COVID-19 Vaccine Hesitancy among English-Speaking Pregnant Women Living in Rural Western United States
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Key Informant Interviews
2.3. Participants and Procedures
2.4. Measures and Instruments
2.5. Ad Design
2.6. Data Analysis
3. Results
3.1. Participants
3.2. Quantitative Analysis of Social Media Ad Reactions Promoting Vaccination
3.2.1. Overview of Ads and Participants Viewing Ads
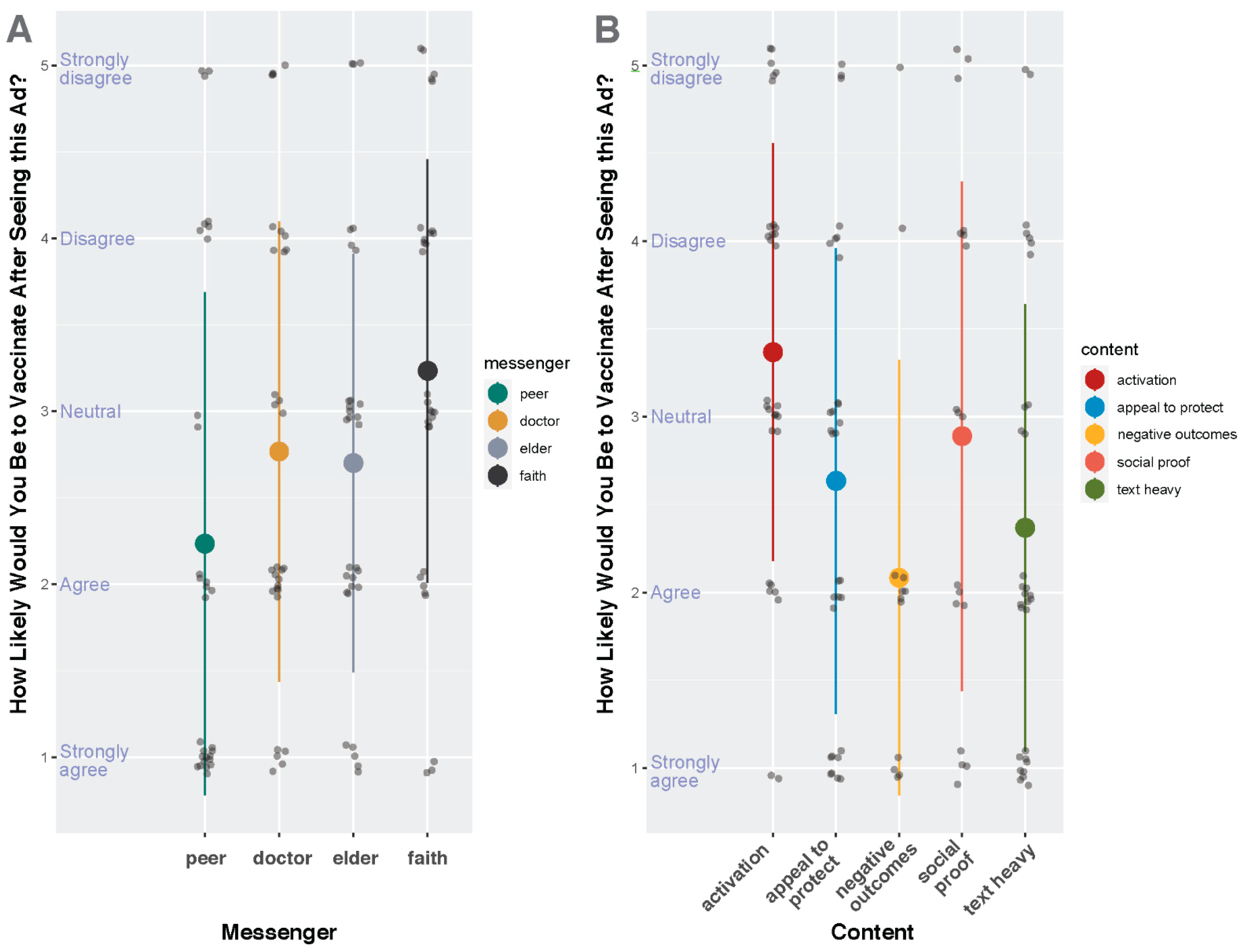
3.2.2. Quantitative Analysis
3.2.3. Qualitative Reactions to Social Media Ads Promoting Vaccination
Participants Favored Ads Featuring Scientific Content
Participants Reacted Negatively toward Ads Featuring Elderly and Faith-Based Messengers and Activation-Based Content
3.3. Qualitative Analysis of Interview Themes
3.3.1. Themes Emerging from the Qualitative Analysis
3.3.2. Facilitators and Barriers to Vaccine Uptake
3.3.3. Self-perceived Risk of COVID
3.3.4. Sources of Information on Health Decision-Making
3.3.5. Vaccine Hesitancy
3.3.6. Relationship with Care Providers Influences Vaccine Acceptance
4. Discussion
4.1. Summary of Study Findings
4.2. Study Findings in the Context of Literature
4.3. Implications for Clinical Care or Public Health Campaigns
4.4. Strengths and Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Centers for Disease Control and Prevention. Maternal and infant outcomes among severely ill pregnant and postpartum women with 2009 pandemic influenza A (H1N1)—United States, April 2009–August 2010. MMWR Morb. Mortal Wkly Rep. 2011, 60, 1193–1196. [Google Scholar]
