Characteristics of Varicella Breakthrough Cases in Jinhua City, 2016–2024
Abstract
1. Introduction
2. Data and Methods
2.1. Data Sources
2.2. Case Extraction and Vaccination Data Matching
2.3. Definitions
2.4. Breakthrough Interval Calculation
2.5. Heatmap Construction
2.6. Bivariate Cubic Regression Modeling
2.7. Statistical Analysis
3. Results
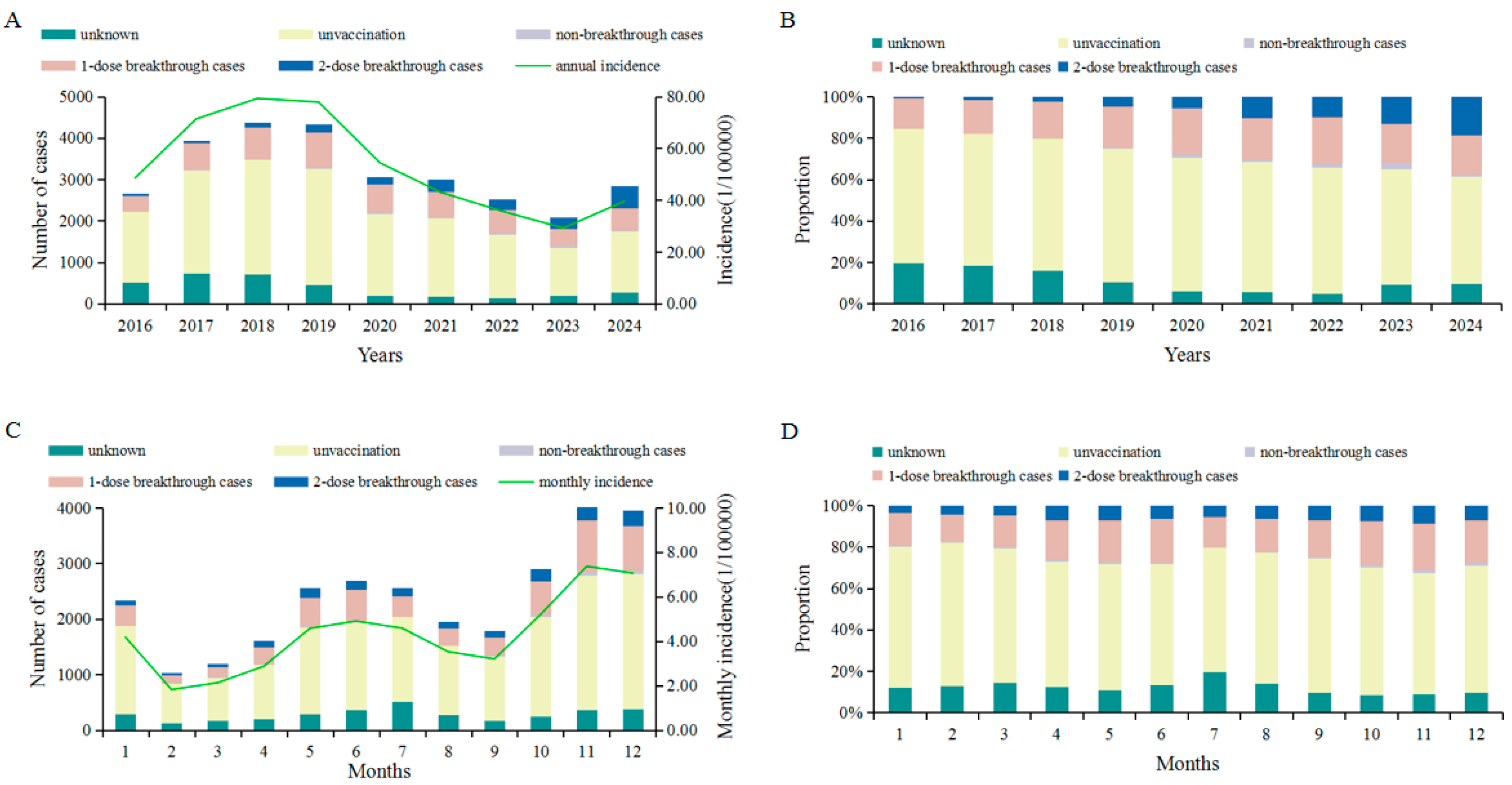
3.1. A Rising Trend in the Annual Proportion of Breakthrough Cases Was Observed in Jinhua City
3.2. Breakthrough Cases Were Primarily Observed Among School-Aged Children in Jinhua City
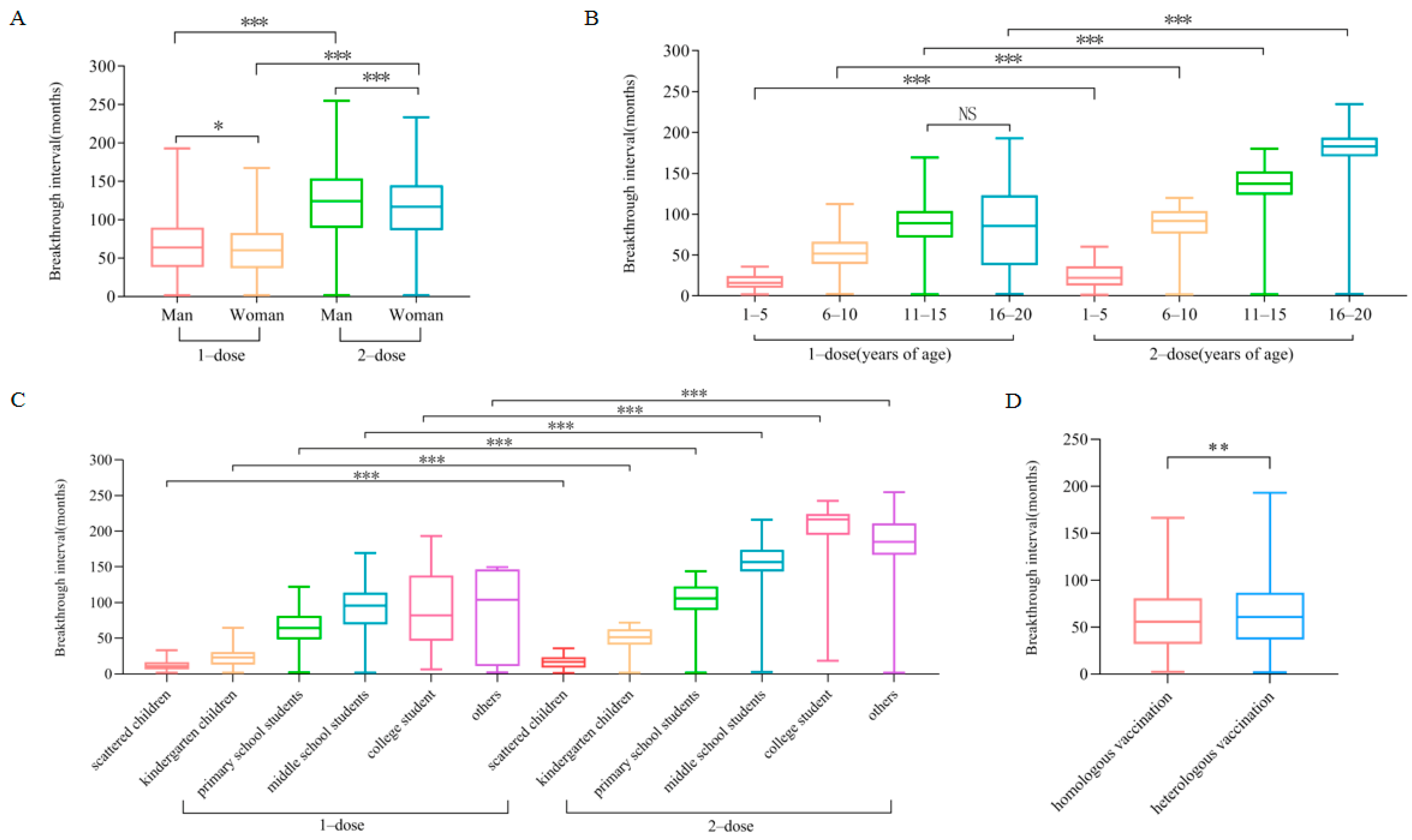
3.3. Breakthrough Interval Was Influenced by Dose, Sex, Age, Population Category, and Vaccination Type
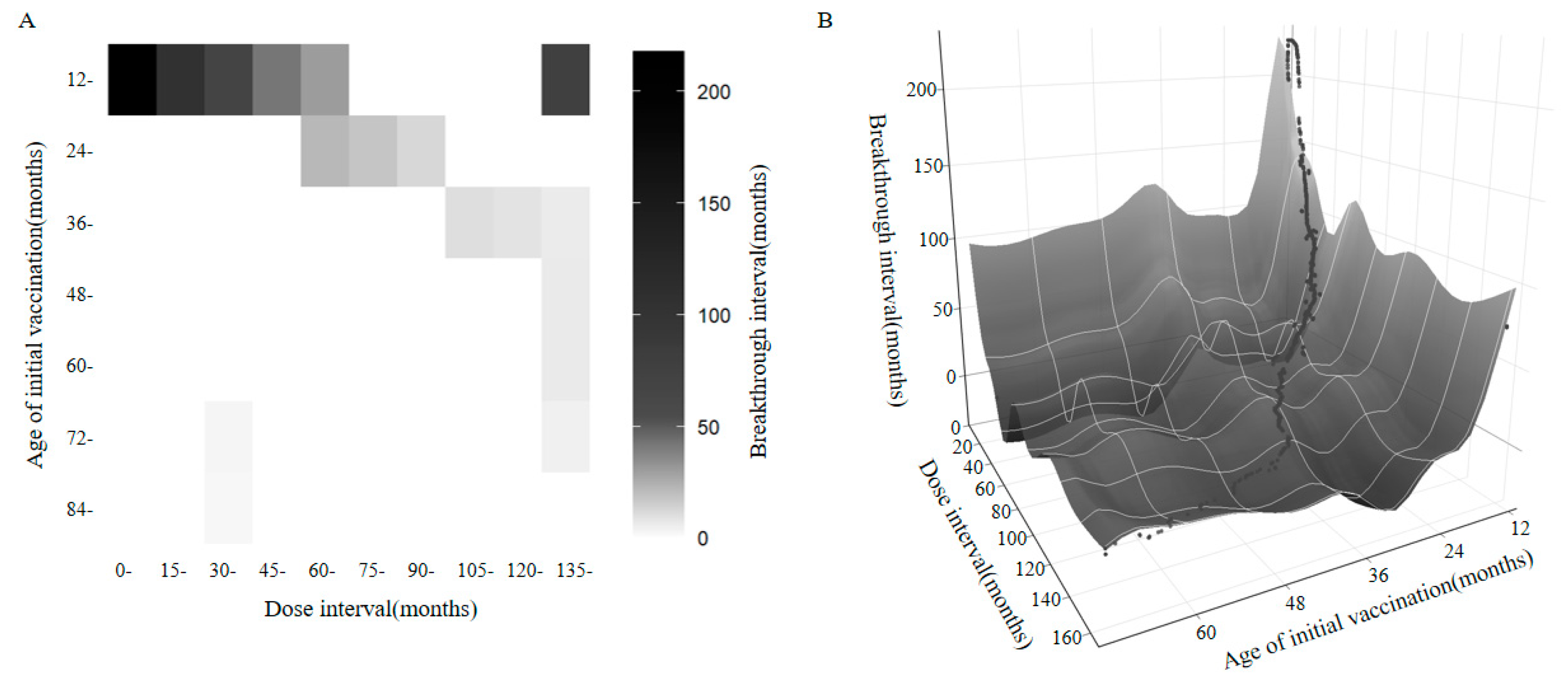
3.4. Combined Effect of Initial Vaccination Age and Dose Interval on Breakthrough Interval
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Heininger, U.; Seward, J.F. Varicella. Lancet 2006, 368, 1365–1376. [Google Scholar] [CrossRef] [PubMed]
- Arvin, A.M. Varicella-zoster virus. Clin. Microbiol. Rev. 1996, 9, 361–381. [Google Scholar] [CrossRef]
- Otani, N.; Shima, M.; Yamamoto, T.; Okuno, T. Effect of Routine Varicella Immunization on the Epidemiology and Immunogenicity of Varicella and Shingles. Viruses 2022, 14, 588. [Google Scholar] [CrossRef]
- Marin, M.; Güris, D.; Chaves, S.S.; Schmid, S.; Seward, J.F. Prevention of varicella: Recommendations of the Advisory Committee on Immunization Practices (ACIP). MMWR. Recommendations and reports: Morbidity and mortality weekly report. Recomm. Rep. 2007, 56, 1–40. [Google Scholar]
- Wooding, E.L.; Kadambari, S.; Warris, A. Varicella: Is it time for a global vaccination programme? Arch. Dis. Child. 2025, 110, 586–591. [Google Scholar] [CrossRef]
- Leung, J.; Broder, K.R.; Marin, M. Severe varicella in persons vaccinated with varicella vaccine (breakthrough varicella): A systematic literature review. Expert Rev. Vaccines 2017, 16, 391–400. [Google Scholar] [CrossRef]
