Evaluation of the Safety and Immunogenicity of Fractional Intradermal COVID-19 Vaccines as a Booster: A Pilot Study
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Design and Participants
2.2. Chemiluminescent Microparticle Assay (CMIA) for Anti-SARS-CoV-2 Binding Antibody
2.3. 50% Plaque Reduction Neutralization Test (PRNT50) against SARS-CoV-2 Strains
2.4. IFN-γ ELISpot
2.5. Statistical Analysis
3. Results
3.1. Baseline Characteristics of Study Participants
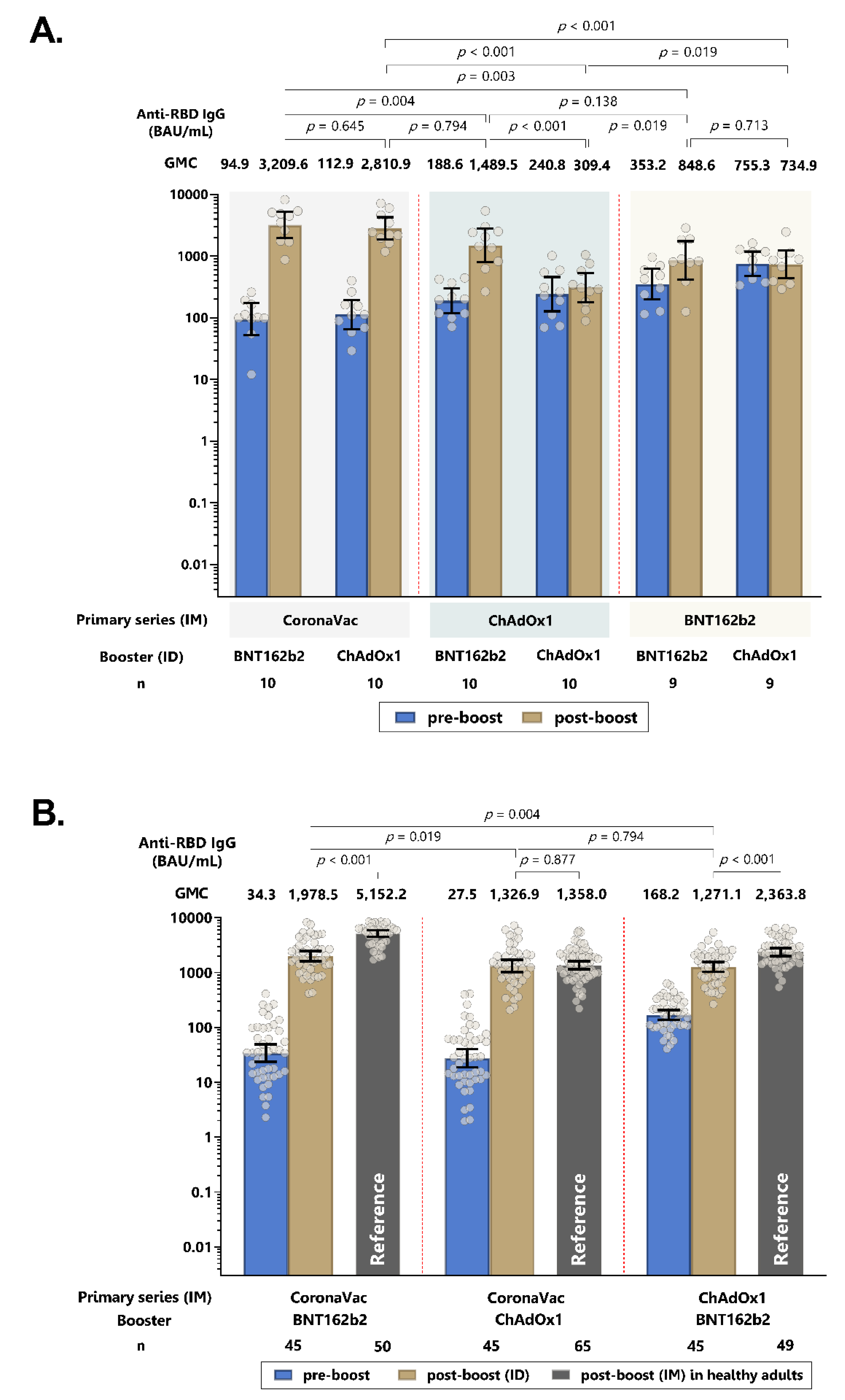
3.2. Humoral Immune Responses of Intradermal ChAdOx1 and BNT162b2 Booster
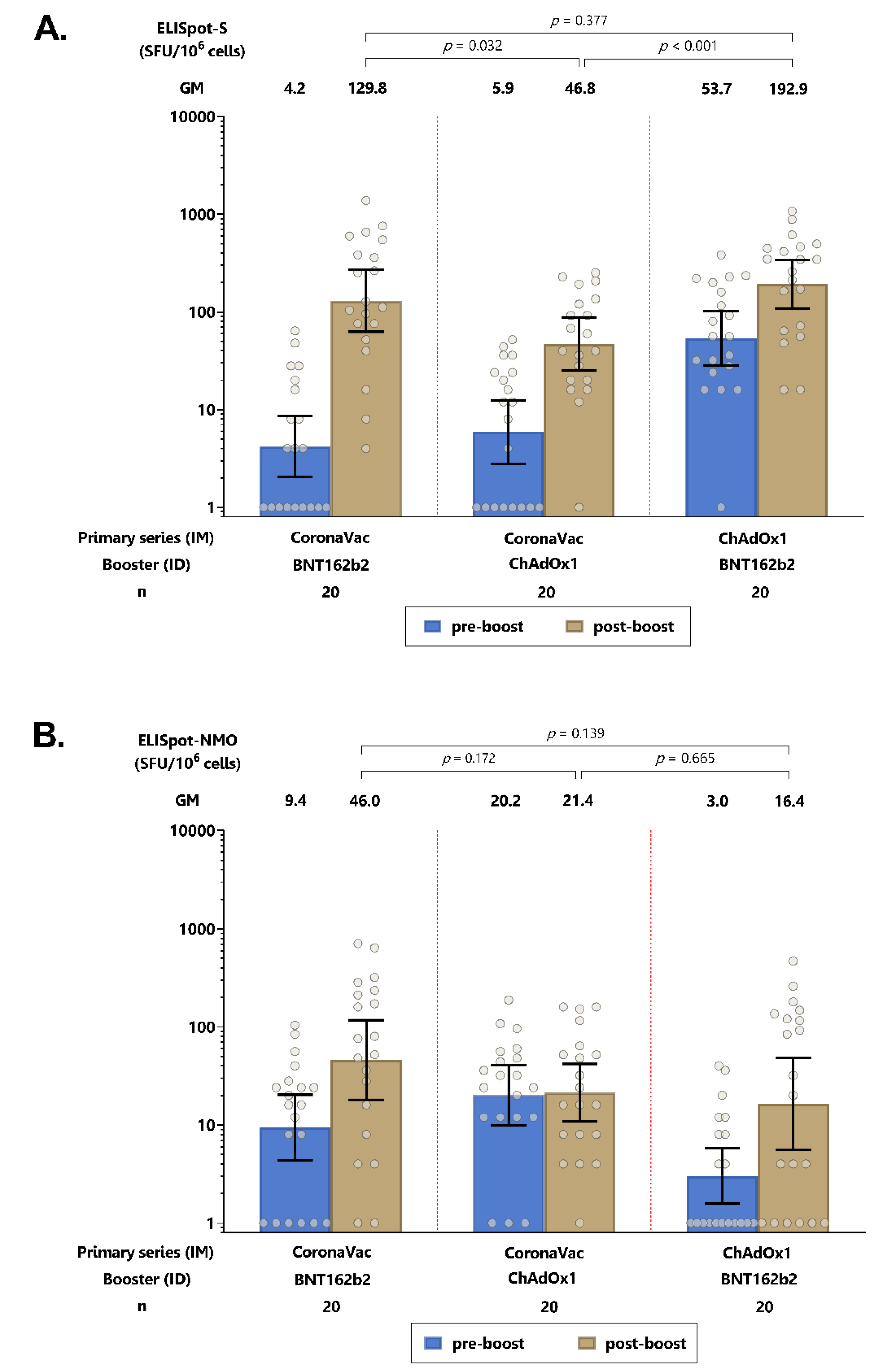
3.3. Cellular Immune Responses of Intradermal ChAdOx1 and BNT162b2 Booster