- Bianchi-Jassir, F.; Seale, A.C.; Kohli-Lynch, M.; Lawn, J.E.; Baker, C.J.; Bartlett, L.; Cutland, C.; Gravett, M.G.; Heath, P.T.; Ip, M.; et al. Preterm Birth Associated with Group B Streptococcus Maternal Colonization Worldwide: Systematic Review and Meta-analyses. Clinical infectious diseases: An official publication of the Infectious Diseases Society of America. Clin. Infect. Dis. 2017, 65 (Suppl. 2), S133–S142. [Google Scholar] [CrossRef] [PubMed]
- DeSisto, C.L.; Wallace, B.; Simeone, R.M.; Polen, K.; Ko, J.Y.; Meaney-Delman, D.; Ellington, S.R. Risk for Stillbirth Among Women with and Without COVID-19 at Delivery Hospitalization—United States, March 2020–September 2021. MMWR Morb. Mortal Wkly. Rep. 2021, 70, 1640–1645. [Google Scholar] [CrossRef]
- Coler, B.; Cervantes, O.; Li, M.; Coler, C.; Li, A.; Shivakumar, M.; Every, E.; Schwartz, D.; Adams Waldorf, K.M. Common pathways targeted by viral hemorrhagic fever viruses to infect the placenta and increase the risk of stillbirth. Placenta 2022. [Google Scholar] [CrossRef] [PubMed]
- McClure, E.M.; Saleem, S.; Goudar, S.S.; Garces, A.; Whitworth, R.; Esamai, F.; Patel, A.B.; Tikmani, S.S.; Mwenechanya, M.; Chomba, E.; et al. Stillbirth 2010–2018: A prospective, population-based, multi-country study from the Global Network. Reprod. Health 2020, 17 (Suppl. 2), 146. [Google Scholar] [CrossRef]
- World Health Organization. Child Mortality (Under 5 Years). Available online: https://www.who.int/news-room/fact-sheets/detail/levels-and-trends-in-child-under-5-mortality-in-2020 (accessed on 6 June 2023).
