Transcriptomic Analysis of Immune Tolerance Induction in NOD Mice Following Oral Vaccination with GAD65-Lactococcus lactis
Abstract
1. Introduction
2. Materials and Methods
2.1. Ethics Statement
2.2. Animals, Experimental Design, and Sampling
2.3. ELISA
2.3.1. GAD65-Specific IgA and IgG Detection
2.3.2. Serum TGF-β Detection
2.4. Flow Cytometry Assay
2.5. Histology
2.6. RNA Extraction and Sequencing
2.7. Real-Time RT-PCR
2.8. Statistical Analyses
3. Results
3.1. Oral GAD65 L. lactis Alleviated Hyperglycemia and Diabetes
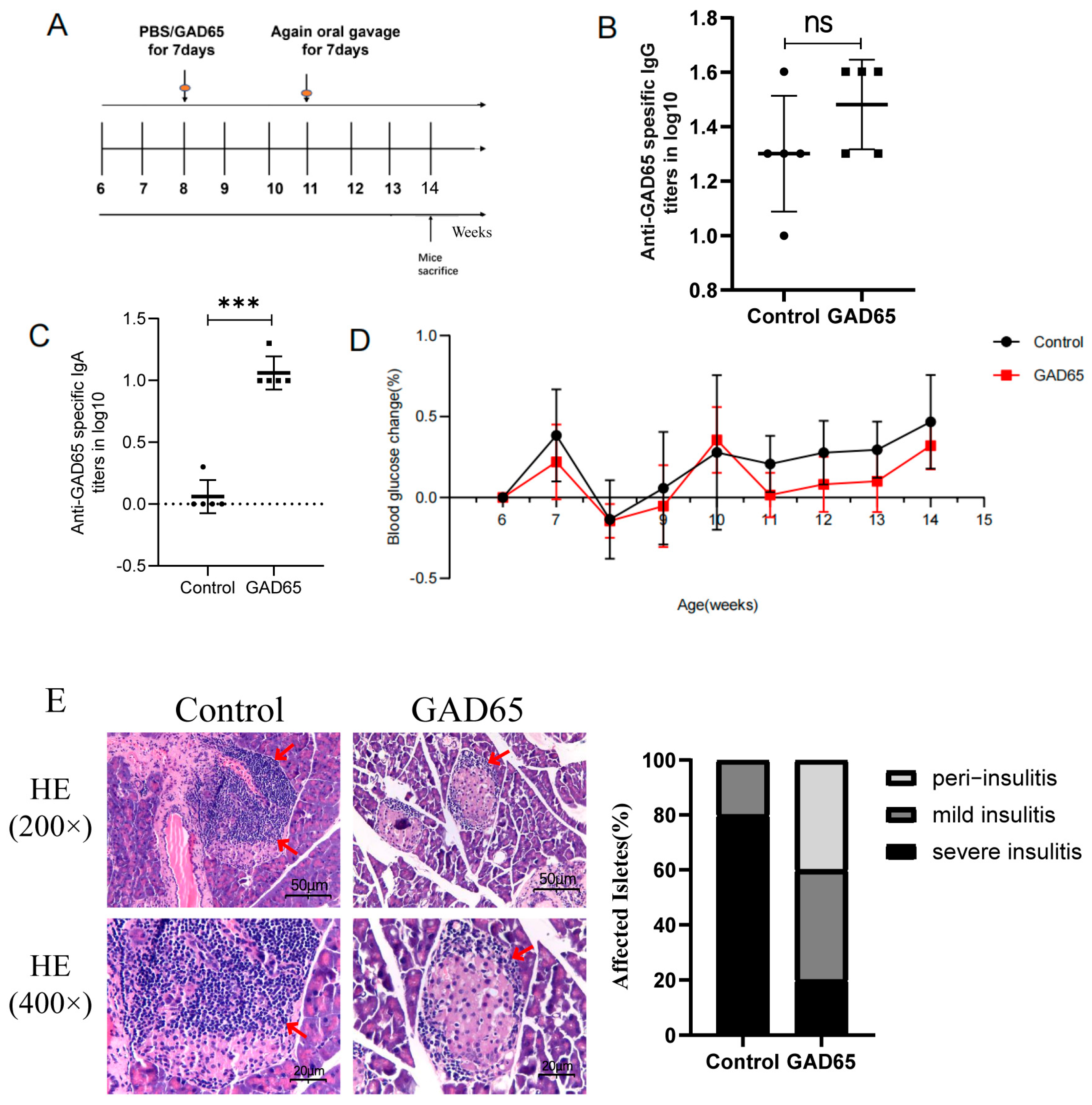
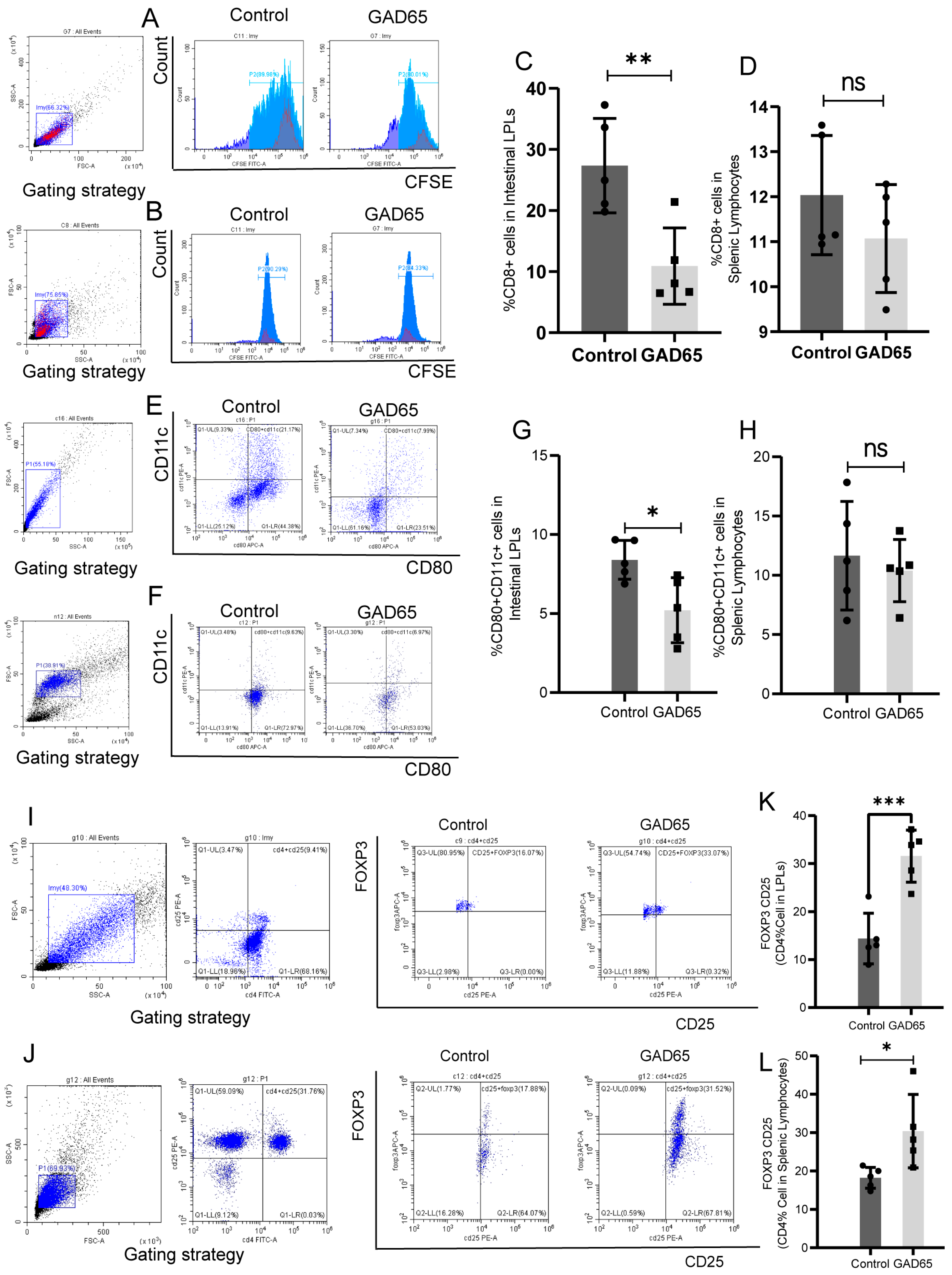
3.2. The Difference in Lymphocyte Responses in the Intestinal Lamina Propria and Spleen After Oral GAD65 L. lactis
3.3. Oral GAD65 L. lactis Induced Significant Change of Transcriptome in the Intestinal LPLs and Splenic Lymphocytes
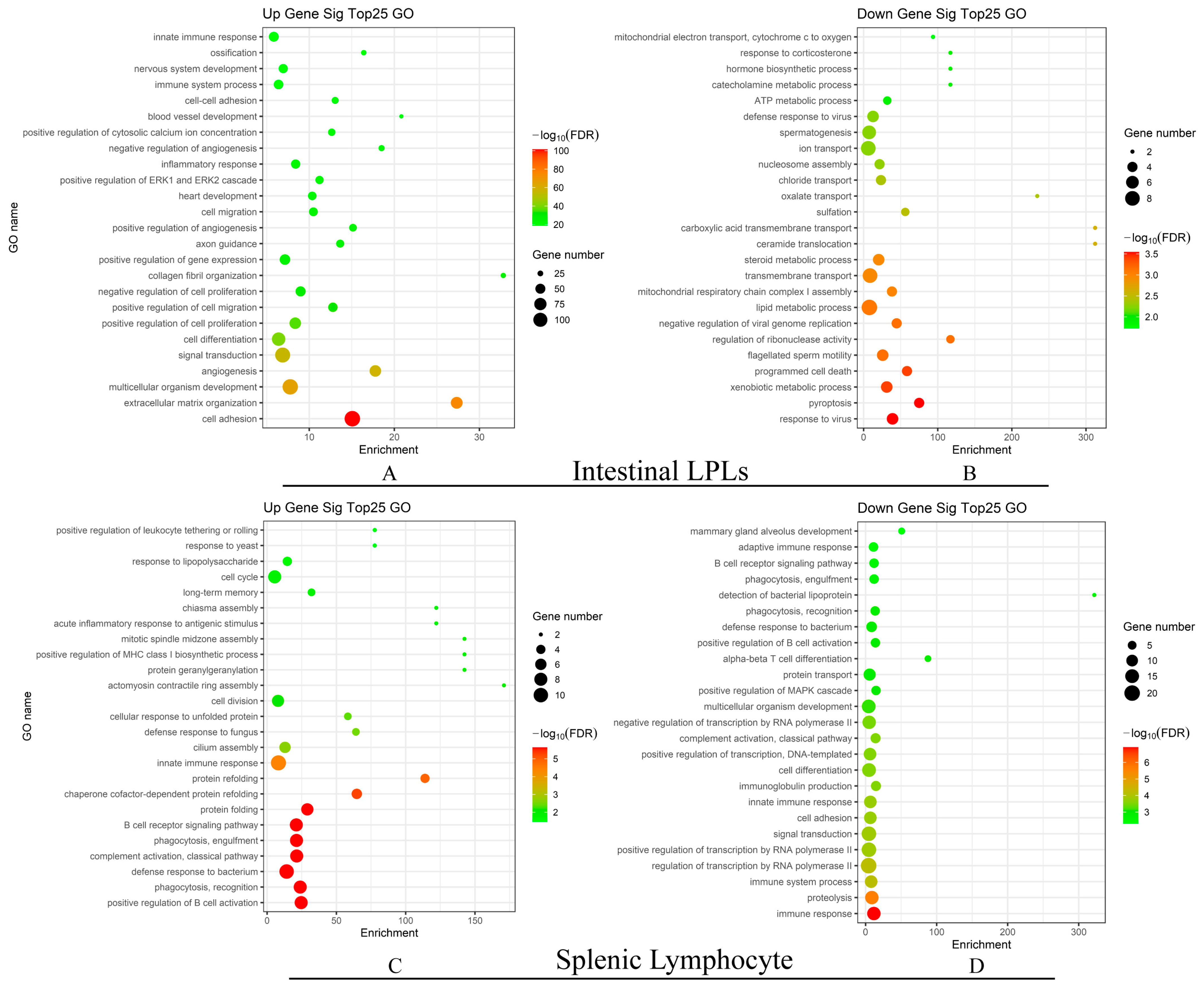
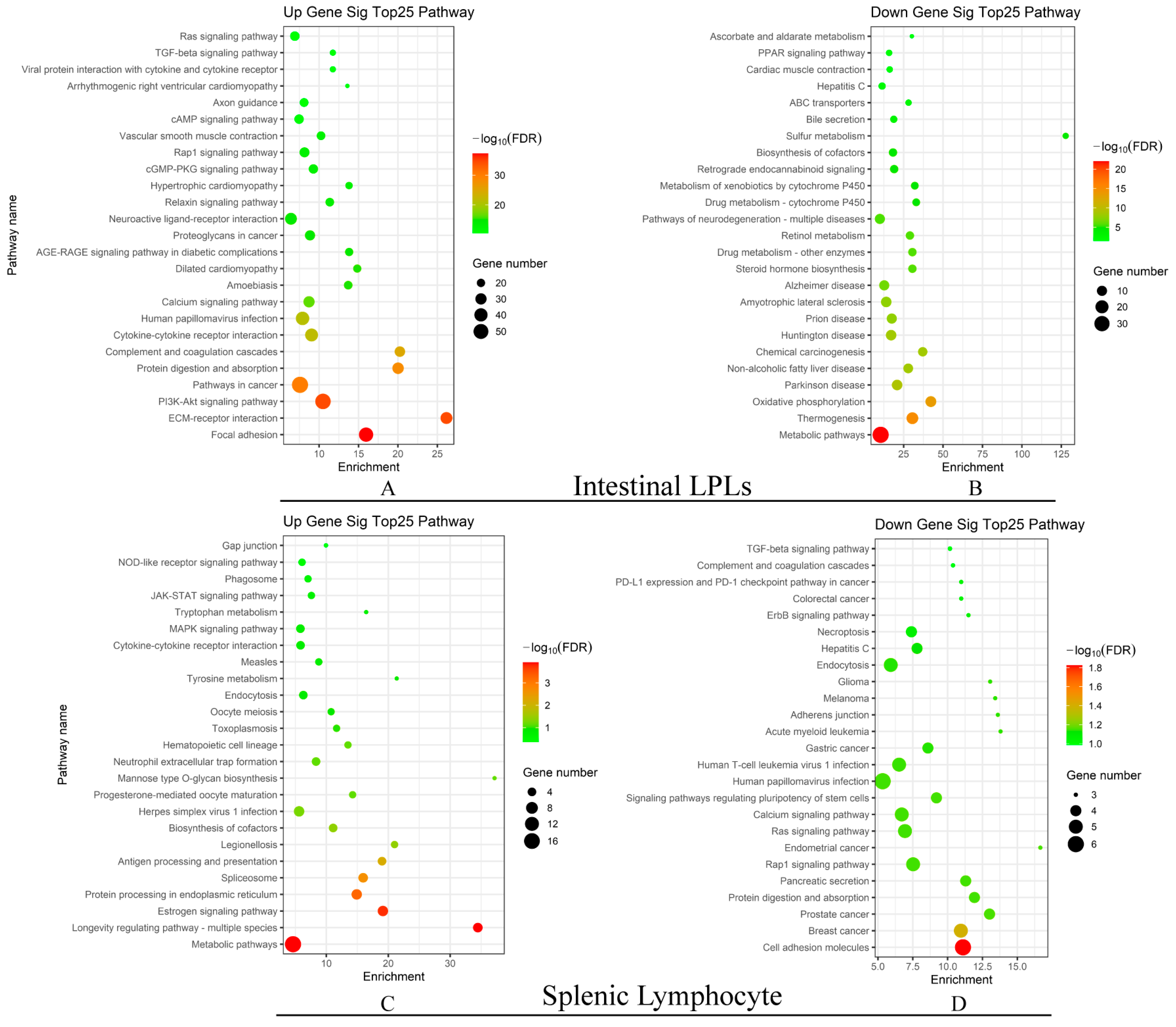
3.4. Oral GAD65 L. lactis Induced Different Biological Processes in the Intestinal LPLs and Splenic Lymphocytes
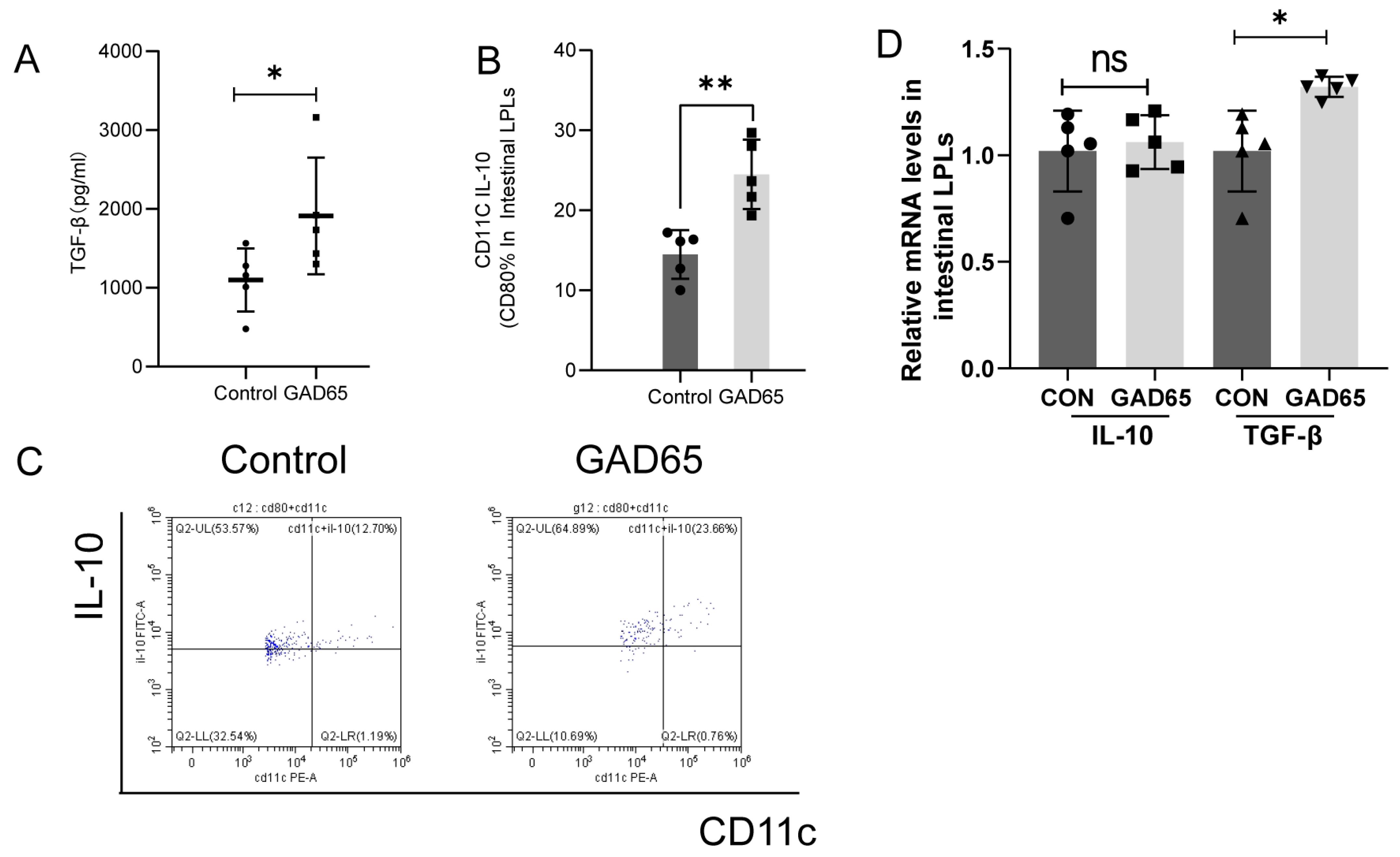
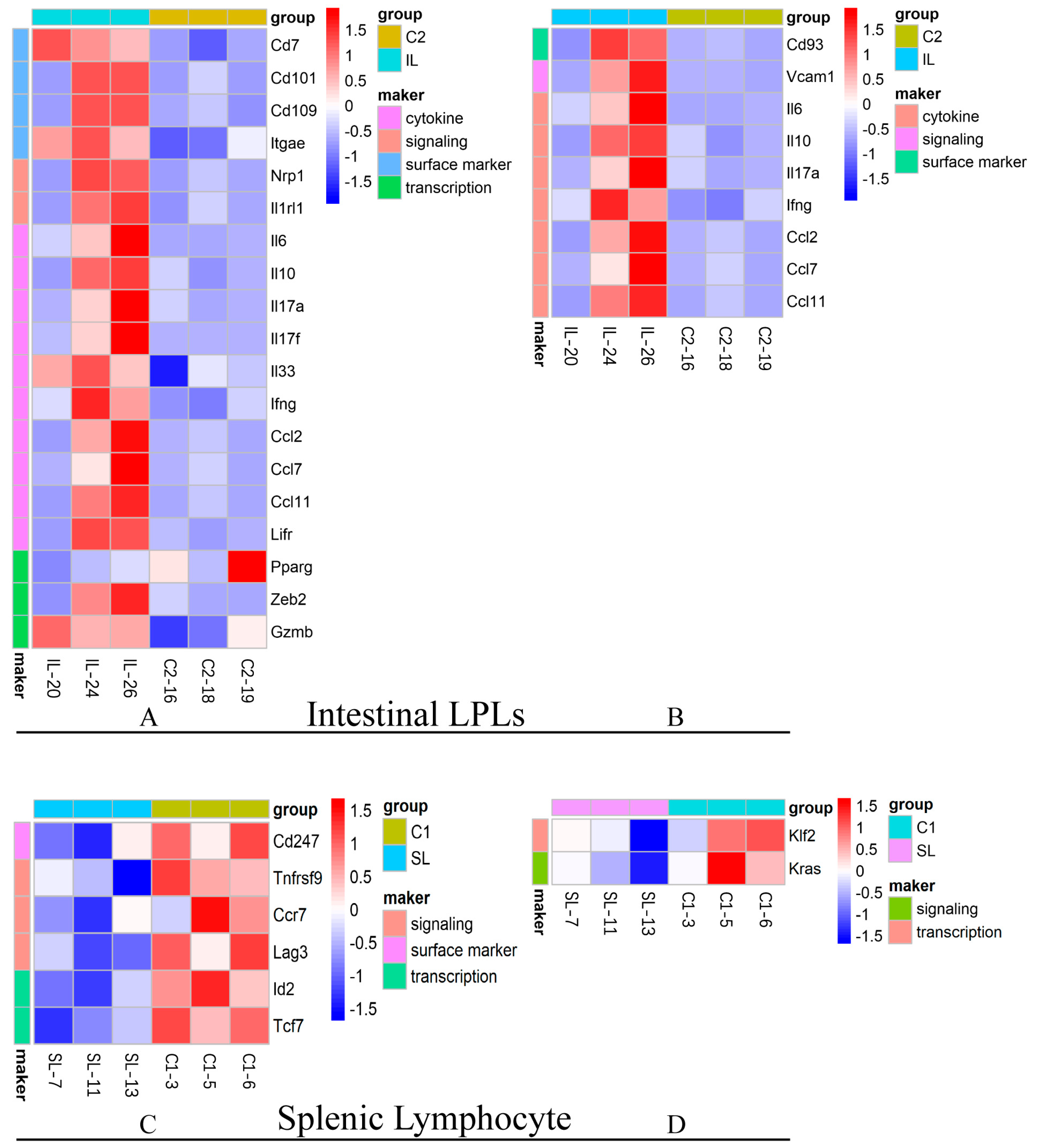
3.5. Oral GAD65 L. lactis Promoted Different T and B Cell Immune Responses in the Intestinal LPLs and Splenic Lymphocytes
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Conflicts of Interest
Abbreviations
| GAD65 | Glutamic Acid Decarboxylase 65 |
| LPLs | Intestinal Lamina Propria Lymphocytes |
| NOD | Non-Obese Diabetic |
| T1D | Type 1 Diabetes |
| DCs | Dendritic Cells |
| Tregs | Regulatory T Cells |
| ELISA | Enzyme-Linked Immunosorbent Assay |
| PBS | Phosphate-Buffered Saline |
| FBS | Fetal Bovine Serum |
| RIN | RNA Integrity Number |
| GO | Gene Ontology |
| KEGG | Kyoto Encyclopedia of Genes and Genomes |
| GSEA | Gene Set Enrichment Analysis |
| CFSE | Carboxyfluorescein Succinimidyl Ester |
| OD | Optical Density |
| HRP | Horseradish Peroxidase |
| TGF-β | Transforming Growth Factor-β |
| sIgA | Secretory Immunoglobulin A |
| GALT | Gut-Associated Lymphoid Tissue |
| AhR | Aryl Hydrocarbon Receptor |
| PP | Peyer’s Patches |
| mLN | Mesenteric Lymph Nodes |
| SL | Splenic Lymphocytes in GAD65 group |
| IL | Intestinal LPLs in GAD65 group |
| RVM | Random Variance Model |
| DSS | Dextran Sulfate Sodium |
References
- Joglekar, M.V.; Kaur, S.; Pociot, F.; Hardikar, A.A. Prediction of progression to type 1 diabetes with dynamic biomarkers and risk scores. Lancet Diabetes Endocrinol. 2024, 12, 483–492. [Google Scholar] [CrossRef]