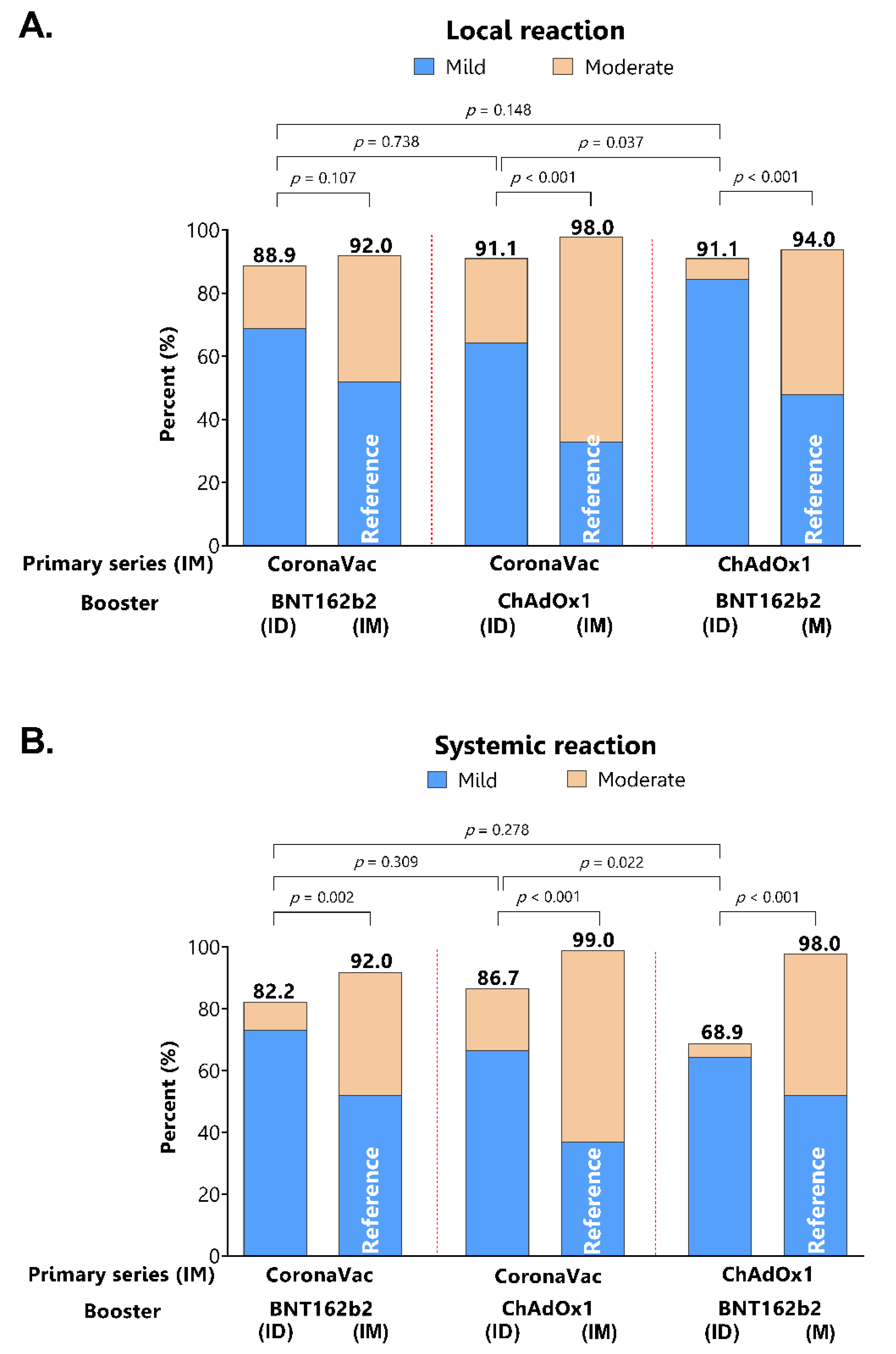
3.4. Reactogenicity of Intradermal Booster Injections
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- The World Health Organization. 11 Vaccines Granted Emergency Use Listing (EUL) by WHO. Available online: https://www.who.int/news/item/19-05-2022-who-validates-11th-vaccine-for-covid-19 (accessed on 20 June 2022).
