F. Emerging influenza viruses and the prospect of a universal influenza virus vaccine. Biotechnol. J. 2015, 10, 690–701. [Google Scholar] [CrossRef] [PubMed]
- Krammer, F. Novel universal influenza virus vaccine approaches. Curr. Opin. Virol. 2016, 17, 95–103. [Google Scholar] [CrossRef] [PubMed]
- Minor, P.D.; Engelhardt, O.G.; Wood, J.M.; Robertson, J.S.; Blayer, S.; Colegate, T.; Fabry, L.; Heldens, J.G.; Kino, Y.; Kistner, O.; et al. Current challenges in implementing cell-derived influenza vaccines: Implications for production and regulation, July 2007, NIBSC, Potters Bar, UK. Vaccine 2009, 27, 2907–2913. [Google Scholar] [CrossRef] [PubMed]
- Gregersen, J.P. A quantitative risk assessment of exposure to adventitious agents in a cell culture-derived subunit influenza vaccine. Vaccine 2008, 26, 3332–3340. [Google Scholar] [CrossRef] [PubMed]
- Ulmer, J.B. Influenza DNA vaccines. Vaccine 2002, 20, 74–76. [Google Scholar] [CrossRef]
- Dormitzer, P.R. Rapid production of synthetic influenza vaccines. Curr. Top. Microbiol. Immunol. 2015, 386, 237–273. [Google Scholar] [CrossRef] [PubMed]
- Bardiya, N.; Bae, J.H. Influenza vaccines: Recent advances in production technologies. Appl. Microbiol. Biotechnol. 2005, 67, 299–305. [Google Scholar] [CrossRef] [PubMed]
| Manufacturer | Name of Product | Vaccine Type | Adjuvant | Produced in | Age Recommended |
|---|---|---|---|---|---|
| Abbot healthcare | Trivalent: Influvac Imuvac | Inactivated | None | Egg | From 6 months |
| AstraZeneca | Quadrivalent: Fluenz Tetra (Flumist quadrivalent) Trivalent: Fluarix Alpharix Influsplit | Live attenuated | None | Egg | From 24 months to 17 years Trivalent: From 6 months |
| GlaxoSmithKline | Quadrivalent: Fluarix Tetra Alpharix Tetra Influsplit Tetra | Inactivated/split | None | Egg | From 3 years |
| Novartis | Trivalent: Agrippal Fluvirin Fluad | Inactivated/subunit | None None Squalene (MF59) | Egg | From 6 years From 4 years From 65 years |
| Pfizer/CSl Australia | Trivalent: Afluria Enzira | Inactivated | None | Egg | From 5 years |
| Sanofi Pasteur | Trivalent: Vaxigrip Intanza 9 µg Intanza 15µg | Inactivated | None | Egg | From 6 months From 18-59 years From 60 years |
| Manufacturer | Name of Product | Vaccine Type | Adjuvant | Produced in | Age Recommended |
|---|---|---|---|---|---|
| GlaxoSmithKline | Quadrivalent: Fluarix | Inactivated/split | None | Egg | From 3 years |
| ID Biomedical Corp. of Quebec (distributed by GlaxoSmithKline) | Quadrivalent: FluLaval | Inactivated/split | None | Egg | From 3 years |
| Sanofi Pasteur | Quadrivalent: Fluzone Quadrivalent: Fluzone Intradermal Trivalent: Fluzone high dose | Inactivated/split Inactivated/split/intradermal Inactivated/split | None | Egg | From 6 through 35 months From 36 through 8 years From 9 years From 18 through 64 years From 65 years |
| Seqirus | Trivalent: Afluria Fluvirin Fluad | Inactivated/subunit Inactivated/subunit/adjuvanted | None Yes | Egg | From 5 through 8 years From 4 years From 65 years |
| MedImmune | Quadrivalent: FluMist | Live/attenuated | None | Egg | From 2 through 49 years |
| Vaccine | Year of Study Data | Phase | Trial Participants | Results: Immunogenicity |
|---|---|---|---|---|
| Optaflu | 2002 | I and II, randomized, observer-blind, controlled, single-center | 240 healthy adults aged 18–40 years and 120 elderly aged > 61 years | Both Optaflu and control vaccine met all CHMP criteria in hemagglutination inhibition assay |
| Optaflu | 2004–2005 | III, randomized, observer-blind, controlled, multicenter | 1300 healthy adults aged 18–40 years and 1354 elderly aged > 61 years | Both Optaflu and control vaccine met all CHMP criteria. |
| Optaflu | 2005–2006 | III, randomized, observer-blind, controlled, multicenter, lot-to-lot variability | 1200 healthy adults aged 18–40 years | Both Optaflu lots and control vaccine met all three CHMP criteria |
| Celtura | 2009 | I, single center | 176 adults | Satisfactory immune responses |
| Vaccine | Year of Study Data | Phase | Trial Participants | Results: Immunogenicity |
|---|---|---|---|---|
| Celvapan | 2009 | I and II, prospective, randomized, open label, multicentre | 200 adults and 200 elderly | Vaccine met criteria for immunogenicity responses |
| Preflucel | 2008-2009 | III, multicenter, randomized, double-blind, | 7250 adults, 3210 older | Vaccine met criteria for immunogenicity responses |
| Vaccine | Year of Study Data | Phase | Trial Participants | Results: Immunogenicity |
|---|---|---|---|---|
| n/a | 2009 | I, randomized, open label | 60 adults | Poor immunogenicity and humoral immune responses |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Manini, I.; Trombetta, C.M.; Lazzeri, G.; Pozzi, T.; Rossi, S.; Montomoli, E. Egg-Independent Influenza Vaccines and Vaccine Candidates. Vaccines 2017, 5, 18. https://doi.org/10.3390/vaccines5030018
Manini I, Trombetta CM, Lazzeri G, Pozzi T, Rossi S, Montomoli E. Egg-Independent Influenza Vaccines and Vaccine Candidates. Vaccines. 2017; 5(3):18. https://doi.org/10.3390/vaccines5030018
Chicago/Turabian StyleManini, Ilaria, Claudia Maria Trombetta, Giacomo Lazzeri, Teresa Pozzi, Stefania Rossi, and Emanuele Montomoli. 2017. "Egg-Independent Influenza Vaccines and Vaccine Candidates" Vaccines 5, no. 3: 18. https://doi.org/10.3390/vaccines5030018
APA StyleManini, I., Trombetta, C. M., Lazzeri, G., Pozzi, T., Rossi, S., & Montomoli, E. (2017). Egg-Independent Influenza Vaccines and Vaccine Candidates. Vaccines, 5(3), 18. https://doi.org/10.3390/vaccines5030018