- Carey, S.T.; Bridgeman, C.; Jewell, C.M. Biomaterial Strategies for Selective Immune Tolerance: Advances and Gaps. Adv. Sci. 2023, 10, e2205105. [Google Scholar] [CrossRef] [PubMed]
- Ludvigsson, J. GAD65: A prospective vaccine for treating Type 1 diabetes? Expert Opin. Biol. Ther. 2017, 17, 1033–1043. [Google Scholar] [CrossRef] [PubMed]
- Wherrett, D.K.; Bundy, B.; Becker, D.J.; DiMeglio, L.A.; Gitelman, S.E.; Goland, R.; Gottlieb, P.A.; Greenbaum, C.J.; Herold, K.C.; Marks, J.B.; et al. Antigen-based therapy with glutamic acid decarboxylase (GAD) vaccine in patients with recent-onset type 1 diabetes: A randomised double-blind trial. Lancet 2011, 378, 319–327. [Google Scholar] [CrossRef]
- Nowak, C.; Lind, M.; Sumnik, Z.; Pelikanova, T.; Nattero-Chavez, L.; Lundberg, E.; Rica, I.; Martínez-Brocca, M.A.; Ruiz de Adana, M.; Wahlberg, J.; et al. Intralymphatic GAD-Alum (Diamyd®) Improves Glycemic Control in Type 1 Diabetes With HLA DR3-DQ2. J. Clin. Endocrinol. Metab. 2022, 107, 2644–2651. [Google Scholar] [CrossRef]
- Rezende, R.M.; Weiner, H.L. History and mechanisms of oral tolerance. Semin. Immunol. 2017, 30, 3–11. [Google Scholar] [CrossRef] [PubMed]
- Voelker, R. Drug Approved to Aid Youths With Peanut Allergy. JAMA 2020, 323, 817. [Google Scholar] [CrossRef]
- Weiner, H.L. Current issues in the treatment of human diseases by mucosal tolerance. Ann. N. Y. Acad. Sci. 2004, 1029, 211–224. [Google Scholar] [CrossRef]
- Azizpour, M.; Hosseini, S.D.; Jafari, P.; Akbary, N. Lactococcus lactis: A New Strategy for Vaccination. Avicenna J. Med. Biotechnol. 2017, 9, 163–168. [Google Scholar]
- Cook, D.P.; Gysemans, C.; Mathieu, C. Lactococcus lactis As a Versatile Vehicle for Tolerogenic Immunotherapy. Front. Immunol. 2017, 8, 1961. [Google Scholar] [CrossRef]
- Craig, K.; Dai, X.; Li, A.; Lu, M.; Xue, M.; Rosas, L.; Gao, T.Z.; Niehaus, A.; Jennings, R.; Li, J. A Lactic Acid Bacteria (LAB)-Based Vaccine Candidate for Human Norovirus. Viruses 2019, 11, 213. [Google Scholar] [CrossRef]
- Levit, R.; Cortes-Perez, N.G.; de Moreno de Leblanc, A.; Loiseau, J.; Aucouturier, A.; Langella, P.; LeBlanc, J.G.; Bermúdez-Humarán, L.G. Use of genetically modified lactic acid bacteria and bifidobacteria as live delivery vectors for human and animal health. Gut Microbes 2022, 14, 2110821. [Google Scholar] [CrossRef]
- Mobergslien, A.; Vasovic, V.; Mathiesen, G.; Fredriksen, L.; Westby, P.; Eijsink, V.G.; Peng, Q.; Sioud, M. Recombinant Lactobacillus plantarum induces immune responses to cancer testis antigen NY-ESO-1 and maturation of dendritic cells. Hum. Vaccin. Immunother. 2015, 11, 2664–2673. [Google Scholar] [CrossRef] [PubMed]
