Evaluation of T-Cell Responses Following Sequential Vaccination with PCV13 and PPSV23 Against Streptococcus pneumoniae in Patients with Psoriasis
Abstract
1. Introduction
2. Materials and Methods
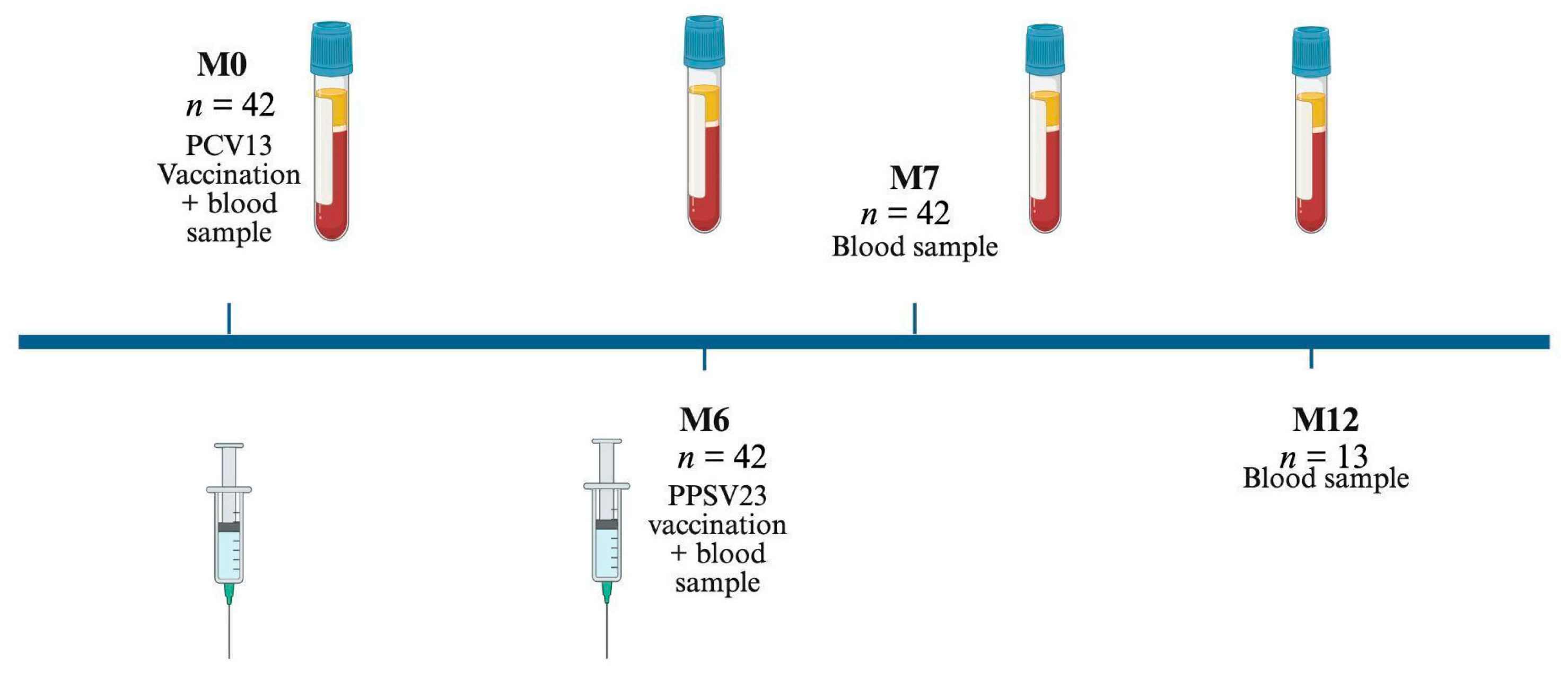
2.1. Study Population
2.2. Vaccines
2.3. Measurement of Cellular Immunity Against S. pneumoniae
2.4. Pneumococcal Antibody ELISA
2.5. Statistical Analysis
3. Results
3.1. Clinical Course of the Study Population
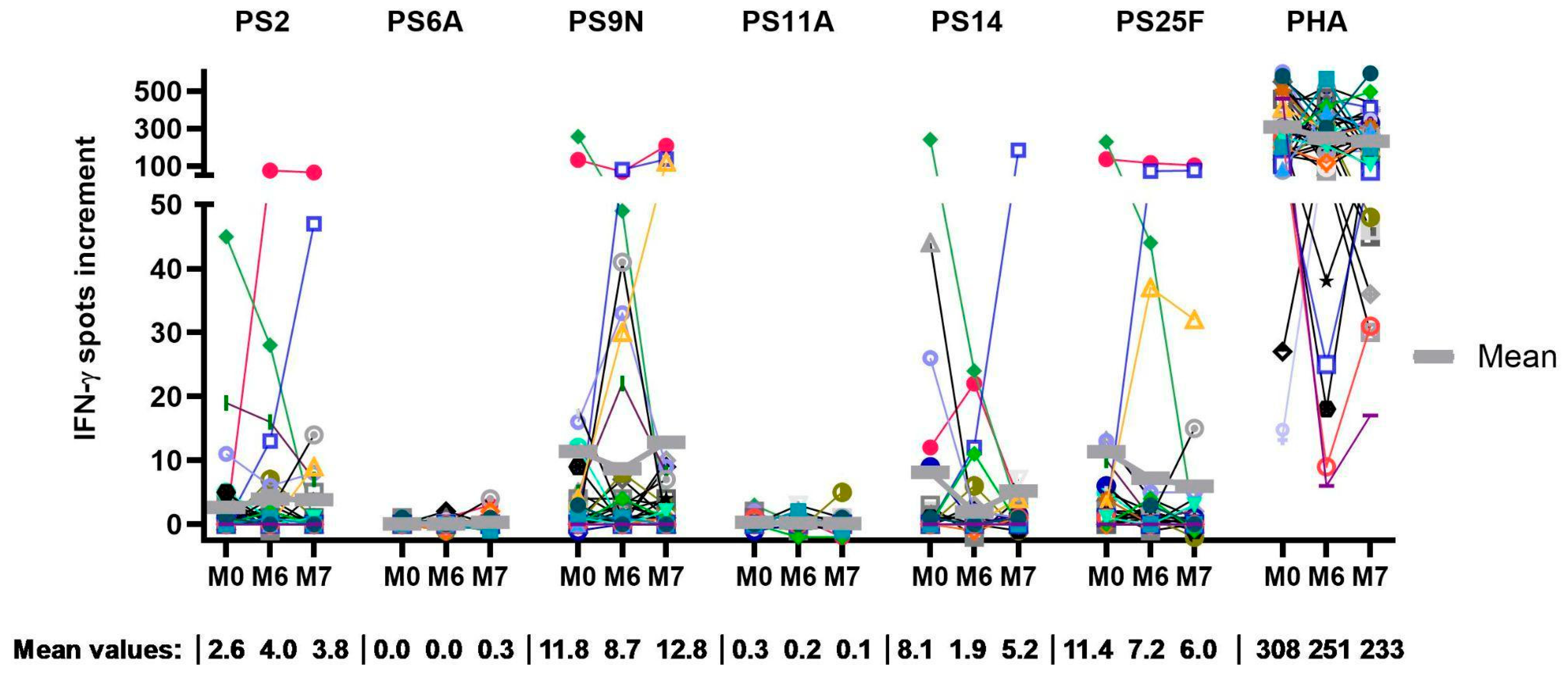
3.2. Cellular Immune Response to Pneumococcus-Specific ELISpot
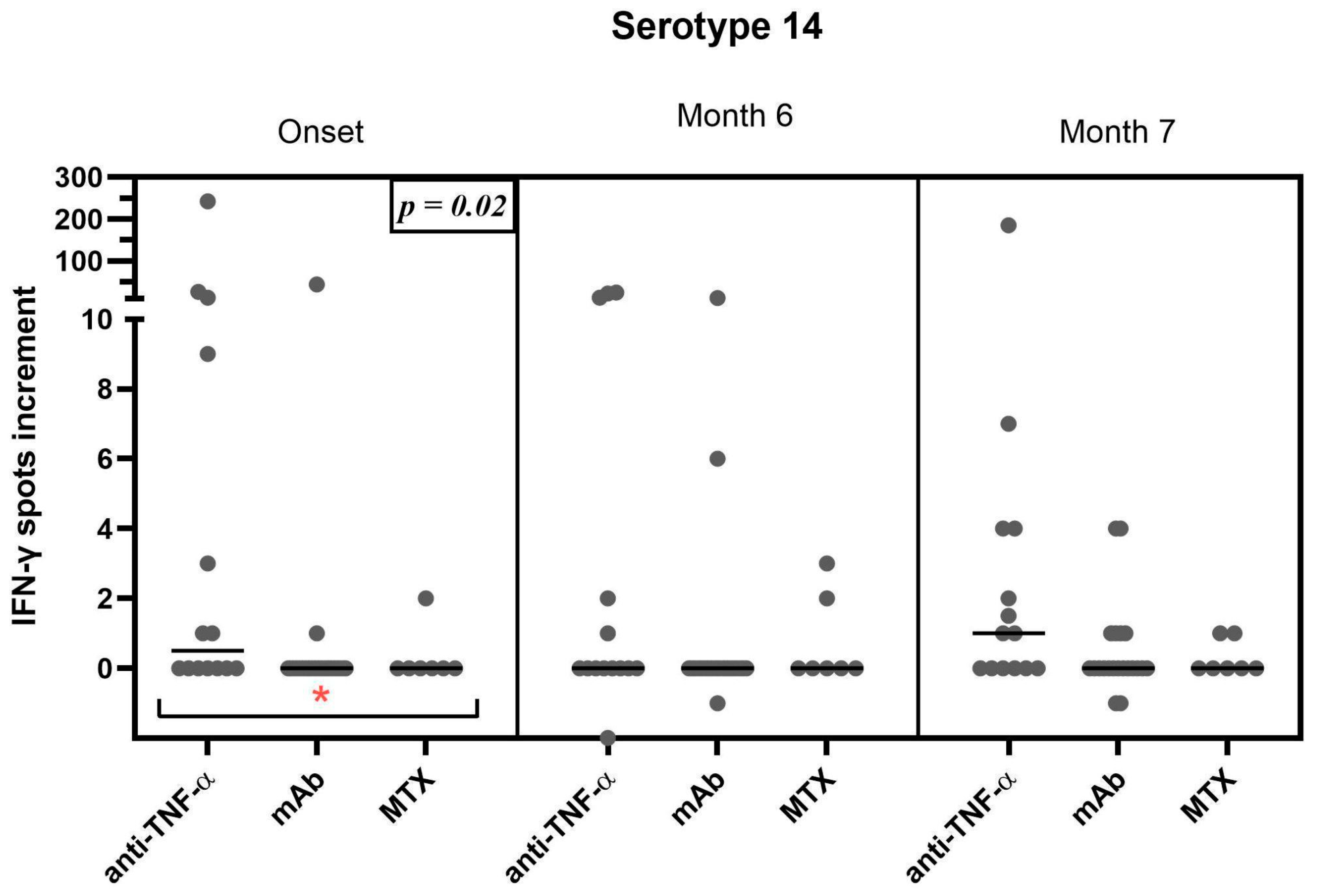
3.3. Correlation of the Therapeutic Regimen with the Cellular Response
3.4. Correlation of the Therapeutic Regimen with the Humoral Response
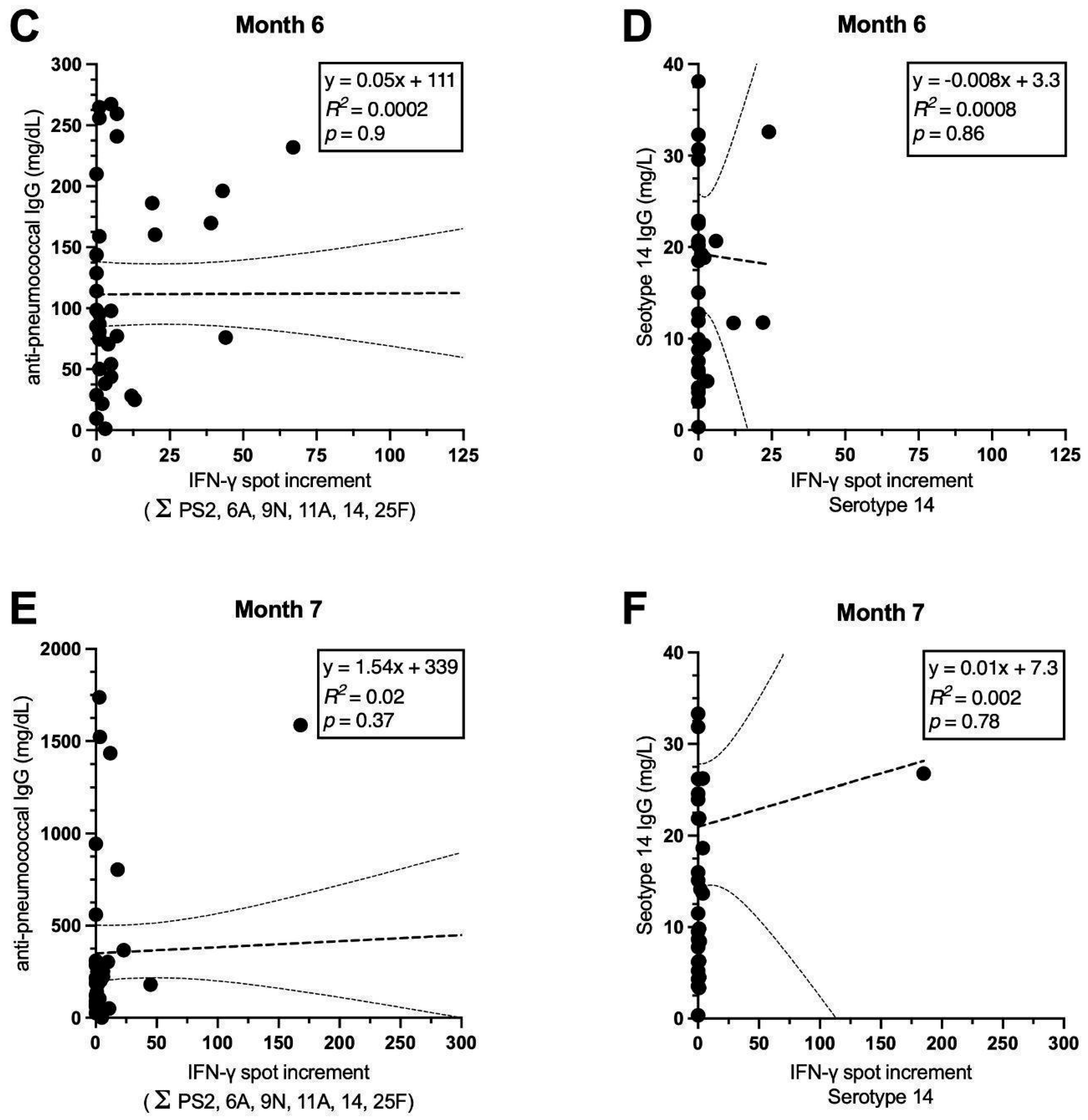
3.5. Correlation of Cellular Immune Responses and Antibody Production
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| BSA | Body Surface Area |
| CAP | Community-Acquired Pneumonia |
| CD4 | Cluster of Differentiation 4 |
| CD45RO | Cluster of Differentiation 45 Isoform RO |
| DLQI | Dermatology Life Quality Index |
| ELISA | Enzyme-Linked-Immunosorbent Assay |
| ELISpot | Enzyme-Linked Immunosorbent Spot Assay |
| IFN-γ | Interferon-γ |
| IgG/M | Immunoglobulin G/M |
| IL | Interleukin |
| MTX | Methotrexate |
| PASI | Psoriasis Area and Severity Index |
| PCV13 | Prevenar 13 |
| PPSV23 | Pneumovax 23 |
| PS | Pneumococcal Serotype |
| RKI | Robert-Koch Institute |
| TNF | Tumor Necrosis Factor |
References
- Christophers, E. Psoriasis—Epidemiology and clinical spectrum. Clin. Exp. Dermatol. 2001, 26, 314–320. [Google Scholar] [CrossRef]
- Mrowietz, U.; Lauffer, F.; Sondermann, W.; Gerdes, S.; Sewerin, P. Psoriasis as a Systemic Disease. Dtsch. Arztebl. Int. 2024, 121, 467–472. [Google Scholar] [CrossRef]
- Gerdes, S.; Mrowietz, U.; Boehncke, W.H. Comorbidity in psoriasis. Hautarzt 2016, 67, 438–444. [Google Scholar] [CrossRef]
