Influenza Vaccination Appropriateness: Insights from the Local Health Unit of Catania During the 2023/2024 and 2024/2025 Seasons
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Study Variables
2.3. Statistical Analysis
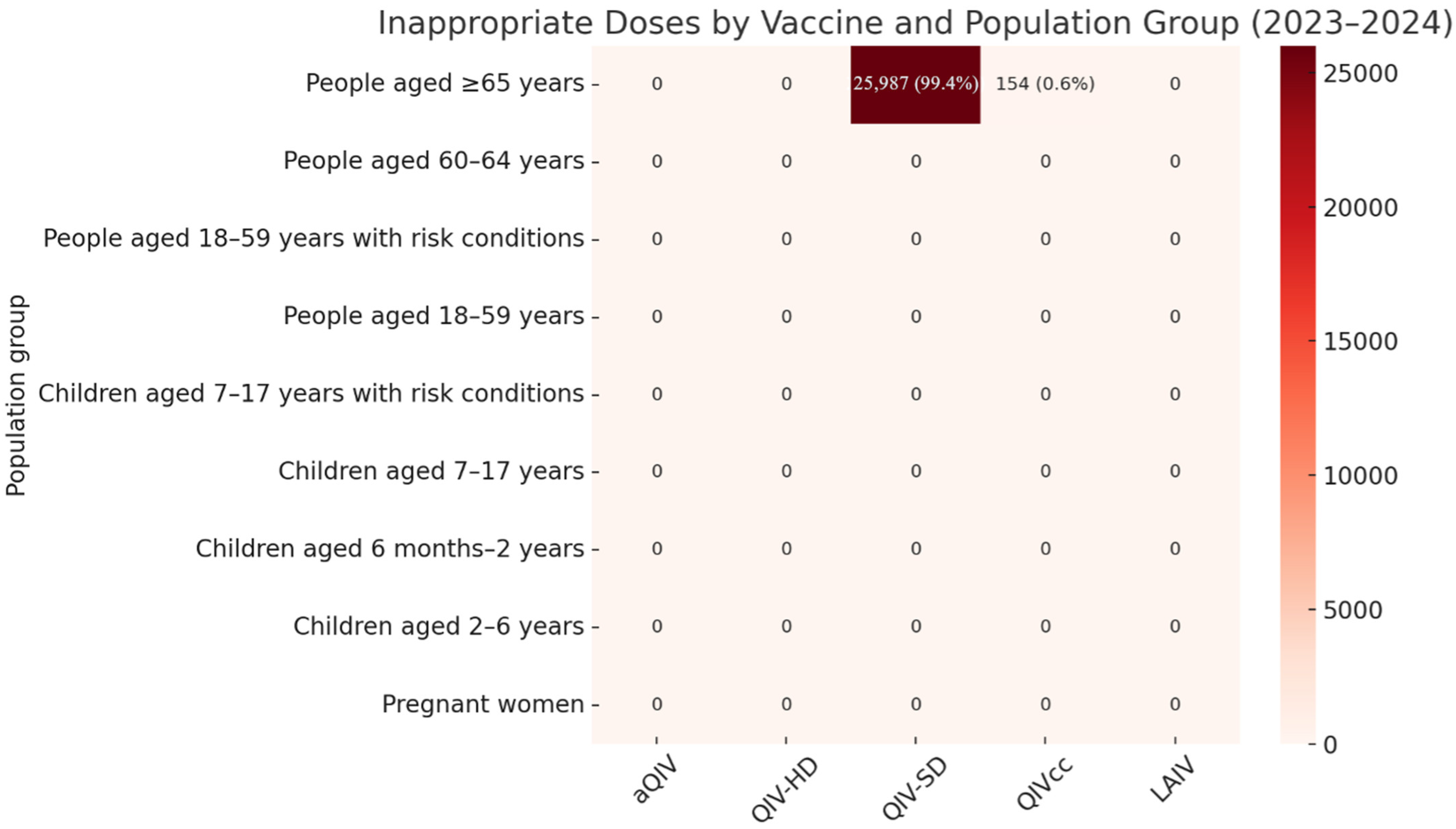
3. Results
3.1. 2023–2024 Campaign
3.2. 2024–2025 Campaign
4. Discussion
4.1. Older Adults (≥65 Years)
4.2. Population Aged 60–64 Years
4.3. Adults (18–59 Years) with and Without Risk Conditions
4.4. Pediatric Population and Pregnant/Postpartum Women
5. Study Limitations
6. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Abbreviations
References
- Javanian, M.; Barary, M.; Ghebrehewet, S.; Koppolu, V.; Vasigala, V.; Ebrahimpour, S. A Brief Review of Influenza Virus Infection. J. Med. Virol. 2021, 93, 4638–4646. [Google Scholar] [CrossRef]
- Nypaver, C.; Dehlinger, C.; Carter, C. Influenza and Influenza Vaccine: A Review. J. Midwifery Womens Health 2021, 66, 45–53. [Google Scholar] [CrossRef]
- Ryu, S.; Cowling, B. Human Influenza Epidemiology. Cold Spring Harb. Perspect. Med. 2021, 11, a038356. [Google Scholar] [CrossRef] [PubMed]
- Lampejo, T. Influenza and Antiviral Resistance: An Overview. Eur. J. Clin. Microbiol. Infect. Dis. 2020, 39, 1201–1208. [Google Scholar] [CrossRef]
- Kim, Y.-H.; Hong, K.-J.; Kim, H.; Nam, J.-H. Influenza Vaccines: Past, Present, and Future. Rev. Med. Virol. 2022, 32, e2243. [Google Scholar] [CrossRef]