- Elahi, S.; Ertelt, J.M.; Kinder, J.M.; Jiang, T.T.; Zhang, X.; Xin, L.; Chaturvedi, V.; Strong, B.S.; Qualls, J.E.; Steinbrecher, K.A.; et al. Immunosuppressive CD71+ erythroid cells compromise neonatal host defence against infection. Nature 2013, 504, 158–162. [Google Scholar] [CrossRef]
- Bunis, D.G.; Bronevetsky, Y.; Krow-Lucal, E.; Bhakta, N.R.; Kim, C.C.; Nerella, S.; Jones, N.; Mendoza, V.F.; Bryson, Y.J.; Gern, J.E.; et al. Single-Cell Mapping of Progressive Fetal-to-Adult Transition in Human Naive T Cells. Cell Rep. 2021, 34, 108573. [Google Scholar] [CrossRef]
- Chu, H.Y.; Englund, J.A. Maternal immunization. Birth Defects Res. 2017, 109, 379–386. [Google Scholar] [CrossRef]
- Perl, S.H.; Uzan-Yulzari, A.; Klainer, H.; Asiskovich, L.; Youngster, M.; Rinott, E.; Youngster, I. SARS-CoV-2-Specific Antibodies in Breast Milk After COVID-19 Vaccination of Breastfeeding Women. JAMA 2021, 325, 2013–2014. [Google Scholar] [CrossRef]
- Alcendor, D.J.; Matthews-Juarez, P.; Smoot, D.; Hildreth, J.E.K.; Tabatabai, M.; Wilus, D.; Brown, K.Y.; Juarez, P.D. The COVID-19 Vaccine and Pregnant Minority Women in the US: Implications for Improving Vaccine Confidence and Uptake. Vaccines 2022, 10, 2122. [Google Scholar] [CrossRef]
- Bhattacharya, O.; Siddiquea, B.N.; Shetty, A.; Afroz, A.; Billah, B. COVID-19 vaccine hesitancy among pregnant women: A systematic review and meta-analysis. BMJ Open 2022, 12, e061477. [Google Scholar] [CrossRef] [PubMed]
- Battarbee, A.N.; Stockwell, M.S.; Varner, M.; Newes-Adeyi, G.; Daugherty, M.; Gyamfi-Bannerman, C.; Tita, A.T.; Vorwaller, K.; Vargas, C.; Subramaniam, A.; et al. Attitudes Toward COVID-19 Illness and COVID-19 Vaccination among Pregnant Women: A Cross-Sectional Multicenter Study during August-December 2020. Am. J. Perinatol. 2022, 39, 75–83. [Google Scholar] [CrossRef] [PubMed]
- Smith, E.R.; Oakley, E.; Grandner, G.W.; Ferguson, K.; Farooq, F.; Afshar, Y.; Ahlberg, M.; Ahmadzia, H.; Akelo, V.; Aldrovandi, G.; et al. Adverse maternal, fetal, and newborn outcomes among pregnant women with SARS-CoV-2 infection: An individual participant data meta-analysis. BMJ Glob. Health 2023, 8, e009495. [Google Scholar] [CrossRef] [PubMed]
- Schwartz, D.A.; Avvad-Portari, E.; Babál, P.; Baldewijns, M.; Blomberg, M.; Bouachba, A.; Camacho, J.; Collardeau-Frachon, S.; Colson, A.; Dehaene, I.; et al. Placental Tissue Destruction and Insufficiency From COVID-19 Causes Stillbirth and Neonatal Death From Hypoxic-Ischemic Injury: A Study of 68 Cases With SARS-CoV-2 Placentitis From 12 Countries. Arch. Pathol. Lab. Med. 2022, 146, 660–676. [Google Scholar] [CrossRef] [PubMed]
- Papageorghiou, A.T.; Deruelle, P.; Gunier, R.B.; Rauch, S.; Garcia-May, P.K.; Mhatre, M.; Usman, M.A.; Abd-Elsalam, S.; Etuk, S.; Simmons, L.E.; et al. Preeclampsia and COVID-19: Results from the INTERCOVID prospective longitudinal study. Am. J. Obstet. Gynecol. 2021, 225, 289-e1–289-e17. [Google Scholar] [CrossRef]