- Saraiva, T.D.; Morais, K.; Pereira, V.B.; de Azevedo, M.; Rocha, C.S.; Prosperi, C.C.; Gomes-Santos, A.C.; Bermudez-Humaran, L.; Faria, A.M.; Blottiere, H.M.; et al. Milk fermented with a 15-lipoxygenase-1-producing Lactococcus lactis alleviates symptoms of colitis in a murine model. Curr. Pharm. Biotechnol. 2015, 16, 424–429. [Google Scholar] [CrossRef] [PubMed]
- Robert, S.; Gysemans, C.; Takiishi, T.; Korf, H.; Spagnuolo, I.; Sebastiani, G.; Van Huynegem, K.; Steidler, L.; Caluwaerts, S.; Demetter, P.; et al. Oral delivery of glutamic acid decarboxylase (GAD)-65 and IL10 by Lactococcus lactis reverses diabetes in recent-onset NOD mice. Diabetes 2014, 63, 2876–2887. [Google Scholar] [CrossRef] [PubMed]
- Mathieu, C.; Wiedeman, A.; Cerosaletti, K.; Long, S.A.; Serti, E.; Cooney, L.; Vermeiren, J.; Caluwaerts, S.; Van Huynegem, K.; Steidler, L.; et al. A first-in-human, open-label Phase 1b and a randomised, double-blind Phase 2a clinical trial in recent-onset type 1 diabetes with AG019 as monotherapy and in combination with teplizumab. Diabetologia 2024, 67, 27–41. [Google Scholar] [CrossRef]
- Lee, J.S.; Oka, K.; Obara, M.; Nishimukai, M.; Yoo, Y.C.; Yamada, K.; Tsukahara, T.; Nakayama, K.; Hara, H.; Ishizuka, S. Improved isolation methods for mucosal leukocytes from small and large intestines in rats. Biosci. Biotechnol. Biochem. 2009, 73, 1732–1740. [Google Scholar] [CrossRef]
- Green, E.A.; Eynon, E.E.; Flavell, R.A. Local expression of TNFalpha in neonatal NOD mice promotes diabetes by enhancing presentation of islet antigens. Immunity 1998, 9, 733–743. [Google Scholar] [CrossRef]
- Sakaguchi, S.; Mikami, N.; Wing, J.B.; Tanaka, A.; Ichiyama, K.; Ohkura, N. Regulatory T Cells and Human Disease. Annu. Rev. Immunol. 2020, 38, 541–566. [Google Scholar] [CrossRef]
- Gouirand, V.; Habrylo, I.; Rosenblum, M.D. Regulatory T Cells and Inflammatory Mediators in Autoimmune Disease. J. Investig. Dermatol. 2022, 142 Pt B, 774–780. [Google Scholar] [CrossRef]
- Wang, X.; Sherman, A.; Liao, G.; Leong, K.W.; Daniell, H.; Terhorst, C.; Herzog, R.W. Mechanism of oral tolerance induction to therapeutic proteins. Adv. Drug Deliv. Rev. 2013, 65, 759–773. [Google Scholar] [CrossRef]
- Weiner, H.L.; da Cunha, A.P.; Quintana, F.; Wu, H. Oral tolerance. Immunol. Rev. 2011, 241, 241–259. [Google Scholar] [CrossRef]
