Reemergence of Measles in the Americas: The Genotype B3 2011–2012 Outbreak in Ecuador
Abstract
:1. Introduction
2. Materials and Methods
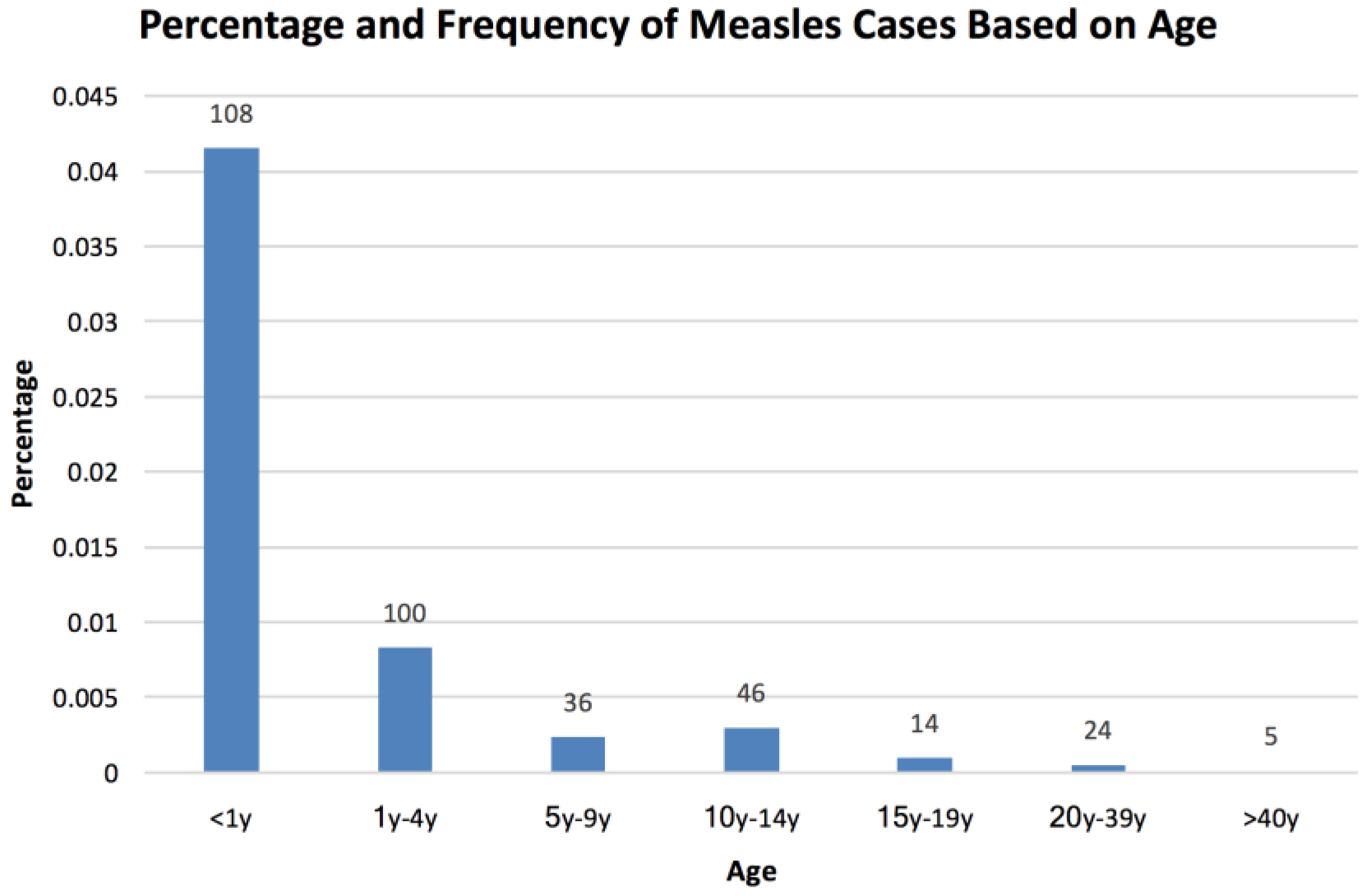