- Hölsken, S.; Krefting, F.; Schedlowski, M.; Sondermann, W. Common Fundamentals of Psoriasis and Depression. Acta Derm. Venereol. 2021, 101, adv00609. [Google Scholar] [CrossRef]
- Mrowietz, U.; Kragballe, K.; Reich, K.; Spuls, P.; Griffiths, C.E.; Nast, A.; Franke, J.; Antoniou, C.; Arenberger, P.; Balieva, F.; et al. Definition of treatment goals for moderate to severe psoriasis: A European consensus. Arch. Dermatol. Res. 2011, 303, 1–10. [Google Scholar] [CrossRef]
- Parisi, R.; Iskandar, I.Y.K.; Kontopantelis, E.; Augustin, M.; Griffiths, C.E.M.; Ashcroft, D.M. National, regional, and worldwide epidemiology of psoriasis: Systematic analysis and modelling study. BMJ 2020, 369, m1590. [Google Scholar] [CrossRef]
- Parisi, R.; Symmons, D.P.; Griffiths, C.E.; Ashcroft, D.M. Global epidemiology of psoriasis: A systematic review of incidence and prevalence. J. Investig. Dermatol. 2013, 133, 377–385. [Google Scholar] [CrossRef] [PubMed]
- Sbidian, E.; Chaimani, A.; Garcia-Doval, I.; Doney, L.; Dressler, C.; Hua, C.; Hughes, C.; Naldi, L.; Afach, S.; Le Cleach, L. Systemic pharmacological treatments for chronic plaque psoriasis: A network meta-analysis. Cochrane Database Syst. Rev. 2021, 4, CD011535. [Google Scholar] [CrossRef] [PubMed]
- Kao, L.-T.; Lee, C.-Z.; Liu, S.-P.; Tsai, M.-C.; Lin, H.-C. Psoriasis and the Risk of Pneumonia: A Population-Based Study. PLoS ONE 2014, 9, e116077. [Google Scholar] [CrossRef] [PubMed]
- Chiricozzi, A.; Gisondi, P.; Bellinato, F.; Girolomoni, G. Immune Response to Vaccination in Patients with Psoriasis Treated with Systemic Therapies. Vaccines 2020, 8, 769. [Google Scholar] [CrossRef]
- Wakkee, M.; de Vries, E.; van den Haak, P.; Nijsten, T. Increased risk of infectious disease requiring hospitalization among patients with psoriasis: A population-based cohort. J. Am. Acad. Dermatol. 2011, 65, 1135–1144. [Google Scholar] [CrossRef]