- Mao, R.F.; Chen, Y.Y.; Zhang, J.; Chang, X.; Wang, Y.F. Type 1 diabetes mellitus and its oral tolerance therapy. World J. Diabetes 2020, 11, 400–415. [Google Scholar] [CrossRef]
- Mayer, L.; Shao, L. Therapeutic potential of oral tolerance. Nat. Rev. Immunol. 2004, 4, 407–419. [Google Scholar] [CrossRef] [PubMed]
- Faria, A.M.; Weiner, H.L. Oral tolerance: Therapeutic implications for autoimmune diseases. Clin. Dev. Immunol. 2006, 13, 143–157. [Google Scholar] [CrossRef]
- Ramirez, J.E.V.; Sharpe, L.A.; Peppas, N.A. Current state and challenges in developing oral vaccines. Adv. Drug Deliv. Rev. 2017, 114, 116–131. [Google Scholar] [CrossRef] [PubMed]
- Kwong, K.W.; Xin, Y.; Lai, N.C.; Sung, J.C.; Wu, K.C.; Hamied, Y.K.; Sze, E.T.; Lam, D.M. Oral Vaccines: A Better Future of Immunization. Vaccines 2023, 11, 1232. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.J.; Davidson, L.; Eisenbarth, G.; Weiner, H.L. Suppression of diabetes in nonobese diabetic mice by oral administration of porcine insulin. Proc. Natl. Acad. Sci. USA 1991, 88, 10252–10256. [Google Scholar] [CrossRef]
- Pozzilli, P.; Pitocco, D.; Visalli, N.; Cavallo, M.G.; Buzzetti, R.; Crinò, A.; Spera, S.; Suraci, C.; Multari, G.; Cervoni, M.; et al. No effect of oral insulin on residual beta-cell function in recent-onset type I diabetes (the IMDIAB VII). IMDIAB Group. Diabetologia 2000, 43, 1000–1004. [Google Scholar] [CrossRef]
- Chaillous, L.; Lefèvre, H.; Thivolet, C.; Boitard, C.; Lahlou, N.; Atlan-Gepner, C.; Bouhanick, B.; Mogenet, A.; Nicolino, M.; Carel, J.C.; et al. Oral insulin administration and residual beta-cell function in recent-onset type 1 diabetes: A multicentre randomised controlled trial. Diabète Insuline Orale group. Lancet 2000, 356, 545–549. [Google Scholar] [CrossRef]
- Mege, J.L. Dendritic cell subtypes: A new way to study host-pathogen interaction. Virulence 2016, 7, 5–6. [Google Scholar] [CrossRef] [PubMed]
- Ergun-Longmire, B.; Marker, J.; Zeidler, A.; Rapaport, R.; Raskin, P.; Bode, B.; Schatz, D.; Vargas, A.; Rogers, D.; Schwartz, S.; et al. Oral insulin therapy to prevent progression of immune-mediated (type 1) diabetes. Ann. N. Y. Acad. Sci. 2004, 1029, 260–277. [Google Scholar] [CrossRef]
- Bergerot, I.; Arreaza, G.A.; Cameron, M.J.; Burdick, M.D.; Strieter, R.M.; Chensue, S.W.; Chakrabarti, S.; Delovitch, T.L. Insulin B-chain reactive CD4+ regulatory T-cells induced by oral insulin treatment protect from type 1 diabetes by blocking the cytokine secretion and pancreatic infiltration of diabetogenic effector T-cells. Diabetes 1999, 48, 1720–1729. [Google Scholar] [CrossRef]