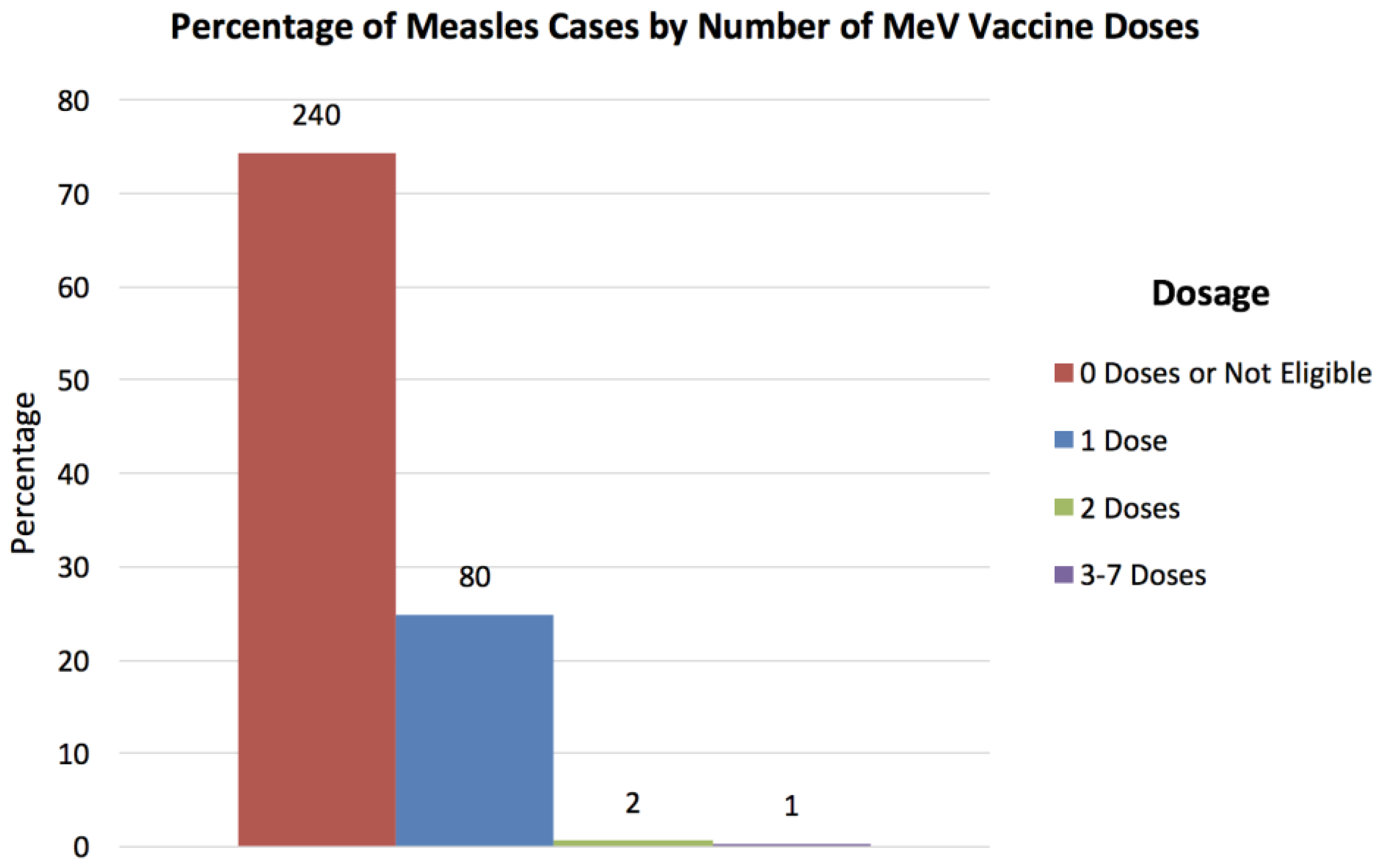
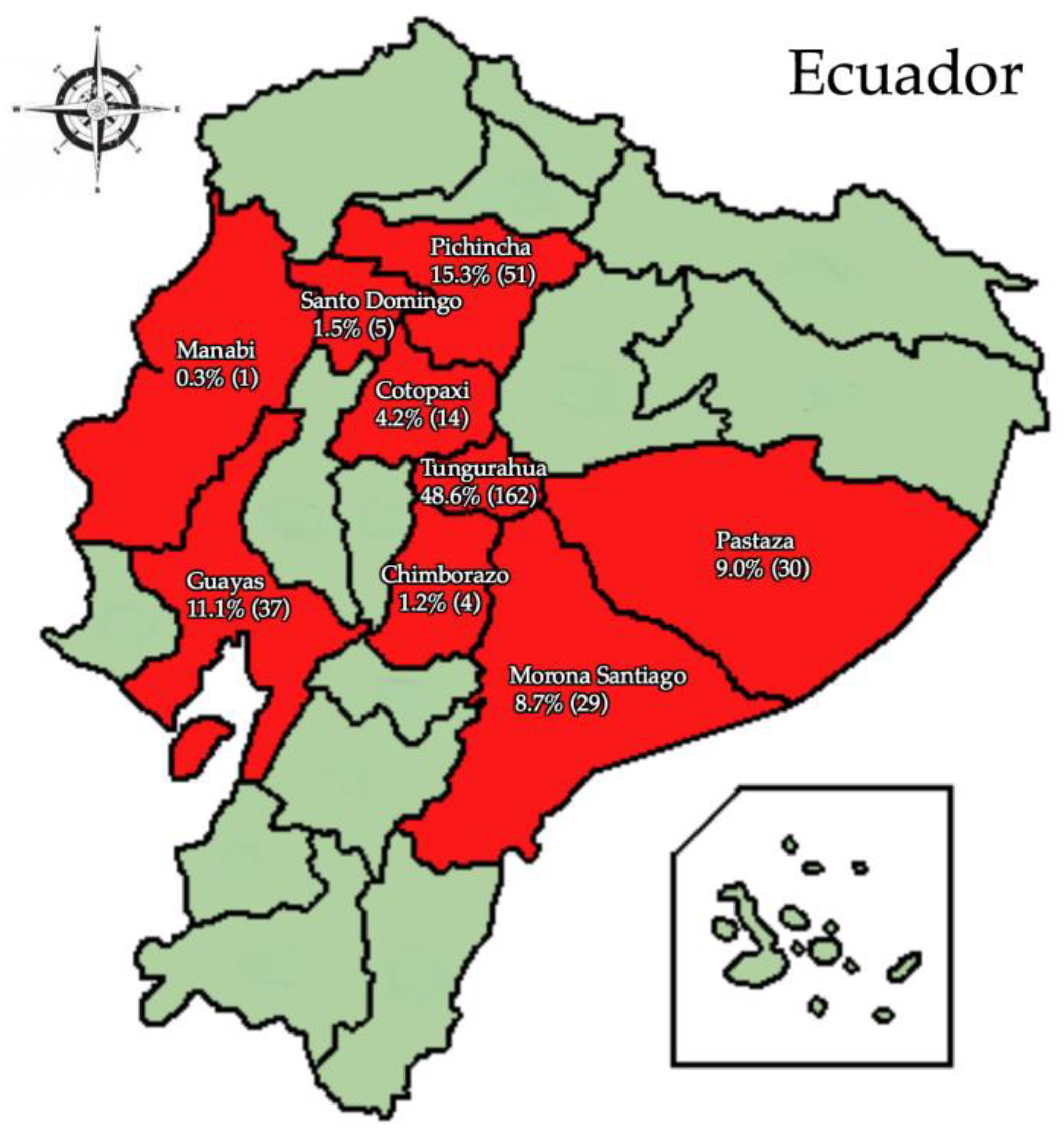
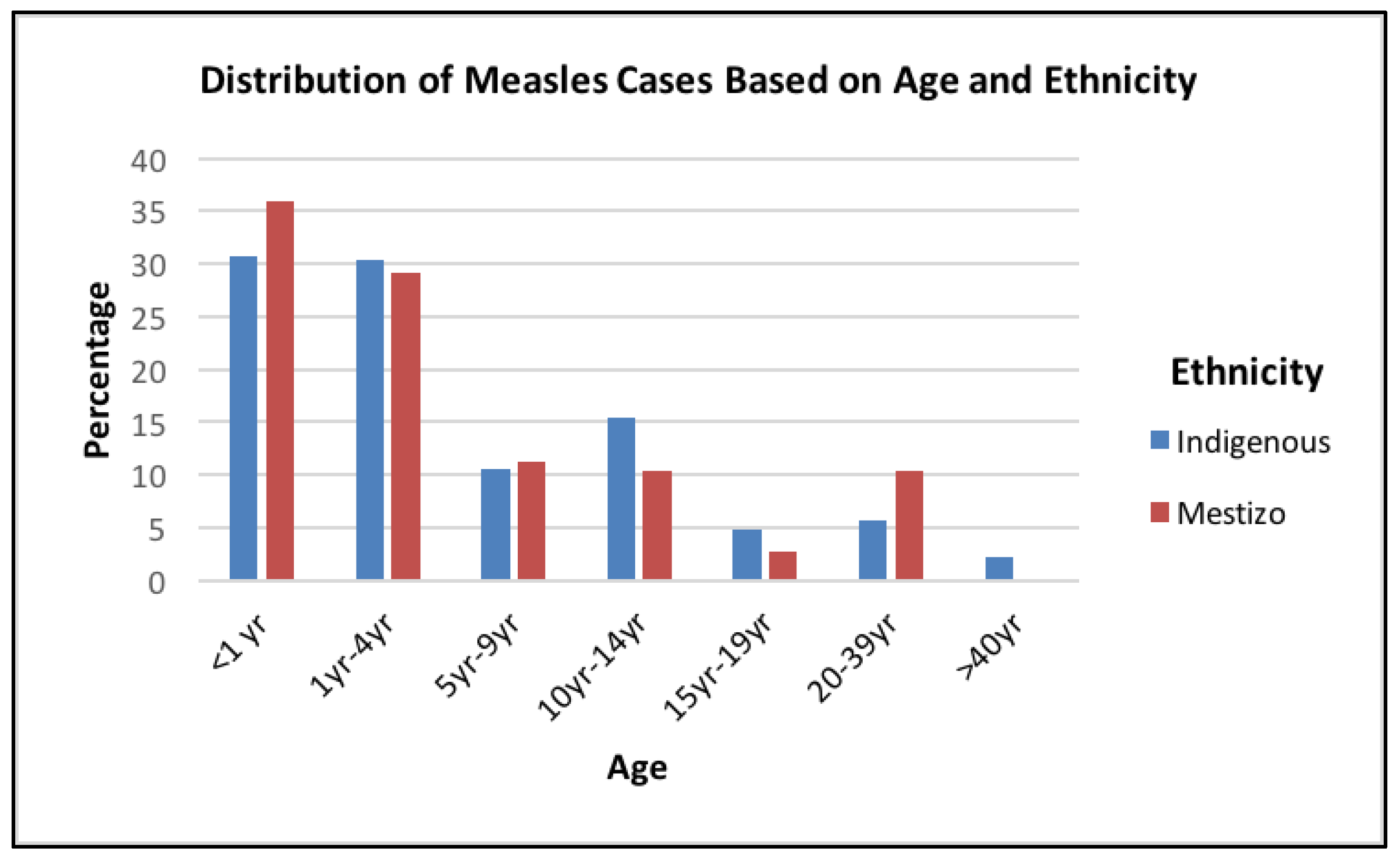
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Pan American Health Organization. Health in the Americas; Pan American Health Organization: Washington, DC, USA, 2007; Volume 1. [Google Scholar]
- National Immunization Program (Centers for Disease Control and Prevention). Epidemiology and Prevention of Vaccine-Preventable Diseases Course Textbook. Available online: https://www.cdc.gov/vaccines/pubs/pinkbook/downloads/meas.pdf (accessed on 31 May 2016).