- Wine-Lee, L.; Keller, S.C.; Wilck, M.B.; Gluckman, S.J.; Van Voorhees, A.S. From the Medical Board of the National Psoriasis Foundation: Vaccination in adult patients on systemic therapy for psoriasis. J. Am. Acad. Dermatol. 2013, 69, 1003–1013. [Google Scholar] [CrossRef]
- Falkenhorst, G.; Remschmidt, C.; Harder, T.; Wichmann, O.; Glodny, S.; Hummers-Pradier, E.; Ledig, T.; Bogdan, C. Background paper to the updated pneumococcal vaccination recommendation for older adults in Germany. Infektionsepidemiologie 2016, 59, 1623–1657. [Google Scholar] [CrossRef] [PubMed]
- Bonten, M.J.; Huijts, S.M.; Bolkenbaas, M.; Webber, C.; Patterson, S.; Gault, S.; van Werkhoven, C.H.; van Deursen, A.M.; Sanders, E.A.; Verheij, T.J.; et al. Polysaccharide conjugate vaccine against pneumococcal pneumonia in adults. N. Engl. J. Med. 2015, 372, 1114–1125. [Google Scholar] [CrossRef]
- Centers for Disease Control and Prevention. Use of 13-valent pneumococcal conjugate vaccine and 23-valent pneumococcal polysaccharide vaccine for adults with immunocompromising conditions: Recommendations of the Advisory Committee on Immunization Practices (ACIP). MMWR Morb. Mortal. Wkly. Rep. 2012, 61, 816–819. [Google Scholar]
- Mülling, N.; van de Sand, L.; Völk, K.; Aufderhorst, U.W.; van der Linden, M.; Horn, P.A.; Kribben, A.; Wilde, B.; Krawczyk, A.; Witzke, O.; et al. Follow-Up of Serotype-Specific Antibodies after Sequential Vaccination with Two Pneumococcal Vaccines in Kidney Transplant Recipients. HLA 2023, 102, 4. [Google Scholar] [CrossRef]
- Chilson, E.; Scott, D.A.; Schmoele-Thoma, B.; Watson, W.; Moran, M.M.; Isturiz, R. Immunogenicity and safety of the 13-valent pneumococcal conjugate vaccine in patients with immunocompromising conditions: A review of available evidence. Hum. Vaccin. Immunother. 2020, 16, 2758–2772. [Google Scholar] [CrossRef]