- Reveneau, N.; Geoffroy, M.C.; Locht, C.; Chagnaud, P.; Mercenier, A. Comparison of the immune responses induced by local immunizations with recombinant Lactobacillus plantarum producing tetanus toxin fragment C in different cellular locations. Vaccine 2002, 20, 1769–1777. [Google Scholar] [CrossRef]
- Oh, S.H.; Kim, S.H.; Jeon, J.H.; Kim, E.B.; Lee, N.K.; Beck, S.; Choi, Y.J.; Kang, S.K. Cytoplasmic expression of a model antigen with M Cell-Targeting moiety in lactic acid bacteria and implication of the mechanism as a mucosal vaccine via oral route. Vaccine 2021, 39, 4072–4081. [Google Scholar] [CrossRef] [PubMed]
- Heystek, H.C.; Moulon, C.; Woltman, A.M.; Garonne, P.; van Kooten, C. Human immature dendritic cells efficiently bind and take up secretory IgA without the induction of maturation. J. Immunol. 2002, 168, 102–107. [Google Scholar] [CrossRef]
- Ng, S.T.H.; Price, M.J.; Richardson, N.; Nawaf, M.; Copland, A.; Streeter, H.B.; Narendran, P.; Wraith, D.C. Preclinical Development of a Tolerogenic Peptide From Glutamate Decarboxylase as a Candidate for Antigen-Specific Immunotherapy in Type 1 Diabetes. Diabetes 2025, 74, 384–397. [Google Scholar] [CrossRef]
- Reeves, P.L.S.; Rudraraju, R.; Wong, F.S.; Hamilton-Williams, E.E.; Steptoe, R.J. Antigen presenting cell-targeted proinsulin expression converts insulin-specific CD8(+) T-cell priming to tolerance in autoimmune-prone NOD mice. Eur. J. Immunol. 2017, 47, 1550–1561. [Google Scholar] [CrossRef]
- Worbs, T.; Bode, U.; Yan, S.; Hoffmann, M.W.; Hintzen, G.; Bernhardt, G.; Förster, R.; Pabst, O. Oral tolerance originates in the intestinal immune system and relies on antigen carriage by dendritic cells. J. Exp. Med. 2006, 203, 519–527. [Google Scholar] [CrossRef] [PubMed]
- Weiberg, D.; Basic, M.; Smoczek, M.; Bode, U.; Bornemann, M.; Buettner, M. Participation of the spleen in the IgA immune response in the gut. PLoS ONE 2018, 13, e0205247. [Google Scholar] [CrossRef]
- Boehm, T.; Hess, I.; Swann, J.B. Evolution of lymphoid tissues. Trends Immunol. 2012, 33, 315–321. [Google Scholar] [CrossRef]
- Neely, H.R.; Flajnik, M.F. Emergence and Evolution of Secondary Lymphoid Organs. Annu. Rev. Cell Dev. Biol. 2016, 32, 693–711. [Google Scholar] [CrossRef]
- Dillon, A.; Lo, D.D. M Cells: Intelligent Engineering of Mucosal Immune Surveillance. Front. Immunol. 2019, 10, 1499. [Google Scholar] [CrossRef] [PubMed]