- Shapiro, E.D.; Marin, M. The Effectiveness of Varicella Vaccine: 25 Years of Postlicensure Experience in the United States. J. Infect. Dis. 2022, 226 (Suppl. S4), S425–S430. [Google Scholar] [CrossRef]
- Lee, Y.H.; Choe, Y.J.; Cho, S.I.; Park, H.; Bang, J.H.; Lee, J.-K. Effects of One-dose Varicella Vaccination on Disease Severity in Children during Outbreaks in Seoul, Korea. J. Korean Med. Sci. 2019, 34, e83. [Google Scholar] [CrossRef]
- Siedler, A.; Dettmann, M.; Tolksdorf, K.; Polte, C.; Walter, C.; Ehlers, B. Laboratory investigations of vaccinated patients with varicella. Vaccine 2015, 33, 1968–1973. [Google Scholar] [CrossRef]
- Suo, L.; Lu, L.; Wang, Q.; Yang, F.; Wang, X.; Pang, X.; Marin, M.; Wang, C. Varicella outbreak in a highly-vaccinated school population in Beijing, China during the voluntary two-dose era. Vaccine 2017, 35, 4368–4373. [Google Scholar] [CrossRef] [PubMed]
- Fu, J.; Wang, J.; Jiang, C.; Shi, R.; Ma, T. Outbreak of varicella in a highly vaccinated preschool population. Int. J. Infect. Dis. 2015, 37, 14–18. [Google Scholar] [CrossRef]
- Li, Y.; Xu, F.; Liu, M.; Teng, S.; Liang, F.; Wang, F. Effectiveness of two-dose vs. one-dose varicella vaccine in children in Shanghai, China: A prospective cohort study. Front. Public Health 2024, 12, 1320407. [Google Scholar] [CrossRef]
- Zhu, Y.F.; Li, Y.F.; Du, Y.; Zeng, M. Epidemiological characteristics of breakthrough varicella infection during varicella outbreaks in Shanghai, 2008–2014. Epidemiol. Infect. 2017, 145, 2129–2136. [Google Scholar] [CrossRef] [PubMed]
- Liu, B.; Li, X.; Yuan, L.; Sun, Q.; Fan, J.; Jing, Y.; Meng, S. Analysis on the epidemiological characteristics of varicella and breakthrough case from 2014 to 2022 in Qingyang City. Hum. Vaccines Immunother. 2023, 19, 2224075. [Google Scholar] [CrossRef]