- Li, J.; Chen, Y.; Wang, X.; Yu, H. Influenza-Associated Disease Burden in Mainland China: A Systematic Review and Meta-Analysis. Sci. Rep. 2021, 11, 2886. [Google Scholar] [CrossRef]
- Kumari, R.; Sharma, S.D.; Kumar, A.; Ende, Z.; Mishina, M.; Wang, Y.; Falls, Z.; Samudrala, R.; Pohl, J.; Knight, P.R.; et al. Antiviral Approaches against Influenza Virus. Clin. Microbiol. Rev. 2023, 36, e00040-22. [Google Scholar] [CrossRef]
- Smyk, J.; Szydłowska, N.; Szulc, W.; Majewska, A. Evolution of Influenza Viruses—Drug Resistance, Treatment Options, and Prospects. Int. J. Mol. Sci. 2022, 23, 12244. [Google Scholar] [CrossRef]
- Cheng, Y.; Cao, X.; Cao, Z.; Xu, C.; Sun, L.; Gao, Y.; Wang, Y.; Li, S.; Wu, C.; Li, X.; et al. Effects of Influenza Vaccination on the Risk of Cardiovascular and Respiratory Diseases and All-Cause Mortality. Ageing Res. Rev. 2020, 62, 101124. [Google Scholar] [CrossRef] [PubMed]
- Nuwarda, R.F.; Alharbi, A.A.; Kayser, V. An Overview of Influenza Viruses and Vaccines. Vaccines 2021, 9, 1032. [Google Scholar] [CrossRef] [PubMed]
- Mangas-Moro, A.; Zamarrón, E.; Carpio, C.; Álvarez-Sala, R.; Arribas-López, J.R.; Prados, C. Influenza Vaccination Mitigates Severe Complications in Hospitalized Patients: A Ten-Year Observational Study, Spain, 2009–2019. Am. J. Infect. Control 2024, 52, 563–569. [Google Scholar] [CrossRef]
- Ministero Della Salute. Prevenzione e Controllo Dell’influenza: Raccomandazioni per La Stagione 2024–2025. Available online: https://www.epicentro.iss.it/influenza/raccomandazioni-ministero (accessed on 6 March 2025).