- Wells, J.M.; Mercenier, A. Mucosal delivery of therapeutic and prophylactic molecules using lactic acid bacteria. Nat. Rev. Microbiol. 2008, 6, 349–362. [Google Scholar] [CrossRef]
- Takiishi, T.; Cook, D.P.; Korf, H.; Sebastiani, G.; Mancarella, F.; Cunha, J.P.; Wasserfall, C.; Casares, N.; Lasarte, J.J.; Steidler, L.; et al. Reversal of Diabetes in NOD Mice by Clinical-Grade Proinsulin and IL-10-Secreting Lactococcus lactis in Combination With Low-Dose Anti-CD3 Depends on the Induction of Foxp3-Positive T Cells. Diabetes 2017, 66, 448–459. [Google Scholar] [CrossRef] [PubMed]
- Gmoshinski, I.V.; Shipelin, V.A.; Trusov, N.V.; Apryatin, S.A.; Mzhelskaya, K.V.; Shumakova, A.A.; Timonin, A.N.; Riger, N.A.; Nikityuk, D.B. Effects of Tyrosine and Tryptophan Supplements on the Vital Indicators in Mice Differently Prone to Diet-Induced Obesity. Int. J. Mol. Sci. 2021, 22, 5956. [Google Scholar] [CrossRef]
- Shipelin, V.A.; Trusov, N.V.; Apryatin, S.A.; Shumakova, A.A.; Balakina, A.S.; Riger, N.A.; Gmoshinski, I.V.; Nikityuk, D.B. Effects of Tyrosine and Tryptophan in Rats with Diet-Induced Obesity. Int. J. Mol. Sci. 2021, 22, 2429. [Google Scholar] [CrossRef] [PubMed]
- Cervantes-Barragan, L.; Colonna, M. AHR signaling in the development and function of intestinal immune cells and beyond. Semin. Immunopathol. 2018, 40, 371–377. [Google Scholar] [CrossRef]
- Ehrlich, A.K.; Pennington, J.M.; Bisson, W.H.; Kolluri, S.K.; Kerkvliet, N.I. TCDD, FICZ, and Other High Affinity AhR Ligands Dose-Dependently Determine the Fate of CD4+ T Cell Differentiation. Toxicol. Sci. 2018, 161, 310–320. [Google Scholar] [CrossRef]
- Boule, L.A.; Burke, C.G.; Jin, G.B.; Lawrence, B.P. Aryl hydrocarbon receptor signaling modulates antiviral immune responses: Ligand metabolism rather than chemical source is the stronger predictor of outcome. Sci. Rep. 2018, 8, 1826. [Google Scholar] [CrossRef]
- Li, X.; Wang, Q.; Ding, L.; Wang, Y.X.; Zhao, Z.D.; Mao, N.; Wu, C.T.; Wang, H.; Zhu, H.; Ning, S.B. Intercellular adhesion molecule-1 enhances the therapeutic effects of MSCs in a dextran sulfate sodium-induced colitis models by promoting MSCs homing to murine colons and spleens. Stem Cell Res. Ther. 2019, 10, 267. [Google Scholar] [CrossRef] [PubMed]
- Bénard, A.; Mittelstädt, A.; Klösch, B.; Glanz, K.; Müller, J.; Schoen, J.; Nüse, B.; Brunner, M.; Naschberger, E.; Stürzl, M.; et al. IL-3 orchestrates ulcerative colitis pathogenesis by controlling the development and the recruitment of splenic reservoir neutrophils. Cell Rep. 2023, 42, 112637. [Google Scholar] [CrossRef] [PubMed]