- Froneman, C.; Kelleher, P.; Jose, R.J. Pneumococcal Vaccination in Immunocompromised Hosts: An Update. Vaccines 2021, 9, 536. [Google Scholar] [CrossRef]
- Schlaberg, J.; Vygen-Bonnet, S.; Falman, A.; Wilhelm, J.; Hummers, E.; Ledig, T.; Bogdan, C. Aktualisierung der Empfehlungen der STIKO zur Standardimpfung von Personen ≥60 Jahre sowie zur Indikationsimpfung von Risikogruppen gegen Pneumokokken und die dazugehörige wissenschaftliche Begründung. Epidemiol. Bull. 2023, 39, 3–44. [Google Scholar]
- Robert Koch-Institut. Wissenschaftliche Begründung für die Aktualisierung der Empfehlungen zur Indikationsimpfung gegen Pneumokokken für Risikogruppen. Robert Koch-Institut 2016. Available online: https://www.rki.de/DE/Aktuelles/Publikationen/Epidemiologisches-Bulletin/2016/37_16.pdf?__blob=publicationFile&v=3&utm_source=chatgpt.com (accessed on 6 October 2023).
- McCool, T.L.; Weiser, J.N. Limited role of antibody in clearance of Streptococcus pneumoniae in a murine model of colonization. Infect. Immun. 2004, 72, 5807–5813. [Google Scholar] [CrossRef]
- Crnkic Kapetanovic, M.; Saxne, T.; Jonsson, G.; Truedsson, L.; Geborek, P. Rituximab and abatacept but not tocilizumab impair antibody response to pneumococcal conjugate vaccine in patients with rheumatoid arthritis. Arthritis Res. Ther. 2013, 15, R171. [Google Scholar] [CrossRef]
- Kapetanovic, M.C.; Roseman, C.; Jonsson, G.; Truedsson, L.; Saxne, T.; Geborek, P. Antibody response is reduced following vaccination with 7-valent conjugate pneumococcal vaccine in adult methotrexate-treated patients with established arthritis, but not those treated with tumor necrosis factor inhibitors. Arthritis Rheum. 2011, 63, 3723–3732. [Google Scholar] [CrossRef] [PubMed]