- Wang, M.; Li, X.; You, M.; Wang, Y.; Liu, X.; Li, Z.; Zhao, W.; Jiang, Z.; Hu, Y.; Yin, D.; et al. Epidemiological Characteristics of Varicella Outbreaks—China, 2006–2022. China CDC Wkly. 2023, 5, 1161–1166. [Google Scholar] [CrossRef]
- Zhang, H.; Lai, X.; Patenaude, B.N.; Jit, M.; Fang, H. Adding new childhood vaccines to China’s National Immunization Program: Evidence, benefits, and priorities. Lancet Public Health 2023, 8, e1016–e1024. [Google Scholar] [CrossRef]
- Duan, C.; Zhang, Y.; Zhang, Q.; Zhang, S.; Zhong, P.; Gong, G.; Zhu, Y.; Fei, J.; Zhao, J.; Sun, Y.; et al. Impact of natural and socio-economic factors on varicella incidence in children in Shanghai, 2013–2022. Front. Public Health 2025, 13, 1565717. [Google Scholar] [CrossRef]
- Yue, C.; Li, Y.; Wang, Y.; Cao, L.; Zhu, X.; Martin, K.; Wang, H.; An, Z. The varicella vaccination pattern among children under 5 years old in selected areas in China. Oncotarget 2017, 8, 45612–45618. [Google Scholar] [CrossRef]
- Sun, X.; Wang, Y.; Zhang, L.; Liu, Y.; Xu, L.; Chen, Q.; Sun, H.; Wang, F.; Wang, Z.; Wang, W. Explore the optimal timing for administering the second dose of the varicella vaccine in China. J. Med. Virol. 2023, 95, e29119. [Google Scholar] [CrossRef]
- Lin, M.; Yang, T.; Deng, P.; Yang, L.; Xue, C. Analysis on the Epidemiological Characteristics of Breakthrough Varicella Cases and Incremental Effectiveness of 2-Dose Varicella Vaccine in China. Vaccines 2025, 13, 160. [Google Scholar] [CrossRef]
- Hattori, F.; Higashimoto, Y.; Miura, H.; Kawamura, Y.; Yoshikawa, A.; Ihira, M.; Yoshikawa, T. Epidemiology and clinical features of breakthrough varicella in the 9 years after universal vaccination began in Japan. Vaccine 2025, 59, 127274. [Google Scholar] [CrossRef]
- Quinn, H.E.; Gidding, H.F.; Marshall, H.S.; Booy, R.; Elliott, E.J.; Richmond, P.; Crawford, N.; McIntyre, P.B.; Macartney, K.K. Varicella vaccine effectiveness over 10 years in Australia; moderate protection from 1-dose program. J. Infect. 2019, 78, 220–225. [Google Scholar] [CrossRef]