- Luerce, T.D.; Gomes-Santos, A.C.; Rocha, C.S.; Moreira, T.G.; Cruz, D.N.; Lemos, L.; Sousa, A.L.; Pereira, V.B.; de Azevedo, M.; Moraes, K.; et al. Anti-inflammatory effects of Lactococcus lactis NCDO 2118 during the remission period of chemically induced colitis. Gut Pathog. 2014, 6, 33. [Google Scholar] [CrossRef]
- Liu, Y.; Cai, Y.; Liu, L.; Wu, Y.; Xiong, X. Crucial biological functions of CCL7 in cancer. PeerJ 2018, 6, e4928. [Google Scholar] [CrossRef]
- Luo, Z.; Lin, Y.; Meng, Y.; Li, M.; Ren, H.; Shi, H.; Cheng, Q.; Wei, T. Spleen-Targeted mRNA Vaccine Doped with Manganese Adjuvant for Robust Anticancer Immunity In Vivo. ACS Nano 2024, 18, 30701–30715. [Google Scholar] [CrossRef]
- Yue, Y.; Xu, J.; Li, Y.; Cheng, K.; Feng, Q.; Ma, X.; Ma, N.; Zhang, T.; Wang, X.; Zhao, X.; et al. Antigen-bearing outer membrane vesicles as tumour vaccines produced in situ by ingested genetically engineered bacteria. Nat. Biomed. Eng. 2022, 6, 898–909. [Google Scholar] [CrossRef] [PubMed]

| Cell | Comparison | Upregulated | Downregulated | Total |
|---|---|---|---|---|
| Splenic lymphocytes | lncRNA Vaccinated vs. Control | 467 | 371 | 838 |
| miRNA Vaccinated vs. Control | 7 | 5 | 12 | |
| mRNA Vaccinated vs. Control | 176 | 233 | 409 | |
| intestinal LPLs | lncRNA Vaccinated vs. Control | 600 | 357 | 957 |
| miRNA Vaccinated vs. Control | 46 | 17 | 63 | |
| mRNA Vaccinated vs. Control | 1055 | 162 | 1217 |
| Upregulated | |||||||
|---|---|---|---|---|---|---|---|
| Tissue | GO | GO Pathway | No. of Difference Gene | Tissue | GO | GO Pathway | No. of Difference Gene |
| spleen | GO:0050871 | positive regulation of B cell activation | 8 | intestinal tract | GO:0007155 | cell adhesion | 124 |
| GO:0006910 | phagocytosis, recognition | 8 | GO:0030198 | extracellular matrix organization | 69 | ||
| GO:0042742 | defense response to bacterium | 10 | GO:0007275 | multicellular organism development | 122 | ||
| GO:0006958 | complement activation, classical pathway | 8 | GO:0001525 | angiogenesis | 66 | ||
| GO:0006911 | phagocytosis, engulfment | 8 | GO:0007165 | signal transduction | 115 | ||
| Downregulated | |||||||
| spleen | GO:0006955 | immune response | 14 | intestinal tract | GO:0009615 | response to virus | 5 |
| GO:0006508 | proteolysis | 14 | GO:0070269 | pyroptosis | 4 | ||
| GO:0002376 | immune system process | 12 | GO:0006805 | xenobiotic metabolic process | 5 | ||