- Karasartova, D.; Gazi, U.; Tosun, O.; Gureser, A.S.; Sahiner, I.T.; Dolapci, M.; Ozkan, A.T. Anti-Pneumococcal Vaccine-Induced Cellular Immune Responses in Post-Traumatic Splenectomized Individuals. J. Clin. Immunol. 2017, 37, 388–396. [Google Scholar] [CrossRef]
- Winthrop, K.L.; Korman, N.; Abramovits, W.; Rottinghaus, S.T.; Tan, H.; Gardner, A.; Mukwaya, G.; Kaur, M.; Valdez, H. T-cell-mediated immune response to pneumococcal conjugate vaccine (PCV-13) and tetanus toxoid vaccine in patients with moderate-to-severe psoriasis during tofacitinib treatment. J. Am. Acad. Dermatol. 2018, 78, 1149–1155.e1. [Google Scholar] [CrossRef]
- Wright, A.K.; Bangert, M.; Gritzfeld, J.F.; Ferreira, D.M.; Jambo, K.C.; Wright, A.D.; Collins, A.M.; Gordon, S.B. Experimental human pneumococcal carriage augments IL-17A-dependent T-cell defence of the lung. PLoS Pathog. 2013, 9, e1003274. [Google Scholar] [CrossRef]
- Chaplin, D.D. Overview of the immune response. J. Allergy Clin. Immunol. 2010, 125, S3–S23. [Google Scholar] [CrossRef] [PubMed]
- Helmer, L.; van de Sand, L.; Wojtakowski, T.; Otte, M.; Witzke, O.; Sondermann, W.; Krawczyk, A.; Lindemann, M. Antibody responses after sequential vaccination with PCV13 and PPSV23 in patients with moderate to severe plaque psoriasis under immunosuppressive therapy. mBio 2024, 15, e0048224. [Google Scholar] [CrossRef] [PubMed]
- Aravena, O.; Pesce, B.; Soto, L.; Orrego, N.; Sabugo, F.; Wurmann, P.; Molina, M.C.; Alfaro, J.; Cuchacovich, M.; Aguillón, J.C.; et al. Anti-TNF therapy in patients with rheumatoid arthritis decreases Th1 and Th17 cell populations and expands IFN-γ-producing NK cell and regulatory T cell subsets. Immunobiology 2011, 216, 1256–1263. [Google Scholar] [CrossRef] [PubMed]
- Furiati, S.C.; Catarino, J.S.; Silva, M.V.; Silva, R.F.; Estevam, R.B.; Teodoro, R.B.; Pereira, S.L.; Ataide, M.; Rodrigues, V.; Rodrigues, D.B.R. Th1, Th17, and Treg Responses are Differently Modulated by TNF-α Inhibitors and Methotrexate in Psoriasis Patients. Sci. Rep. 2019, 9, 7526. [Google Scholar] [CrossRef]