- Villar, J.; Ariff, S.; Gunier, R.B.; Thiruvengadam, R.; Rauch, S.; Kholin, A.; Roggero, P.; Prefumo, F.; Do Vale, M.S.; Cardona-Perez, J.A.; et al. Maternal and Neonatal Morbidity and Mortality Among Pregnant Women with and Without COVID-19 Infection: The INTERCOVID Multinational Cohort Study. JAMA Pediatr. 2021, 175, 817–826. [Google Scholar] [CrossRef]
- Lokken, E.M.; Huebner, E.M.; Taylor, G.G.; Hendrickson, S.; Vanderhoeven, J.; Kachikis, A.; Coler, B.; Walker, C.L.; Sheng, J.S.; Al-Haddad, B.J.; et al. Disease Severity, Pregnancy Outcomes and Maternal Deaths among Pregnant Patients with SARS-CoV-2 Infection in Washington State. Am. J. Obstet. Gynecol. 2021, 225, e1–e77. [Google Scholar] [CrossRef]
- Lokken, E.M.; Walker, C.L.; Delaney, S.; Kachikis, A.; Kretzer, N.M.; Erickson, A.; Resnick, R.; Vanderhoeven, J.; Hwang, J.K.; Barnhart, N.; et al. Clinical characteristics of 46 pregnant women with a severe acute respiratory syndrome coronavirus 2 infection in Washington State. Am. J. Obstet. Gynecol. 2020, 223, 911.e1–911.e14. [Google Scholar] [CrossRef]
- Sun, Y.; Monnat, S.M. Rural-urban and within-rural differences in COVID-19 vaccination rates. J. Rural Health 2022, 38, 916–922. [Google Scholar] [CrossRef]
- Cheng, K.J.G.; Sun, Y.; Monnat, S.M. COVID-19 Death Rates Are Higher in Rural Counties with Larger Shares of Blacks and Hispanics. J. Rural Health 2020, 36, 602–608. [Google Scholar] [CrossRef]
- Kaur, R.; Callaghan, T.; Regan, A.K. Disparities in Prenatal Immunization Rates in Rural and Urban US Areas by Indicators of Access to Care. J. Rural Health 2023, 39, 142–152. [Google Scholar] [CrossRef] [PubMed]
- Abbasi, J. Widespread Misinformation About Infertility Continues to Create COVID-19 Vaccine Hesitancy. JAMA 2022, 327, 1013–1015. [Google Scholar] [CrossRef] [PubMed]
- Blakeway, H.; Prasad, S.; Kalafat, E.; Heath, P.T.; Ladhani, S.N.; Le Doare, K.; Magee, L.A.; O’brien, P.; Rezvani, A.; von Dadelszen, P.; et al. COVID-19 vaccination during pregnancy: Coverage and safety. Am. J. Obstet. Gynecol. 2022, 226, 236.e1–236.e14. [Google Scholar] [CrossRef]
- Cui, Y.; Binger, K.S.; Tsaih, S.W.; Palatnik, A. Factors of COVID-19 vaccine hesitancy during pregnancy: A cross-sectional study. Am. J. Obstet. Gynecol. 2022, 226, S306. [Google Scholar] [CrossRef]
- Wilson, R.J.; Paterson, P.; Jarrett, C.; Larson, H.J. Understanding factors influencing vaccination acceptance during pregnancy globally: A literature review. Vaccine 2015, 33, 6420–6429. [Google Scholar] [CrossRef] [PubMed]
- Yasmin, F.; Najeeb, H.; Moeed, A.; Naeem, U.; Asghar, M.S.; Chughtai, N.U.; Yousaf, Z.; Seboka, B.T.; Ullah, I.; Lin, C.Y.; et al. COVID-19 Vaccine Hesitancy in the United States: A Systematic Review. Front. Public Health 2021, 9, 770985. [Google Scholar] [CrossRef]
- Cui, Y.; Binger, K.; Palatnik, A. Attitudes and Beliefs Associated With COVID-19 Vaccination During Pregnancy. JAMA Netw. Open 2022, 5, e227430. [Google Scholar] [CrossRef]
- Walker, K.K.; Head, K.J.; Owens, H.; Zimet, G.D. A qualitative study exploring the relationship between mothers’ vaccine hesitancy and health beliefs with COVID-19 vaccination intention and prevention during the early pandemic months. Hum. Vaccin. Immunother. 2021, 17, 3355–3364. [Google Scholar] [CrossRef]