- De Quadros, C.A.; Olive, J.M.; Hersh, B.S.; Strassburg, M.A.; Henderson, D.A.; Brandling-Bennett, D.; Alleyne, G.A. Measles elimination in the Americas. Evolving strategies. JAMA 1996, 275, 224–229. [Google Scholar] [CrossRef] [PubMed]
- Pan American Health Organization. XXIV Meeting of the Pan American Sanitary Conference, Washington, DC., USA, 27 September–1 October, 1993; Pan American Health Organization: Washington, DC, USA, 1993. [Google Scholar]
- Pan American Health Organization. Measles Eradication Field Guide; ProQuest: Washington, DC, USA, 1999; p. 28. [Google Scholar]
- Pan American Sanitary Bureau. Health in the Americas. In PAHO Scientific and Technical Publications, 1993–1996 ed.; Pan American Health Organization, Pan American Sanitary Bureau, Regional Office of the World Health Organization: Washington, DC, USA, 1998. [Google Scholar]
- Pan American Health Organization. Special program of vaccines and immunization. Proccedings of the Conclusions and Recommendations: XII Technical Advisory Group Meeting on Vaccine-Preventable Diseases, Guatemala, Guatemala, 8–12 September 1997. [Google Scholar]
- Pan American Health Organization. Health in the Americas, 2007 ed.; Pan American Health Organization: Washington, DC, USA, 2007; Volume 2. [Google Scholar]
- Centers for Disease Control and Prevention. Measles Cases and Outbreaks. Available online: http://www.cdc.gov/measles/cases-outbreaks.html (accessed on 31 May 2016).
- Pan American Health Organization. Division of vaccines and immunization. Proccedings of the Conclusions and Recommendations: XIII Technical Advisory Group Meeting on Vaccine-Preventable Diseases, Gatineau, QC, Canada, 12–16 April 1999; p. 5. [Google Scholar]
- Pan American Health Organization. Measles outbreak in an isolated community in Bolivia. EPI Newslett. 2000, 22, 1–3. [Google Scholar]
- Pan American Health Organization. Health in the Americas: Regional Outlook and Country Profiles, 2012 ed.; Pan American Health Organization: Washington, DC, USA, 2012. [Google Scholar]
- Sato, H.; Albrecht, P.; Reynolds, D.W.; Stagno, S.; Ennis, F.A. Transfer of measles, mumps, and rubella antibodies from mother to infant: Its effect on measles, mumps, and rubella immunization. Am. J. Dis. Child. 1979, 133, 1240–1243. [Google Scholar] [CrossRef] [PubMed]
- Rota, P.A.; Rota, J.S.; Redd, S.B.; Papania, M.J.; Bellini, W.J. Genetic analysis of measles viruses isolated in the United States between 1989 and 2001: Absence of an endemic genotype since 1994. J. Infect. Dis. 2004, 189, S160–S164. [Google Scholar] [CrossRef] [PubMed]
- De Quadros, C.A.; Izurieta, H.; Venczel, L.; Carrasco, P. Measles eradication in the Americas: Progress to date. J. Infect. Dis. 2004, 189, S227–S235. [Google Scholar] [CrossRef] [PubMed]
- Pan American Health Organization. Health in the Americas, 2002 ed.; Pan American Health Organization: Washington, DC, USA, 2002. [Google Scholar]
- Puertas, B.; Schlesser, M. Assessing community health among indigenous populations in ecuador with a participatory approach: Implications for health reform. J. Community Health 2001, 26, 133–147. [Google Scholar] [CrossRef] [PubMed]
- Sánchez, J. Inequality, Ethnicity and Social Disorder: The Ecuadorian Case; Third World Bank Conference on Inequality: Washington, DC, USA, 2006. [Google Scholar]
- UNICEF and WHO. Immunization Summary—A Statistical Reference Containing Data through 2010, 2012 ed.; Unicef: New York, NY, USA, 2012. [Google Scholar]
- Margheritis, A. “Todos somos migrantes” (we are all migrants): The paradoxes of the innovative state-led transnationalism in Ecuador. Int. Political Sociol. 2011, 5, 198–217. [Google Scholar] [CrossRef]
- Jokisch, B.D. Ecuador: From Mass Emigration to Return Migration? Available online: http://www.migrationpolicy.org/article/ecuador-mass-emigration-return-migration (accessed on 1 June 2016).