| GO:0006357 | regulation of transcription by RNA polymerase II | 20 | GO:0012501 | programmed cell death | 4 | ||
| GO:0045944 | positive regulation of transcription by RNA polymerase II | 17 | GO:0030317 | flagellated sperm motility | 5 | ||
| Upregulated | |||||||
|---|---|---|---|---|---|---|---|
| Tissue | KEGG | KEGG Pathway | No. of Difference Gene | Tissue | KEGG | KEGG Pathway | No. of Difference Gene |
| spleen | ko1100 | Metabolic pathways | 17 | intestinal tract | ko04510 | Focal adhesion | 46 |
| ko 04213 | Longevity regulating pathway—multiple species | 5 | ko04512 | ECM-receptor interaction | 33 | ||
| ko 04915 | Estrogen signaling pathway | 6 | ko04151 | PI3K-Akt signaling pathway | 54 | ||
| ko 04141 | Protein processing in the endoplasmic reticulum | 6 | ko05200 | Pathways in cancer | 59 | ||
| ko03040 | Spliceosome | 5 | ko04974 | Protein digestion and absorption | 31 | ||
| Downregulated | |||||||
| spleen | ko04514 | Cell adhesion molecules | 6 | intestinal tract | ko01100 | Metabolic pathways | 35 |
| ko05224 | Breast cancer | 5 | ko04714 | Thermogenesis | 15 | ||
| ko5215 | Prostate cancer | 4 | ko00190 | Oxidative phosphorylation | 12 | ||
| ko04974 | Protein digestion and absorption | 4 | ko05012 | Parkinson disease | 11 | ||
| ko04972 | Pancreatic secretion | 4 | ko04932 | Non-alcoholic fatty liver disease | 9 | ||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xie, M.; Ma, C.; Wang, X.; Li, T.; Zhang, S.; Shi, J.; Sun, J.; Hu, Y. Transcriptomic Analysis of Immune Tolerance Induction in NOD Mice Following Oral Vaccination with GAD65-Lactococcus lactis. Vaccines 2025, 13, 927. https://doi.org/10.3390/vaccines13090927
Xie M, Ma C, Wang X, Li T, Zhang S, Shi J, Sun J, Hu Y. Transcriptomic Analysis of Immune Tolerance Induction in NOD Mice Following Oral Vaccination with GAD65-Lactococcus lactis. Vaccines. 2025; 13(9):927. https://doi.org/10.3390/vaccines13090927
Chicago/Turabian StyleXie, Mengxin, Chunli Ma, Xinyi Wang, Tengjiao Li, Shihan Zhang, Jiandong Shi, Jing Sun, and Yunzhang Hu. 2025. "Transcriptomic Analysis of Immune Tolerance Induction in NOD Mice Following Oral Vaccination with GAD65-Lactococcus lactis" Vaccines 13, no. 9: 927. https://doi.org/10.3390/vaccines13090927
APA StyleXie, M., Ma, C., Wang, X., Li, T., Zhang, S., Shi, J., Sun, J., & Hu, Y. (2025). Transcriptomic Analysis of Immune Tolerance Induction in NOD Mice Following Oral Vaccination with GAD65-Lactococcus lactis. Vaccines, 13(9), 927. https://doi.org/10.3390/vaccines13090927