- Dube, E.; Ward, J.K.; Verger, P.; MacDonald, N.E. Vaccine Hesitancy, Acceptance, and Anti-Vaccination: Trends and Future Prospects for Public Health. Annu. Rev. Public Health 2021, 42, 175–191. [Google Scholar] [CrossRef]
- Balhotra, K.; Chahal, K.; Silver, M.; Atallah, F.; Narayanamoorthy, S.; Minkoff, H. COVID-19 Vaccine Hesitancy in an Underrepresented Minority Community. J. Community Health 2023, 48, 489–495. [Google Scholar] [CrossRef]
- Ehrlich, S.F.; Maples, J.; Burnette, S.; Rand, B.; Zite, N.; Paudel, A.; Fortner, K. Individual-level determinants of COVID-19 vaccination in pregnant people in East Tennessee. Int. J. Gynaecol. Obstet. 2023. [CrossRef] [PubMed]
- Hahn, M.B.; Fried, R.L.; Cochran, P.; Eichelberger, L.P. Evolving perceptions of COVID-19 vaccines among remote Alaskan communities. Int. J. Circumpolar. Health 2022, 81, 2021684. [Google Scholar] [CrossRef] [PubMed]
- Fairchild, A.L.; Bayer, R.; Green, S.H.; Colgrove, J.; Kilgore, E.; Sweeney, M.; Varma, J.K. The Two Faces of Fear: A History of Hard-Hitting Public Health Campaigns Against Tobacco and AIDS. Am. J. Public Health 2018, 108, 1180–1186. [Google Scholar] [CrossRef] [PubMed]

| Characteristic | Categories | N (%) |
|---|---|---|
| Race and Ethnicity * | White Black of African American Hispanic/Latino American Indian/Alaskan Native | 25 (83.3) 4 (13.3) 3 (10) 1 (3.3) |
| Pregnancy Status | Currently Pregnant Pregnant within Last Six Months | 10 (33.3) 20 (66.7) |
| Number of Children | 0 1 2–4 >4 Prefer Not to Say | 4 (13.3) 14 (46.7) 9 (30) 2 (6.7) 1 (3.3) |
| Marital Status | Married Not Married, Living with Partner | 29 (96.7) 1 (3.3) |
| Level of Education | Some High School High School Bachelor’s Degree Master’s Degree Trade School Prefer not to Say | 1 (3.3) 8 (26.7) 12 (40) 7 (23.3) 1 (3.3) 1 (3.3) |
| Employment Status | Employed Full-Time Employed Part-Time Seeking Opportunities Other | 10 (33.3) 8 (26.7) 4 (13.3) 8 (26.7) |
| Annual Household Income | <25,000 25,000–50,000 50,000–100,000 100,000–200,000 | 6 (20) 7 (23.3) 12 (40) 5 (16.7) |
| Religion | Not Religious Christian (Protestant) Christian (Catholic) Christian (Any other denomination) Other | 13 (43.3) 4 (13.3) 3 (10) 8 (26.7) 2 (6.7) |
| Political Affiliation | Very Liberal Slightly Liberal Slightly Conservative Very Conservative Prefer not to Say | 6 (20) 6 (20) 10 (33.3) 6 (20) 2 (6.7) |
| Vaccination Status | Received a COVID-19 Vaccine Not Vaccinated | 21 (70) 9 (30) |
| Type of COVID-19 Vaccine Received | Type of COVID-19 Vaccine Received (N = 21) ** Johnson & Johnson Moderna Pfizer | 3 (14.3) 12 (57.1) 10 (47.6) |
| Number of COVID-19 Boosters Received | 1st Booster 2nd Booster 3rd Booster | 12 (40) 6 (20) 2 (6.7) |
| Messenger | Ad Number | Content | Number of Views |
|---|---|---|---|
| Peer | 1 | Appeal to Protect | 5 |
| 2 | Text-Heavy | 5 | |
| 3 | Social Proof | 10 | |
| 4 | Information (Negative outcomes) | 5 | |