- Ssentongo, P.; Ssentongo, A.E.; Voleti, N.; Groff, D.; Sun, A.; Ba, D.M.; Nunez, J.; Parent, L.J.; Chinchilli, V.M.; Paules, C.I. SARS-CoV-2 vaccine effectiveness against infection, symptomatic and severe COVID-19: A systematic review and meta-analysis. BMC Infect. Dis. 2022, 22, 439. [Google Scholar] [CrossRef] [PubMed]
- Accorsi, E.K.; Britton, A.; Fleming-Dutra, K.E.; Smith, Z.R.; Shang, N.; Derado, G.; Miller, J.; Schrag, S.J.; Verani, J.R. Association Between 3 Doses of mRNA COVID-19 Vaccine and Symptomatic Infection Caused by the SARS-CoV-2 Omicron and Delta Variants. JAMA 2022, 327, 639–651. [Google Scholar] [CrossRef] [PubMed]
- Khoury, D.S.; Cromer, D.; Reynaldi, A.; Schlub, T.E.; Wheatley, A.K.; Juno, J.A.; Subbarao, K.; Kent, S.J.; Triccas, J.A.; Davenport, M.P. Neutralizing antibody levels are highly predictive of immune protection from symptomatic SARS-CoV-2 infection. Nat. Med. 2021, 27, 1205–1211. [Google Scholar] [CrossRef] [PubMed]
- Moss, P. The T cell immune response against SARS-CoV-2. Nat. Immunol. 2022, 23, 186–193. [Google Scholar] [CrossRef] [PubMed]
- Ritchie, H.; Mathieu, E.; Rodés-Guirao, L.; Appel, C.; Giattino, C.; Ortiz-Ospina, E.; Hasell, J.; Macdonald, B.; Beltekian, D.; Roser, M. Coronavirus Pandemic (COVID-19). Our World Data 2020. Available online: https://ourworldindata.org/covid-vaccinations?country=JPN~USA (accessed on 16 June 2022).
- Roozen, G.V.T.; Roukens, A.H.E.; Roestenberg, M. COVID-19 vaccine dose sparing: Strategies to improve vaccine equity and pandemic preparedness. Lancet Glob. Health 2022, 10, e570–e573. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Shaw, R.H.; Stuart, A.S.V.; Greenland, M.; Aley, P.K.; Andrews, N.J.; Cameron, J.C.; Charlton, S.; Clutterbuck, E.A.; Collins, A.M.; et al. Safety and immunogenicity of heterologous versus homologous prime-boost schedules with an adenoviral vectored and mRNA COVID-19 vaccine (Com-COV): A single-blind, randomised, non-inferiority trial. Lancet 2021, 4, 856–869. [Google Scholar] [CrossRef]
- Stuart, A.S.V.; Shaw, R.H.; Liu, X.; Greenland, M.; Aley, P.K.; Andrews, N.J.; Cameron, J.C.; Charlton, S.; Clutterbuck, E.A.; Collins, A.M.; et al. Immunogenicity, safety, and reactogenicity of heterologous COVID-19 primary vaccination incorporating mRNA, viral-vector, and protein-adjuvant vaccines in the UK (Com-COV2): A single-blind, randomised, phase 2, non-inferiority trial. Lancet 2022, 399, 802. [Google Scholar] [CrossRef]
- Atmar, R.L.; Lyke, K.E.; Deming, M.E.; Jackson, L.A.; Branche, A.R.; El Sahly, H.M.; Rostad, C.A.; Martin, J.M.; Johnston, C.; Rupp, R.E.; et al. Homologous and Heterologous Covid-19 Booster Vaccinations. N. Engl. J. Med. 2022, 386, 1046–1057. [Google Scholar] [CrossRef] [PubMed]
- Tenbusch, M.; Schumacher, S.; Vogel, E.; Priller, A.; Held, J.; Steininger, P.; Beileke, S.; Irrgang, P.; Brockhoff, R.; Salmanton-García, J.; et al. Heterologous prime-boost vaccination with ChAdOx1 nCoV-19 and BNT162b2. Lancet Infect. Dis. 2021, 21, 1212–1213. [Google Scholar] [CrossRef]
- Costa, S.A.; Weckx, L.; Clemens, R.; Mendes, A.V.A.; Souza, A.R.; Silveira, M.B.; da Guarda, S.N.F.; de Nobrega, M.M.; de Moraes Pinto, M.I.; Gonzalez, I.G.S.; et al. Heterologous versus homologous COVID-19 booster vaccination in previous recipients of two doses of CoronaVac COVID-19 vaccine in Brazil (RHH-001): A phase 4, non-inferiority, single blind, randomised study. Lancet 2022, 399, 521–529. [Google Scholar] [CrossRef]
- Levin, C.; Perrin, H.; Combadiere, B. Tailored immunity by skin antigen-presenting cells. Hum Vaccin Immunother 2015, 11, 27–36. [Google Scholar] [CrossRef]
- Meijer, W.J.; Wensing, A.M.J.; Bos, A.A.; Kuiphuis, J.C.F.; Hagelen, E.M.M.; Wilschut, J.C.; de Vries, M.J.T.; Riezebos-Brilman, A. Influenza vaccination in healthcare workers; comparison of side effects and preferred route of administration of intradermal versus intramuscular administration. Vaccine 2017, 35, 1517–1523. [Google Scholar] [CrossRef]
- The World Health Organization. Interim Recommendations for Heterologous COVID-19 Vaccine Schedules; The World Health Organization: Geneva, Switzerland, 2021; Available online: https://apps.who.int/iris/bitstream/handle/10665/350635/WHO-2019-nCoV-vaccines-SAGE-recommendation-heterologous-schedules-2021.1-eng.pdf?sequence=1 (accessed on 26 January 2022).