- Calabrò, G.E.; Vitale, F.; Rizzo, C.; Pugliese, A.; Boccalini, S.; Bechini, A.; Panatto, D.; Amicizia, D.; Domnich, A.; Amodio, E.; et al. Il Nuovo Vaccino Coniugato Antipneumococcico 15-Valente per La Prevenzione Delle Infezioni Da S. Pneumoniae in Età Pediatrica: Una Valutazione Di HTA. J. Prev. Med. Hyg. 2023, 64, E1–E160. [Google Scholar] [CrossRef]
- Trombetta, C.M.; Kistner, O.; Montomoli, E.; Viviani, S.; Marchi, S. Influenza Viruses and Vaccines: The Role of Vaccine Effectiveness Studies for Evaluation of the Benefits of Influenza Vaccines. Vaccines 2022, 10, 714. [Google Scholar] [CrossRef]
- Almadhoon, H.; Hamdallah, A.; Elsayed, S.; Hagrass, A.; Hasan, M.; Fayoud, A.; Al-Kafarna, M.; Elbahnasawy, M.; Alqatati, F.; Ragab, K.; et al. The Effect of Influenza Vaccine in Reducing the Severity of Clinical Outcomes in Patients with COVID-19: A Systematic Review and Meta-Analysis. Sci. Rep. 2022, 12, 14266. [Google Scholar] [CrossRef]
- Boccalini, S.; de Waure, C.; Martorella, L.; Orlando, P.; Bonanni, P.; Bechini, A. The Evolution of Annual Immunization Recommendations Against Influenza in Italy: The Path to Precision Vaccination. Vaccines 2025, 13, 356. [Google Scholar] [CrossRef]
- Domnich, A.; Icardi, G.; Panatto, D.; Scarpaleggia, M.; Trombetta, C.-S.; Ogliastro, M.; Stefanelli, F.; Bruzzone, B.; Orsi, A. Influenza Epidemiology and Vaccine Effectiveness during the 2023/2024 Season in Italy: A Test-Negative Case-Control Study. Int. J. Infect. Dis. 2024, 147, 107202. [Google Scholar] [CrossRef] [PubMed]
- Antonelli Incalzi, R.; Consoli, A.; Lopalco, P.; Maggi, S.; Sesti, G.; Veronese, N.; Volpe, M. Influenza Vaccination for Elderly, Vulnerable and High-Risk Subjects: A Narrative Review and Expert Opinion. Intern. Emerg. Med. 2024, 19, 619–640. [Google Scholar] [CrossRef] [PubMed]
- Trucchi, C.; D’Amelio, M.; Amicizia, D.; Orsi, A.; Loiacono, I.; Tosatto, R.; Piazza, M.F.; Paganino, C.; Pitrelli, A.; Icardi, G.; et al. Lowering the Recommended Age for the Free and Active Offer of Influenza Vaccination in Italy: Clinical and Economic Impact Analysis in the Liguria Region. Hum. Vaccin. Immunother. 2021, 17, 1387–1395. [Google Scholar] [CrossRef] [PubMed]
- Bonanni, P.; Boccalini, S.; Zanobini, P.; Dakka, N.; Lorini, C.; Santomauro, F.; Bechini, A. The Appropriateness of the Use of Influenza Vaccines: Recommendations from the Latest Seasons in Italy. Hum. Vaccin. Immunother. 2018, 14, 699–705. [Google Scholar] [CrossRef]
- Pestarino, L.; Domnich, A.; Orsi, A.; Bianchi, F.; Cannavino, E.; Brasesco, P.C.; Russo, G.; Valbonesi, S.; Vallini, G.; Ogliastro, M.; et al. Rollout of the 2022/2023 Seasonal Influenza Vaccination and Correlates of the Use of Enhanced Vaccines among Italian Adults. Vaccines 2023, 11, 1748. [Google Scholar] [CrossRef]
- Buchy, P.; Badur, S. Who and When to Vaccinate against Influenza. Int. J. Infect. Dis. 2020, 93, 375–387. [Google Scholar] [CrossRef]