- Kurugöl, Z.; Gökçe, Ş. Outbreak of varicella in preschool children despite one-dose vaccination. Turk. J. Pediatr. 2018, 60, 56–62. [Google Scholar] [CrossRef] [PubMed]
- Arvin, A.M. Insights From Studies of the Genetics, Pathogenesis, and Immunogenicity of the Varicella Vaccine. J. Infect. Dis. 2022, 226 (Suppl. S4), S385–S391. [Google Scholar] [CrossRef]
- Huber, A.; Gazder, J.; Dobay, O.; Mészner, Z.; Horváth, A. Attitudes towards varicella vaccination in parents and paediatric healthcare providers in Hungary. Vaccine 2020, 38, 5249–5255. [Google Scholar] [CrossRef]
- Sun, X.; Zhu, Y.; Sun, H.; Xu, Y.; Zhang, L.; Wang, Z. Comparison of varicella outbreaks in schools in China during different vaccination periods. Hum. Vaccines Immunother. 2022, 18, 2114255. [Google Scholar] [CrossRef] [PubMed]
- Wu, Q.S.; Wang, X.; Liu, J.Y.; Chen, Y.-F.; Zhou, Q.; Wang, Y.; Sha, J.-D.; Xuan, Z.-L.; Zhang, L.-W.; Yan, L.; et al. Varicella outbreak trends in school settings during the voluntary single-dose vaccine era from 2006 to 2017 in Shanghai, China. Int. J. Infect. Dis. 2019, 89, 72–78. [Google Scholar] [CrossRef]
- Obi, O.A. Varicella in the 21st Century. NeoReviews 2024, 25, e274–e281. [Google Scholar] [CrossRef] [PubMed]
- Mahmud, A.S.; Metcalf, C.J.; Grenfell, B.T. Comparative dynamics, seasonality in transmission, and predictability of childhood infections in Mexico. Epidemiol. Infect. 2017, 145, 607–625. [Google Scholar] [CrossRef]
- Barbieri, E.; Cocchio, S.; Furlan, P.; Scamarcia, A.; Cantarutti, L.; Dona’, D.; Giaquinto, C.; Baldo, V. A population database analysis to estimate the varicella vaccine effectiveness in children < 14 years in a high vaccination coverage area from 2004 to 2022. Vaccine 2024, 42, 126387. [Google Scholar] [CrossRef]
- Kim, E.Y.; Park, C.; Lee, G.; Jeong, S.; Song, J.; Lee, D.-H. Epidemiological characteristics of varicella outbreaks in the Republic of Korea, 2016–2020. Osong Public Health Res. Perspect. 2022, 13, 133–141. [Google Scholar] [CrossRef]