- Chan, E.S.; Cronstein, B.N. Molecular action of methotrexate in inflammatory diseases. Arthritis Res. Ther. 2002, 4, 266. [Google Scholar] [CrossRef]
- Gäckler, A.; Mülling, N.; Völk, K.; Wilde, B.; Eisenberger, U.; Rohn, H.; Horn, P.A.; Witzke, O.; Lindemann, M. Establishment of an ELISpot Assay to Detect Cellular Immunity against S. pneumoniae in Vaccinated Kidney Transplant Recipients. Vaccines 2021, 9, 1438. [Google Scholar] [CrossRef] [PubMed]
- Mahil, S.K.; Bechman, K.; Raharja, A.; Domingo-Vila, C.; Baudry, D.; Brown, M.A.; Cope, A.P.; Dasandi, T.; Graham, C.; Lechmere, T.; et al. The effect of methotrexate and targeted immunosuppression on humoral and cellular immune responses to the COVID-19 vaccine BNT162b2: A cohort study. Lancet Rheumatol. 2021, 3, e627–e637. [Google Scholar] [CrossRef]
- Thümmler, L.; Gäckler, A.; Bormann, M.; Ciesek, S.; Widera, M.; Rohn, H.; Fisenkci, N.; Otte, M.; Alt, M.; Dittmer, U.; et al. Cellular and Humoral Immunity against Different SARS-CoV-2 Variants Is Detectable but Reduced in Vaccinated Kidney Transplant Patients. Vaccines 2022, 10, 1348. [Google Scholar] [CrossRef]
- Morgans, H.A.; Bradley, T.; Flebbe-Rehwaldt, L.; Selvarangan, R.; Bagherian, A.; Barnes, A.P.; Bass, J.; Cooper, A.M.; Fischer, R.; Kleiboeker, S.; et al. Humoral and cellular response to the COVID-19 vaccine in immunocompromised children. Pediatr. Res. 2023, 94, 200–205. [Google Scholar] [CrossRef]
- Wuorimaa, T.; Käyhty, H.; Eskola, J.; Bloigu, A.; Leroy, O.; Surcel, H.M. Activation of cell-mediated immunity following immunization with pneumococcal conjugate or polysaccharide vaccine. Scand. J. Immunol. 2001, 53, 422–428. [Google Scholar] [CrossRef]