- The World Health Organization. Intradermal Delivery of Vaccines; The World Health Organization: Geneva, Switzerland, 2009; Available online: https://media.path.org/documents/TS_opt_idd_review.pdf?_gl=1*14mu43d*_ga*MTk5OTI1MzI5Ny4xNjU3Nzg1MzQ5*_ga_YBSE7ZKDQM*MTY1Nzc4NTM0OC4xLjAuMTY1Nzc4NTM0OC4w (accessed on 3 August 2021).
- Egunsola, O.; Clement, F.; Taplin, J.; Mastikhina, J.; Li, J.B.; Lorenzetti, D.L.; Dowsett, L.E.; Noseworthy, T. Immunogenicity and Safety of Reduced-Dose Intradermal vs Intramuscular Influenza Vaccines: A Systematic Review and Meta-analysis. JAMA Netw. Open 2021, 4, e2035693. [Google Scholar] [CrossRef]
- Angkasekwinai, N.; Sewatanon, J.; Niyomnaitham, S.; Phumiamorn, S.; Sukapirom, K.; Sapsutthipas, S.; Sirijatuphat, R.; Wittawatmongkol, O.; Senawong, S.; Mahasirimongkol, S.; et al. Comparison of safety and immunogenicity of CoronaVac and ChAdOx1 against the SARS-CoV-2 circulating variants of concern (Alpha, Delta, Beta) in Thai healthcare workers. Vaccine X 2022, 10, 100153. [Google Scholar] [CrossRef]
- Angkasekwinai, N.; Niyomnaitham, S.; Sewatanon, J.; Phumiamorn, S.; Sukapirom, K.; Senawong, S.; Mahasirimongkol, S.; Toh, Z.Q.; Umrod, P.; Somporn, T.; et al. The Immunogenicity against Variants of Concern and Reactogenicity of Four COVID-19 Booster Vaccinations Following CoronaVac or ChAdOx1 nCoV-19 Primary Series. 2022. Available online: https://www.medrxiv.org/content/10.1101/2021.11.29.21266947v2.article-metrics (accessed on 20 June 2022).