- Walensky, R.P.; Bunnell, R.; Layden, J.; Kent, C.K.; Gottardy, A.J.; Leahy, M.A.; Martinroe, J.C.; Spriggs, S.R.; Yang, T.; Doan, Q.M.; et al. Prevention and Control of Seasonal Influenza with Vaccines: Recommendations of the Advisory Committee on Immunization Practices-United States, 2022-23 Influenza Season. MMWR Recomm. Rep. 2022, 71, 1–28. [Google Scholar]
- Havers, F.P.; Moro, P.L.; Hunter, P.; Hariri, S.; Bernstein, H. Use of Tetanus Toxoid, Reduced Diphtheria Toxoid, and Acellular Pertussis Vaccines: Updated Recommendations of the Advisory Committee on Immunization Practices-United States, 2019. Morb. Mortal. Wkly. Rep. 2020, 69, 77–83. [Google Scholar] [CrossRef]
- Amodio, E.; D’Anna, A.; Verso, M.G.; Leonforte, F.; Genovese, D.; Vitale, F. Rotavirus Vaccination as a Public Health Strategy to Reduce the Burden of Hospitalization: The Field Experience of Italy (2008–2018). J. Med. Virol. 2023, 95, e29000. [Google Scholar] [CrossRef]
- Calabrò, G.E.; Boccalini, S.; Panatto, D.; Rizzo, C.; Di Pietro, M.L.; Abreha, F.M.; Ajelli, M.; Amicizia, D.; Bechini, A.; Giacchetta, I.; et al. The New Quadrivalent Adjuvanted Influenza Vaccine for the Italian Elderly: A Health Technology Assessment. Int. J. Environ. Res. Public Health 2022, 19, 4166. [Google Scholar] [CrossRef]
- Italian Medicines Agency (AIFA). AIFA: Influenza Vaccines for the 2024–2025 Season. AIFA, 16 September 2024. Available online: https://www.aifa.gov.it/en/-/influenza_8_i_vaccini_autorizzati_per_la_stagione_2024-2025 (accessed on 27 August 2025).
- Soni, D.; Van Haren, S.; Idoko, O.; Evans, J.; Diray-Arce, J.; Dowling, D.; Levy, O. Towards Precision Vaccines: Lessons From the Second International Precision Vaccines Conference. Front. Immunol. 2020, 11, 590373. [Google Scholar] [CrossRef] [PubMed]
- Boccalini, S.; Tacconi, F.M.; Lai, P.L.; Bechini, A.; Bonanni, P.; Panatto, D. Appropriateness and preferential use of different seasonal influenza vaccines: A pilot study on the opinion of vaccinating physicians in Italy. Vaccine 2019, 37, 915–918. [Google Scholar] [CrossRef] [PubMed]
- Rumi, F.; Basile, M.; Cicchetti, A. Analisi di costo-efficacia e budget impact per il vaccino antinfluenzale quadrivalente ad alto dosaggio nella popolazione anziana Italiana. Glob. Reg. Health Technol. Assess. 2021, 8, 105–113. [Google Scholar] [CrossRef]
- Domnich, A.; Lapi, F.; Orsi, A.; Grattagliano, I.; Rossi, A.; Cricelli, C. Declining number of general practitioners can impair influenza vaccination uptake among Italian older adults: Results from a panel analysis. BMC Health Serv. Res. 2024, 24, 950. [Google Scholar] [CrossRef]
- Schmid, P.; Rauber, D.; Betsch, C.; Lidolt, G.; Denker, M.L. Barriers of Influenza Vaccination Intention and Behavior—A Systematic Review of Influenza Vaccine Hesitancy, 2005–2016. PLoS ONE 2017, 12, e0170550. [Google Scholar] [CrossRef]