- Gazi, U.; Tosun, O.; Kursat Derici, M.; Karasartova, D.; Semra Gureser, A.; Taylan Ozkan, A. Importance of NK Cells in Cellular and Humoral Responses Triggered by Pneumococcus Vaccination. Int. Arch. Allergy Immunol. 2024, 185, 362–369. [Google Scholar] [CrossRef]
- Ramos-Sevillano, E.; Ercoli, G.; Brown, J.S. Mechanisms of Naturally Acquired Immunity to Streptococcus pneumoniae. Front. Immunol. 2019, 10, 358. [Google Scholar] [CrossRef]
- Tin Tin Htar, M.; Christopoulou, D.; Schmitt, H.J. Pneumococcal serotype evolution in Western Europe. BMC Infect Dis. 2015, 15, 419. [Google Scholar] [CrossRef] [PubMed]
- Mureithi, M.W.; Finn, A.; Ota, M.O.; Zhang, Q.; Davenport, V.; Mitchell, T.J.; Williams, N.A.; Adegbola, R.A.; Heyderman, R.S. T cell memory response to pneumococcal protein antigens in an area of high pneumococcal carriage and disease. J. Infect Dis. 2009, 200, 783–793. [Google Scholar] [CrossRef]
- Visvanathan, S.; Keenan, G.F.; Baker, D.G.; Levinson, A.I.; Wagner, C.L. Response to pneumococcal vaccine in patients with early rheumatoid arthritis receiving infliximab plus methotrexate or methotrexate alone. J. Rheumatol. 2007, 34, 952–957. [Google Scholar] [PubMed]


| Parameter 1 | Median (Range) or Number (No. 1) |
|---|---|
| Median age (range), years | 49 (18–67) |
| Patient sex (male/female) | 23/19 |
| Median interval since first diagnosis (range), years | 26 (1–50) |
| Mean PASI (Month 0) | 2.2 (0–15) |
| Mean DLQI (Month 0) | 3.9 (0–23) |
| Immunosuppression, no. 1 | |
| MTX > 10 mg/week | 7 |
| TNF-alpha-inhibitor (adalimumab/infliximab/etanercept/certolizumab), MTX-co-medication max. 10 mg/week) | 14 |
| IL-17-inhibitor (secukinumab/ixekizumab) | 6 |
| IL-12/23-inhibitor (ustekinumab) | 4 |
| IL-23-inhibitor (guselkumab) | 11 |
| Serotype | 2 | 6A | 9N | 11A | 14 | 25F |
| Mean value of spot increments Median value of spot increments | ||||||