- Schweiger, M. Intradermal covid-19 vaccination could solve supply problems. BMJ 2021, 374, n1980. [Google Scholar] [CrossRef]
- Pinpathomrat, N.; Intapiboon, P.; Seepathomnarong, P.; Ongarj, J.; Sophonmanee, R.; Hengprakop, J.; Surasombatpattana, S.; Uppanisakorn, S.; Mahasirimongkol, S.; Sawaengdee, W.; et al. Immunogenicity and safety of an intradermal ChAdOx1 nCoV-19 boost in a healthy population. NPJ Vaccines 2022, 7, 52. [Google Scholar] [CrossRef] [PubMed]
- Tawinprai, K.; Siripongboonsitti, T.; Porntharukchareon, T.; Wittayasak, K.; Thonwirak, N.; Soonklang, K.; Sornsamdang, G.; Auewarakul, C.; Mahanonda, N. Immunogenicity and safety of an intradermal fractional third dose of ChAdOx1 nCoV-19/AZD1222 vaccine compared with those of a standard intramuscular third dose in volunteers who previously received two doses of CoronaVac: A randomized controlled trial. Vaccine 2022, 40, 1761–1767. [Google Scholar] [CrossRef]
- Intapiboon, P.; Seepathomnarong, P.; Ongarj, J.; Surasombatpattana, S.; Uppanisakorn, S.; Mahasirimongkol, S.; Sawaengdee, W.; Phumiamorn, S.; Sapsutthipas, S.; Sangsupawanich, P.; et al. Immunogenicity and Safety of an Intradermal BNT162b2 mRNA Vaccine Booster after Two Doses of Inactivated SARS-CoV-2 Vaccine in Healthy Population. Vaccines 2021, 9, 1375. [Google Scholar] [CrossRef] [PubMed]
- Jara, A.; Undurraga, E.A.; Zubizarreta, J.R.; Gonzalez, C.; Pizarro, A.; Acevedo, J.; Leo, K.; Paredes, F.; Bralic, T.; Vergara, V.; et al. Effectiveness of homologous and heterologous booster doses for an inactivated SARS-CoV-2 vaccine: A large-scale prospective cohort study. Lancet Glob. Health 2022, 10, e798–e806. [Google Scholar] [CrossRef]


| Primary Series (IM-IM)-Booster (ID) | Types of Vaccines | ||||
|---|---|---|---|---|---|
| All | CoronaVac- BNT162b2 | CoronaVac- ChAdOx1 | ChAdOx1-BNT162b2 | p-Value | |
| Number of subjects, n (%) | 135 (100.0) | 45 (33.3) | 45 (33.3) | 45 (33.3) | - |
| Age (years), median (IQR) | 39.0 (30.0, 46.0) | 38.0 (29.0, 44.0) | 39.0 (29.0, 45.0) | 43.0 (30.0, 50.0) | 0.186 |
| Male, n (%) | 60 (44.4) | 18 (30.0) | 23 (38.3) | 19 (31.7) | 0.533 |
| Body mass index: BMI (kg/m2), median (IQR) | 24.0 (21.5, 26.6) | 24.4 (22.3, 26.8) | 23.6 (20.4, 26.5) | 23.9 (21.5, 26.4) | 0.719 |
| Interval between last dose of primary series and booster (weeks), median (IQR) | 9.9 (7.4, 12.0) | 11.6 (8.9, 16.3) | 10.3 (9.1, 12.6) | 7.4 (6.0, 9.6) | 0.083 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Niyomnaitham, S.; Chatsiricharoenkul, S.; Toh, Z.Q.; Senawong, S.; Pheerapanyawaranun, C.; Phumiamorn, S.; Licciardi, P.V.; Chokephaibulkit, K. Evaluation of the Safety and Immunogenicity of Fractional Intradermal COVID-19 Vaccines as a Booster: A Pilot Study. Vaccines 2022, 10, 1497. https://doi.org/10.3390/vaccines10091497
Niyomnaitham S, Chatsiricharoenkul S, Toh ZQ, Senawong S, Pheerapanyawaranun C, Phumiamorn S, Licciardi PV, Chokephaibulkit K. Evaluation of the Safety and Immunogenicity of Fractional Intradermal COVID-19 Vaccines as a Booster: A Pilot Study. Vaccines. 2022; 10(9):1497. https://doi.org/10.3390/vaccines10091497
Chicago/Turabian StyleNiyomnaitham, Suvimol, Somruedee Chatsiricharoenkul, Zheng Quan Toh, Sansnee Senawong, Chatkamol Pheerapanyawaranun, Supaporn Phumiamorn, Paul V. Licciardi, and Kulkanya Chokephaibulkit. 2022. "Evaluation of the Safety and Immunogenicity of Fractional Intradermal COVID-19 Vaccines as a Booster: A Pilot Study" Vaccines 10, no. 9: 1497. https://doi.org/10.3390/vaccines10091497
APA StyleNiyomnaitham, S., Chatsiricharoenkul, S., Toh, Z. Q., Senawong, S., Pheerapanyawaranun, C., Phumiamorn, S., Licciardi, P. V., & Chokephaibulkit, K. (2022). Evaluation of the Safety and Immunogenicity of Fractional Intradermal COVID-19 Vaccines as a Booster: A Pilot Study. Vaccines, 10(9), 1497. https://doi.org/10.3390/vaccines10091497