- Klein, N.P.; Holmes, T.H.; Sharp, M.A.; Heineman, T.C.; Schleiss, M.R.; Bernstein, D.I.; Kemble, G.; Arvin, A.M.; Dekker, C.L. Variability and gender differences in memory T cell immunity to varicella-zoster virus in healthy adults. Vaccine 2006, 24, 5913–5918. [Google Scholar] [CrossRef]
- Fischinger, S.; Boudreau, C.M.; Butler, A.L.; Streeck, H.; Alter, G. Sex differences in vaccine-induced humoral immunity. Semin. Immunopathol. 2019, 41, 239–249. [Google Scholar] [CrossRef]
- Fink, A.L.; Klein, S.L. The evolution of greater humoral immunity in females than males: Implications for vaccine efficacy. Curr. Opin. Physiol. 2018, 6, 16–20. [Google Scholar] [CrossRef] [PubMed]
- Al-Qaisi, T.S.; Abumsimir, B. Vaccination strategies, the power of the unmatched double hits. Future Sci. OA 2023, 9, Fso887. [Google Scholar] [CrossRef]
- Eisen, H.N. Affinity enhancement of antibodies: How low-affinity antibodies produced early in immune responses are followed by high-affinity antibodies later and in memory B-cell responses. Cancer Immunol. Res. 2014, 2, 381–392. [Google Scholar] [CrossRef] [PubMed]
- Doherty, T.M.; Weinberger, B.; Didierlaurent, A.; Lambert, P.-H. Age-related changes in the immune system and challenges for the development of age-specific vaccines. Ann. Med. 2025, 57, 2477300. [Google Scholar] [CrossRef] [PubMed]
- Palm, A.E.; Henry, C. Remembrance of Things Past: Long-Term B Cell Memory After Infection and Vaccination. Front. Immunol. 2019, 10, 1787. [Google Scholar] [CrossRef]
| Variables | Varicella Cases | Breakthrough Cases | χ2 | p | ||
|---|---|---|---|---|---|---|
| 1-Dose | 2-Dose | Total | ||||
| Sex | 0.034 | 0.085 | ||||
| Man | 16,110 | 3253 (20.19) | 1148 (7.13) | 4401 (27.32) | ||
| Woman | 12,668 | 2191 (17.30) | 781 (6.16) | 2972 (23.46) | ||
| Age (years) | 189.090 | <0.001 | ||||