| Month 0 | 2.6 | 0.0 | 11.4 | 0.3 | 8.1 | 11.4 |
| n = 42 | 0.0 | 0.0 | 1.0 | 0.0 | 0.0 | 1.0 |
| Month 6 | 4.0 | 0.0 | 8.7 | 0.2 | 1.9 | 7.2 |
| n = 42 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.5 |
| Month 7 | 3.8 | 0.3 | 12.8 | 0.1 | 5.2 | 6.0 |
| n = 42 | 0.0 | 0.0 | 1.0 | 0.0 | 0.0 | 0.0 |
| Month 12 | 4.5 | 1.4 | 5.4 | 1.2 | 3.8 | 4.8 |
| n = 13 | 1.0 | 1.0 | 1.5 | 0.0 | 2.5 | 2.5 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wojtakowski, T.; van de Sand, L.; Helmer, L.; Mokanis, M.; Witzke, O.; Horn, P.A.; Krawczyk, A.; Sondermann, W.; Lindemann, M. Evaluation of T-Cell Responses Following Sequential Vaccination with PCV13 and PPSV23 Against Streptococcus pneumoniae in Patients with Psoriasis. Vaccines 2025, 13, 920. https://doi.org/10.3390/vaccines13090920
Wojtakowski T, van de Sand L, Helmer L, Mokanis M, Witzke O, Horn PA, Krawczyk A, Sondermann W, Lindemann M. Evaluation of T-Cell Responses Following Sequential Vaccination with PCV13 and PPSV23 Against Streptococcus pneumoniae in Patients with Psoriasis. Vaccines. 2025; 13(9):920. https://doi.org/10.3390/vaccines13090920
Chicago/Turabian StyleWojtakowski, Thea, Lukas van de Sand, Lorena Helmer, Mona Mokanis, Oliver Witzke, Peter A. Horn, Adalbert Krawczyk, Wiebke Sondermann, and Monika Lindemann. 2025. "Evaluation of T-Cell Responses Following Sequential Vaccination with PCV13 and PPSV23 Against Streptococcus pneumoniae in Patients with Psoriasis" Vaccines 13, no. 9: 920. https://doi.org/10.3390/vaccines13090920
APA StyleWojtakowski, T., van de Sand, L., Helmer, L., Mokanis, M., Witzke, O., Horn, P. A., Krawczyk, A., Sondermann, W., & Lindemann, M. (2025). Evaluation of T-Cell Responses Following Sequential Vaccination with PCV13 and PPSV23 Against Streptococcus pneumoniae in Patients with Psoriasis. Vaccines, 13(9), 920. https://doi.org/10.3390/vaccines13090920