- Mankertz, A.; Mulders, M.N.; Shulga, S.; Kremer, J.R.; Brown, K.E.; Santibanez, S.; Muller, C.P.; Tikhonova, N.; Lipskaya, G.; Jankovic, D.; et al. Molecular genotyping and epidemiology of measles virus transmission in the World Health Organization European Region, 2007–2009. J. Infect. Dis. 2011, 204, S335–S342. [Google Scholar] [CrossRef] [PubMed]
- Freier, L.F. A Gateway to the Americas: Ecuador’s “Policy of Open Doors” as a Natural Experiment. Available online: http://paperroom.ipsa.org/papers/view/18248 (accessed on 29 May 2016).
- World Health Organization. Measles Outbreaks: REGIONS of the Americas, Europe and Africa. Available online: http://www.who.int/csr/don/2011_10_07/en/ (accessed on 29 May 2016).
- Pan American Health Organization. Paving the way for immunization. In Proceedings of the XX Technical Advisory Group Meeting on Vaccine-Preventable Diseases; Washington, DC, USA, 17–19 October 2012.
- Centers for Disease Control and Prevention. Increased Transmission and Outbreaks of Measles—European Region, 2011. Available online: https://www.cdc.gov/mmwr/preview/mmwrhtml/mm6047a1.htm (accessed on 31 May 2016).
- Andrus, J.K.; Vicari, A.; Tambini, G.; Periago, M.R. The global inter-relatedness of disease control. Lancet Infect. Dis. 2007, 7, 176. [Google Scholar] [CrossRef]
- Williams, C.J.; Schenkel, K.; Eckmanns, T.; Altmann, D.; Krause, G. FIFA World Cup 2006 in Germany: Enhanced surveillance improved timeliness and detection. Epidemiol. Infect. 2009, 137, 597–605. [Google Scholar] [CrossRef] [PubMed]
- Hasan, H. Measles Initiative Partners Gear up to Tackle Challenges Ahead; WHO: Geneva, Switzerland, 2011. [Google Scholar]
- 55th Directing Council of the 68th Session of the Regional Committee of WHO for the Americas. Available online: https://www.google.ch/url?sa=t&rct=j&q=&esrc=s&source=web&cd=2&ved=0ahUKEwjms-68jpnUAhXpIMAKHZ_TBSYQFggqMAE&url=http%3A%2F%2Firis.paho.org%2Fxmlui%2Fbitstream%2Fhandle%2F123456789%2F33696%2FCD55-DIV-12-e.pdf%3Fsequence%3D1%26isAllowed%3Dy&usg=AFQjCNHyUw5xiTuofDaWn0ZhajaN3d9KFQ&cad=rja (accessed on 31 May 2016).
| Vaccine Status | Indigenous | Mestizo | ||
|---|---|---|---|---|
| Percentage | Frequency | Percentage | Frequency | |
| Did not receive measles vaccine | 57.85% | 280 | 49.67% | 1375 |
| Received measles vaccine | 42.15% | 204 | 50.34% | 1393 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Le, N.K.; Mhaskar, R.; Hoare, I.; Espinel, M.; Fernanda Rivadeneira, M.; Malavade, S.; Izurieta, R. Reemergence of Measles in the Americas: The Genotype B3 2011–2012 Outbreak in Ecuador. Vaccines 2017, 5, 15. https://doi.org/10.3390/vaccines5020015
Le NK, Mhaskar R, Hoare I, Espinel M, Fernanda Rivadeneira M, Malavade S, Izurieta R. Reemergence of Measles in the Americas: The Genotype B3 2011–2012 Outbreak in Ecuador. Vaccines. 2017; 5(2):15. https://doi.org/10.3390/vaccines5020015
Chicago/Turabian StyleLe, Nicole K., Rahul Mhaskar, Ismael Hoare, Mauricio Espinel, María Fernanda Rivadeneira, Sharad Malavade, and Ricardo Izurieta. 2017. "Reemergence of Measles in the Americas: The Genotype B3 2011–2012 Outbreak in Ecuador" Vaccines 5, no. 2: 15. https://doi.org/10.3390/vaccines5020015
APA StyleLe, N. K., Mhaskar, R., Hoare, I., Espinel, M., Fernanda Rivadeneira, M., Malavade, S., & Izurieta, R. (2017). Reemergence of Measles in the Americas: The Genotype B3 2011–2012 Outbreak in Ecuador. Vaccines, 5(2), 15. https://doi.org/10.3390/vaccines5020015