| <1 | 735 | 0 (0.00) | 0 (0.00) | 0 (0.00) | ||
| 1~5 | 2061 | 535 (25.96) | 205 (9.94) | 740 (35.90) | ||
| 6~10 | 5671 | 1652 (29.13) | 848 (14.95) | 2500 (44.08) | ||
| 11~15 | 8739 | 2514 (28.77) | 772 (8.83) | 3286 (37.60) | ||
| 16~20 | 4750 | 730 (15.37) | 104 (2.19) | 834 (17.56) | ||
| 21~25 | 1707 | 13 (0.76) | 0 (0.00) | 13 (0.76) | ||
| ≥26 | 5115 | 0 (0.00) | 0 (0.00) | 0 (0.00) | ||
| Demographic Groups | 198.135 | <0.001 | ||||
| Scattered children | 1970 | 280 (14.21) | 61 (3.10) | 341 (17.31) | ||
| Kindergarten children | 1449 | 306 (21.12) | 204 (14.08) | 510 (35.20) | ||
| Primary school students | 8661 | 2653 (30.63) | 1126 (13.00) | 3779 (43.63) | ||
| Middle school students | 8842 | 2031 (22.97) | 464 (5.25) | 2495 (28.22) | ||
| College student | 2401 | 99 (4.12) | 69 (2.87) | 168 (6.99) | ||
| Others | 5455 | 75 (1.37) | 5 (0.09) | 80 (1.46) | ||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Du, Z.-p.; Long, Z.-p.; Chen, M.-a.; Sheng, W.; He, Y.; Zhang, G.-m.; Wu, X.-h.; Pang, Z.-f. Characteristics of Varicella Breakthrough Cases in Jinhua City, 2016–2024. Vaccines 2025, 13, 842. https://doi.org/10.3390/vaccines13080842
Du Z-p, Long Z-p, Chen M-a, Sheng W, He Y, Zhang G-m, Wu X-h, Pang Z-f. Characteristics of Varicella Breakthrough Cases in Jinhua City, 2016–2024. Vaccines. 2025; 13(8):842. https://doi.org/10.3390/vaccines13080842
Chicago/Turabian StyleDu, Zhi-ping, Zhi-ping Long, Meng-an Chen, Wei Sheng, Yao He, Guang-ming Zhang, Xiao-hong Wu, and Zhi-feng Pang. 2025. "Characteristics of Varicella Breakthrough Cases in Jinhua City, 2016–2024" Vaccines 13, no. 8: 842. https://doi.org/10.3390/vaccines13080842
APA StyleDu, Z.-p., Long, Z.-p., Chen, M.-a., Sheng, W., He, Y., Zhang, G.-m., Wu, X.-h., & Pang, Z.-f. (2025). Characteristics of Varicella Breakthrough Cases in Jinhua City, 2016–2024. Vaccines, 13(8), 842. https://doi.org/10.3390/vaccines13080842