| 5 | Activation | 5 | |
| Elder | 6 | Appeal to Protect | 13 |
| 7 | Text-Heavy | 6 | |
| 8 | Activation | 9 | |
| Doctor | 9 | Appeal to Protect | 7 |
| 10 | Text-Heavy | 6 | |
| 11 | Social Proof | 9 | |
| 12 | Information (Negative outcomes) | 6 | |
| 13 | Activation | 2 | |
| Faith | 14 | Appeal to Protect | 4 |
| 15 | Text-Heavy | 11 | |
| 16 | Activation | 14 |
| Messenger | How Likely | Content | How Likely | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Predictors | Estimates | SE | t | p | Predictors | Estimates | SE | t | p |
| (Intercept) a | 2.23 | 0.2 | 9.4 | <0.001 | (Intercept) b | 2.04 | 0.4 | 5.8 | <0.001 |
| Doctor | 0.53 | 0.3 | 1.9 | 0.063 | Activation | 1.32 | 0.4 | 3.5 | 0.001 |
| Elder | 0.47 | 0.3 | 1.6 | 0.103 | Social Proof | 0.87 | 0.4 | 2 | 0.047 |
| Faith | 1.00 | 0.3 | 3.5 | 0.001 | Text Heavy | 0.32 | 0.4 | 0.9 | 0.395 |
| Appeal to Protect | 0.59 | 0.4 | 1.6 | 0.123 | |||||
| Random Effects | |||||||||
| σ2 (residual variance) | 1.21 | σ2 (residual variance) | 1.15 | ||||||
| τ00 (random intercept variance) | 0.50 | τ00 (random intercept variance) | 0.52 | ||||||
| N | 30 | N | 30 | ||||||
| Observations | 120 | Observations | 120 | ||||||
| Marginal R2 | 0.069 | Marginal R2 | 0.103 | ||||||
| Conditional R2 | 0.340 | Conditional R2 | 0.382 | ||||||
| Themes | Sub-Themes |
|---|---|
| Vaccine Uptake | Facilitators Barriers |
| Self-perceived Risk of COVID-19 During Pregnancy | Greater Perceived Risk No Perceived Change in Risk |
| Information Source for Health Decision-Making | Trusted Sources Distrusted Sources |
| Vaccine Hesitancy | Vaccine Hesitant Vaccine Positive |
| Relationship with Care Provider | Positive Relationship Negative Relationship |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cox, E.; Sanchez, M.; Baxter, C.; Crary, I.; Every, E.; Munson, J.; Stapley, S.; Stonehill, A.; Taylor, K.; Widmann, W.; et al. COVID-19 Vaccine Hesitancy among English-Speaking Pregnant Women Living in Rural Western United States. Vaccines 2023, 11, 1108. https://doi.org/10.3390/vaccines11061108
Cox E, Sanchez M, Baxter C, Crary I, Every E, Munson J, Stapley S, Stonehill A, Taylor K, Widmann W, et al. COVID-19 Vaccine Hesitancy among English-Speaking Pregnant Women Living in Rural Western United States. Vaccines. 2023; 11(6):1108. https://doi.org/10.3390/vaccines11061108
Chicago/Turabian StyleCox, Elizabeth, Magali Sanchez, Carly Baxter, Isabelle Crary, Emma Every, Jeff Munson, Simone Stapley, Alex Stonehill, Katherine Taylor, Willamina Widmann, and et al. 2023. "COVID-19 Vaccine Hesitancy among English-Speaking Pregnant Women Living in Rural Western United States" Vaccines 11, no. 6: 1108. https://doi.org/10.3390/vaccines11061108
APA StyleCox, E., Sanchez, M., Baxter, C., Crary, I., Every, E., Munson, J., Stapley, S., Stonehill, A., Taylor, K., Widmann, W., Karasz, H., & Adams Waldorf, K. M. (2023). COVID-19 Vaccine Hesitancy among English-Speaking Pregnant Women Living in Rural Western United States. Vaccines, 11(6), 1108. https://doi.org/10.3390/vaccines11061108