- Gavazzi, G.; Fougère, B.; Hanon, O.; Leroux-Roels, I.; Brochot, E.; Blanchard, E.; Russell, C.A.; Paccalin, M. Enhanced influenza vaccination for older adults in Europe: A review of the current situation and expert recommendations for the future. Expert Rev. Vaccines 2025, 24, 350–364. [Google Scholar] [CrossRef]
- Lee, J.K.H.; Lam, G.K.L.; Yin, J.K.; Loiacono, M.M.; Samson, S.I. High-Dose Influenza Vaccine in Older Adults by Age and Seasonal Characteristics: Systematic Review and Meta-Analysis Update. Vaccine X 2023, 14, 100327. [Google Scholar] [CrossRef] [PubMed]
- Italian Ministry of Health. Circular of April 21, 2023—Prevention and Control of Influenza: Recommendations for the 2023–2024Season. (21-04-2023). Available online: https://www.trovanorme.salute.gov.it/norme/renderNormsanPdf?anno=2023&codLeg=93294&parte=1%20&serie=null (accessed on 27 August 2025).
- European Centre for Disease Prevention and Control (ECDC). Seasonal Influenza—Annual Epidemiological Report for 2023/2024; ECDC: Stockholm, Sweden, 2024.
- Tan, P.; Patone, M.; Clift, A.; Dambha-Miller, H.; Saatci, D.; Ranger, T.; Garriga, C.; Zaccardi, F.; Shah, B.; Coupland, C.; et al. Factors Influencing Influenza, Pneumococcal and Shingles Vaccine Uptake and Refusal in Older Adults: A Population-Based Cross-Sectional Study in England. BMJ Open 2023, 13, e058705. [Google Scholar] [CrossRef] [PubMed]
- Han, K.; Hou, Z.; Tu, S.; Liu, M.; Chantler, T.; Larson, H. Factors Influencing Childhood Influenza Vaccination: A Systematic Review. Vaccines 2024, 12, 233. [Google Scholar] [CrossRef] [PubMed]
- Veroniki, A.; Thirugnanasampanthar, S.; Konstantinidis, M.; Dourka, J.; Ghassemi, M.; Neupane, D.; Khan, P.; Nincic, V.; Corry, M.; Robson, R.; et al. Trivalent and Quadrivalent Seasonal Influenza Vaccine in Adults Aged 60 and Older: A Systematic Review and Network Meta-Analysis. BMJ Evid. Based Med. 2024, 29, 239–254. [Google Scholar] [CrossRef]
- Zhang, L.; Wang, W.; Wang, S. Effect of Vaccine Administration Modality on Immunogenicity and Efficacy. Expert Rev. Vaccines 2015, 14, 1509–1523. [Google Scholar] [CrossRef]
- Morse-Brady, J.; Hart, A.M. Prevalence and Types of Vaccination Errors from 2009 to 2018: A Systematic Review of the Medical Literature. Vaccine 2020, 38, 1623–1629. [Google Scholar] [CrossRef]
- Brillo, E.; Ciampoletti, M.; Tosto, V.; Buonomo, E. Exploring Tdap and Influenza Vaccine Uptake and Its Determinants in Pregnancy: A Cross-Sectional Study. Ann. Ig. 2022, 34, 358–374. [Google Scholar] [CrossRef]
- Italian Ministry of Health. Circular of May 21, 2024—Prevention and Control of Influenza: Recommendations for the 2024–2025 Season. (20-05-2024). Available online: https://www.trovanorme.salute.gov.it/norme/renderNormsanPdf?anno=2024&codLeg=100738&parte=1%20&serie=null (accessed on 27 August 2025).
- Seravalli, V.; Romualdi, I.; Ammar, O.; De Blasi, C.; Boccalini, S.; Bechini, A.; Di Tommaso, M. Vaccination Coverage during Pregnancy and Factors Associated with Refusal of Recommended Vaccinations: An Italian Cross Sectional Study. Vaccine X 2024, 18, 100483. [Google Scholar] [CrossRef]
- Filia, A.; Bella, A.; D’Ancona, F.; Fabiani, M.; Giambi, C.; Rizzo, C.; Ferrara, L.; Pascucci, M.G.; Rota, M.C. Childhood Vaccinations: Knowledge, Attitudes and Practices of Paediatricians and Factors Associated with Their Confidence in Addressing Parental Concerns, Italy, 2016. Eurosurveillance 2019, 24. [Google Scholar] [CrossRef]

| Target Population | QIV-SD | QIVcc | aQIV | QIV-HD | LAIV |
|---|---|---|---|---|---|
| People aged ≥65 years | S | S | R | R | - |
| People aged 60–64 years | S | S | S | S | - |
| People aged 18–59 years with risk conditions | S | S | S | - | - |
| Adults aged 18–59 years | S | S | S | - | - |
| Children aged 7–17 years with risk conditions | S | S | - | S | S |
| Children aged 7–17 years | S | S | - | - | S |
| Children aged 2–6 years | S | S | - | - | S |
| Children aged 6 months–2 years | S | - | - | - | - |
| Pregnant and postpartum women | S | S | - | - | - |
| Season | Population Group | n Vaccinated | Inappropriate Doses n (%) | Reference Group | RR (95% CI) |
|---|---|---|---|---|---|
| 2023–2024 | ≥65 years | 123,281 | 26,141 (21.2%) | Adults 60–64 years | 0.23 (0.225–0.231) |
| Adults 60–64 years | 20,599 | 0 (0.0%) | — | Reference | |
| 2024–2025 | ≥65 years | 124,733 | 4915 (3.9%) | Adults 60–64 years | 0.12 (0.118–0.128) |
| Adults 60–64 years | 18,508 | 0 (0.0%) | — | Reference |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Leonforte, F.; Fiorilla, C.; Giorgianni, G.; Nicosia, V.; Contarino, F.; Genovese, C.; Genovese, G.; Morlino, G.; Chimienti, M.; Mistretta, A. Influenza Vaccination Appropriateness: Insights from the Local Health Unit of Catania During the 2023/2024 and 2024/2025 Seasons. Vaccines 2025, 13, 925. https://doi.org/10.3390/vaccines13090925
Leonforte F, Fiorilla C, Giorgianni G, Nicosia V, Contarino F, Genovese C, Genovese G, Morlino G, Chimienti M, Mistretta A. Influenza Vaccination Appropriateness: Insights from the Local Health Unit of Catania During the 2023/2024 and 2024/2025 Seasons. Vaccines. 2025; 13(9):925. https://doi.org/10.3390/vaccines13090925
Chicago/Turabian StyleLeonforte, Francesco, Claudio Fiorilla, Gabriele Giorgianni, Vito Nicosia, Fabio Contarino, Cristina Genovese, Giovanni Genovese, Giustino Morlino, Martina Chimienti, and Antonio Mistretta. 2025. "Influenza Vaccination Appropriateness: Insights from the Local Health Unit of Catania During the 2023/2024 and 2024/2025 Seasons" Vaccines 13, no. 9: 925. https://doi.org/10.3390/vaccines13090925
APA StyleLeonforte, F., Fiorilla, C., Giorgianni, G., Nicosia, V., Contarino, F., Genovese, C., Genovese, G., Morlino, G., Chimienti, M., & Mistretta, A. (2025). Influenza Vaccination Appropriateness: Insights from the Local Health Unit of Catania During the 2023/2024 and 2024/2025 Seasons. Vaccines, 13(9), 925. https://doi.org/10.3390/vaccines13090925







